


Trans. Nonferrous Met. Soc. China 22(2012) 1620-1628
Fabrication and photocatalytic properties of Cu2S/T-ZnOw heterostructures via simple polyol process
WU De-zhi, FAN Xi-mei, TIAN Ke, DAI Jia, LIU Hua-rong
Key Laboratory of Advanced Technologies of Materials of Ministry of Education,
School of Materials Science and Engineering, Southwest Jiaotong University, Chengdu 610031, China
Received 23 September 2011; accepted 5 January 2012
Abstract: The Cu2S/tetrapod-like ZnO whisker (T-ZnOw) heterostructures were successfully synthesized via a simple polyol process employing the poly(vinyl pyrrolidone) (PVP) as a surfactant. The as-prepared heterostructures were characterized by X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared (FTIR). The photocatalytic properties of Cu2S/T-ZnOw nanocomposites synthesized with different PVP concentrations were evaluated by photodegradation of methyl orange (MO) under UV irradiation. The results show that the Cu2S/T-ZnOw nanocomposites exhibit remarkable improved photocatalytic property compared with the pure T-ZnOw. The sample prepared with 3.0 g/L PVP shows an excellent photocatalytic property and the highest photodegradation rate of MO is 97% after UV irradiation for 120 min. Besides, the photocatalytic activity of the photocatalyst has no evident decrease even after four cycles, which demonstrates that the Cu2S/T-ZnOw photocatalyst exhibits an excellent photostability. Moreover, the photocatalytic mechanism of the Cu2S/T-ZnOw nanocomposites was also discussed.
Key words: Cu2S/T-ZnOw heterostructure; photocatalytic property; Cu2S nanoparticle
1 Introduction
A great deal of efforts have been devoted in recent years to developing semiconductor photocatalysts with high photocatalytic activities for environmental protection purposes such as air purification, water disinfection, hazardous waste remediation, and water purification [1-4]. Among various semiconductor photocatalysts, ZnO has been recognized to be a preferable material for a variety of environmental applications, due to its high photosensitivity, non-toxic nature, low cost and chemical stability [5-7]. However, despite its great potential, the fast recombination of the photogenerated electron-hole pairs in the single phase ZnO leads to a very low photocatalytic efficiency and hinders the commercialization of this technology. Compared with single-phase photocatalysts, the heterostructures possess significant advantages for promoting the separation of electron-hole pairs and keeping reduction and oxidation reactions at two different reaction sites [8]. Recently, coupled semiconductors composed of ZnO and other metal oxides or sulfides have also been studied, such as combining ZnO with SnO2, In2O3, CeO2 and CdS semiconductor materials [9-13]. These results demonstrated that the nanocomposites fabricated by the coupling of different semiconductor materials will exhibit collective and enhanced property by mutual transfer of charge carriers (electrons and holes) from one semiconductor to another under irradiation and, consequently, achieving a higher photocatalytic activity [14-16].
Cu2S is well known as a p-type semiconductor material with a narrow band gap (1.2 eV) [17]. It can be used in photosensitizer of various wide band-gap semiconductor photoanodes [18,19], electronic and optoelectronic chips [20-22], etc. The energy band structures of Cu2S and ZnO are adequate to promote the electron transfer process where the photogenerated electrons can flow from Cu2S to ZnO [23,24], and the charge carriers become physically separated upon generation. Therefore, the Cu2S/tetrapod-like ZnO whisker (T-ZnOw) composites were selected as a target product to prepare for the photocatalytic degradation of organic pollutants.
It is widely acknowledged that organic surfactants, such as poly(vinyl pyrrolidone) PVP, have an important role in controlling the synthesis of nanomaterials, which determine not only the shape but also the size of the particles [25]. For example, LI et al [26] successfully synthesized Bi2WO6 by a hydrothermal process with the assistance of surfactant PVP. XU et al [27] prepared the size-controllable YVO4:Eu3+ spheres by using PVP. SUN et al [28] successfully synthesized the silver nanowires employing PVP as the shape-direction agent. The studies showed that PVP played an important role in controlling the size of the nanomaterials.
The aim of this work is to investigate the role of Cu2S in improving the photocatalytic property of ZnO. In this study, Cu2S nanoparticles were grown on the surface of T-ZnOw by a simple polyol process, in which PVP was used as a growth-directing agent. In order to achieve the aforementioned purpose, the effects of different PVP concentrations on the microstructure and photocatalytic property of the Cu2S/T-ZnOw nanocomposites were discussed.
