
Effect of trace Sn on corrosion behaviors of high voltage anode aluminum foil
SONG Jing-bo(宋婧波)1, MAO Wei-min(毛卫民)1, 2, YANG Hong(杨 宏)1, FENG Hui-ping(冯惠平)1
1. Department of Materials, University of Science and Technology Beijing, Beijing 100083, China;
2. State Key Laboratory for Advanced Metals and Materials, University of Science and Technology Beijing,
Beijing 100083, China
Received 20 September 2007; accepted 16 January 2008
Abstract: The cube texture and the surface corrosion structure of aluminum anode foil for high voltage electrolytic capacitor containing trace Sn were investigated based on quantitative texture analysis and microstructure observation under SEM. High volume fraction of cube texture over 95% and obviously higher specific capacity are obtained in the foils with less than 0.002% Sn. It is indicated that the corrosion behavior of trace Sn on aluminum surface is similar with that of Pb. Higher content of Sn over 0.002% reduces the cube texture component and therefore the specific capacity. Sn, as an eco-friendly microelement, can be applied to replace Pb in improving the homogenous pitting behaviors of high voltage aluminum foils.
Key words: electrolytic capacitor; aluminum foil; Sn; Pb; corrosion
1 Introduction
The electrochemical etching of aluminum foils is an important process used for the enlargement of the surface area of aluminum electrolytic capacitors. High-purity Al-foils for high voltage applications are DC-etched in electrolytes, which causes the growth of tunnels oriented along <001>-directions[1]. It is known that many characteristics of raw foil, such as surface state[2], grain orientation[3], grain size and grain boundary density[4], the nature and distribution of impurities[5-6] could influence the electrochemical etching behaviors. Much research indicated that the Pb impurity had important influences on the tunnel etching, while it remained in the level of about 1×10-6[7]. The Pb addition promotes the development of regular distribution of fine tunnels, and therefore the specific capacity[8]. However, Pb is harmful to the human beings. The European Parliament made a declaration illustrating in 2006 that microelements such as Pb, Hg, Cr were prohibited in the electric products. Recently, Sn was added to aluminum foil instead of Pb as an eco-friendly microelement, while the influence of Sn on recrystallization texture of aluminum foil has been investigated[9]. It was found that micro-alloyed Sn could be assumed as an ecological element substituted for Pb and also guaranteed the high volume fraction of cube texture. However, the effect of the trace Sn on the etching behavior was not revealed. Therefore, it is necessary to investigate in details the effect of Sn on the corrosion behaviors, which is requested by the commercialization of the eco-friendly high voltage anode aluminum foil.
2 Experimental
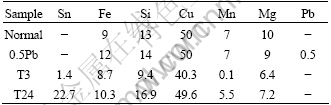
Two types of aluminum foils (sample T1 and T24) with Sn contents of 1.4×10-6 and 22.7×10-6 (mass fraction) respectively were prepared. The contents of Fe, Si and other impurities were at the level of commercial foils for high voltage capacitors. The foil materials were prepared by melting high Al-Sn master alloys, with subsequent casting and homogenization at 600 ℃ for 20 h. Then the ingots were hot-rolled to 6.0 mm in thickness. The hot bands were cold-rolled with a thickness reduction of 98%, to a final thickness of 110 μm. Two other kinds of cold-rolled aluminum foils for high voltage electrolytic capacitors with the thickness of 110 μm were taken from the market, in which sample Normal contains only Fe, Si, Cu and Mg, while sample 0.5Pb contains 0.5×10-6 (mass fraction) Pb besides Fe, Si, Cu and Mg. The chemical compositions of all foils are listed in Table 1. The cold rolled samples were annealed finally in a vacuum furnace at 520 ℃ for 4 h, and then cooled in air.
Table 1 Chemical compositions of aluminum foils (mass fraction, 10-6)
The cube texture fraction of all annealed foils was examined with a X-ray diffraction goniometer (Siemens D5000) and corresponding calculation[10]. The grain size of the foils was determined using orientation mapping technology based on electron backscattering diffraction[11]. The annealed foils were electrochemically etched by an etching and forming factory in a sulfuric acid solution. Then the specific capacities were determined at 375 V forming voltage. The surface corrosion structure was also observed with scanning electron microscope.
3 Results and discussion
3.1 Influence of Sn content on cube texture and corrosion morphology
Table 2 gives the average grain size, the cube texture fraction and the values of specific capacity of the samples. The strong cube texture is obtained in all samples except sample T24. The specific capacity of sample 0.5Pb is the highest, and that of sample T1 containing trace Sn is the next. It should be noticed that the sample Normal has the lowest specific capacity among all samples though its cube texture is the strongest.
