����ԭ��-���ӹ������۵�ȫŨ��Ti-Al��Ԫ��������ѧ���ʼ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���6��
�������ߣ������� ʯ�� ������ ������ ����
����ҳ�룺1256 - 1264
�ؼ��ʣ�Ti-Al��Ԫϵ��Ti��Al��������ӣ�����ѧģ�ͣ���������Ũ�ȣ�ԭ��-���ӹ�������
Key words��Ti-Al binary system; Ti; Al; activity coefficient; thermodynamic model; mass action concentration; atom and molecule coexistence theory
ժ Ҫ������ԭ��-���ӹ������۽��������¶�Ϊ1973~2273 K��ȫŨ�ȷ�ΧTi-Al��Ԫ������������Ũ��Ni������ѧģ�͡����ò�ͬ�¶�(1973��2073��2173��2273 K)�µ�ģ�ͼ��������õ�������ϡ��Һ(0
Abstract: The results of predicting thermodynamic properties in the full composition range of Ti-Al binary melts in a temperature range from 1973 to 2273 K were obtained by coupling with the developed thermodynamic model for calculating mass action concentration Ni of structural units in Ti-Al system based on the atom and molecule coexistence theory (AMCT). Temperature dependence of the activity coefficients of Ti and Al in natural logarithmic form in the infinitely dilute solution (0
Trans. Nonferrous Met. Soc. China 28(2018) 1256-1264
Sheng-chao DUAN1,2, Xiao SHI1,2, Wen-sheng YANG1,2, Han-jie GUO1,2, Jing GUO1,2
1. School of Metallurgical and Ecological Engineering, University of Science and Technology Beijing, Beijing 100083, China;
2. Beijing Key Laboratory of Special Melting and Preparation of High-end Metal Materials, University of Science and Technology Beijing, Beijing 100083, China
Received 11 January 2017; accepted 16 May 2017
Abstract: The results of predicting thermodynamic properties in the full composition range of Ti-Al binary melts in a temperature range from 1973 to 2273 K were obtained by coupling with the developed thermodynamic model for calculating mass action concentration Ni of structural units in Ti-Al system based on the atom and molecule coexistence theory (AMCT). Temperature dependence of the activity coefficients of Ti and Al in natural logarithmic form in the infinitely dilute solution (0
Key words: Ti-Al binary system; Ti; Al; activity coefficient; thermodynamic model; mass action concentration; atom and molecule coexistence theory
1 Introduction
Titanium and its alloys have been the key materials used in various fields due to the attractive mechanical properties, including the lightweight, high specific strength, high ductility, and low thermal conductivity [1-4]. Therefore, it is necessary to ascertain the thermodynamic properties of the alloys, which are important database in order to know phase stabilities and the bonding forces of components and also to predict ternary and high order phase diagrams based on Ti-Al system [5-8]. For these reasons, many efforts have been made to study the thermodynamic properties of Ti-Al system in the past several decades [8-10]. Having in mind the presented literature survey, however, it is clear that many studies on the Ti-Al system concern the investigation on its alloys in the solid state, only few articles describe the thermodynamic behavior in the liquid phase [11]. The experimental techniques employed by the investigators are summarized in Table 1.
In order to overcome the limitation of these studies, investigations on the reaction abilities of elements, especially activity coefficient of Ti (��Ti) and activity coefficient of Al (��Al) in Ti-Al binary melts were also carried out. In the recent past, the activity of Al in molten Ti-Al alloy was measured by MAEDA et al [4] using free evaporation method at 2073 K in an electron beam vacuum furnace. They reported that ��Al was about 0.03 under conditions of xAl��0.1, and it was about 0.1 at xAl��0.2. The experimental measurement of thermodynamic parameter is costly and difficult in the operation due to the systems containing highly reactive element such as titanium. It is therefore very necessary to develop a reliable model for predicting thermodynamic properties of Ti-Al binary melts. GUO et al [6] applied the Midema model to predict formation heat of Ti-Al binary system, which was used to calculate the activity of aluminum in molten Ti-Al alloy. On this basis, they studied the evaporation controlling mode of Al in the binary system during the cold crucible induction skull melting (ISM) process. KOSTOV et al [8] calculated the activity of titanium and aluminum in Ti-Al alloys using the FactSageTM thermochemical software and databases in a temperature range between 1773 and 2273 K. To the knowledge of the present authors, few prediction models were established to predict or evaluate the activity of Ti and Al in liquid Ti-Al alloys. Several prediction models were widely employed to describe thermodynamic properties of metallic melts with regressed artificial parameters obtained by experimental results [12], including but not limited to the regular solution model [13], Miedema model [14], molecular interaction volume model (MIVM) [15], non-random two-liquid (NRTL) equation [16], and Wilson equation [17]. Considering the fact that experimental difficulties and other factors, however, some parameters embodied in the foregoing theoretical models cannot be measured or estimated accurately so that the prediction models do not work well [18].