2 Experimental
2.1 Sample synthesis
The T-ZnOw was prepared by equilibrium gas controlling method using metallic zinc as the main raw material, which has been reported in our former research [29]. Cu2S/T-ZnOw nanocomposites were prepared by a simple polyol process in which T-ZnOw, copper acetate (analytical grade), thiourea, diethylene glycol (DEG) and PVP were used. The fabrication process was as follows: firstly, 0.2 g of copper acetate was added into 80 mL of DEG with PVP concentrations of 0.6, 0.8, 1.0, 3.0 and 5.0 g/L, respectively and the solution was magnetically stirred for about 30 min; then, 2 g T-ZnOw was poured into the obtained solutions; subsequently, 20 mL thiourea solution (0.005 mol/L) was slowly dropped into the above mixed solutions, respectively. Thereafter, the mixed suspension was maintained at 180 �� for 30 min. After the system was cool to room temperature naturally, the precipitates were collected after being washed with distilled water and ethanol three times, respectively, and then dried in a vacuum oven at 60 �� for 8 h. The Cu2S/T-ZnOw compound was obtained.
2.2 Sample characterization
The microstructure of the as-fabricated samples was investigated by X-ray diffraction (Panalytical X'pert PRO) with a Cu target and a monochronmator at 40 kV and 40 mA. Field emission scanning electron microscopy (FESEM, Fei Quanta 200, USA) with an accelerating voltage of 20 kV was conducted to analyze the morphologies of the photocatalysts. X-ray photoelectron spectroscopy (XPS) was performed with a PHI 5600 multitechnique system by a monochromatic Al K�� X-ray source. All core level spectra were referenced to the C 1s neutral carbon peak at 284.8 eV. FTIR spectra were recorded in the range from 450 to 4000 cm-1 using Fourier transform infrared spectrophotometer with KBr as the reference.
2.3 Photocatalytic property test
The photocatalytic properties of the samples were determined by measuring the photodegradation efficiency of MO aqueous solution. The photocatalytic degradation of MO was tested in our home-made instruments. Firstly, 50 mg of the photocatalysts prepared with different PVP concentrations were added into five beakers filled with 50 mL of MO aqueous solution (10 mg/L), respectively. Then, the obtained mixtures was stirred in dark for 20 min, and meanwhile irradiated by a UV-lamp (Hangzhou, China) with 254 nm emission wavelength at 25 ��. The solution was sampled every 20 min during UV irradiation in order to determine the degradation of MO, which was done by measuring the absorbance at 466 nm using a UV-Vis 2550 spectrophotometer. To further evaluate the photostability of the Cu2S/T-ZnOw, recycled experiments for the photodegradation of MO were also conducted.
3 Results and discussion
3.1 Structure and morphology
The X-ray diffraction patterns of pure T-ZnOw, neat Cu2S and Cu2S/T-ZnOw nanocomposites fabricated with different PVP concentrations were measured, respectively. As shown in Fig. 1, the diffraction peaks at 2��=31.88��, 34.41��, 36.26��, 47.52�� and 56.62�� are ascribed to the typical wurtzite structure of ZnO (JCPDS card No. 36��1451). This indicates that the crystal structure of ZnO is intact during the fabrication process. The characteristic peaks at 2��=27.86��, 32.29��, 46.31��, 54.82�� correspond to the (111), (200), (220), (311) crystal planes of neat Cu2S cubic phase (JCPDS card No. 84��1770). However, these diffraction peaks of Cu2S are absence in the XRD patterns of Cu2S/T-ZnOw nanocomposites, which may be attributed to the very low amount of Cu2S nanoparticles loaded on the surface of T-ZnOw. This will be further discussed by the FESEM test and XPS analysis.

Fig. 1 XRD patterns of pure T-ZnOw, neat Cu2S and Cu2S/T-ZnOw nanocomposites prepared with PVP concentrations of 0.6, 0.8, 1.0, 3.0 and 5.0 g/L, respectively
FESEM tests were carried out to observe the amount and morphology of Cu2S in the Cu2S/T-ZnOw nanocomposites. Figure 2 shows the FESEM images of the pure T-ZnOw and Cu2S/T-ZnOw nanocomposites synthesized with the different PVP concentrations. Figure 2(a) gives the morphology of pure T-ZnOw for a comparison. The length of the tetrapod arm of T-ZnOw is 20-40 ��m. Figures 2(b�Cf) show the morphologies of Cu2S/T-ZnOw nanocomposites with PVP concentrations of 0.6, 0.8, 1.0, 3.0 and 5.0 g/L, respectively. It was found that lots of nanoparticles were deposited on the surface of T-ZnOw. When the concentration of PVP was relatively low, many nanoparticles and little agglomerates were obviously observed on the surface of T-ZnOw. Besides, with the increase of PVP concentration from 0.8 to 3.0 g/L, the agglomerates gradually disappeared and the nanoparticles uniformly dispersed on the arms surface of T-ZnOw. However, with further increasing the amount of PVP to 5.0 g/L, the nanoparticle amounts obviously decreased and many nanoparticles failed to deposit on the surface of T-ZnOw (see Fig. 2(f)). The results can confirm that the appropriate amount of PVP is crucial to the synthesis of Cu2S/T-ZnOw nanomaterials. On one hand, an appropriate amount of PVP could efficiently prevent the aggregation of the nanoparticles and play an important role in obtaining monodispersed nanomaterials. On the other hand, when the excessive amount of PVP was used, the amount of Cu2S nanoparticle on the surface of T-ZnOw was reduced due to the strong absorption of PVP which hindered the deposition of Cu2S nanoparticle on the surface of T-ZnOw [24,25].

