Ti(Cu,Pt)2��Ti(Cu,Pt)3��ľ���ṹ�͵�������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���7��
�������ߣ���ά�� ���� ����ɽ ������ �̸�÷ ��չ��
����ҳ�룺1839 - 1848
�ؼ��ʣ�Ti-Cu-Pt�ࣻ����ṹ��X�������䣻��һ��ԭ�����㣻��������
Key words��Ti-Cu-Pt phase; crystal structure; X-ray diffraction; first-principle calculations; elastic properties
ժ Ҫ������X���߷�ĩ�������ݽ��Rietveld�ṹ�������о���ȷ����������Ԫ��Ti(Cu,Pt)2 �� Ti(Cu,Pt)3�ľ���ṹ��ʹ�õ���̽��(EPMA)�����Ʒ�ijɷ֣�ͬʱ�������ѹ�ۼ����͵�һ��ԭ��������䵯�����ܽ����о����о����֣�Ti(Cu,Pt)2�Ŀռ�ȺΪAmm2 (No. 38)����VAu2������ͬ�Ľṹ���͡�Ti(Cu,Pt)3�ĽṹΪ�ķ���ϵ��AlPt3�ṹ���ͣ�����P4/mmm�ռ�Ⱥ (No. 123)������ѹ�۲����͵�һ��ԭ�����������Ti(Cu,Pt)2�ĵ���ģ����Pt����������������Ȼ���С����Ti(Cu,Pt)3�ĵ���ģ����Pt���������Ӽ������������ӡ�
Abstract: This paper focused on the crystal structures of two new ternary phases, Ti(Cu,Pt)2 and Ti(Cu,Pt)3, which were studied by X-ray powder diffraction data using Rietveld method. Electron probe microanalysis was used for sample composition examination. Elastic properties of these phases were further measured by nano-indentation, and meanwhile calculated with first-principle (FP) calculations. It is found that the crystal structure of Ti(Cu,Pt)2 is of orthorhombic cell space group Amm2 (No. 38) with structural prototype of VAu2. The resolved structure of Ti(Cu,Pt)3 is of tetragonal AlPt3 type, belonging to the space group P4/mmm (No. 123). The nano-indentation measurement and FP calculations show that the elastic modulus of Ti(Cu,Pt)2 increases firstly then decreases with Pt content, whereas that of Ti(Cu,Pt)3 almost linearly increases with Pt content.
Trans. Nonferrous Met. Soc. China 30(2020) 1839-1848
Wei-jing ZENG, Kun HU, Hua-shan LIU, Hai-long PENG, Ge-mei CAI, Zhan-peng JIN
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 22 July 2019; accepted 8 June 2020
Abstract: This paper focused on the crystal structures of two new ternary phases, Ti(Cu,Pt)2 and Ti(Cu,Pt)3, which were studied by X-ray powder diffraction data using Rietveld method. Electron probe microanalysis was used for sample composition examination. Elastic properties of these phases were further measured by nano-indentation, and meanwhile calculated with first-principle (FP) calculations. It is found that the crystal structure of Ti(Cu,Pt)2 is of orthorhombic cell space group Amm2 (No. 38) with structural prototype of VAu2. The resolved structure of Ti(Cu,Pt)3 is of tetragonal AlPt3 type, belonging to the space group P4/mmm (No. 123). The nano-indentation measurement and FP calculations show that the elastic modulus of Ti(Cu,Pt)2 increases firstly then decreases with Pt content, whereas that of Ti(Cu,Pt)3 almost linearly increases with Pt content.
Key words: Ti-Cu-Pt phase; crystal structure; X-ray diffraction; first-principle calculations; elastic properties
1 Introduction
Ti-Cu intermetallic compounds have been used as implantable devices and dental prostheses because of their good biocompatibility [1], high hardness and strength, low melting temperature, and strong antibacterial ability [2-5]. The addition of other elements can improve their physical properties because of formation of solutions or new ternary compounds. For example, six ternary phases, ��1-Ti(CuxNi1-x)2, ��2-Ti2(CuxNi1-x)3_H, ��3-Ti2(CuxNi1-x)3_L, ��4-Ti5CuNi14, ��5-TiCuNi2 (Cu3Sb type), and ��6-TiCuNi2 (TiAl3 type) [6-10] were found in the Ti-Cu-Ni system, and TiCu0.5Fe0.5 [11], Ti6CuCo23 [12], TiCuZr [13], TiCu2Zr [14], TiCuPd6 [15] and Ti4CuAg [16], were also reported in other systems. The hardness (H) and elastic modulus (E) of TiCu2Zr can reach (7.5��0.3) GPa and 128.4 GPa, respectively, which are significantly higher than those of most binary Ti-Cu or Zr-Cu phases [14]. As a non-toxic element, Pt is commonly added to improve the physical property of cp-Ti, and Ti-Pt binary compounds are promising candidates for dental applications due to their grindability and mechanical compatibility with bone tissue than cp-Ti [17]. In light of alloying theory, it is expected that property of the binary Ti-Cu compounds may be improved and new compounds may be stabilized by adding Pt in Ti-Cu binary system.
