J. Cent. South Univ. Technol. (2008) 15(s1): 080-083
DOI: 10.1007/s11771-008-319-x
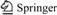
Effect of salt solutions on chain structure of partially hydrolyzed polyacrylamide
ZHANG Qing(张 青), ZHOU Ji-sheng(周吉生), ZHAI Yong-ai(翟永爱),
LIU Feng-qi(刘凤岐), GAO Ge(高 歌)
(College of Chemistry and MacDiarmid Laboratory, Jilin University, Changchun 130021, China)
Abstract: The effect of salt solutions (NaCl, Na2SO4 and CaCl2) on the conformational properties of partially hydrolyzed polyacrylamide (HPAM) was investigated by using static laser light scattering (SLLS). The special interaction between CaCl2 solution and HPAM was also researched. Experimental results show that the chain structure of HPAM is interrelated with the charge density, the kind and the concentration of salt solutions. The mean-square radius of gyration (Rz) and the second virial coefficient (A2) of HPAM decrease with increasing concentration of salt solutions, and the salt effect tends towards the maximum when the concentration of salt solution is increased to some amount.
Key words: partially hydrolyzed polyacrylamide (HPAM); salt effect; chain structure; static laser light scattering (SLLS)
1 Introduction
Over the past three decades, partially hydrolyzed polyacrylamide (HPAM) and their derivatives have attracted a great deal of attention due to their potential applications in different fields, such as enhanced oil recovery[1-2], wastewater treatment[3], and paper manufacturing[4]. HPAM was widely used in Daqing oilfield, and played a very important role in tertiary oil recovery system because it could be used as a viscosity modifier[5-11] to increase sweep efficiency of water flood and chemical flood efficiency. However, there exist different demands on the molecular conformation and relative molecular mass of HPAM owing to the diversities of geology conditions and the differences of content and kind of ions in the underground water. So how to evaluate and determine the molecular conformation and chain structure of HPAM in different micro-circumstances is very important for explaining the mechanism of polymer flood and making good use of the current resource.
HPAM is an anionic polymer with a large number of charges along the polymer chains. Owing to the electrostatic repulsion among anions, the molecule chains of HPAM in solution tend to unfolding. However, the addition of salt to the solution may cause the conformational changes of HPAM.
Static laser light scattering (SLLS) is considered one of effective measures for characterizing polymers and can provide more accurate information compared with the classic viscosimetry with the extremely dilute concentration. Some reports have appeared concerning the effect of salt on polyelectrolyte[12-13]. To our knowledge, effect of salt solutions under different conditions on the conformation of HPAM and the studies on its sensitive range have not been reported.
The main aim of this paper is to establish an absolute method for determining molecular mass of HPAM by SLLS, and to research the effect of salt solutions on the molecule chain structure of HPAM.
2 Experimental
2.1 Materials
HPAM with 25% of degree of hydrolysis was purchased from Daqing Chemical Agent Plant. NaCl, Na2SO4 and CaCl2 aqueous solutions were prepared with deionized water. All solutions were filtered by a 0.20 μm pore-size filter and then kept in sealed glass bottles before use. The resistivity of the deionized water used in this study was 18.3 MΩ?cm.
2.2 SLLS test
Laser light scattering measurements were performed on a Wyatt Dawn DSP laser photometer. The refractive index increments (dn/dc) of samples were determined by an Optilab DSP interferometric refractometer (Wyatt Technology Corporation) under the wavelength of 690 nm at 25 ℃. Astra software was utilized for the data acquisition and analysis. After a series of experiments, the secondary structure data of HPAM under different concentrations of NaCl, Na2SO4 and CaCl2 aqueous solutions were obtained.
3 Results and discussion
HPAM molecule chains contained amide groups and carboxylic acid groups. Carboxylic acid groups ionized in aqueous solution and the electrostatic repulsion among the anions caused the molecule chains to expand. As for HPAM, the reaction mechanisms between different metal ions and carboxylic acid groups of molecule chains were also different. Monovalent cations could effectively shield electrolyte effect, which resulted in shrinking of molecule chain, thus increasing the flexibility and decreasing hydrodynamic radius. However, the reaction mechanism between multivalent cations (Ca2+, Al3+) and carboxylic acid groups is more complex, because the multivalent cations can act as the cross-linking agents to interconnect the HPAM chains and further to influence the conformation and rheological behaviors of HPAM[14-16].
Fig.1 shows a typical Zimm plot of HPAM in 0.5 mol/L of Na2SO4 aqueous solution at 25 ℃, where the concentration of HPAM ranged from 2.3×10-5 to 8.9× 10-5 g/mL. The values of Mw, Rz and A2 were 4.898×106, 137.4 nm and 3.82×10-4 mol・mL/g2, respectively.
