
Recovery of valuable elements from spent YBa2Cu3O7-x/Ag composite superconductor bulks
Li Feng-hua(李凤华), Li Ying-nan(李英楠), Zhang Cong-cong(张聪聪), Fan Zhan-guo(樊占国)
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: Silver as a highly conductive metal is usually doped in YBa2Cu3O7-x superconductor bulks to improve critical current density of YBa2Cu3O7-x superconductor. The valuable metal elements silver, yttrium, barium and copper in waste YBa2Cu3O7-x/Ag composite superconductor bulks were recovered, respectively. Silver was recovered with process at first, the waste was dissolved by nitric acid and silver chloride was precipitated by adding chloride acid, then silver pig was obtained by melting silver chloride together with sodium carbonate at 1 000 ℃. The effective factors on recovery ratio and purity of silver were studied. The chemical analysis proves that the purity of silver ingot is 95.86%. The recovery ratio of silver is calculated to be 92.56%. The loss of silver may be due to the loss of silver chloride during filtering and the volatilization of silver when silver chloride and sodium carbonate are smelted at high temperature. For other three metal elements, Y3+, Ba2+ and Cu2+, in the surplus waste liquid after recovering silver, they were separated with the sequence of barium, copper and then yttrium step by step. First, sulfate acid was used to precipitate barium sulfate. Then, sodium sulfide was added to the surplus solution so that copper could be separated as copper sulfide. During this separation procedure, it was important to control the pH value to be 1-2. After that, oxalic acid was added into the surplus solution to obtain yttrium oxalate. Finally, yttrium oxide was formed by burning yttrium oxalate. The XRD results indicate that the final products are all single-phase compounds as BaSO4, CuS and Y2O3, respectively.
Key words: recovery; waste YBa2Cu3O7-x/Ag composite; leaching; smelting; precipitation
_____________________________________________________________________________________________________
1 Introduction
Studies on the superconductor YBa2Cu3O7-x (Y123) were stimulated by the basic concept of a considerable enhancement of critical current density (Jc) by eliminating the weak links in grain boundary due to the introduction of highly conductive metallic inclusions into the ceramic materials. Thus, during the practical preparation of YBa2Cu3O7-x superconductor bulks or tapes, Ag is usually doped into YBa2Cu3O7-x matrix[1-3]. Ag particles with size of 10-50 μm homogeneously scattered in YBa2Cu3O7-x superconductor bulks or films can relieve the internal micro-cracks by acting as non-reactive conducting bridges to connect YBa2Cu3O7-x crystalline grains. Also Ag has fluxing action on YBa2Cu3O7-x which enhances the liquid phase initiation and the decomposition of Y123. It eventually comes out that the melting temperature of Y123 reduces[4], which is favorable for high temperature heat treatment process, especially for the preparation of YBa2Cu3O7-x tapes on silver substrate with the limitation that the melting point of silver is lower than that of Y123.
Waste YBa2Cu3O7-x/Ag composite superconductor bulks are usually abandoned as solid waste without comprehensive utilization. The content of silver in waste YBa2Cu3O7-xAg composite superconductor bulks is 10%-30%, which is considerably high[4-7], compared with other wastes or nature minerals bearing silver to which the process of intensive milling for the pretreatment of minerals has been applied frequently. Thus, the recovery of silver from waste YBa2Cu3O7-x/Ag composite superconductor bulks is of practical importance.
This study describes laboratory treatment to waste YBa2Cu3O7-x/Ag composites. A process consisted of chemical leaching and fire metallurgy is proposed to separate silver from waste superconductor. Some related factors which have effects on recovery ratio and purity of silver during the process are discussed. The elemental compositions in the liquid after leaching silver are mainly Ba(NO3)2, Cu(NO3)2 and Y(NO3)3. If they are recovered, it can not only alleviate environmental pollution of solid waste residue, but also make them useful as secondary resource. Thus, for the filtrate, sulfate acid, sodium sulfide and oxalic acid are added to the solution successively to separate barium, copper and yttrium oxalate.
