Trans. Nonferrous Met. Soc. China 23(2013) 1421-1427
Enhanced thermal and electrical properties of poly (D,L-lactide)/ multi-walled carbon nanotubes composites by in-situ polymerization
Qing-hua LI1, Qin-hua ZHOU2, Dan DENG3, Qiao-zhen YU3, Li GU3, Ke-da GONG3, Ke-hang XU3
1. School of Materials Science and Engineering, Changzhou University, Changzhou 213164, China;
2. College of Medicine, Jiaxing University, Jiaxing 314001, China;
3. College of Material and Textile Engineering, Jiaxing University, Jiaxing 314001, China
Received 27 March 2012; accepted 11 January 2013
Abstract: Biodegradable poly (D,L-lactide) (PLA)/carboxyl-functionalized multi-walled carbon nanotubes (c-MWCNTs) composites were achieved via in-situ polymerization. These as-prepared composite materials were characterized with FT-IR, XRD, TG, DSC, SEM, and high insulation resistance meter. The results demonstrate that the multi-walled carbon nanotube was carboxyl functionalized, which improved the collection between c-MWCNTs and PLA, and further realized the graft copolymerization of c-MWCNTs and PLA. There is a higher glass transition temperature and a lower pyrolysis temperature of PLA/c-MWCNTs composites than pure PLA. The c-MWCNTs gave a better dispersion than unmodified MWCNTs in the PLA matrix, and an even coating of PLA on the surface of c-MWCNTs was obtained, which increased the interfacial interaction. High insulation resistance analysis showed that the addition of c-MWCNTs increased the electric conductivity, and c-MWCNTs performed against the large dielectric coefficient and electrostatic state of PLA. These results demonstrated that c-MWCNTs modified PLA composites were beneficial for potential application in the development of heat-resisting and conductivity plastic engineering.
Key words: in-situ polymerization; multi-walled carbon nanotubes; polylactide; thermal properties; electrical conductivity
1 Introduction
Polylactide(PLA) is a synthetic aliphatic polyester derived from renewable resources, such as corn, sugar beet and cassava [1,2], which can ultimately be degraded under composting conditions [3]. PLA assumes unique properties, such as biodegradability, biocompatibility and good mechanical properties [4,5], and has been used in biomedical and food packaging applications [6,7]. However, there are some drawbacks for PLA, such as brittleness, low crystallization rate, low impact strength, low glass transition temperature and poor electric conductivity [8], which has limited the applications of PLA as common plastic materials.
Many studies have been attempted to improve the properties of PLA [9], particularly the thermal stability and electric conductivity properties [10], by blending with other heat-resistant materials, such as montmorillonite [11], zinc oxide nano-crystal whisker, calcium carbonate, nano silicon dioxide [12] and carbon nano-tubes[13]. Carbon nano-tubes are an interesting molecular form of carbon in the fullerene family, discovered by IIJIMA [14].
Carbon nanotubes have extremely low electrical resistance which is derived from their 1D character and the peculiar electronic structure of graphite. Carbon nanotubes (CNTs) are the stiffest known fiber, with a measured elastic modulus of 1.4 TPa [15]. CNTs have now shown a thermal conductivity at least twice that of diamond [16]. CNTs have the unique property of feeling cold to the touch, like metal, on the sides with the tube ends exposed, but similar to wood on the other sides [17]. With their unique physical properties, many investigators have endeavored to fabricate advanced CNT composite materials that exhibit one or more of these properties such as conductive filler in polymers. For example, polyimide/carbon nanotube (PI/CNT) composites with different proportions of CNT were fabricated by in-situ process [18], polylactide/carbon nanotube composites [19]. The above modification means for PLA were about L-PLA; however, no research about the improvement of heat-resistant and electric conductivity properties for D,L-PLA has been performed, which is a relatively cheaper biodegradable plastic [20,21]. In this work, an organic functional group was introduced into MWCNTs after being treated with mixed acids, to form carboxyl-functionalized carbon nanotubes (c-MWCNTs). With D,L-lactide and c-MWCNTs as monomers, and stannous octoate as initiator, in-situ polymerization was performed, and c-MWCNTs modified poly (D,L-lactide) was achieved, and the properties of PLA/c-MWCNTs composites were characterized.
2 Experimental
2.1 Materials
Main reagents included concentrated sulfuric acid (98%), nitric acid (60%), D,L-lactide (86%), zinc oxide, acetone, acetic acid, tetrahydrofuran, chloroform (all were analytically pure, Shanghai National Medicine), stannous octoate (95%, Sigma), de-ionized water (prepared in the laboratory), and multi-walled carbon nanotubes with diameter of 10-20 nm, length of 5-15 μm and specific area of 40-300 m2/g (95%, Shenzhen Nanotech Port Co., Ltd.).