Fig. 2 FESEM images of pure T-ZnOw (a) and Cu2S/T-ZnOw nanocomposites synthesized with different PVP concentrations of 0.6 g/L (b), 0.8 g/L (c), 1.0 g/L (d), 3.0 g/L (e) and 5.0 g/L (f), respectively, and EDS spectrum (g) taken from region marked by matrix
In order to reveal the chemical compositions of the nanoparticles, the area marked by a matrix in Fig. 2(b) was analyzed by the energy dispersive X-ray spectroscopy (EDS) and the result is shown in Fig. 2(g). It can be seen that the elements in the nanoparticles are Zn, O, Cu and S. These results suggest that the nanoparticles may be composed of Cu2S and ZnO, and the surface chemical states will be further confirmed by XPS analysis. Moreover, the observed morphologies also indicate that the loading amount of Cu2S nanoparticles is small, which matches the XRD results.
3.2 Surface chemical states and FTIR studies
The XPS spectra of Cu2S/T-ZnOw photocatalyst synthesized with 3.0 g/L PVP are shown in Fig. 3. All binding energy values in the XPS spectra were calibrated according to the information of C 1s (284.6 eV). From Fig. 3(a), Zn, O, Cu, S and C elements were observed and there are no peaks of other elements. The presence of C element mainly originated from the oil pump owing to vacuum treatment. Therefore, it was concluded that the Cu2S/T-ZnOw nanocomposites were composed of the Zn, O, Cu and S elements. The binding energy of S 2p for Cu2S/T-ZnOw nanocomposites is 162 eV (Fig. 3(b)). The binding energy is smaller than that of sulfur and related compounds (S0: 164.0 eV; chemisorbed SO2: 163�C165.5 eV; SO32-: 166.4 eV; SO42-: 168�C170 eV), which is typical for S2- ions [30]. The binding energy peaks of Cu 2p3/2 and Cu 2p1/2 for Cu2S/T-ZnOw nanocomposites locate at about 932.2 and 952.3 eV, respectively, and no ��shake-up�� peaks are found in the higher binding energy direction (Fig. 3(c)), which demonstrated that the copper in the nanocomposites was present in the +1 oxidation state (Cu+) [31]. These results further confirmed that the nanoparticles on the surface of T-ZnOw were composed of Cu2S crystals, agreeing with the above-mentioned EDS results.
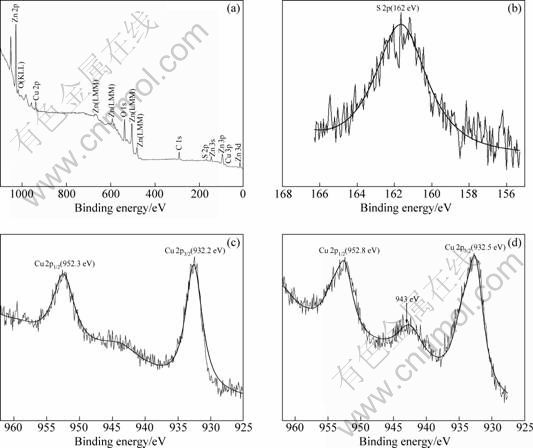
Fig. 3 XPS patterns of Cu2S/T-ZnOw nanocomposites prepared with 3.0 g/L PVP after the fourth recycled photocatalytic experiment: (a) Full spectrum; (b) Spectrum for S 2p; (c) Spectra for Cu 2p3/2 and Cu 2p1/2; (d) Spectra for Cu 2p3/2 and Cu 2p1/2
In order to further investigate the photostability of the Cu2S/T-ZnOw nanocomposites, XPS of the sample with 3.0 g/L PVP after the fourth recycled photocatalytic experiment is also presented in Fig. 3(d). It can be seen that there is a ��shake-up�� characteristic peak at about 943 eV associated with both Cu 2p3/2 and Cu 2p1/2 lines, which is generally assigned to the +2 oxidation state copper [31,32]. Therefore, the XPS results confirm the phase transformation from a little Cu2S to CuS on the face of the Cu2S/T-ZnOw nanocomposites in MO solution after the fourth recycled UV light experiment.