2 Experimental
Waste bulks were ground into powder and then weighed and dissolved in nitric acid in 80 ℃ water bath for 30 min. The upper solution would appear blue because of the existence of copper nitrate. When x value in YBa2Cu3O7-x was 0.5, the reactions of YBa2Cu3O7-x/Ag composites with nitric acid were as follows:
YBa2Cu3O6.5+HNO3→Y(NO3)3+Ba(NO3)2+Cu(NO3)2+H2O (1)
Ag+HNO3(Dilute)→AgNO3+NO(g)+H2O (2)
Ag+HNO3(Hot and dense)→AgNO3+NO2(g)+H2O (3)
When hydrochloride was added in the blue solution, white silver chloride sediment appeared:
AgNO3+HCl→AgCl(s)+HNO3 (4)
After filtering, the sediment silver chloride was dried at 105 ℃ for 2 h. The silver chloride could be easily converted to pure (99.99%) silver metal by smelting at 1 100 ℃ with sodium carbonate as flux[8]. Silver chloride and sodium carbonate with the stoichiometric proportion were mixed and made into bulks. Then, they were smelted in a corundum crucible. A layer of excess sodium carbonate was previously put on the bottom of the corundum crucible to avoid the adhesion of silver on it. The bulks in the crucible were heated in muffle furnace at 1 000 ℃ for 5 min by thorough roasting. Sodium carbonate reacted sufficiently with silver chloride and the reactions were as follows:
Na2CO3+AgCl→Ag2CO3+NaCl (5)
Ag2CO3→Ag2O+CO2(g) (6)
Ag2O→Ag+O2(g) (7)
The final product, silver ingot, was washed by de-ionized water to remove the soluble sodium chloride in it, then dried and weighed to calculate the recovery ratio. A little piece of the silver ingot was rolled into plate for X-ray diffraction.
As for the blue liquid after leaching silver, with cations of Ba2+, Cu2+ and Y3+, sulfate acid was the first choice to separate barium, since Ba2+ is easy to combine with SO42- as indissoluble barium sulfate while Cu2+ and Y3+ are not. After filtering, Ba2+ was eliminated as barium sulfate. The reaction was as follows:
Ba2++SO42-→BaSO4↓ (8)
Then, the white precipitation was dried at 105 ℃ for 2 h and barium sulfate powder was achieved.
The next step was to prepare copper sulfide. It is indissoluble in water, hydrochloric acid and sulfate acid, but can dissolve in hot concentrated nitric acid. As rare earth sulfide, Y2S3, is also indissoluble in the water; however, it is easy to react with acid to produce hydrogen sulphide. Thus, sodium sulfide solution is chosen to separate copper and yttrium in this work.
As the acidity of the filtrate after filtering barium sulfate was very strong, the pH value of the solution should be adjusted to be about 1. Sodium sulfide solution, the precipitator, with a low concentration should be slowly added. This solution was boiled for 30 min so that the sulfide particle agglomerated and was easy to be leached. The black precipitates were filtrated and dried at 105 ℃ for 2 h. In order to eliminate rare earth hydroxide in the precipitates, this precipitates were dissolved with HCl, then the black CuS precipitates were filtrated and dried again.
Rare earth oxalate is indissoluble in acid medium, and rare earth oxide can be obtained by burning rare earth oxalate at 800-1 000 ℃ for 2 h. The reactions were as follows:
Y3++H2C2O4+H2O→Y2(C2O4)3・nH2O+H+ (9)
Y2(C2O4)3→Y2O3+CO2↑+CO↓ (10)
Regulate pH value of the filtrate to 1-2 with ammonia. Heat the beaker to 80-90 ℃. The dosage of the oxalic acid usually was 1.2-1.4 times that of rare earth oxide. Leach and wash the white precipitates after holding for 30 min. Then, dry the white precipitates at 105 ℃ for 2 h. Transfer the white precipitates to a clean melting pot, and yttrium oxide powder was obtained by calcining at 900 ℃ for 2 h.
3 Results and discussion
3.1 Recovery of silver
3.1.1 Leaching ratio of silver
The leaching ratios of silver for three samples were calculated to be 95.47%, 97.83% and 96.14%, respectively (Table 1).