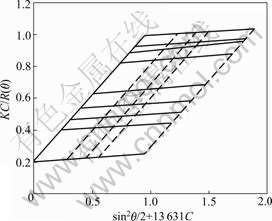
Fig.1 Zimm plot of HPAM in 0.5 mol/L of Na2SO4 aqueous solution at 25 ℃
The addition of Na+ could effectively neutralize the negative charge, which brought about the shrinkage of molecule chains and the decrease of hydrodynamic radius. From Fig.2 and Fig.3 we could see that Rz and A2 of HPAM decrease with the increase of Na+ concentration. Because of the increase of ionic strength by adding NaCl aqueous solution, the double electrical layers on HPAM molecular chains were compressed, and electrostatic repulsion among the anions was shielded. In addition, as shown in the figures, Rz and A2 of HPAM reduced suddenly as the Na+ concentration was increased to 0.45 mol/L. The reason was that Na+ ions first gathered around the macromolecule to balance the charges and form an ion enrichment region. Afterwards, Na+ diffused into other parts of aqueous solution when the charge was balanced. Therefore, Rz of HPAM decreased by 25% when the concentration of Na+ increased from 0.02 to 0.2 mol/L, and however, it reduced only by 30% when the concentration increased from 0.02 to 1.00 mol/L. Comparing the effects of Na2SO4 and NaCl aqueous solution on the chain structure of HPAM, we found that the salt effects had nothing to do with the type of anions as for strong electrolyte solutions.
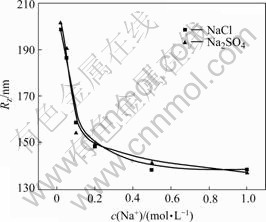
Fig.2 Na+ concentration dependence of Rz of HPAM
Fig.3 Na+ concentration dependence on A2 of HPAM
Interaction of Ca2+ with HPAM was more complex, and usually, intramolecular and intermolecular crosslinkings could be formed. Fig.4 and Fig.5 show Ca2+ concentration dependence of Rz and A2 of HPAM respectively. Rz of HPAM decreased with the increase of concentration of CaCl2 solution, which was similar to the influence of NaCl solution, but the shrinking effect of chain dimension was more obvious. This is because the complex reaction occurred between Ca2+ and carboxyl groups of HPAM, which not only shielded the electrostatic repulsion among anions but also effectively reduced charge densities around the macromolecule chain. As shown in Fig.4, Rz of HPAM had a slight increase after the concentration of Ca2+ arrived at 0.08 mol/L. The reason was that multi-molecule crosslinking happened under the action of Ca2+ which caused the increase of molecular dimension.
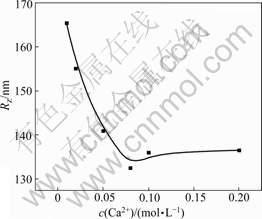
Fig.4 Ca2+ concentration dependence of Rz of HPAM
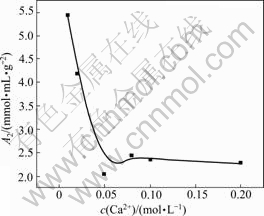
Fig.5 Ca2+ concentration dependence of A2 of HPAM
The variation of Mw as a function of Ca2+ concentration is presented in Fig.6. Mw of HPAM increased persistently with the increase of Ca2+ concentration, which confirmed that the complex reaction occurred between Ca2+ and carboxyl groups of HPAM. In the range of low concentration, the Mw of HPAM was approaching to the molecular weight of single molecule (Fig.1) and the complex reaction mainly occurred in intramoleculary. When the concentration of Ca2+ increased to 0.02 mol/L, the Mw was about twice the molecular mass of single molecule, because at this point crosslinking reaction began to take place in intermoleculary.
In addition, combining the above results we found
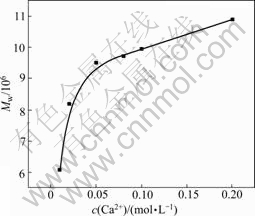
Fig.6 Ca2+ concentration dependence of apparent molecular mass (Mw) of HPAM
that in the certain concentration range of CaCl2 solution, the molecule of HPAM still shrank with increasing concentration of CaCl2 solution. Therefore, the effect of Ca2+ on the molecular dimension of HPAM was mainly resulted from the action of crosslinking.