Recently, two new ternary phases of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 have been reported by our research group [18]. The Pt contents for Ti(Cu,Pt)2 and Ti(Cu,Pt)3 are (4.0-47.6) at.% and (42.2-65.6) at.% at 1073 K, respectively. However, crystal structure and related properties of these two phases were not studied in detail. In this work, crystal structures and elastic properties of these two phases were investigated.
2 Experimental
2.1 Materials and synthesis
Eight alloys of the Ti(Cu1-xPtx)2 (x=0.05, 0.2, 0.35, 0.5) and Ti(Cu1-xPtx)3 (x=0.6, 0.7, 0.8, 0.9) compounds with a mass of 2 g were arc-melted from high-purity metals, i.e. 99.99 Ti, 99.99 Cu and 99.99 Pt (wt.%), in a chamber filled with argon gas. These alloys were remelted four times to ensure complete fusion and composition homogeneity before being enclosed in an evacuated quartz tube and annealed at 1073 K for 60 d. The annealed alloys were then instantaneously quenched in water to room temperature.
2.2 Structure characterization and hardness measurement
The elemental compositions of the Ti(Cu,Pt)2 and Ti(Cu,Pt)3 compounds were obtained through wavelength-dispersive spectroscopy (WDS) on a JEOL JXA-8800R electron probe micro analyzer (EPMA).
Powder X-ray diffraction patterns of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 compounds were collected at room temperature using a Rigaku D/max 2550V powder diffractometer equipped with a Cu K�� radiation and a diffracted-beam graphite monochromator. The diffractometer was operated at 40 kV and 250 mA, the 2�� scan range was from 10�� to 100�� with a step size of 0.02�� and a count time of 2 s per step. The Materials Data Inc. software Jade 5.0 [19] was used for phase identification. After data collection, the stability of both the X-ray source and the samples was checked by recording the diffraction lines again at low angles.
The H and E were measured on single phase or polycrystalline samples with nano-indentor (UNHT+MCT+MST, CSM, Switzerland). Test was repeated five times on each of the target phase. The surface of each sample was polished before the test.
3 First-principle calculations
Structural optimization and elastic constant calculations were performed with the VASP code [20,21], of which the generalized gradient approximation (GGA) with the Perdew-Burke- Ernzerh (PBE) functional [22] was used as the exchange-correlation potential. Integration over the Brillouin zone was performed using the Monkhorst- Pack set [23] with 450 eV of energy cutoff. The structural parameters of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 were acquired from experimental values of lattice parameter, and the terminal phase models of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 were constructed from a unit cell in which the mixed sites are filled either with Cu or with Pt. The k-point meshes of Ti(Cu1-xPtx)2 (x=0, 1) and Ti(Cu1-xPtx)3 (x=0, 1) were set as 15��9��15 and 15��15��15, respectively. The tolerances of total energy difference for geometry optimization were 10-5 eV/atom, the maximum force of 0.001 eV/ , and the maximum displacement of 1.0��10-2
, and the maximum displacement of 1.0��10-2  . Bulk modulus and volume dependence of the total energies were calculated by fitting the energy against volume data with Birche-Murnaghan equation of states.
. Bulk modulus and volume dependence of the total energies were calculated by fitting the energy against volume data with Birche-Murnaghan equation of states.
4 Results and discussion
4.1 Crystal structure of Ti(Cu0.65Pt0.35)2 phase
Electron probe microanalysis (EPMA) was employed to analyze whether the annealed alloys of Ti(Cu1-xPtx)2 (x=0.05, 0.2, 0.35, 0.5) are single phase or not. Figure 1 shows the phases present in the annealed alloys. It is seen that only the Ti(Cu1-xPtx)2 (x=0.35) alloy is single phase (Fig. 1(c)), while other samples contain at least two phases. So the single-phase sample Ti(Cu0.65Pt0.35)2 was chosen to determine its crystal structure of Ti(Cu,Pt)2.
High-quality X-ray diffraction patterns of the Ti(Cu0.65Pt0.35)2 alloy powder were collected, as illustrated in Fig. 2. By using the Jade 5 program [19], the crystal structure of the Ti(Cu0.65Pt0.35)2 phase was successfully indexed and it has an orthorhombic unit cell with the following lattice parameters: a=4.5688(2)  , b=8.1822(2)
, b=8.1822(2)  , c=4.5603(3)
, c=4.5603(3)  . Reflection conditions (hkl: k+l=2n; 0kl: k+l=2n; h0l: l=2n; hk0: k=2n; h00: no conditions; 0k0: k=2n; 00l: l=2n) indicated the possible space groups: Cmcm (No. 63), Amm2 (No. 38), Cmm2 (No. 35), C222 (No. 21). By comparing the lattice parameters and PXRD pattern of the Ti(Cu0.65Pt0.35)2 phase with those presented in the structure-type database, it was found that Ti(Cu0.65Pt0.35)2 and VAu2 [24] have the same structure type. So the space group Amm2 (No. 38) and the atomic position parameters of VAu2 were taken as the starting values to refine the structural parameters of Ti(Cu0.65Pt0.35)2. The structural refinement of Ti(Cu0.65Pt0.35)2 was then performed using the Fullprof_suite program [25]. The pseudo- Voigt function was used for the simulation of the peak shapes. There are 27 refined parameters, including scale factor, cell parameters, atomic coordinates, full width at half maximum (FWHM), preferred orientation, and isotropic displacement parameters. Background correction used a 12-term polynomial. When Ti atoms occupy 2a and 2b positions of V atoms, Cu and Pt atoms statistically occupy 4d and 4e positions, final refinement converged to rather good residual value (profile factor RP=5.07%, weighted profile factor RWP= 6.83%, Bragg factor RB=7.85%, crystallographic RF-factor RF=5.63%) and yielded a formula of Ti33.33Cu43.29Pt23.38 which was in good agreement with the composition of a new phase of Ti(Cu0.65Pt0.35)2 (32.95 at.% Ti, 43.20 at.% Cu, and 23.85 at.% Pt) by EPMA.