Figure 4 shows the FTIR spectra of pure T-ZnOw and Cu2S/T-ZnOw nanocomposites. The characteristic absorption peak at 504 cm-1 was assigned to Zn��O stretching vibration [33]. The absorption band at 3410 cm-1 corresponds to the stretching vibration of the hydroxyl, which came from the surface of pure ZnO particles. By comparison, the absorption peak at 1380 cm-1 was attributed to the stretching vibration of Cu��S in the Cu2S/T-ZnOw nanocomposites [34]. In addition, the hydroxyl stretching vibration in the Cu2S/T-ZnOw nanocomposites disappeared, which indicates that the Cu2S nanoparticles were strongly bonded to the surface of T-ZnOw. Besides, the characteristic peaks of the C=O, C��N and C��H groups from PVP did not appear in the FTIR spectrum, suggesting that the organic surfactant PVP had been removed completely from the Cu2S/T-ZnOw nanocomposites.

Fig. 4 FTIR spectra of pure T-ZnOw and Cu2S/T-ZnOw nanocomposites prepared with 3.0 g/L PVP
3.3 UV-Vis diffuse re?ectance spectroscopy
Figure 5 shows the UV-Vis diffuse reflectance spectra of T-ZnOw and Cu2S/T-ZnOw photocatalysts. It can be seen that the optical absorption edges of the samples prepared with 0.6, 3.0 and 5.0 g/L PVP are shifted towards longer wavelength located at 393, 388 and 386 nm, respectively. Besides, the diffuse reflectance spectrum of the samples shows a long absorption band between 400 and 800 nm, while there is only a UV absorption edge located at about 381 nm for T-ZnOw. The band gap energies (Eg) of the samples and T-ZnOw were determined to be 3.15, 3.20, 3.21 and 3.25 eV, respectively, according to the following equation, Eg=1239.8/��g [35], where ��g is the wavelength of the optical absorption edge which was obtained from the intersection of two tangents of the absorption curve.

Fig. 5 UV-Vis diffuse re?ectance spectra of T-ZnOw and Cu2S/T-ZnOw photocatalysts prepared with 0.6, 3.0 and 5.0 g/L PVP, respectively
3.4 Photocatalytic property
Figure 6 shows the absorption curves of MO aqueous solution in the presence of Cu2S/T-ZnOw nanocomposites synthesized with 3.0 g/L PVP. Obviously, the absorbance peaks of MO aqueous solution weakened with the increase of the irradiation time, and the maximum absorbance peak located at 466 nm almost disappeared completely after UV irradiation for 120 min, due to the demethylation and hydroxylation of MO during the photocatalytic experiment [36].
The photocatalytic properties of Cu2S/T-ZnOw nanocomposites synthesized with different concentrations of PVP were evaluated in terms of the photodegradation efficiency of MO solution under UV light irradiation. In the present work, the degradation efficiency (Rd) is defined by the following equation [37]:
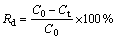
where C0 is the initial concentration of MO solution and Ct is the concentration of MO solution after the irradiation time of t. As shown in Fig. 7, the degradation efficiency of MO solution without any photocatalyst is less than 3% within 120 min under UV irradiation. In other words, the single UV irradiation has a minimal influence on the degradation of MO solution. For comparison, the photocatalytic experiment of neat T-ZnOw was also introduced. The photodegradation efficiency of MO solution with the Cu2S/T-ZnOw nanocomposites is higher than that of the neat T-ZnOw after UV irradiation for 120 min. The photocatalytic property of Cu2S/T-ZnOw nanocomposites increases with the increase of PVP concentration up to 3.0 g/L. However, with further increasing the PVP concentration to 5.0 g/L, the degradation efficiency of MO solution declines obviously after irradiation for 120 min. Besides, the sample synthesized with 3.0 g/L PVP exhibits the highest photodegradation rate (97%) in photodegradation of MO for 120 min. In the photocatalytic experiments, the commercial TiO2 (Degussa P-25) was used as the photocatalytic reference. It can be seen that the highest photodegradation rate of the sample almost reached that of P-25 after UV irradiation for 120 min in our home-made instruments.
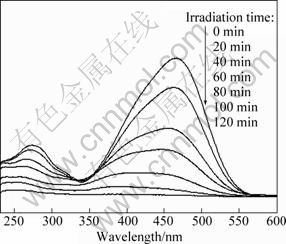
Fig. 6 Absorption curves of MO solution in the presence of Cu2S/T-ZnOw synthesized with 3.0 g/L PVP under different UV irradiation time

Fig. 7 Photodegradation curves of MO solution in the presence of Cu2S/T-ZnOw nanocomposites synthesized with different PVP concentrations
The photoactivity enhancement for the coupled semiconductor/semiconductor heterostructure or metal/ semiconductor heterostructure was attributed to the heterojuction existing in the composites, which might act as a rapid separation site for the photogenerated electrons and holes due to the difference in the energy level of their conduction bands and valence bands [38�C41]. For the Cu2S/T-ZnOw heterostructure catalyst, the enhanced photoactivity was contributed to the many heterojuctions existing in the coupled Cu2S/T-ZnOw semiconductors. When increasing the PVP concentration, the Cu2S nanoparticles uniformly dispersed on the surface of T-ZnOw and thus increased the heterojuctions existing at the interface of Cu2S and T-ZnOw, which were beneficial to the photoactivity enhancement. However, when an excessive amount of PVP was used, the Cu2S amount was reduced due to the strong absorption of PVP on the surface of T-ZnOw, which resulted in reducing the amount of heterojuctions and thus reduced the photoactivity. Therefore, an appropriate PVP concentration was beneficial to enhancing the photoactivity of the Cu2S/T-ZnOw heterostructure catalysts.