4 Conclusions
Based on our experiment results, the conclusions were given as follows. The shielding effect of monovalent inorganic salts on HPAM caused the shrinkage of molecule chain, and this effect tended to an extreme with increasing concentration of the salt. As for strong electrolyte solutions, the salt effects had nothing to do with the type of anions. Rz and A2 of HPAM had remarkable changes in a very narrow concentration range of Na+, which indicated that volume phase transition would occur via altering the concentration of monovalent cations. The shielding effect and crosslinking action of Ca2+ on HPAM occurred simultaneously, however, the crosslinking action played a more important role in the influence of molecular dimension.
References
[1] Wang Yi-yang, DAI Yu-hua, Zhang Lu, LUO Lan, CHU Yan-ping, ZHAO Sui, LI Miao-zhen, WANG Er-jian, YU Jia-yong. Hydrophobically modified associating polyacrylamide solutions: Relaxation processes and dilational properties at the oil-water interface [J]. Macromolecules, 2004, 37(8): 2930-2937.
[2] BOCK J, Schulz D N, McCormick C L. Water soluble polymers: Encyclopedia of polymer science and engineering [M]. New York: Wiley and Sons, 1986: 730-784.
[3] WEI Shi-tang, MA De-rong, WANG Run-sheng. Study on the treatment of radioactive waste water using partially hydrolyzed polyacrylamide as a flocculant [J]. Shanghai Environmental Sciences, 1982, 1(1): 37-42. (in Chinese)
[4] ALLEN L H, PELTON R H. The effects of some electrolytes on flocculation with a cationic polyacrylamide [J]. Colloid Polym Sci, 1983, 261(6): 485-492.
[5] HOU Ji-rui, LIU Zhong-chun, ZHANG Shu-fen, YUE Xiang-an, YANG Jin-zong. The role of viscoelasticity of alkali/surfactant/ polymer solutions in enhanced oil recovery [J]. J Petrol Sci Eng, 2005, 47(4): 219-235.
[6] FLEW S, SELLIN R H J. Non-Newtonian flow in porous media―A laboratory study of polyacrylamide solutions [J]. J Non-Newton Fluid Mech, 1993, 47: 169.
[7] Zolfaghari R, Katbab A A, Nabavizadeh J, Tabasi R Y, Nejad M H. Preparation and characterization of nanocomposite hydrogels based on polyacrylamide for enhanced oil recovery applications [J]. J Appl Polym Sci, 2006, 100(3): 2096-2103.
[8] ZHANG Lu-hong, ZHANG Dan, JIANG Bin. The rheological behavior of salt tolerant polyacrylamide solutions [J]. Chem Eng Technol, 2006, 29(3): 395-400.
[9] Shaikh S, AsrofAli S, Hamad E Z. Synthesis and solution properties of poly(acrylamide-styrene) block copolymers with high hydrophobic content [J]. Polym Eng Sci, 1999, 39(10): 1962-1968.
[10] Guerrero S J, Boldarino P, Zurimendi J A. Characterization of polyacrylamides used in enhanced oil recovery [J]. J Appl Polym Sci, 1985, 30(3): 955-967.
[11] Khune G D, Donaruma L G, Hatch M J, Kilmer N H, Shepitka J S, Martin F D. Modified acrylamide polymers for enhanced oil recovery [J]. J Appl Polym Sci, 1985, 30(2): 875-885.
[12] Dautzenberg H, Karibyants N. Polyelectrolyte complex formation in highly aggregating systems. Effect of salt: Response to subsequent addition of NaCl [J]. Macromol Chem Phys, 1999, 200(1): 118-125.
[13] GUO Liang, TOM K C, jankins R D. Effects of salt on the intrinsic viscosity of model alkali-soluble associative polymers [J]. Macromol Chem Phys, 1998, 199(6): 1175-1184.
[14] PENG Xiao-hong, SHEN Jia-rui. Studies on salting effects of DMC-AM-AA terpolymers in aqueous solution [J]. Acta Polym Sinica, 1999, 6: 736-740. (in Chinese)
[15] YE Mei-ling, HAN Dong, SHI Liang-he. Studies on determination of molecular weight for ultrahigh molecular weight partially hydrolyzed polyacrylamide [J]. J Appl Poly Sci, 1996, 60(3): 317-322.
[16] PENG Shu-fu, WU Chi. Light scattering study of the formation and structure of partially hydrolyzed poly(acrylamide)/calcium(II) complexes [J]. Macromolecules, 1999, 32(3): 585-589.
(Edited by CHEN Wei-ping)
Foundation item: Project(50673033) supported by the National Natural Science Foundation of China
Received date: 2008-06-25; Accepted date: 2008-08-05
Corresponding author: GAO Ge, Professor; Tel: +86-431-82214252; E-mail: gaoge@jlu.edu.cn