. Reflection conditions (hkl: k+l=2n; 0kl: k+l=2n; h0l: l=2n; hk0: k=2n; h00: no conditions; 0k0: k=2n; 00l: l=2n) indicated the possible space groups: Cmcm (No. 63), Amm2 (No. 38), Cmm2 (No. 35), C222 (No. 21). By comparing the lattice parameters and PXRD pattern of the Ti(Cu0.65Pt0.35)2 phase with those presented in the structure-type database, it was found that Ti(Cu0.65Pt0.35)2 and VAu2 [24] have the same structure type. So the space group Amm2 (No. 38) and the atomic position parameters of VAu2 were taken as the starting values to refine the structural parameters of Ti(Cu0.65Pt0.35)2. The structural refinement of Ti(Cu0.65Pt0.35)2 was then performed using the Fullprof_suite program [25]. The pseudo- Voigt function was used for the simulation of the peak shapes. There are 27 refined parameters, including scale factor, cell parameters, atomic coordinates, full width at half maximum (FWHM), preferred orientation, and isotropic displacement parameters. Background correction used a 12-term polynomial. When Ti atoms occupy 2a and 2b positions of V atoms, Cu and Pt atoms statistically occupy 4d and 4e positions, final refinement converged to rather good residual value (profile factor RP=5.07%, weighted profile factor RWP= 6.83%, Bragg factor RB=7.85%, crystallographic RF-factor RF=5.63%) and yielded a formula of Ti33.33Cu43.29Pt23.38 which was in good agreement with the composition of a new phase of Ti(Cu0.65Pt0.35)2 (32.95 at.% Ti, 43.20 at.% Cu, and 23.85 at.% Pt) by EPMA.

Fig. 1 Backscattered electron (BSE) images of Ti(Cu1-xPtx)2 (x=0.05-0.5) alloys annealed at 1023 K for 60 d
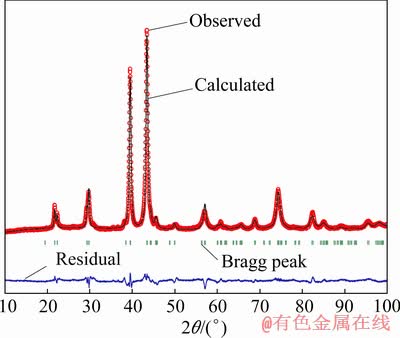
Fig. 2 Observed, calculated and residual powder XRD patterns and Bragg peak positions of Ti(Cu0.65Pt0.35)2 alloy
The crystal data, structural refinement, standardized atomic positions and isotropic atomic displacement parameters (Beq) of the Ti(Cu0.65Pt0.35)2 phase are listed in Table 1.
A set of interatomic distances in Ti(Cu0.65Pt0.35)2 are given in Table 2. The shortest bond length in the structure is dTi1�CCu2(Pt2)=2.6076  , which corresponds to 95.6% of the sum of the atomic radii. The crystal structure of Ti(Cu0.65Pt0.35)2 is shown in Fig. 3.
, which corresponds to 95.6% of the sum of the atomic radii. The crystal structure of Ti(Cu0.65Pt0.35)2 is shown in Fig. 3.
It should be pointed out that CENZUAL et al [26] also reported another structural type of Au2V which crystallized in orthorhombic space group Cmcm (No. 63). The unit cell has 12 atoms, 4 V atoms occupy 4c positions and 8 Au atoms occupy 8g positions. The results of structural refinement of Ti(Cu0.65Pt0.35)2 are Rp=6.46% and RWP=7.76% which are not as good as the results obtained with Amm2. This may be related to the quenching in water of the samples at 1073 K.