Table 1 Summary of previous investigations on thermodynamic activities in Ti-Al system

The atom and molecule coexistence theory (AMCT) has been reliably applied to predicting reaction abilities of multicomponent alloy system containing compound, eutecticum, metatectic, and solid solution [19,20]. In addition to this, investigators in Ref. [19,21-28] have proved that the calculated mass action concentration by means of AMCT-Ni model can be used for substituting the measured activities of corresponding elements in Fe-Si, Ge-Al, Au-Pb, Ag-Pd, Ca-Al-Si, and In-Sb-Cu systems relative to pure liquid matter as standard state. Also, it has been demonstrated in the authors�� most recent study, based on the reported values of activity of Ti and Al in the full composition range of Ti-Al binary melts [9], that 1) atoms of Ti and Al and three molecules as TiAl3, TiAl, and Ti5Al11 could exist in Ti-Al binary melts; 2) the calculated mass action concentration of free Ti (NTi) or mass action concentration of free Al (NAl) in the full composition range of Ti-Al binary melts by solving the nonlinear multivariable equations with MatlabTM, which has good 1:1 corresponding relationship with the reported activity of Ti (aR,Ti) or reported activity of Al (aR,Al) relative to pure liquid Ti(l) or Al(l) as standard state (see Fig. 1); 3) a rod-type relationship between the calculated mass action concentration (Ni) and the calculated equilibrium mole number (ni) of TiAl3, and Ti5Al11 in Ti-Al binary melts has been found. At the same time, a spindle-type relationship between the calculated mass action concentation (NTiAl) and the calculated equilibrium mole number (nTiAl) in this binary melt has also been investigated [9].
In consideration of the great practical significance, the aim of this paper is to give a contribution to thermodynamic study of Ti-Al binary melts by coupling with thermodynamic model, i.e., AMCT-Ni model, for calculating mass action concentration of structural units (Ni) based on the atom and molecule coexistence theory (AMCT) in the full composition range of Ti-Al binary melts in the temperature range from 1973 to 2273 K, as well as to show the possibility of application of the used model in thermodynamic description of the system, such as the thermodynamic properties of Ti-Al binary melts: the Raoultian activity coefficient (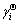 ), the standard molar Gibbs free energy change (
), the standard molar Gibbs free energy change (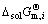 ) of dissolving pure liquid i(l) for forming 1% (mass fraction) of element i in binary metallic melts, and excess molar mixing thermodynamic properties.
) of dissolving pure liquid i(l) for forming 1% (mass fraction) of element i in binary metallic melts, and excess molar mixing thermodynamic properties.
2 Hypotheses
It has been briefly demonstrated in Section 1 that atoms of Ti and Al, and molecules of TiAl3, TiAl, and Ti5Al11 can coexist in Ti-Al binary melts. The hypotheses of the developed AMCT-Ni model for Ti-Al binary melts can be summarized as follows [19]: 1) the Ti-Al binary melts at elevated temperature are composed of five structural units, including two atoms of Ti and Al and three molecules of TiAl3, TiAl, and Ti5Al11 according to the phase diagram of Ti-Al binary system; 2) each structural unit occupies its independent position in Ti-Al binary melts; 3) the elements of Ti and Al in Ti-Al binary melts will take part in reactions of forming three molecules TiAl3, TiAl, and Ti5Al11 in the form of atoms; 4) the reactions of forming molecules of TiAl3, TiAl, and Ti5Al11 are under the chemical dynamic equilibrium between the simple atoms of Ti and Al; 5) three structural units in Ti-Al binary melts as TiAl3, TiAl, and Ti5Al11 bear the structural continuity in the investigated composition range; 6) the chemical reactions of forming three molecules of TiAl3, TiAl, and Ti5Al11 from Ti and Al obey the mass action law.
3 Results and discussion
3.1 Results of AMCT-Ni thermodynamic model for Ti-Al binary melts
The establishment of AMCT-Ni thermodynamic model and determination of mass action concentration of structural units in Ti-Al binary melts at elevated temperatures have been given in detail elsewhere [9]. Figure 1 shows the comparison between model- calculated mass action concentration Ni and determined activities ai of Ti and Al in Ti-Al melts, from which it can be observed that the calculated Ni can be applied to substituting the measured ai relative to pure liquid matter as standard state in Ti-Al binary system. The standard molar Gibbs free energy change 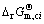 (1973 K��T��2273 K) of reactions for forming TiAl3, TiAl, and Ti5Al11 from Ti and Al relative to pure liquid matter as standard state is listed as follows.
(1973 K��T��2273 K) of reactions for forming TiAl3, TiAl, and Ti5Al11 from Ti and Al relative to pure liquid matter as standard state is listed as follows.