Table 2 Average grain size, volume fraction of cube texture and specific capacity of samples at 375 V
 * Tested at 375 V
* Tested at 375 V
Fig.1 shows the corresponding surface corrosion structures of the annealed foils. Samples 0.5Pb and T1 exhibit relatively uniform distribution of etching pits (Figs.1(c) and (e)). However, sample Normal, containing neither Pb nor Sn, has obviously fewer etching pores with uneven distribution (Fig.1(a)). It is found that sample T24, containing higher content of Sn, has a coarse surface etching morphology. Fig.1(h) shows the detailed view of corrosion structures for sample T24. It is observed that the etching pits consist of cubic pores and non-cubic pores. In addition, large channels appear along the grain boundaries (Fig.1(h)), which lead to the inhomogeneity of corrosion structure and the reduction of specific capacity (Table 2). However, it is difficult to find non-cubic pores and the corrosion along grain boundaries in samples Normal, 0.5Pb and T1 (Figs.1(b), (d) and (f)).

Fig.1 Surface corrosion morphologies of aluminum foils annealed at 520 ℃ for 4 h: (a), (b) Sample Normal; (c), (d) Sample 0.5Pb; (e), (f) Sample T1; (g), (h) Sample T24
3.2 Distribution and effects of Pb and Sn
The contents of Fe, Si, Mg and Cu in samples Normal and 0.5Pb are almost the same according to the chemical analysis (Table 1). In this case, the difference in Pb between samples Normal and 0.5Pb causes the difference of corrosion morphologies (Fig.1(a) and (c)) and the corresponding capacitance. The surface energy of high purity aluminum (980 mJ/m2) is the main driving force for the microelement concentration in the foil surface[12]. The alloyed elements can easily become concentrated at the external surface by annealing at high temperature. ASHITAKA et al[13] claimed that Pb could segregate to a metal layer located immediately beneath the thermally formed oxide film. The segregation of Pb, which could provide anode sites to form local cells with aluminum base, may cause electrochemical activation [14]. Thus, it is believed that the contact between the areas with and without enriched Pb promotes initial pitting sites and consequently improves dense distribution of etching sites. Therefore, the more homogenous corrosion pores in sample 0.5Pb (Fig.1(d)) should be induced by Pb. The density of electric current in each individual corrosion pit is hereby reduced and the current distribution becomes more homogenous under the same electrochemical etching condition, which should result in finer and more dispersive corrosion pits followed by the higher specific capacity[4].
The etching pores on sample T1 (Figs.1(e) and (f)) also distribute uniformly over the entire surface in comparison with the corrosion morphology of sample 0.5Pb. It should be noticed that the contents of Mg, Fe, Cu and Si in samples T1 and T24 are approximately the same with those in samples Normal and 0.5Pb(Table 1). The relatively uniform distribution of etching pores (Figs.1(e) and (f)) may not be due to the concentration of those four kinds of atoms on the surface. It is assumed that Sn can act as the same role as Pb in the channel etching process. Both Sn and Pb are in the same element group ⅣA. The atom radii of Al and microelement Pb,Sn are listed in Table 3, in which both Pb and Sn atoms are larger than Al atom and can make positive mismatches in aluminum crystalline lattice. It seems that the surface energy can be more effectively released if the larger size atoms come towards the foil surface despite of their low density in the matrix. The diffusion coefficients[15] of microelement Pb, Sn and other impurity elements as a function of temperature are shown in Fig.2. It is found that the diffusion coefficients of both Pb and Sn are higher than those of any other microelements. Therefore, the concentration of Sn atoms near the surface would be the same with that of Pb atoms. The potentials of both Pb and Sn are much more positive than that of aluminum (Table 3), of which the standard electrode potential could give briefly an expression on the chemical activity of the atoms. Sn may behave in the similar manner with Pb in the aspect of causing the electrochemical activation, since Sn can also form a local cell with the aluminum base. Thus, the Sn atoms, which are accumulated around the surface area, could provide large number of sites for pitting corrosion. This should be the reason that the amount of corrosion pores is much denser (Fig.1(e) and (g)) than that in sample Normal (Fig.1(a)).
Table 3 Size mismatch between atoms of Al and microelements, and standard electrode potential of corresponding ions at 25 ℃

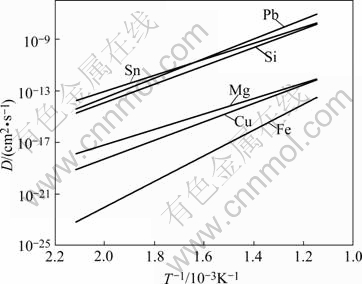
Fig.2 Diffusion coefficient of microelements in Al matrix
3.3 Limitation of Sn content
Table 2 shows that the specific capacity decreases in the order from Sample T1 to T24. It was reported that the structure characteristics of the raw foil, such as grain orientation[3] and gain size[2], have great influence on the corrosion behaviors. In present work, the differences in grain size (Table 2) are relatively small among all samples, implying that they are not the major factors responsible for the difference in surface morphology of the etched foils. It was pointed out[9] that the much higher content of Sn and the annealing at 520 ℃ for longer time would induce an abnormal growth and reduce the cube texture component. The lower volume fraction of cube texture in sample T24 (Table 2) would drastically weaken the increase of the effective surface area, and therefore the specific capacity. On the other hand, there would be more high angle boundaries with higher energy as the cube component decreases, therefore, the higher defect density around those high angle grain boundaries induces the preferred corrosion of the boundaries (Fig.1(h)), which reduces the specific capacity. Thus, Sn can be applied to replace Pb in improving the homogenous corrosion behaviors and enhancing the capacity of high voltage aluminum foils only if Sn content keeps relatively low, in which the volume fraction of cube texture is not reduced.