Table 1 Crystal data and structural refinement parameters for Ti(Cu0.65Pt0.35)2 phase

Table 2 Typical interatomic distance for Ti(Cu0.65Pt0.35)2 phase


Fig. 3 Schematic illustration of crystal structure of Ti(Cu,Pt)2 phase
4.2 Crystal structure of Ti(Cu0.3Pt0.7)3
Microstructures of the alloys with compositions of Ti(Cu1-xPtx)3 (x=0.6, 0.7, 0.8, 0.9) are presented in Fig. 4. As can be seen, Ti(Cu0.3Pt0.7)3 alloy (Fig. 4(b)) is almost single phase. The elemental composition (25.08 at.% Ti, 22.52 at.% Cu, and 52.40 at.% Pt) of the compound was obtained from EPMA.
Powder XRD pattern of the alloy Ti(Cu0.3Pt0.7)3 was recollected by step-scanning mode with relatively high resolution as shown in Fig. 5. The indexing result of diffraction pattern indicated a tetragonal cell, with the lattice parameters a=3.9097(2)  and c=3.7227(3)
and c=3.7227(3)  . The analysis of systematic extinctions (no conditions) suggested that the possible space groups were P4/mmm,
. The analysis of systematic extinctions (no conditions) suggested that the possible space groups were P4/mmm,  ,
, 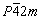 , P4mm and P422. Among them, the P4/mmm has a symmetric centre and high symmetry. We also chose trial-and-error and isostructural method to determine the crystal structure of Ti(Cu0.3Pt0.7)3 phase. Finally, the best fit was found for AlPt3 [27] structure with space group P4/mmm (No. 123). The reliability R factors for the finally achieved parameter set were RP=8.41%, RWP=9.95%, RF=7.94%, RB=6.56%. The observed, calculated, and residuals X-ray powder diffraction patterns of Ti(Cu0.3Pt0.7)3 are shown in Fig. 5.
, P4mm and P422. Among them, the P4/mmm has a symmetric centre and high symmetry. We also chose trial-and-error and isostructural method to determine the crystal structure of Ti(Cu0.3Pt0.7)3 phase. Finally, the best fit was found for AlPt3 [27] structure with space group P4/mmm (No. 123). The reliability R factors for the finally achieved parameter set were RP=8.41%, RWP=9.95%, RF=7.94%, RB=6.56%. The observed, calculated, and residuals X-ray powder diffraction patterns of Ti(Cu0.3Pt0.7)3 are shown in Fig. 5.
In Refs. [28,29], AlPt3 compound crystallizes not only the space group P4/mmm (No. 123), but also P4/mbm (No. 127) and 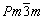 (No. 221). The results of structural refinement of Ti(Cu0.3Pt0.7)3 for space group P4/mbm are not acceptable.
(No. 221). The results of structural refinement of Ti(Cu0.3Pt0.7)3 for space group P4/mbm are not acceptable.
The crystal structure data of the Ti(Cu0.3Pt0.7)3 phase are listed in Table 3. As can be seen that Ti atoms occupy 1a (0, 0, 0) site, while Cu and Pt atoms jointly occupy 1c (0.5, 0.5, 0) and 2e (0, 0.5, 0.5) sites. The interatomic distances of this phase are given in Table 4. All these bond lengths are reasonable with respect to atomic radii. The crystal structure of Ti(Cu0.3Pt0.7)3 is a face tetragonal structure, as illustrated in Fig. 6.
4.3 Elastic properties
4.3.1 Nano-indentation measurement
The H of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 phases were measured through nano-indentation technique.

Fig. 4 Backscattered electron images (BSE) of Ti(Cu1-xPtx)3 (x=0.6-0.9) alloys annealed at 1023 K for 60 d
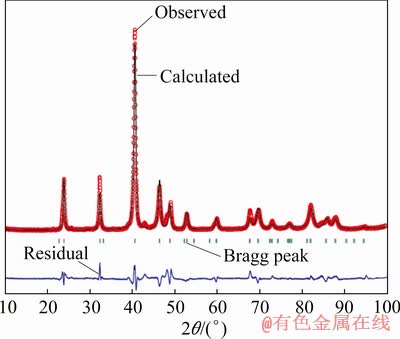
Fig. 5 Observed, calculated and residual powder XRD patterns and Bragg peak positions of Ti(Cu0.3Pt0.7)3 alloy
For Ti(Cu1-xPtx)2 (x=0.05, 0.2, 0.35, 0.5) and Ti(Cu1-xPtx)3 (x=0.6, 0.7, 0.8, 0.9) alloys that contain multiple phases, indentation was performed on the target phase only, i.e. Ti(Cu,Pt)2 or Ti(Cu,Pt)3 phase.
Table 3 Crystal data and refined parameters for Ti(Cu0.3Pt0.7)3 phase

Table 4 Typical interatomic distance for Ti(Cu0.3Pt0.7)3 phase


Fig. 6 Schematic illustration of crystal structure of Ti(Cu0.3Pt0.7)3 phase
A mathematical model was proposed to assess nano-hardness and modulus through indentation load-displacement data [30]. Usually, H is defined as the ratio of the maximum applied load (Pmax) to the projected contact area of the indented impression (A):
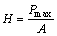 (1)
(1)
And the elastic modulus of the materials can be calculated as
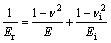 (2)
(2)
where Er is the reduced elastic modulus which takes into account the elastic contributions of the sample and the indenter tip; E and �� are respectively the elastic modulus and Poisson ratio of the tested material, while Ei and ��i are those for the indenter.