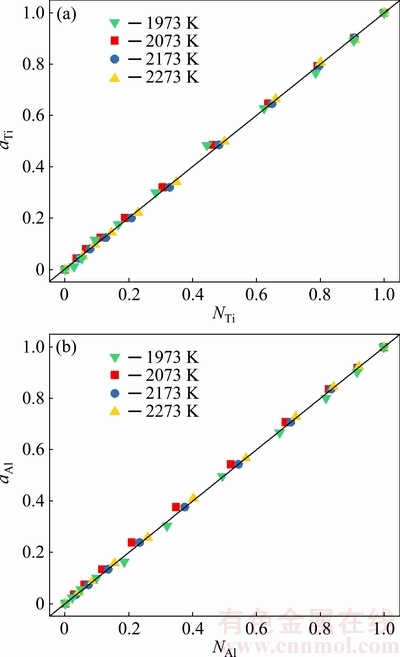
Fig. 1 Relationship between calculated mass action concentration Ni and activity ai in full composition range of Ti-Al binary melts in temperature range from 1973 to 2273 K
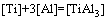
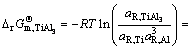
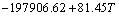 (1)
(1)
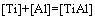
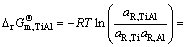
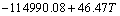 (2)
(2)
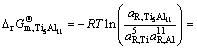
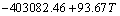 (3)
(3)
where R is the mole gas constant (8.314 J/(mol��K)), T is the temperature (K), and aR,i is the activity relative to pure liquid i as standard state (-).
As a representative, the relationship between mole fraction of Ti (xTi) and calculated mass action concentrations (Ni) of five structural units as Ti, Al, TiAl3, TiAl, and Ti5Al11 in the full composition range of Ti-Al binary melts in the temperature range from 1973 to 2273 K is shown in Fig. 2.
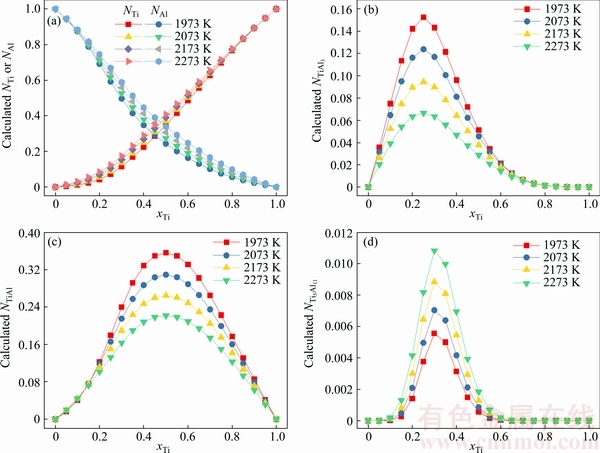
Fig. 2 Relationship between mole fraction of Ti (xTi) and calculated mass action concentrations (Ni) of structural units as Ti, Al, TiAl3, TiAl, and Ti5Al11 in full composition range of Ti-Al binary melts in temperature range from 1973 to 2273 K, respectively
Figure 2(a) shows the variation of the calculated mass action concentrations of Ti (NTi) and Al (NAl) as a function of mole fraction of Ti (xTi) in the temperature range from 1973 to 2273 K. As can be observed in Fig. 2(a), at a given temperature, the calculated mass action concentration of Ti (NTi) shows a sluggish increase tendency with an increase of mole fraction of Ti (xTi) from 0 to 0.2, and then displays a drastic increase trend with an increase of mole fraction of Ti (xTi) from 0.2 to 1.0 at the above mentioned four temperatures. However, an opposite variation relationship of the calculated NAl against xTi can be found at 1973, 2073, 2173, and 2273 K. Meanwhile, for a fixed concentration, as temperature increases, the activity of both components increases, which means that increasing temperature can promote the reaction ability.
The reverse V-type relationships between mole fraction xTi and calculated mass action concentrations of TiAl3, TiAl, and Ti5Al11 in the full composition range of Ti-Al binary melts at 1973, 2073, 2173, and 2273 K are shown in Figs. 2(b)-(d), respectively. As shown in Figs. 2(b) and (c), increasing temperature from 1973 to 2273 K can result in a decrease of the maximum value of calculated mass action concentration NTiAl3 from 0.1525 to 0.0664 and NTiAl from 0.3560 to 0.2215, respectively. However, the maximum value of calculated mass action concentration NTi5Al11 increases from 0.0056 to 0.0108 with an increase in the temperature range. It should be specially emphasized that the sum of the calculated mass action concentration Ni of five structural units as Ti, Al, TiAl3, TiAl, and Ti5Al11 in Ti-Al binary melts should be unity. Therefore, it can be deduced that the calculated mass action concentration Ni of five structural units in Ti-Al binary melts should be competitive or coupled each other [28].
3.2 Determination of standard molar Gibbs free energy change 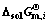 of dissolving pure liquid i for forming 1% element i in Ti-Al binary melts
of dissolving pure liquid i for forming 1% element i in Ti-Al binary melts
According to the definition of Ni, the physical meaning of the Ni is equilibrium mole fraction of structural unit i in a closed system. The basic meaning of Ni is almost consistent with the traditionally applied activity ai of component i in metallic melts, in which pure liquid matter is chosen as the standard state and mole faction is selected as a concentration unit [19]. Meanwhile, it has been pointed out by the present authors [9] that the calculated mass action concentration of Ti (NTi) or that of Al (NAl) had been validated to substitute the reported activity of Ti (aR,Ti) or that of Al (aR,Al) in Ti-Al binary melts in full composition range in a temperature range from 1973 to 2273 K.