The photostability of the Cu2S/T-ZnOw photocatalyst was also studied by cycled tests of the catalysts in fresh MO solution under UV light irradiation. Figure 8 shows the change of photodegradation efficiency from the first cycle to the fourth cycle using the sample with 3.0 g/L PVP. It can be seen that the degradation efficiency of the sample slightly drops after every cycle, which is mainly ascribed to the phase transformation of a little Cu2S to CuS. This agrees well with the XPS results (Fig. 3(d)). However, the photodegradation efficiency of the sample even after the fourth cycle is still much higher than that of pure T-ZnOw, which demonstrates that the Cu2S/T-ZnOw photocatalyst possesses an excellent photostability.
The enhanced activity of the coupled Cu2S/T-ZnOw nanocomposites under UV irradiation can be interpreted using a schematic diagram of the energy band structure of Cu2S/T-ZnOw heterojunction, as shown in Fig. 9. It is reported that interparticle transfer of charge carriers contributes to the enhanced photocatalytic efficiency of coupled semiconductors when the energies of valence and conduction bands properly match [42]. The conduction band (CB) edge of Cu2S (-4.44 eV vs absolute vacuum scale (AVS)) is higher compared with ZnO (-4.19 eV vs AVS); the valence band (VB) edges of Cu2S and ZnO are located at -3.34 eV and -0.99 eV (vs AVS), respectively [24,43,44]. On the basis of the different positions of their band gaps, the photoexcited electrons can transfer from the conduction band of Cu2S to that of ZnO, contrarily, the photoexcited holes immigrate from the valence band of ZnO to that of Cu2S under UV irradiation. Besides, the interface between the Cu2S/T-ZnOw heterostructure might act as a rapid separation site for the photogenerated electrons and holes, which resulted in enhancing the separation efficiency of photogenerated electrons and holes, and thus enhanced the photocatalytic activity in the photocatalytic process [38,41,45,46]. The photocatalytic reactions were possibly proposed as follows:
Cu2S/ZnO+hv��Cu2S(e+h+)/ZnO(e+h+)��
Cu2S(h+)/ZnO(e) (1)
e+O2��?O2- (2)
h++OH-��?OH (3)
?O2-+H2O��?HO2+OH- (4)
?HO2+H2O��?H2O2+?OH (5)
H2O2��2?OH (6)
?OH+MO��Oxidation products (7)
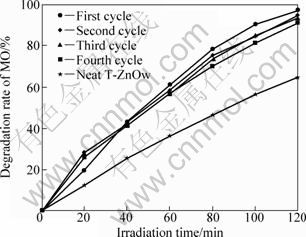
Fig. 8 Photodegradation curves of MO solution in the presence of Cu2S/T-ZnOw synthesized with 3.0 g/L PVP for different recycled experiments
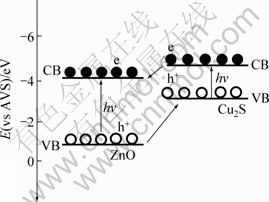
Fig. 9 Schematic representation of excitation and separation of electrons and holes for Cu2S/T-ZnOw heterostructure under UV irradiation
Under UV light irradiation, the coupled semiconductor catalysts were excited to generate the electron (e)-hole (h+) pairs. The photogenerated electrons emigrated to the surface of ZnO, and the photogenerated holes transferred to that of Cu2S. Then the electrons reacted with the dissolved oxygen to yield the superoxide anion radical (?O2-), which continuously participated in the photocatalytic reaction to generate the hydroperoxy (?HO2) and then generated the hydroxyl radical (?OH) which was responsible for the oxidation decomposition of methyl orange. Besides, the holes were trapped by the surface hydroxyl to produce the hydroxyl radical. Finally, the model pollutant (MO) was oxidized by the hydroxyl radical in the photocatalytic process [47]. Of course, the higher hydroxyl radical content was beneficial to enhancing the photoactivity of the Cu2S/T-ZnOw catalyst.