Table 5 lists the H and E of Ti(Cu,Pt)2 and Ti(Cu,Pt)3. Both H and E of these two phases increase with increasing content of Pt. For Ti(Cu,Pt)2, whose composition formula is denoted as Ti(Cu1-xPtx)2, with x increasing from 0.08 to 0.48, E increases from 86.7 to 163.5 GPa and H increases from 4.5 to 6.1 GPa. It is worth noting that E of Ti(Cu0.52Pt0.48)2 phase reaches 163.5 GPa, higher than the highest E that has been reported for binary compound TiCu, 161.1 GPa [31]. For Ti(Cu,Pt)3, E increases from 218.4 to 262.2 GPa and H increases from 9.4 to 13.6 GPa with x of Ti(Cu1-xPtx)3 increasing from 0.62 to 0.88.
4.3.2 Elastic property calculation
First-principle calculations have been successfully used in disclosing the correlation between the crystal structure and the elastic properties [31-34]. Here, the elastic properties of the terminal members TiCu2 and TiPt2 of Ti(Cu,Pt)2, and TiCu3 and TiPt3 of Ti(Cu,Pt)3, were calculated using the same approach.
For the single-crystal material, elastic constant (Cij) can be defined as means of a Taylor expansion of total energy, E(V,��), in terms of the infinite small change of strain (��) for the primitive cell volume (V) [35]:
 (3)
(3)
where the indices i and j will run over 1, 2, 3, 4, 5 and 6, the volume of the unstrained system is denoted V0, E(V0, 0) is the corresponding total energy, ��i is an element in the stress tensor and ��i is the factor which takes the value 1 if the Voigt numbers are 1, 2, or 3, and takes the value 2 if the Voigt numbers are 4, 5, or 6.
The bulk modulus B, shear modulus G, elastic modulus E and Poisson ratio �� for the polycrystalline materials can be found from the calculated elastic constants of single-crystal material by using the Voigt-Reuss-Hill (VRH) approximations [36-38]. In light of Ref. [36], for the orthorhombic and tetragonal structures, Voigt bulk modulus (BV) and shear modulus (GV) can be calculated as follows:
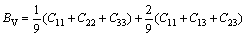 (4)
(4)
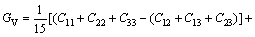
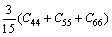 (5)
(5)
Table 5 Measured hardness (H) and elastic modulus (E) of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 phases

while Reuss bulk modulus (BR) and Reuss shear modulus (GR) are defined as [39]
 (6)
(6)

 (7)
(7)
where S is the compliance matrix and C is the elastic constant matrix related by S=C-1.
HILL [38] considered that the average over Voigt and Reuss terms is considered to be the best approximation for finding the theoretical elastic modulus of polycrystalline materials:
B=(BV+BR)/2, G=(GV+GR)/2 (8)
For polycrystalline materials, E and 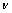 can thus be obtained using the following relations:
can thus be obtained using the following relations:
 ,
, 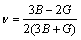 (9)
(9)
The calculated equilibrium lattice parameters, volumes of terminal phases TiCu2 and TiPt2 of Ti(Cu,Pt)2 are listed in Table 6, together with the experimental data of Ti(Cu0.65Pt0.35)2. The calculated single-crystal elastic constants for terminal phases TiCu2 and TiPt2 of Ti(Cu,Pt)2 are listed in Table 7, and the B, G, E and 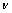 are listed in Table 8. It is clear that these phases are elastically stable according to the mechanical stability criteria for orthorhombic structure, which are: C11>0, C22>0, C33>0, C44>0, C55>0, C66>0, C11+C22+C33+2(C12+ C13+C23)>0, C11+C22-2C12>0, C11+C33-2C13>0, and C22+C33-2C23>0 [39].
are listed in Table 8. It is clear that these phases are elastically stable according to the mechanical stability criteria for orthorhombic structure, which are: C11>0, C22>0, C33>0, C44>0, C55>0, C66>0, C11+C22+C33+2(C12+ C13+C23)>0, C11+C22-2C12>0, C11+C33-2C13>0, and C22+C33-2C23>0 [39].
Table 6 Calculated lattice parameters, unit-cell volumes of terminal phases TiCu2 and TiPt2 and experimental value of Ti(Cu0.65Pt0.35)2
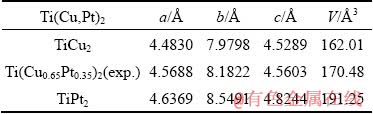
Table 7 Calculated elastic constants for Ti(Cu,Pt)2 phase

Table 8 Calculated bulk modulus B, shear modulus G, elastic modulus E and Poisson ratio 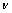 for Ti(Cu,Pt)2 phase
for Ti(Cu,Pt)2 phase
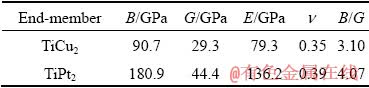
The ratio of B/G proposed by PUGH [40] can be used to predict the brittle or ductile behavior of a material. When B/G is larger than 1.75, the material is ductile, otherwise it is brittle. In Table 8, the B/G ratios of TiCu2 and TiPt2 are 3.10 and 4.07, respectively, implying that Ti(Cu,Pt)2 is a ductile phase.