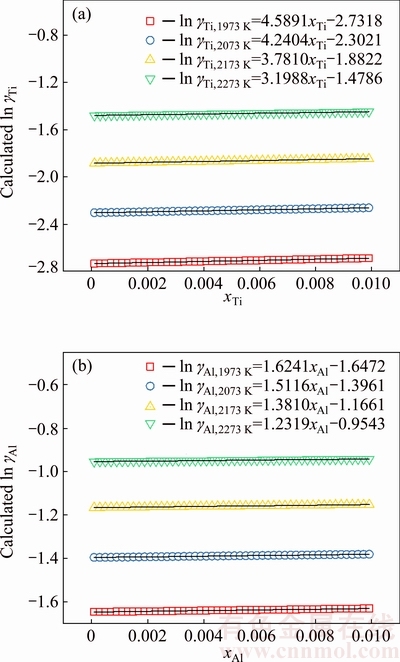
Fig. 3 Relationship between mole fraction of Ti (xTi) and calculated activity coefficient of Ti in logarithmic form (ln ��Ti) (a), and relationship between mole fraction of Al (xAl) and calculated activity coefficient of Al in logarithmic form (ln ��Al) (b) in temperature range from 1973 to 2273 K
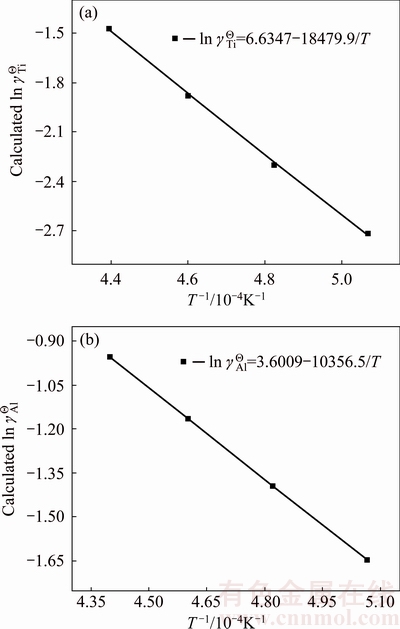
Fig. 4 Relationship between reciprocal of temperature and calculated activity coefficient of Ti in logarithmic form (ln ��Ti) (a), and calculated activity coefficient of Al in logarithmic form (ln ��Al) (b) in full composition range of Ti-Al binary melts in temperature range from 1973 to 2273 K
The activity coefficient of Ti (��Ti) or that of Al (��Al) in Ti-Al binary melts with changing mole fraction of Ti (xTi) or that of Al (xAl) from 0 to 0.01 at an interval of xi as 0.0001 at temperatures of 1973, 2073, 2173, and 2273 K can be determined as ��i=Ni/xi based on the developed AMCT-Ni model for Ti-Al binary melts [25,26,28]. The relationship between mole fraction of Ti (xTi) and the calculated activity coefficient of Ti in natural logarithmic form (ln ��Ti) in Ti-Al binary melts is illustrated in Fig. 3(a). The corresponding relationship at above mentioned four different temperatures can be expressed by the linear equations as follows:
ln ��Ti, 1973 K=4.5891xTi-2.7138 (0
ln ��Ti, 2073 K=4.2404xTi-2.3021 (0
ln ��Ti, 2173 K=3.7810xTi-1.8822 (0
ln ��Ti, 2273 K=3.1988xTi-1.4786 (0
The relationship between mole fraction of Al (xAl) and the calculated activity coefficient of Al in natural logarithmic form (ln ��Al) in Ti-Al binary melts is illustrated in Fig. 3(b). The corresponding relationship at above mentioned four different temperatures can be expressed by the linear equations as follows:
ln ��Al,1973 K=1.6241xAl-1.6472 (0
ln ��Al,2073 K=1.5116xAl-1.3961 (0
ln ��Al,2173 K=1.3810xAl-1.1661 (0
ln ��Al,2273 K=1.2319xAl-0.9543 (0
In view of Wagner equation [29], activity coefficient of component i (��i) in binary metallic melts referring to pure matter as standard state can be expressed as follows:
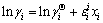 (12)
(12)
where 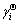 is the activity coefficient of component i in the infinitely dilute solution (-),
is the activity coefficient of component i in the infinitely dilute solution (-), 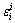 is the first-order activity interaction coefficient of element i (-), and xi is the mole fraction of element i (-). Therefore, the intercepts of linear functions in Eq. (12) and in Fig. 3(a) can be treated as the values of the Raoultian activity coefficient of Ti (
is the first-order activity interaction coefficient of element i (-), and xi is the mole fraction of element i (-). Therefore, the intercepts of linear functions in Eq. (12) and in Fig. 3(a) can be treated as the values of the Raoultian activity coefficient of Ti (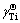 ) in infinitely dilute Ti-Al binary melts at the above mentioned four temperatures. The dependence of the calculated Raoultian activity coefficient of Ti in natural logarithmic form (
) in infinitely dilute Ti-Al binary melts at the above mentioned four temperatures. The dependence of the calculated Raoultian activity coefficient of Ti in natural logarithmic form (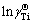 ) in the infinitely dilute Ti-Al binary melts on the temperature range of interest is illustrated in Fig. 4(a) and regressed as
) in the infinitely dilute Ti-Al binary melts on the temperature range of interest is illustrated in Fig. 4(a) and regressed as
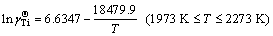 (13)
(13)