Table 1 Experimental data of silver recovery process from YBa2Cu3O7-x/Ag composites
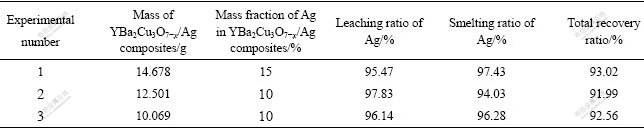
There are three main factors concerned with the leaching ratio of silver chloride. The first is the concentration and quantity of nitric acid. Dense nitric acid can raise the leaching ratio of silver but its evaporation will result in a low usage and leaching of impurities. However, the dilute nitric acid will slow down the reaction speed. In this work, different concentrations of nitric acid, 2, 4, 6, 8 and 10 mol/L were employed, respectively. Finally, 6 mol/L of nitric acid was decided as the optimum concentration, for this concentration of resulted in a proper reaction speed and relatively less evaporation. It has been proved that the leaching ratio would increase when leaching silver from silver sulfide with increasing the amount of nitric acid, and the final addition of nitric acid is double theoretical amount in Ref.[9]. In this work, nitric acid of 6 mol/L was serially added, the total addition amount of nitric acid was double the theoretical addition, nearly the same as the use level in Ref.[9].
The second factor on the leaching ratio of silver chloride is the reacting temperature. Theoretically, higher temperature can improve the leaching ratio of silver, but the leaching speed of other elements will also be accelerated, which in the end become impurities in the product of silver chloride. In this work, the reaction temperature is controlled at 80 ℃. It is necessary of the solution being kept boiling to make silver chloride granulate and reduce the loss of silver chloride.
The third factor is the mass ratio of solid to liquid. The leaching ratio decreases with the increase of ratio of solid to liquid, since more nitric acid can make the solid dissolve thoroughly. In this work, the leaching ratio nearly reached the maximum when the ratio was 1?4. Taking the efficiency into account, it is not necessary to use excess nitric acid.
3.1.2 Recovery ratio of silver
According to Table 1, the average recovery ratio of the three groups is 92.56%, lower than 97% that was from processed radiographic films in the Ref.[10]. It is considered that the loss of silver originates from the segments, i.e. small amount of silver chloride left in the filter residue, on the filter paper, as well as those left in the crucible and volatized at high temperature. The small content of silver in the insoluble residue can be leached by adding more nitric acid, but it is inefficient for the recovery cost. It is recommended that the mixture of silver chloride and sodium carbonate is sintered at a temperature near the melting point (851 ℃) of sodium carbonate to avoid the volatilization of silver. In order to increase the recovery ratio, excess sodium carbonate is also needed for a thorough reaction with silver chloride.
3.1.3 Purity of silver
The X-ray diffraction pattern of the filter sediment silver chloride indicates that there is not any impurity in silver chloride, namely, the content of the impurity is too little to be examined by the X-ray diffraction.
The purity of recovered silver ingot is analyzed by chemical method and the results of two samples are 95.59% and 95.86%, respectively. This indicates that there still exist over 4% impurities in the silver samples. The X-ray diffraction pattern of silver plate rolled from silver ingot is shown in Fig.1.

Fig.1 X-ray diffraction pattern of silver plate recovered from waste YBa2Cu3O7-x/Ag composites showing that small content of impurity possibly of silver chloride
According to Fig.1, there is only a little peak at 31.9315? of 2θ, indicating the impurity in silver plate. The impurity of silver chloride can be removed by adding excess sodium carbonate in the smelting process of silver chloride and sodium carbonate. It is suggested that over 99.9% pure silver can be obtained with electrolysis method directly when the content of silver in silver ingot is over 90 %[11].
As each experimental amount of waste YBa2Cu3O7-x/Ag materials is just about 10 g and silver amount is 1-2 g, by comparing with batch size in some references, it makes the calculative recovery ratio lower than that in Ref.[10].
3.2 Recovery of barium
Fig.2 shows the X-ray diffraction result of the dried white precipitates, indicating that the precipitate is pure barium sulfate, i.e., the content of the impurity is lower than the detection limit.

Fig.2 X-ray diffraction pattern of obtained pure barium sulfate
During the precipitation, as Ba2+ and SO42- combined quickly, the reaction easily occurred without heating up. On the contrary, heating might bring some adverse effects. The solubility of Y2(SO4)3 is 9.76 g in 100 g water at 20 ℃, and the solubility decreases when the temperature increases[12]. For example, the solubility is 4.9 g in 100 g water at 40 ℃. If the solution is heated up, some yttrium sulfate might be crystallized in the product and thus affect the purity of barium sulfate and the recovery of yttrium oxide. Besides, it is not suggested to use sodium sulfate in this work. Because yttrium sulfate as rare earth sulfate reacts with alkali metal sulfate easily to generate sulfate double salt[12], such as Na2SO4・Y2(SO4)3, while rare earth sulfate double salt is relatively indissoluble normally. Thus, its existence may also affect the purity of barium sulfate and the recovery of yttrium oxide. In addition, if sulfate acid is excessive during the separation of barium and there is surplus SO42- in the solution, yttrium will also combine with sodium easily to generate indissoluble alkali metal sulfate which remains in the white precipitates and thus affects the subsequent separation of Cu2+ and Y3+ when adding sodium sulfide during the separation of copper.