2.2 Preparation of D,L-lactide
With D,L-lactic acid as material, D,L-lactide was prepared by reduced pressure distillation technique. 300 mL of D,L- lactic acid was put into a three-neck flask, the free water was removed by keeping the reaction system at the temperature of 80 °C, by reducing pressure and stirring simultaneously, then 3.06 g of La-Ti composite oxides were added into the system as catalyst to promote polycondensation of lactic acid, the temperature was kept at 140 °C for 4-5 h, and the reaction system was turning from white into light yellow. After stopping vacuum and stirring swiftly, replacing the receiving flask, the reaction system was warmed up to 200 °C, and was kept under fine vacuum, then the light yellow crude lactide was distilled from reaction flask. White and pure lactide was obtained by recrystallization using acetic ester as solvent.
2.3 Surface modification of multi-walled carbon nanotubes
10 g of multi-walled carbon nanotubes were put into round bottom flask, in which mixed acid (with volume ratio of concentrated sulfuric acid to nitric acid of 3:1) was added. After 2 h of ultrasonic dispersion, the reaction system was kept stirring at of 50 °C for 3 h, and was cooled down to room temperature, into which a large amount of de-ionized water was added to increase pH of the system. Then the system was kept stewing for several hours, and assumed lamination, and clear liquid of the upper layer was removed, the bottom was centrifuged till the upper clear liquid was neutral, and the deposition was dried in vacuum at 60 °C for 12 h.
2.4 Preparation of PLA/c-MWCNTs composite
Certain amounts of D,L-lactide and multi-walled carbon nanotubes were mixed evenly and put into a flask, then chloroform solution with stannous octoate was added as initiator. After being kept under ultrasonic vibration for 20 min, the flask was depressed at 35 °C for 2 h and sealed under vacuum, and then the flask was kept in a vacuum drying oven at 160 °C for 18 h. After being purified with tetrahydrofuran (C4H8O), filtrated by depressing to remove the residual carbon nanotubes, and deposited with de-ionized water, the dark PLA/ c-MWCNTs composite was accomplished.
2.5 Analysis and characterization
IR spectra of the original MWCNTs, as-prepared modified MWCNTs and PLA/c-MWCNTs composite were recorded by a Nicolet-IR550 infrared spectrometer. Crystal structure of MWCNTs and crystallinity of PLA/c-MWCNTs were characterized by X-ray diffractometry (Cu Kα, λ=1.54178 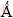 , Rigaku D/MAX- rotation). The glass transition temperature of PLA/ c-MWCNTs was recorded by differential scanning calorimetry (200PC, Netzsch, heating rate of 10 °C/min, temperature range of 30-100 °C). TGA was performed to record the heat decomposition of MWCNTs, PLA, and PLA/c-MWCNTs with a Netzsch STA-409PC apparatus under nitrogen gas at a flow rate of 25 L/min. Pattern observation of MWCNTs and PLA/c-MWCNTs was performed on a scanning electron microscope (SEM, Hitachi S-570). Surface resistivity and volume resistivity of PLA/c-MWCNTs composite were recorded on a digital high resistance meter (Shanghai Jinghong) at detection voltages of 25, 50, 100, 250, 500, and 1000 V, respectively.
, Rigaku D/MAX- rotation). The glass transition temperature of PLA/ c-MWCNTs was recorded by differential scanning calorimetry (200PC, Netzsch, heating rate of 10 °C/min, temperature range of 30-100 °C). TGA was performed to record the heat decomposition of MWCNTs, PLA, and PLA/c-MWCNTs with a Netzsch STA-409PC apparatus under nitrogen gas at a flow rate of 25 L/min. Pattern observation of MWCNTs and PLA/c-MWCNTs was performed on a scanning electron microscope (SEM, Hitachi S-570). Surface resistivity and volume resistivity of PLA/c-MWCNTs composite were recorded on a digital high resistance meter (Shanghai Jinghong) at detection voltages of 25, 50, 100, 250, 500, and 1000 V, respectively.
3 Results and discussion
Figure 1 shows the IR spectra of MWCNTs, modified MWCNTs and the prepared PLA/c-MWCNTs composite. In IR spectrum (a), the peaks at 2916.76 cm-1 and 2851.89 cm-1 originate from the stretching vibration of C―H of MWCNT. In IR spectrum (b), the peaks at 1726.64 cm-1 and 3440.38 cm-1 originate from the stretching vibration of C=O and O―H of the modified MWCNTs, respectively. In IR spectrum (c), the peaks at 3002.60 cm-1 and 2949.19 cm-1 originate from the stretching vibration of ―CH3 and C―H of PLA, respectively; the peak at 1758.79 cm-1 originates from the stretching vibration of C=O; the peaks at 1091.02 cm-1 , 1194.52 cm-1, 1269.29 cm-1 originate from the stretching vibration of C―O. All the characteristic peaks in Fig. 2 demonstrate the accomplishment of carboxylated MWCNTs, and chemical bonding of PLA and carboxylated MWCNTs.