Similarly, the temperature dependence of the calculated Raoultian activity coefficient of Al in natural logarithmic form (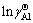 ) in the infinitely dilute Ti-Al binary melts is shown in Fig. 4(b) and regressed as
) in the infinitely dilute Ti-Al binary melts is shown in Fig. 4(b) and regressed as
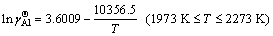 (14)
(14)
The standard molar Gibbs free energy change 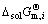 of dissolving liquid Ti or Al can be acquired by inserting the relationship between a%,i and aR,i [29]:
of dissolving liquid Ti or Al can be acquired by inserting the relationship between a%,i and aR,i [29]:
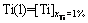
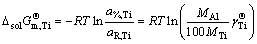 (15)
(15)
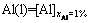
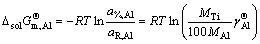 (16)
(16)
where a%,i or aR,i is activity relative to 1% (mass fraction) of element i as standard state or pure liquid i as standard state (-), and Mi is relative molecular mass (kg/mol). Accordingly, the influence of changing temperature from 1973 to 2273 K on the determined standard molar dissolve Gibbs free energy 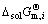 can be obtained by inserting the determined Raoultian activity coefficient
can be obtained by inserting the determined Raoultian activity coefficient 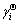 in Eqs. (13) and (14) into Eqs. (15) and (16) as
in Eqs. (13) and (14) into Eqs. (15) and (16) as
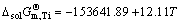
(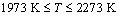 ) (17)
) (17)
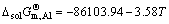
(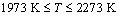 ) (18)
) (18)
3.3 Determination of excess molar mixing thermo- dynamic properties
The excess molar mixing Gibbs free energy change of Ti-Al binary melts can be defined as [29]
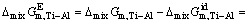

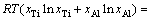
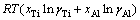 (19)
(19)
where 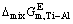 is the excess molar Gibbs free energy change of Ti-Al binary melts as a real solution comparing with ideal solution as a basis (J/mol),
is the excess molar Gibbs free energy change of Ti-Al binary melts as a real solution comparing with ideal solution as a basis (J/mol), 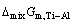 is the molar mixing Gibbs free energy change of Ti-Al binary melts as a real solution (J/mol),
is the molar mixing Gibbs free energy change of Ti-Al binary melts as a real solution (J/mol), 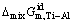 is the molar mixing Gibbs free energy change of Ti-Al binary melts as an ideal solution (J/mol). The excess molar mixing entropy change
is the molar mixing Gibbs free energy change of Ti-Al binary melts as an ideal solution (J/mol). The excess molar mixing entropy change 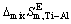 of Ti-Al binary melts can be derived from Eq. (19) as
of Ti-Al binary melts can be derived from Eq. (19) as
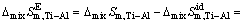
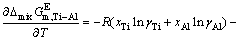
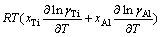 (20)
(20)
The excess molar mixing enthalpy change 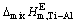 of Ti-Al binary melts can be derived from Eq. (19) and Eq. (20) as
of Ti-Al binary melts can be derived from Eq. (19) and Eq. (20) as
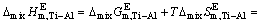
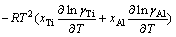 (21)
(21)
The quantity of  ln ��i/
ln ��i/ T shown in Eqs. (20) and (21) can be calculated by means of the method reported by YANG et al [28] in the study of Fe-Si binary system.
T shown in Eqs. (20) and (21) can be calculated by means of the method reported by YANG et al [28] in the study of Fe-Si binary system.