LIU et al [41] proposed a method for modeling the effect of alloying elements on the elastic coefficients. As for the E of Ti(Cu,Pt)2, we have
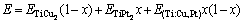 (10)
(10)
Figure 7 shows the E of Ti(Cu,Pt)2 as a function of Pt content. Dotted line means the changing trend between calculated and measured values, fitting result shows E a non-monotonic behavior with Pt content, i.e., it increases firstly then decreases. This nonlinearity fits well with Eq. (10): E=136.22x+78.92(1-x)+ 235.77x(1-x).

Fig. 7 Relationship between Pt content and elastic modulus for target phase Ti(Cu1-xPtx)2 (x=0, 0.08, 0.22, 0.35, 0.48, 1)
In the case of Ti(Cu,Pt)3, the calculated equilibrium lattice parameters, volumes of terminal phase TiCu3 and TiPt3 of Ti(Cu,Pt)3 are listed in Table 9, together with the experimental data of Ti(Cu0.3Pt0.7)3. The elastic constants and moduli of TiCu3 and TiPt3 are listed in Tables 10 and 11, respectively. Similarly, the elastic constants also satisfy the mechanical stability criteria of tetragonal structure (C11>0, C33>0, C44>0, C66>0, (C11-C12)>0, (C11+C33-2C13)>0 and [2(C11+C12)+C33+4C13]>0) [42]. From Table 11, the B/G values of TiCu3 and TiPt3 are 5.74 and 2.12, respectively, both of which are larger than 1.75, indicating ductility of Ti(Cu,Pt)3 phase.
From Fig. 8, it is seen that E of Ti(Cu,Pt)3 shows approximately linear relation with Pt content, which meets the linear function E=210.23x+77.68. In Fig. 8 dashed lines show the possible variation tendency among calculated and measured values of E. It is worth noting that this curve is constructed by combining the experimental and calculated values of E of Ti(Cu,Pt)3, further confirmation and discussion on the actual variation trend are still needed.
Table 9 Calculated lattice parameters, unit-cell volumes of terminal phases TiCu3 and TiPt3 and experimental values of Ti(Cu0.3Pt0.7)3

Table 10 Calculated elastic constants for Ti(Cu,Pt)3 phase

Table 11 Calculated bulk modulus B, shear modulus G, elastic modulus E and Poisson ratio 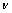 for Ti(Cu,Pt)3 phase
for Ti(Cu,Pt)3 phase


Fig. 8 Relationship between Pt content and elastic modulus for target phase Ti(Cu1-xPtx)3 (x=0, 0.62, 0.70, 0.78, 0.88, 1)
5 Conclusions
(1) The crystal structure and elastic properties of two new phases, Ti(Cu,Pt)2 and Ti(Cu,Pt)3, were investigated. Ti(Cu,Pt)2 has VAu2-type structure, belonging to space group Amm2 (No. 38). Cu and Pt atoms share 4d and 4e sites, while Ti atoms occupy 2a and 2b sites. Ti(Cu,Pt)3 has tetragonal AlPt3-type structure of space group P4/mmm (No. 123). Ti atoms occupy 1a site alone, and Cu and Pt occupy 1b and 2e sites.
(2) The elastic moduli of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 are in the range of 79.3-163.5 GPa and 77.3-286.9 GPa, respectively. The elastic modulus for Ti(Cu,Pt)2 phase exhibits a maximum value with increasing Pt content, while that for Ti(Cu,Pt)3 approximately increases linearly with content of Pt. Additionally, the B/G ratios of Ti(Cu,Pt)2 and Ti(Cu,Pt)3 are found in the range of 3.10-4.07 and 2.12-5.74, implying the good ductility of both phases.
References
[1] ZHANG Er-Lin, ZHENG Lan-lan, LIU Jie, BAI Bing, LIU Cong. Influence of Cu content on the cell biocompatibility of Ti-Cu sintered alloys [J]. Materials Science and Engineering C, 2015, 46: 148-157.
[2] KIKUCHI M, TAKADA Y, KIYOSUE S, YODA M, WOLDU M, CAI Z, OKUNO O, OKABE T. Mechanical properties and microstructures of cast Ti-Cu alloys [J]. Dental Materials, 2003, 19: 174-181.
[3] OHKUBO C, SHIMULA I, AOKI T, HANATANI S, HOSOI T, HATTORI M, ODA Y, OKABE T. Wear resistance of experimental Ti-Cu alloys [J]. Biomaterials, 2003, 24: 3377-3381.
[4] MASATOSHI T, MASAFUMI K, YUKYO T, OSAMU O. Mechanical properties and microstructures of dental cast Ti-Ag and Ti-Cu alloys [J]. Dental Materials Journal, 2002, 21: 270-280.