Furthermore, the significant phenomenon is that the specific capacity of sample T1 is still lower than that of sample 0.5Pb. It is believed that the specific capacity of high voltage foils is dependent on both the microstructure of aluminum foils and the corrosion process. Thus, the relatively lower capacity of sample T1 may be partially attributed to the current corrosion process in the current foil etching factories, which always matches high voltage anode aluminum foils containing Pb. It is believed that a more suitable corrosion process for anode foils with trace Sn needs to be developed.
4 Conclusions
The harmless Sn atoms exhibit anodic behavior interpreted as electrochemical activation as Pb to prompt surface etching of Al foil. Therefore, Sn can be used as an eco-friendly element substituting for Pb to ensure homogenous initial pitting pores, since it does not concern environment problem.
However, higher content of Sn over 0.002% can reduce the volume fraction of cube texture and therefore the specific capacity. More suitable corrosion process for high voltage anode foils with trace Sn needs to be developed.
References
[1] LEE D Y. Tecture and related phenomena [M]. Seoul: The Korean Institute of Metals and Materials, 2007: 554-559.
[2] JIANG H, MAO W, YANG P, FENG H. Influence of surface state on the pitting corrosion behaviors of low voltage aluminum anode foil [J]. Engineering Materials, 2005(2): 22-25. (in Chinese)
[3] KAJIHARA K, TOKUDA K, SUGIZAKI Y. Development of cube texture during industrial processing of high purity aluminum foil obtained with 99% volume fraction of cube component [J]. Materials Science Forum, 2002, 408/412(1): 791-796.
[4] MAO W, CHEN L, SA L, YU Y, LI Y. Influence of grain boundaries on corrosion structure of low voltage aluminum foil [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(1): 1-5. (in Chinese)
[5] XU J, MAO W, FENG H, SHU L. Influence of Cu content on the recrystallization texture of aluminum foil for high voltage anode electrolytic capacitor [J]. Journal of University of Science and Technology Beijing, 2004, 24(2): 133-136. (in Chinese)
[6] MAO W, JIANG H, YANG P. Distribution of microelements and their influence on the corrosion behavior of aluminum foil [J]. Journal of Material Science and Technology, 2005, 21(1): 43-46.
[7] ASHITAKA Z, THOMPSON G E, SKELDON P, WOOD G C, HABAZAKI H, SHIMIZU K. Influence of thermal and surface treatments on distributions of lead in Al-Pb foils [J]. Journal of the Electrochemical Society, 2000, 147(1): 132-139.
[8] ZUO H, MAO W. Influence of micro-alloyed Pb and chromate on the corrosion behavior of high voltage anode aluminum foil [J]. Journal of Materials Science and Engineering, 2005, 23(4): 604-606. (in Chinese)
[9] SONG J, MAO W, YANG H, FENG H. Influence of micro-alloyed Sn on the recrystallization texture of aluminum foil for high voltage anode aluminum foil [J]. Metal Heat Treatment, 2007, 32(7): 1-4. (in Chinese)
[10] CHEN N, MAO W, YU Y, YANG H, ZHANG Y. A method of quantitative fiber texture analysis [C]// Liang Z. Proc 11th Inter Conf Textures of Materials. Beijing: International Academic Publisher, 1996: 81-83.
[11] YANG P. Technology and applications of electron backscattering diffraction [M]. Beijing: Metallurgical Industry Press, 2007. (in Chinese)
[12] TADA K, AWAYA K. Influence of oxide films formed by final annealing on the D.C. etching property of aluminum foil for electrolytic capacitors [J]. J Japan Institute of Light Metals, 2000, 50(11): 590-593.
[13] ASHITAKA Z, THOMPSON G E, SKELDON P, WOOD G C, SHIMIZU K. The behavior of copper and lead during heat-treatment and surface treatment of aluminium capacitor foils [J]. J Electrochem Soc, 1999, 146(4): 1380-1385.
[14] LIN W, TU G C, LIN C F. The effect of lead impurity on the DC-etching behavior of aluminum foil for electrolytic capacitor usage [J]. Corrosion Science, 1996, 38(6): 889-907.
[15] MINAMINO Y, YASUDA T, ARAKI H, YAMANE T. Interdiffusion in binary aluminium alloys and atom size effects [J]. Defect and Diffusion Forum, 1990, 66/69: 1251-1256.
Foundation item: Project(50571020) supported by the National Natural Science Foundation of China; Project(20040008010) supported by the Doctoral Foundation of the Education Ministry of China
Corresponding author: MAO Wei-min; Tel: +86-10-62332989; E-mail: wmmao@mater.ustb.edu.cn.
(Edited by YUAN Sai-qian)