4 Conclusions
The Cu2S/T-ZnOw heterostructures were successfully synthesized by a simple polyol process. The heterostructure catalysts show much better photocatalytic activity and photostability in photodegradation of MO under UV light compared with the pure T-ZnOw, and the Cu2S/T-ZnOw heterostructure catalyst with 3.0 g/L PVP content exhibits the optimal photocatalytic activity. In future, the coupled Cu2S/T-ZnOw nanocomposites may be a new photocatalyst to be utilized in environmental pollution control.
References
[1] HOFFMANN M R, MARTIN S T, CHOI W, BAHNEMANN D W. Environmental applications of semiconductor photocatalysis [J]. Chemical Reviews, 1995, 95: 69-96.
[2] HU C, YU J C, HAO Z, WONG P K. Photocatalytic degradation of triazine-containing azo dyes in aqueous TiO2 suspensions [J]. Applied Catalysis B, 2003, 42: 47-55.
[3] YU Y, YU J C, YU J, KWOK Y C, DING L, GE W, CHE Y, ZHAO J, WONG P K. Enhancement of photocatalytic activity of mesoporous TiO2 by using carbon nanotubes [J]. Applied Catalysis A, 2005, 289: 186-196.
[4] YU Y, YU J C, CHAN C Y, CHE Y, ZHAO J, DING L, GE W, WONG P K. Enhancement of adsorption and photocatalytic activity of TiO2 by using carbon nanotubes for the treatment of azo dye [J]. Applied Catalysis B, 2005, 61: 1-11.
[5] SAKTHIVEL S, NEPPOLIAN B, SHANKAR M V, ARABINDOO B, PALANICHAMY M, MOORAGESAN V. Solar photocatalytic degradation of azo dye: Comparison of photocatalytic efficiency of ZnO and TiO2 [J]. Solar Energy Materials & Solar Cells, 2003, 77: 65-82.
[6] KHODJA A A, SEHILI T, PILICHOWSKI F, BOULE P. Photocatalytic degradation of 2-phenylphenol on TiO2 and ZnO in aqueous suspensions [J]. Journal of Photochemstry and Photobiology A, 2001, 141: 231-239.
[7] GU C D, CHENG C, HUANG H Y, WONG T L, WANG N, ZHANG T Y. Growth and photocatalytic activity of dendrite-like ZnO@Ag heterostructure nanocrystals [J]. Crystal Growth & Design, 2009, 9: 3278-3285.
[8] YANG D, PARK S E, LEE J K, LEE S W. Sonochemical deposition of nanosized Au on titanium oxides with different surface coverage and their photocatalytic activity [J]. Journal of Crystal Growth, 2009, 311: 508-511.
[9] ZHENG L R, ZHENG Y H, CHEN C Q, ZHAN Y Y, LIN X Y, ZHENG Q, WEI K M, ZHU J F. Network structured SnO2/ZnO heterojunction nanocatalyst with high photo catalytic activity [J]. Inorganic Chemistry, 2009, 48: 1819-1825.
[10] WANG Z Y, HUANG B B, DAI Y, QIN X Y, ZHANG X Y, WANG P, LIU H X, YU J X. Highly photocatalytic ZnO/In2O3 heteronanostructures synthesized by a coprecipitation method [J]. Journal of Physical Chemistry C, 2009, 113: 4612-4617.
[11] MA T Y, YUAN Z Y, CAO J L. Hydrangea-like meso-/macroporous ZnO-CeO2 binary oxide materials: Synthesis, photocatalysis and CO oxidation [J]. European Journal of Inorganic Chemistry, 2010, 716-724.
[12] WU Y, TAMAKI T, VOLOTINEN T, BELOVA L, RAO K V. Enhanced photoresponse of inkjet-printed ZnO thin ?lms capped with CdS nanoparticles [J]. The Journal of Physical Chemistry Letters, 2010, 1: 89-92.
[13] XU F, VOLKOV V, ZHU Y, BAI H Y, REA A, VALAPPIL N V, SU W, GAO X Y, KUSKOVSKY I L, MATSUI H. Long electron-hole separation of ZnO-CdS core-shell quantum dots [J]. Journal of Physical Chemistry C, 2009, 113: 19419-19423.
[14] ROLISON D R. Catalytic nanoarchitectures-the importance of nothing and the unimportance of periodicity [J]. Science, 2003, 299: 1698-1701.
[15] ARAI T, YANAGIDA M, KONISHI Y, IWASAKI Y, SUGIHARA H, SAYAMA K. Efficient complete oxidation of acetaldehyde into CO2 over CuBi2O4/WO3 composite photocatalyst under visible and UV light irradiation [J]. Journal of Physical Chemistry C, 2007, 111: 7574-7577.
[16] LI B X, WANG Y F. Facile synthesis and photocatalytic activity of ZnO-CuO nanocomposite [J]. Superlattices and Microstructures, 2010, 47: 615-623.
[17] GORAI S, GANGULI D, CHAUDHURI S. Synthesis of 1D Cu2S with tailored morphology via single and mixed ionic surfactant templates [J]. Materials Chemistry and Physics, 2004, 88: 383-387.