[5] ZHANG Er-lin, LI Fang-bing, WANG Hong-ying, LIU Jie, WANG Chun-min, LI Mu-qin, YANG Ke. A new antibacterial titanium-copper sintered alloy: Preparation and antibacterial property [J]. Materials Science and Engineering C, 2013, 33: 4280-4287.
[6] YAKUSHIJI M, KONDO Y, KAMEI K. Phase equilibria in the Cu rich region of the Cu-Ni-Ti system and constitution of the Cu-T quasi-binary system [J]. Journal of the Japan Institute of Metals, 1980, 44: 620-624.
[7] PFEIFER H U, BHAN S, SCHUBERT K. The phase diagram establishment of the system Ti-Ni-Cu and some quasi-homologous alloys [J]. Journal of the Less Common Metals, 1968, 14: 291-302. (in German)
[8] VANLOO F J J, BASTIN G F, LEENEN A J H. Phase relations in the ternary Ti-Ni-Cu system at 800 and 870 ��C [J]. Journal of the Less Common Metals, 1978, 57: 111-121.
[9] VANVUCHT J H N. Influence of radius ratio on the structure of intermetallic compounds of the AB3type [J]. Journal of the Less Common Metals, 1966, 11: 308-322.
[10] SINHA A. Close-packed ordered AB3 structures in ternary alloys of certain transition metals [J]. Transactions of the Metallurgical Society of Aime, 1969, 245: 911-917.
[11] WU Chu-ying, LI Jian-chun. Phase structure of the Ti Cu1-xFex system [J]. Metallurgical and Materials Transactions A, 1989, 20: 981-985.
[12] LIU Y, TAKASUKI T, IZUMI O. Alloying behavior of Co3Ti [J]. Metallurgical and Materials Transactions A, 1986, 17: 1433-1439.
[13] CHEBOTNIKOV V N, MOLOKANOV V V. Structures and properties of amorphous and crystalline alloys in the Ti2Cu-Zr2Cu section in the Ti-Zr-Cu system [J]. Inorganic Materials. 1990, 26: 808-811.
[14] YAN Xin-lin, CHEN Xing-qiu, GRYTSIV A V, ROGL P, PODLOUCKY R, POMJAKUSHIN V, SCHMIDT H, GIESTER G. Crystal structure, phase stability and elastic properties of the Laves phase ZrTiCu2 [J]. Intermetallics. 2008, 16: 651-657.
[15] ZIYA A, TAKAHASHI M, OHSHIMA K. The structure and physical properties of CuMPd6 (M=Ti, V, Mn and Fe) ternary alloys [J]. Journal of Alloys Compounds,2009, 479: 60-64.
[16] EREMENKO V, BUYANOV Y, PANCHENKO N. The liquidus surface of the system titanium-copper-silver [J]. Powder Metallurgy and Metal Ceramics, 1970, 9: 301-304.
[17] SONG H J, HAN M K, JEONG H G, LEE Y T, PARK Y J. Microstructure analysis of Ti-xPt alloys and the effect of Pt content on the mechanical properties and corrosion behavior of Ti alloys [J]. Materials, 2014, 7: 3990-4000.
[18] ZHONG Yue, LIU Hua-shan, CAI Ge-mei, JIN Zhan-peng. Experimental study on phase equilibria in Ti-Cu-Pt system [J]. Journal of Phase Equilibria and Diffusion, 2017, 38: 466-476.
[19] QIN Ping-li, CHEN Ye-qing, LIU Hong-rui, NONG Liang-qin, ZENG Ling-min. Crystal structure, thermal expansion and electrical properties of the new Al0.32ErGe2 compound [J]. Journal of Alloys and Compounds, 2008, 463: 34-37.
[20] KRESSE G, FURTHMULLER J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set [J]. Computational Materials Science, 1996, 6: 15-50.
[21] KRESSE G, FURTHMULLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set [J]. Physical Review B, 1996, 54: 11169-11186.
[22] PERDEW J P, CHEVARY J A, VOSKO S H, JACKSON K A, PEDERSON M R, SINGH D J, FIOLHAIS C. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation [J]. Physical Review B, 1992, 46: 6671-6687.
[23] MONKHORST H J, PACK J D. Special points for Brillouin-zone integrations [J]. Physical Review B, 1976, 13: 5188-5192.
[24] STOLZ E, SCHUBERT K. Structure investigations in some systems quasi-homologous homologous to T4-B1 [J]. Zeitschrift fuer Metallkunde, 1962, 53(7): 433-444. (in German)
[25] CARVAJAL J R. Recent advances in magnetic structure determination by neutron powder diffraction [J]. Physica B: Condensed Matter, 1993, 192: 55-69.
[26] CENZUAL K, GELATO L M, PENZO M, PARTHE E. Inorganic structure types with revised space groups [J]. Acta Crystallographoca: Section B, 1991, 47: 433-439.