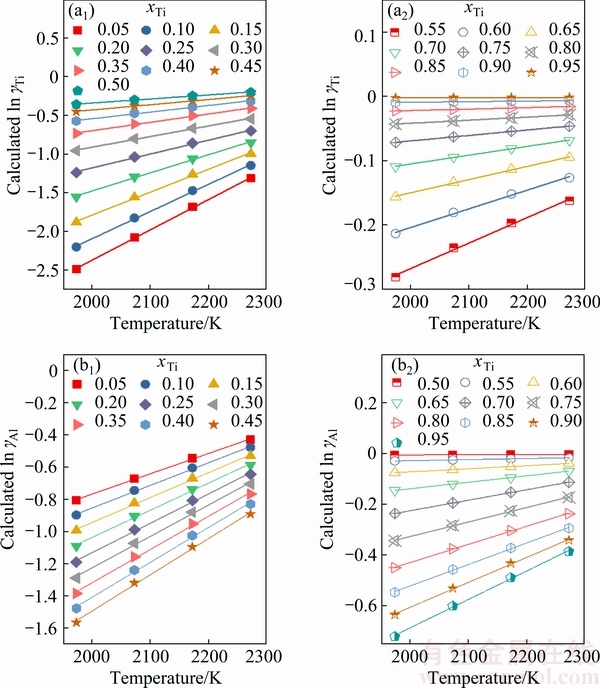
Fig. 5 Calculated activity coefficient of Ti in logarithmic form (ln ��Ti) (a1, a2) or that of Al in logarithmic form (ln ��Al) (b1, b2) in full composition range of Ti-Al binary melts as function of temperature under fixed mole fraction of Ti (xTi) from 0.05 to 0.95 at interval of 0.05
Figure 5 represents the calculated activity coefficient of Ti in natural logarithmic form (ln ��Ti) or that of Al (ln ��Al) in the full composition range of Ti-Al binary melts as a function of temperature under the fixed mole fraction of Ti (xTi) from 0.05 to 0.95 at an interval of 0.05, respectively. By plotting  ln ��i/
ln ��i/ T versus T at different mole fractions xTi, a straight line should be obtained. From the slope of straight line, the values of
T versus T at different mole fractions xTi, a straight line should be obtained. From the slope of straight line, the values of  ln ��Ti/
ln ��Ti/ T or
T or  ln ��Al/
ln ��Al/ T at different mole fractions xTi can be determined. The relationship between the calculated
T at different mole fractions xTi can be determined. The relationship between the calculated  ln ��i/
ln ��i/ T and mole fraction of Ti (xTi) in full composition range in a temperature range from 1973 to 2273 K is illustrated in Fig. 6. The regressed formula of
T and mole fraction of Ti (xTi) in full composition range in a temperature range from 1973 to 2273 K is illustrated in Fig. 6. The regressed formula of  ln ��Ti/
ln ��Ti/ T or
T or  ln ��Al/
ln ��Al/ T against mole fraction of Ti (xTi) can be expressed as
T against mole fraction of Ti (xTi) can be expressed as
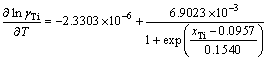 (22)
(22)
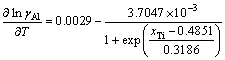 (23)
(23)
To describe the excess thermodynamic properties of Ti-Al binary melts, the excess molar mixing Gibbs free energy 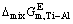 , the excess molar mixing entropy
, the excess molar mixing entropy 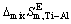 , and the excess molar mixing enthalpy
, and the excess molar mixing enthalpy 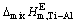 were estimated by inserting the obtained values of
were estimated by inserting the obtained values of 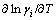 into Eqs. (19)-(21), respectively.
into Eqs. (19)-(21), respectively.
The relationships between the excess thermodynamic properties and mole fraction of Ti (xTi) at temperatures of 1973, 2073, 2173 and 2273 K are well presented in Fig. 7, from which a V-type relationship between the determined 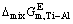 ,
, 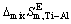 or
or 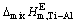 of Ti-Al binary melts and xTi can be seen. Meanwhile, it can be observed that changing temperature from 1973 to 2273 K can give rise to a visible decrease tendency of the determined
of Ti-Al binary melts and xTi can be seen. Meanwhile, it can be observed that changing temperature from 1973 to 2273 K can give rise to a visible decrease tendency of the determined 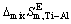 and
and 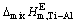 , while the increasing temperature has a little effect on
, while the increasing temperature has a little effect on 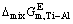 at a fixed mole fraction of Ti.
at a fixed mole fraction of Ti.

Fig. 6 Calculated  ln ��Ti/
ln ��Ti/ T or
T or  ln ��Al/
ln ��Al/ T in Ti-Al binary melts as function of mole fraction of Ti (xTi) from 0.05 to 0.95 at interval of 0.05 in temperature range from 1973 to 2273 K
T in Ti-Al binary melts as function of mole fraction of Ti (xTi) from 0.05 to 0.95 at interval of 0.05 in temperature range from 1973 to 2273 K
Fig. 7 Relationship between mole fraction of Ti (xTi) and calculated excess molar mixing Gibbs free energy change 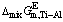 (a), excess molar mixing entropy change
(a), excess molar mixing entropy change 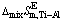 (b), and excess molar mixing enthalpy change
(b), and excess molar mixing enthalpy change 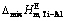 (c) in temperature range from 1973 to 2273 K
(c) in temperature range from 1973 to 2273 K
4 Conclusions
1) A thermodynamic prediction model for determining the thermodynamic properties of Ti-Al binary melts based on the atom and molecule coexistence theory (AMCT) was developed in a temperature range from 1973 to 2273 K.
2) The determined 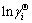 by AMCT model has a good linear relationship with temperature, and the values of the Raoultain activity coefficient of Ti (
by AMCT model has a good linear relationship with temperature, and the values of the Raoultain activity coefficient of Ti (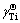 ) and that of Al (
) and that of Al (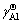 ) in the infinitely dilute solution of Ti-Al binary melts in the temperature range from 1973 to 2273 K can be expressed as
) in the infinitely dilute solution of Ti-Al binary melts in the temperature range from 1973 to 2273 K can be expressed as 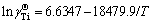 and
and 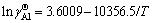 .