[18] SAVELLI M, BOUGNOT T J. Problems of the Cu2S/CdS cell [C]. New York: Springer-Verlag, 1979: 213-256.
[19] DACHRAOUI M, VEDEL J. Improvement of cuprous sulphide stoichiometry by electrochemical and chemical methods [J]. Solar Cells, 1987, 22: 187-194.
[20] PINTILIE I, PENTIA E, PINTILIE L, PETRE D, CONSTANTIN C, BOTILA T. Growth and characterization of PhS deposited on ferroelectric ceramics [J]. Journal of Applied Physics, 1995, 78: 1713-1718.
[21] INDREA E, BARBU A. Indirect photon interaction in PbS photodetectors [J]. Applied Surface Science, 1996, 106: 498-501.
[22] POP I, NASCU C, IONESCU V, INDREA E, BRATU I. Structural and optical properties of PbS thin films obtained by chemical deposition [J]. Thin Solid Films, 1997, 307: 240-244.
[23] PENG M, MA L L, ZHANG Y G, TAN M, WANG J B, YU Y. Controllable synthesis of self-assembled Cu2S nanostructures through a template-free polyol process for the degradation of organic pollutant under visible light [J]. Materials Research Bulletin, 2009, 44: 1834-1841.
[24] XU Y, SCHOONEN M A A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals [J]. American Mineralogist, 2000, 85: 543-556.
[25] YIN Y, ALIVISATOS A P. Colloidal nanocrystal synthesis and the organic-inorganic interface [J]. Nature, 2005, 437: 664-669.
[26] LI Y Y, LIUJ P, HUANG X T, LI G Y. Hydrothermal synthesis of Bi2O6 uniform hierarchical microsheres [J]. Crystal Growth & Design, 2007, 7: 1351-1355.
[27] XU W, WANG Y, BAI X, DONG B, SONG H W. Controllable and synthesis and size-dependent luminescent properties of YVO4: Eu3+ nanosheres and mocrospheres [J]. Journal of Physical Chemistry C, 2010, 114: 14018-14024.
[28] SUN Y G, GATES B, MAYERS B, XIA Y N. Crystalline silver nanowires by soft solution processing [J]. Nano Letters, 2002, 2: 165-168.
[29] FAN Xi-mei, ZHOU Zuo-wan, WANG Jie, TIAN Ke. Morphology and optical properties of tetrapod-like zinc oxide whiskers synthesized via equilibrium gas expanding method [J]. Transactions of Nonferrous Metals Society of China, 2011, 21: 2056-2060.
[30] RODRIGUEN J A, JIRSAK T, CHATURVEDI S, KUHN M. Reaction of SO2 with ZnO  -O and ZnO powders: Photoemission and XANES studies on the formation of SO3 and SO4 [J]. Surface Science, 1999, 442: 400-412.
-O and ZnO powders: Photoemission and XANES studies on the formation of SO3 and SO4 [J]. Surface Science, 1999, 442: 400-412.
[31] WAGNER C D, RIGGS W W, DAVIS L E, MOULDER J F, MUILENBERG G E. Handbook of X-ray photoelectron spectroscopy [M]. Eden Prairie: Perkin-Elmer, 1979.
[32] CHEN P, GU L, CAO X B. From single ZnO multipods to hetero structured ZnO/ZnS, ZnO/ZnSe, ZnO/Bi2S3 and ZnO/Cu2S multipods: controlled synthesis and tunable optical and photoelectronchemical properties [J]. Crystal Engineering Communications, 2010, 12: 3950-3958.
[33] KAUSHIK A, KUMAR J, TIWARI M K, KHAN R, MALHOTRA B D, GUPTA V, SINGH S P. Fabrication and characterization of polyaniline-ZnO hybrid nanocomposite thin film [J]. Journal of Nanoscience and Nanotechnology, 2008, 8: 1757-1761.
[34] TIAN C G, KANG Z H, WANG E, GUO L, WANG C L, XU L, HU C G. Synthesis of dodecanethiolate-protected Cu2S nanoparticles in a two-phase system [J]. Materials Letters, 2005, 59: 1156-1160.
[35] LI X B, WANG L L, LU X H. Preparation of silver-modified TiO2 via microwave-assisted method and its photocatalytic activity for toluence degradation [J]. Journal of Hazard Materials, 2010, 177: 639-647.
[36] CHEN T W, ZHEG Y H, LIN J M, CHEN G N. Study on the photocatalytic degradation of methyl orange in water using Ag/ZnO as catalyst by liquid chromatography electronspray ionization ion-trap mass spectrometry [J]. Journal of American Society for Mass Spectrometry, 2008, 19: 997-1003.