3.3 Recovery of copper
3.3.1 Effects of pH value
pH value has a great effect on the extraction of copper and yttrium because Cu2+ and Y3+ easily combine with OH-:
Y3++3OH-→Y(OH)3↓ (11)
Cu2++3OH-→Cu(OH)2↓ (12)
Once the precipitates is formed, it affects the purity of sequent product. Many data indicate that yttrium hydroxide is white precipitate, the pH value of nitrate precipitate is about 6.95[12], and copper ions begin to generate precipitate at about pH 3. The solution appeared trace turbidity at about pH 1.7 during the experimental process. In order to ensure the purity of copper sulfide, the pH of solution was regulated to be about 1 with ammonia at the beginning of the experiment. If sodium sulfide, a strong base-weak acid salt, is excessive, the solution will easily take hydrolytic reaction as follows and make the pH value of the solution increase slightly:
S2-+H2O→HS-+OH- (13)
HS-+H2O→H2S+OH- (14)
For the reason given above, we can approximately determine the reaction end point according to the variation of pH value. For instance, copper can be considered to precipitate completely under the conditions of stirring for 30 min and pH 1.7. When the pH value is lower than that before reaction, it is suitable to add Na2S. The adding amount of Na2S should not be excessive, otherwise, the pH value may increase to about 10 and most of yttrium and copper will precipitate as hydroxide and cannot be separated. In addition, if the pH value is much higher than that before reaction, even yellow yttrium sulfide will appear, which will hinder the separation of yttrium.
3.3.2 Purification of copper sulfide
The copper sulfide precipitate after filtrating for the first time contains a mass of hydroxide, which is confirmed by Fig.3. The analysis result of the dried precipitate by X-ray diffraction indicates that there are yttrium hydroxide and copper hydroxide in copper sulfide. Hence, hydroxide in the precipitates needs to be eliminated with hydrochloric acid to ensure the purity of copper sulfide and the recovery of yttrium. Fig.4 shows the X-ray diffraction analysis result of the dried precipitates of copper sulfide. It proves that the hydroxide impurity can be removed by hydrochloric acid and pure copper sulfide can be finally obtained.
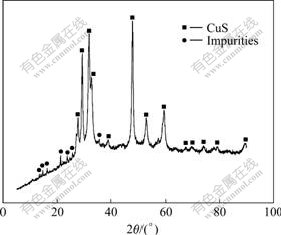
Fig.3 X-ray diffraction pattern of copper sulfide containing impurities before being dissolved with hydrochloric acid

Fig.4 X-ray diffraction pattern of copper sulfide product
3.4 Recovery of yttrium
The white precipitate formed by adding oxalic acid shows to be not theoretic yttrium oxalate but C4H6NO9Y by the X-ray diffraction. However, pure yttrium oxide, as Fig.5 shows, can be obtained by calcining the precipitate at 900 ℃. The impurity in the white precipitate may be hydrocarbons.

Fig.5 X-ray diffraction pattern of obtained pure yttrium oxide
It is necessary of heating, controlling pH value and moderate oxalic acid concentration for precipitating oxalate. If the pH value is too high, the crystallization will be impure. While if the pH value is too low, the precipitation will be incomplete. Ref.[12] pointed out that it is suitable to regulate the pH value to be 1.5. In this experiment, the pH value is determined in the range of 1-2 in order to avoid the generation of hydroxide.
A shortcut process was also attempted, by trying to add oxalic acid first to the liquid after recovering silver to obtain yttrium oxalate under acidic condition, then calcining to obtain yttrium oxide. It was found that the precipitate appeared blue. When the oxalic acid was excessive, the filtrate appeared colorless, indicating that Cu2+ and Y3+ could be co-deposited as CuC2O4 and Y2(C2O4)3. Then, when sulfate acid was added slightly into the solution, white precipitate formed. This indicated that barium was not deposited with oxalic acid, or the solubility of barium oxalate was relatively large under acidic condition. Ref.[13] emphasized that when separating non-rare earth impurity with oxalic acid, the key is to design separation conditions. Keeping the acidity of the solution diminutive may obtain relatively pure rare earth oxide. The experiment proves that CuC2O4 is also slightly dissolved or indissoluble in hydrochloric acid. If continuously adding hydrochloric acid, Y2(C2O4)3 may dissolve, resulting in lowering the recovery ratio of yttrium oxide. Therefore, the separation of copper ion and yttrium ion is the key step if adopting oxalic acid to separate non-rare earth impurity.