[27] STADELMAIER H, HARDY W. Ternary carbon alloys of palladium platinum magnesium, aluminum, zine, gallium, germanium, cadmium, indium, tin, mercury thallium and lead [J]. Zeitschrift fuer Metallkunde, 1961, 52: 391-396. (in German)
[28] BRONGER W, MUELLER P, WRZESIEN K. The structure of platinum-rich aluminum platinum alloys [J]. Zeitschrift fuer Anorganische und Allgemeine Chemie, 1997, 623: 363-368. (in German)
[29] OYA Y, MISHIMA Y, SUZUKI T. The Pt-Al and Pt-Ga phase diagram with emphasis on the polymorphism of Pt3Al and Pt3Ga [J]. Zeitschrift fuer Metallkunde, 1987, 78: 485-490. (in German)
[30] OLIVER W C, PHARR G M. Nanoindentation study on tourmaline [J]. Journal of Materials Research, 1992, 7: 1564-1583.
[31] MA Zhen-ning, WANG Xun, YAN Ting-ting, LI Qian, XU Qi-chen, TIAN Jun-long, WANG Lei. First-principles study on thermodynamic stability and electronic characteristics of long-period stacking ordered phases in Mg-Zn-Y alloys [J]. Journal of Alloys Compounds, 2017, 708: 29-33.
[32] LU Cheng, ZHANG Shuai, ZHANG Yu, ZHOU Da-wei, HE Chao-zheng, LU Zhi-wen. Insights into structural and thermodynamic properties of the intermetallic compound in ternary Mg-Zn-Cu alloy under high pressure and high temperature [J]. Journal of Alloys Compounds, 2014, 597: 119-123.
[33] ZHANG Xu-dong, WANG Feng, JIANG Wei. First- principles investigations of structural, mechanical, electronic and optical properties of U3Si2-type AlSc2Si2 under high pressure [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(1): 148-156.
[34] QIAN Jing, WU Chang-yi, GONG Hao-ran. Phase transition, thermodynamic and elastic properties of ZrC [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(12): 2520-2527.
[35] FAST L, WILLS J, JOHANSSON B, ERIKSSON O. Elastic constants of hexagonal transition metals: Theory [J]. Physical Review B, 1995, 51: 17431-17438.
[36] REUSS A, ANGEW Z. Computation of yield point of mixed crystal due to the plasticity condition for single crystals [J]. Math Mech, 1929, 9: 49-58. (in German)
[37] VOIGT W. On the relation between the elasticity constants of isotropic bodies [J]. Annals of Physcics and Chemistry, 1889, 247: 573-586.
[38] HILL R. The elastic behaviour of a crystalline aggregate [J]. Proceedings of the Physical Society A, 1952, 65: 349-354.
[39] WU Zhi-jian, ZHAO Er-jun, XIANG Hong-ping, HAO Xian-feng, LIU Xiao-juan, MENG Jian. Crystal structures and elastic properties of superhard IrN2 and IrN3 from first principles [J]. Physical Review B, 2007, 76: 054115.
[40] PUGH S F. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals [J]. Philosophical Magazine (Series 7), 1954, 45: 823-843.
[41] LIU Z K, ZHANG H, GANESHAN S, WANG Y, MATHAUDHU S N. Computational modeling of the effects of alloying elements on elastic coefficients [J]. Scripta Material, 2010, 63: 686-691.
[42] BECKSTEIN O, KLEPEIS J E, HART G L W, PANKRATOV O. First-principles elastic constants and electronic structure of ��-Pt2Si and PtSi [J]. Physical Review B, 2001, 63: 134112.
��ά������ ��������ɽ�����������̸�÷����չ��
���ϴ�ѧ ���Ͽ�ѧ�빤��ѧԺ����ɳ 410083
ժ Ҫ������X���߷�ĩ�������ݽ��Rietveld�ṹ�������о���ȷ����������Ԫ��Ti(Cu,Pt)2 �� Ti(Cu,Pt)3�ľ���ṹ��ʹ�õ���̽��(EPMA)�����Ʒ�ijɷ֣�ͬʱ�������ѹ�ۼ����͵�һ��ԭ��������䵯�����ܽ����о����о����֣�Ti(Cu,Pt)2�Ŀռ�ȺΪAmm2 (No. 38)����VAu2������ͬ�Ľṹ���͡�Ti(Cu,Pt)3�ĽṹΪ�ķ���ϵ��AlPt3�ṹ���ͣ�����P4/mmm�ռ�Ⱥ (No. 123)������ѹ�۲����͵�һ��ԭ�����������Ti(Cu,Pt)2�ĵ���ģ����Pt����������������Ȼ���С����Ti(Cu,Pt)3�ĵ���ģ����Pt���������Ӽ������������ӡ�
�ؼ��ʣ�Ti-Cu-Pt�ࣻ����ṹ��X�������䣻��һ��ԭ�����㣻��������
(Edited by Xiang-qun LI)
Foundation item: Projects (2016YFB0701404, 2016YFB0701301) supported by the National Key Research and Development Program of China
Corresponding author: Hua-shan LIU; Tel: +86-13607311970; E-mail: hsliu@csu.edu.cn
DOI: 10.1016/S1003-6326(20)65343-2