[37] TAN T, LI Y, LIU Y, WANG B, SONG X M. Two-step preparation of Ag/tatrapod-like ZnO with photocatalytic activity by thermal evaporation and sputtering [J]. Materials Chemistry and Physics, 2008, 111: 305-308.
[38] YU J G, XIONG J F, CHENG B, LIU S G. Fabrication and characterization of Ag-TiO2 multiphase nanocomposite thin films with enhanced photocatalytic activity [J]. Applied Catalysis B, 2005, 60: 211-221.
[39] TENNAKONE K, BANDARA J. Photocatalytic activity of dye-sensitized tin(IV) oxide nanocrystalline particles attached to zinc oxide particles: Long distance electron transfer via ballistic transport of electrons across nanocrystallites [J]. Applied Catalysis A, 2001, 208: 335-341.
[40] LINSEBIGLER A L, LU G Q, YATES JR J T. Photocatalysis on TiO2 surfaces: Principles, mechanism and selected results [J]. Chemical Reviews, 1995, 95: 735-758.
[41] QI L F, YU J G, JARONIEC M. Preparation and enhanced visible-light photocatalytic H2-production activity of CdS-sensitized Pt/TiO2 nanosheets with exposed (001) facets [J]. Physical Chemistry Chemical Physics, 2011, 13: 8915-8923.
[42] SERPONE N, MARUTHAMUTHU P, PICHAT P, PELIZZETTI E, HIDAKA H. Exploiting the interparticle electron transfer process in the photocatalysed oxidation of phenol, 2-chlorophenol and hole pentachlorophenol: Chemical evidence for electron and hole transfer between coupled semiconductors [J]. Journal of Photochemistry and Photobiology A, 1995, 85: 247-255.
[43] WEI S Q, CHEN Y Y, MA Y Y, SHAO Z G. Fabrication of CuO/ZnO composite films with cathodic co-electrodeposition and their photocatalytic performance [J]. Journal of Molecular Catalysis A, 2010, 331: 112-116.
[44] WANG J, FAN X M, WU D Z, DAI J, LIU H, LIU H R, ZHOU Z W. Fabrication of CuO/T-ZnOw nanocomposites using photo-deposition and their photocatalytic property [J]. Applied Surface Science, 2011, 258: 1797-1805.
[45] ZHANG J, YU J G, ZHANG Y M, LI Q, GONG J R. Visible light photocatalytic H2-production activity of CuS/ZnS porous nanosheets based on photoinduced interfacial charge teansfer [J]. Nano Letters, 2011, 11: 4774-4779.
[46] YU J G, RAN J R. Facile preparation and enhanced photocatalytic H2-production activity of Cu(OH)2 cluster modified TiO2 [J]. Energy Environmental Science, 2011, 4: 1364-1371.
[47] RAJESHWAR K, OSUGI M E, CHANMANEE W, CHENTHAMARAKSHAN C R, ZANONI M V B, KAJITVICHYANUKUl P, KRISHNAN-AYER R. Heterogeneous photocatalytic treatment of organic dyes in air and aqueous media [J]. Journal of Photochemistry and Photobiology C, 2008, 9: 171-192.
����ͭ/����״����п�������ʽ��
��Ԫ�����Ʊ����������
����ǣ���ϣ÷���� �𣬴� �ѣ�������
���Ͻ�ͨ��ѧ ���Ͽ�ѧ�빤��ѧԺ�������Ƚ������������ص�ʵ���ң��ɶ� 610031
ժ Ҫ���Ծ���ϩ������ͪ(PVP)Ϊ������Լ���ͨ����Ԫ�����Ʊ�Cu2S/T-ZnOw���ʽḴ�ϲ��ϣ�����XRD��FESEM��EDS��XPS��FTIR���Է�������Ʒ���б�����ͨ���ⶨ������Һ�Ĺ⽵������������Ʒ�Ĺ�����ԡ����������������������£�Cu2S/T-ZnOw�����ϲ��ϵĹ���������ڴ�����п����ġ���PVP��Ũ��Ϊ3.0 g/Lʱ����Ʒ�Ĺ��������ߣ������������120 min���ȵĽ�����Ϊ97%������4�����ڵĹ��ʵ���Cu2S/T-ZnOw�����Ĺ�����Բ�û�������½���˵������Ʒ��������Ĺ��ȶ��ԡ����⣬������Cu2S/T-ZnOw�����ϲ��ϵĹ��������
�ؼ��ʣ�Cu2S/T-ZnOw���ʽ������ܣ�����ͭ������
(Edited by LI Xiang-qun)
Foundation item: Project (2009AA03Z427) supported by the High-tech Research and Development Program of China; Project (2006z02-006-3) supported by the Science Foundation of Sichuan Province, China
Corresponding author: FAN Xi-mei; Tel: +86-28-87602714; Fax: +86-28-87600454; E-mail: fanximei@126.com
DOI: 10.1016/S1003-6326(11)61365-4