.
3) The standard molar Gibbs free energy change 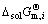 of dissolving pure liquid i(l) for forming 1% element i in Ti-Al binary melts in a temperature range from 1973 to 2273 K can be presented as
of dissolving pure liquid i(l) for forming 1% element i in Ti-Al binary melts in a temperature range from 1973 to 2273 K can be presented as 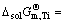 -153641.89+12.11T and
-153641.89+12.11T and 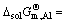 -86103.94-3.58T.
-86103.94-3.58T.
4) The plot of 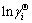 versus T at fixed mole fraction of Ti xTi from 0.05 to 0.95 at an interval 0.05 should give a straight line, and the
versus T at fixed mole fraction of Ti xTi from 0.05 to 0.95 at an interval 0.05 should give a straight line, and the 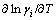 values of Ti or Al in Ti-Al binary melts can be obtained from its slope. The regressed formula of
values of Ti or Al in Ti-Al binary melts can be obtained from its slope. The regressed formula of 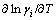 of Ti or Al against mole fraction of Ti (xTi) can be further obtained.
of Ti or Al against mole fraction of Ti (xTi) can be further obtained.
5) The excess thermodynamic properties, such as 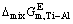 ,
, 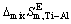 , and
, and 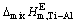 can be accurately predicted by developed AMCT model. Changing temperature from 1973 to 2273 K can give rise to a visible decrease tendency of the determined
can be accurately predicted by developed AMCT model. Changing temperature from 1973 to 2273 K can give rise to a visible decrease tendency of the determined 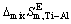 and
and 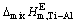 , while the increase of temperature has a little effect on
, while the increase of temperature has a little effect on 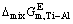 at a fixed mole fraction of Ti (xTi).
at a fixed mole fraction of Ti (xTi).
References
[1] HOCH M, USELL R J. Thermodynamics of titanium alloys: II. Titanium and aluminum activities in the BCC �� phase of the Ti-Al system [J]. Metallurgical Transactions, 1971, 2(9): 2627-2632.
[2] SAMOKHVAL V V, POLESHCHUK P A, VECHER A A. Thermodynamic property of aluminum-titanium and aluminum- vanadium alloys [J]. Russian Journal of Physical Chemistry, 1971, 45(8): 1174-1176.
[3] ECKERT M, BENCZE L, KATH D, NICKEL H, HILPERT K. Thermodynamic activities in the alloys of the Ti-A1 system [J]. Berichte der Bunsengesellschaft f��r Physikalische Chemie, 1996, 100(4): 418-424.
[4] MAEDA M, KIWAKE T, SHIBUYA K, IKEDA T. Activity of aluminum in molten Ti-Al alloys [J]. Materials Science and Engineering A, 1997, 239-240: 276-280.
[5] JACOBSON N S, BRADY M P, MEHROTRA G M. Thermodynamics of selected Ti-Al and Ti-Al-Cr alloys [J]. Oxidation of Metals, 1999, 52(5-6): 537-556.
[6] GUO Jin-jie, JIA Jun, LIU Yuan, LIU Gui-zhong, SU Yan-qing, DING Hong-sheng. Evaporation behavior of aluminum during the cold crucible induction skull melting of titanium aluminum alloys [J]. Metallurgical and Materials Transactions B, 2000, 31(4): 837-844.
[7] REDDY R G, YAHYA A M, BREWER L. Thermodynamic properties of Ti-Al intermetallics [J]. Journal of Alloys and Compounds, 2001, 321(2): 223-227.
[8] KOSTOV A, FRIEDRICH B, ZIVKOVIC D. Predicting thermodynamic properties in Ti-Al binary system by FactSage [J]. Computational Materials Science, 2006, 37(3): 355-360.
[9] DUAN Sheng-chao, CHEN Hun-bin, GUO Han-jie, LIAN Yun-fei. Representation of reaction abilities for Al-Ti binary melts based on the atom-molecule coexistence theory [J]. Chinese Journal of Engineering, 2016, 38(10): 1377-1385. (in Chinese)
[10] CUI Ren-jie, TANG Xiao-xia, GAO Ming, ZHANG Hu, GONG Sheng-kai. Thermodynamic analysis of interactions between Ti-Al alloys and oxide ceramics [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(4): 887-894.
[11] NOVAKOVIC R, GIURANNO D, RICCI E, TUISSI A, WUNDERLICH R, FECHT H, EGRY I. Surface, dynamic and structural properties of liquid Al-Ti alloys [J]. Applied Surface Science, 2012, 258(7): 3269-3275.
[12] XU Shuai, SONG Bin-yi, JIANG Wen-long, YANG Bin. Modeling and calculation of activities of constituent elements in binary alloys [J]. Chinese Journal of Vacuum Science and Technology, 2015(8): 919-925. (in Chinese)
[13] HILDEBRAND J H. A quantitative treatment of deviations from Raoult��s law [J]. Proceedings of the National Academy of Sciences of the United States of America, 1927, 13(5): 267-272.