4 Conclusions
1) Silver in the YBa2Cu3O7-x/Ag composite was recovered by the method of nitric acid leaching. The XRD result indicates that this method can obtain relatively pure AgCl. Silver forms when smelting the mixture of silver chloride and sodium carbonate at 1 000 ℃. The final recovery ratio of silver is 92.56%, and its purity is 95.86%.
2) The liquid after recovering silver mainly contains Ba2+, Cu2+ and Y3+. According to the different chemical properties, sulfate acid, sodium sulfide and oxalic acid are sequentially added into the solution to precipitate barium sulfate, copper sulfide and C4H6NO9Y. Yttrium oxide is obtained by drying and calcining the white C4H6NO9Y precipitate.
3) During the separation procedure, it is important to control the pH value. The XRD results indicate that the final products are all single-phase compounds as anticipated as BaSO4, CuS and Y2O3, respectively. Thus, useful elements in the spent YBa2Cu3O7-x/Ag composite are separated and recovered successfully.
Acknowledgements
The authors wish to express their thanks to the Noble Metals Trial Plant of Northeastern University, China, for the purity analysis of silver ingot.
References
[1] Lee D F, Chaud X, Salama K. Transport current density in bulk oriented-grained YBa2Cu3Ox/silver composites[J]. Phys C, 1991, 181: 81-87.
[2] Mironova M, Lee D F, Salama K. TEM and critical current density studies of melt-textured YBa2Cu3Ox with silver and Y2BaCuO5 additions[J]. Phys C, 1993, 211: 188-204.
[3] Sergeenkov S A. On weak-link behavior of silver-doped high-Tc granular superconductors[J]. Phys C, 1993, 205: 1-13.
[4] Kohayashi S, Yoshizawa S, Miyairi H, WAKANE H, AGAYA S. Large domain growth of Ag-doped YBaCuO-system[J]. Mater Sci Eng B, 1998, 53(1/2): 70-74.
[5] Szalay A, Horvath D, Vajda I, MAMALIS A G, HARNOIS C. Influence of some metallic additives on the flux trapping ability of high temperature superconducting ceramics[J]. J Mater Proc Tech, 2007, 181(1/3): 36-39.
[6] Harnois C, Desgardin G, Laffez I, CHAUD X, BOURGAULT D. High quality weld of melt textured YBCO using Ag doped YBCO junctions[J]. Phys C, 2002, 383: 269-278.
[7] Fujita H, Tomita M, Murakami M, SAKAI N, HIRABAYASHI I, SAWA K. Deformation analysis of electrical contact using YBaCuO bulk superconductors[J]. Phys C, 2006, 445/448: 1123-1126.
[8] Alan J P, Brian W C, Robert P S. A novel process for the recovery of pure silver from impure silver chloride[J]. Hydromet, 1979, 4(3): 233-245.
[9] Holloway P C, Merriam K P, Etsell T H. Nitric acid leaching of silver sulfide precipitates[J]. Hydromet, 2004, 74(3/4): 213-220.
[10] Syed S, Suresha S, Sharma L M, SYED A A. Clean technology for the recovery of silver from processed radiographic films[J]. Hydromet, 2002, 63(3): 277-280.
[11] Sathaiyan N, Nandakumar V, Ramachandran P. Hydrometallurgical recovery of silver from waste silver oxide button cells[J]. J Power Sour, 2006, 161(2): 1463-1468.
[12] Liu G H. Rare earth solid material science[M]. Beijing: Mechanical Engineering Press, 1997. (in Chinese)
[13] Wu W Y. Rare earth metallurgy[M]. Beijing: Chemical Industry Press, 2005. (in Chinese)
___________________________
Foundation item: Project(50702012) supported by the National Natural Science Foundation of China; Project(20060145030) supported by Doctoral Foundation of Chinese Education Ministry
Corresponding author: LI Feng-hua; Tel: +86-24-83687727; E-mail: lifh@smm.neu.edu.cn
(Edited by YANG Hua)