[14] MIEDEMA A R, de CHATEL P F, de BOER F R. Cohesion in alloys-fundamentals of a semi-empirical model [J]. Physica B+C, 1980, 100(1): 1-28.
[15] TAO Dong-ping. A new model of thermodynamics of liquid mixtures and its application to liquid alloys [J]. Thermochimica Acta, 2000, 363(1-2): 105-113.
[16] RENON H, PRAUSNITZ J M. Local compositions in thermodynamic excess functions for liquid mixtures [J]. AIChE Journal, 1968, 14(1): 135-144.
[17] WILSON G M. Vapor-liquid equilibrium. XI: A new expression for the excess free energy of mixing [J]. Journal of the American Chemical Society, 1964, 86(2): 127-130.
[18] YANG Xue-min, ZHANG Meng, LI Peng-cheng, LI Jin-yan, ZHANG Jian. A thermodynamic model for representation reaction abilities of structural units in full composition range of Fe-Si binary melts based on the atom�Cmolecule coexistence theory [J]. Steel Research International, 2013, 84(8): 784-811.
[19] ZHANG Jian. Computational thermodynamics of metallurgical melts and solutions [M]. Beijing: Metallurgical Industry Press, 2007. (in Chinese)
[20] ZHANG Jian. Classification of thermodynamic properties of binary metallic melts according to their phase diagrams [J]. Acta Metallurgica Sinica, 1998, 34(1): 75-85. (in Chinese)
[21] ZHANG Jian. Thermodynamic properties and mixing thermodynamic parameters of Ba-Al, Mg-Al, Sr-Al and Cu-Al metallic melts [J]. Transactions of Nonferrous Metals Society of China, 2004, 14(2): 345-350.
[22] ZHANG Jian. Calculating model of mass action concentrations for Fe-Si melts [J]. Journal of Iron and Steel Research, 1991, 3(2): 7-12. (in Chinese)
[23] ZHANG Jian. On the coexistence theory of metallic structure involving compound formation [J]. Journal of University of Science and Technology Beijing, 1990, 12(3): 201-211. (in Chinese)
[24] ZHANG Jian, WANG Ping. The widespread applicability of the mass action law to metallurgical melts and organic solutions [J]. Calphad, 2001, 25(3): 343-354.
[25] YANG Xue-min, LI Peng-cheng, LI Jin-yan, ZHANG Meng, ZHANG Jian-liang, ZHANG Jian. Representation reaction abilities of structural units and related thermodynamic properties in Fe-P binary melts based on the atom�Cmolecule coexistence theory [J]. Steel Research International, 2014, 85(3): 426-460.
[26] YANG Xue-min, LI Jin-yan, WEI Meng-fang, ZHANG Jian. Thermodynamic evaluation of reaction abilities of structural units in Fe-O binary melts based on the atom�Cmolecule coexistence theory [J]. Metallurgical and Materials Transactions B, 2016, 47(1): 174-206.
[27] YANG Xue-min, ZHANG Meng, LI Peng-cheng, LI Jin-yan, ZHANG Jian-liang, ZHANG Jian. A thermodynamic model for representation reaction abilities of structural units in Fe-S binary melts based on the atom-molecule coexistence theory [J]. Metallurgical and Materials Transactions B, 2012, 43(6): 1358-1387.
[28] YANG Xue-min, LI Jin-yan, LI Peng-cheng, ZHANG Meng, ZHANG Jian. Determination of activity coefficients of elements and related thermodynamic properties of Fe-Si binary melts based on the atom�Cmolecule coexistence theory [J]. Steel Research International, 2014, 85(2): 164-206.
[29] GUO Han-jie. Metallurgical physical chemistry [M]. 2nd ed. Beijing: Metallurgical Industry Press, 2007. (in Chinese).
������1,2��ʯ ��1,2��������1,2��������1,2���� ��1,2
1. �����Ƽ���ѧ ұ������̬����ѧԺ������ 100083��
2. �����Ƽ���ѧ �߶˽������������������Ʊ��������ص�ʵ���ң����� 100083
ժ Ҫ������ԭ��-���ӹ������۽��������¶�Ϊ1973~2273 K��ȫŨ�ȷ�ΧTi-Al��Ԫ������������Ũ��Ni������ѧģ�͡����ò�ͬ�¶�(1973��2073��2173��2273 K)�µ�ģ�ͼ��������õ�������ϡ��Һ(0
�ؼ��ʣ�Ti-Al��Ԫϵ��Ti��Al��������ӣ�����ѧģ�ͣ���������Ũ�ȣ�ԭ��-���ӹ�������
(Edited by Wei-ping CHEN)
Foundation item: Project (U1560203) supported by the Joint Funds of the National Natural Science Foundation of China; Project (51274031) supported by the National Natural Science Foundation of China
Corresponding author: Han-jie GUO; Tel/Fax: +86-10-62334964; E-mail: guohanjie@ustb.edu.cn
DOI: 10.1016/S1003-6326(18)64764-8