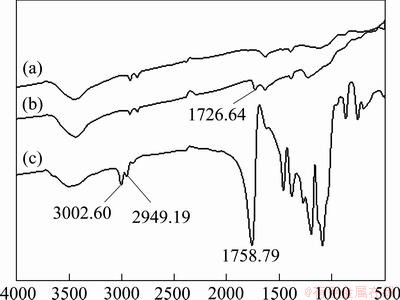
Fig. 1 IR spectra of pure MWNTs (a), modified MWNTs (b) and PLA/c-MWNTs (c)

Fig. 2 XRD patterns of pure MWNTs, modified MWNTs, and PLA/c-MWNTs
The phase and component of the as-prepared product were characterized by powder X-ray diffraction (XRD). Typical XRD patterns of MWNTs, modified MWNTs, and PLA/c-MWCNTs are shown in Fig. 2.
There is not any difference for diffraction location between XRD patterns of pure MWNTs and modified MNNTs, the diffraction peaks at 26.06°, 42.97°, and 53.63° can be readily indexed to planes (002), (100), and (004) of MWCNTs. But there are higher intensity of diffraction peaks and more obvious crystallization peaks for carboxylated MWCNTs (with 0.5% carboxylated MWCNTs) than MWCNTs, and full width at half maximum of diffraction peak at 26.06° changes from 3.41° to 3.46°, suggesting larger size of lattice dimension, better crystallinity, and more complete structure for carboxylated MWCNTs. The possible reason is that acidification of MWCNTs advances removing amorphous carbon, carbon nanoparticles and metal particles. In XRD pattern of PLA/c-MWNTs, there are not any diffraction peaks of MWCNTs, and the only one peak curve bread demonstrates an amorphous sample, suggesting the imbedding of c-MWCNTs by PLA.
Figure 3 illustrates the DSC curves for PLA and PLA/c-MWCNTs, the glass transition temperature (Tg) of PLA is 50 °C; while the glass transition temperature of PLA/c-MWCNTs with 0.5% carboxylated MWCNTs is 74 °C, suggesting that the introduction of carbon nano-tubes increases Tg of PLA.
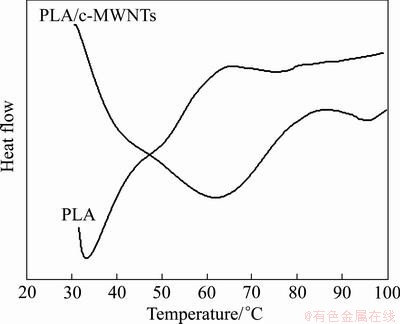
Fig. 3 DSC curves corresponding to PLA and PLA/c-MWNTs
TG curves corresponding to the pure multi-walled carbon nano-tubes, carboxylated MWCNTs, PLA/ c-MWCNTs, and PLA are shown in Fig. 4. These curves represent the mass loss as a function of the temperature. The pure MWCNTs samples degrade little throughout the heating stage (from 25 °C to about 425 °C), and assume 2.6% of mass loss, as a result of certain water inclusion. The carboxylated MWCNTs samples degrade smoothly from 25 °C to about 425 °C, and assume 20% of mass loss for the decomposition of carboxyl and a spot of water. The PLA/c-MWCNTs samples degrade in an obvious single step from 25 °C to about 425 °C, and present 98% of mass loss for the pyrolysis of graft polymer of PLA. The PLA samples degrade in a big single step from 25 °C to about 425 °C, and present 99% of mass loss for the pyrolysis of pure PLA.
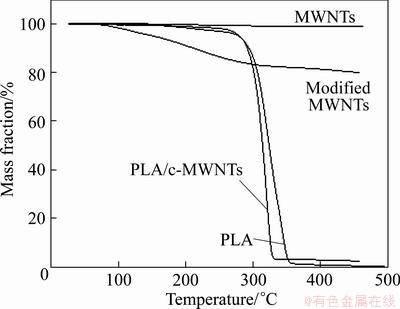
Fig. 4 TGA curves corresponding to pure MWNTs, modified MWNTs, PLA/c-MWNTs, and PLA
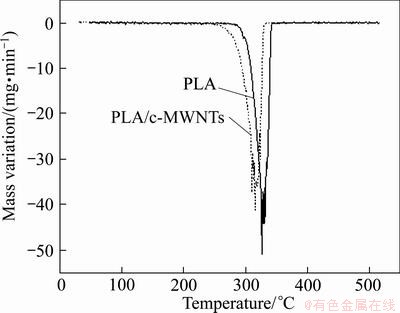
Fig. 5 DTG curves corresponding to PLA and PLA/c-MWNTs
DTG curves corresponding to the pure PLA samples and PLA/c-MWCNTs are shown in Fig. 5. A comparative study of the influence of the introduction of carboxylated MWCNTs and pure PLA shows that with the increase of amount (0-0.75%) of carboxylated MWCNTs into PLA, there is an obvious reduction for starting combustion temperature, ending combustion temperature, and the maximum rate of combustion temperature for the PLA/c-MWCNTs composite, in which the maximum rate of combustion temperature steps down from 326.13 °C (PLA) to 315.98 °C (PLA/c-MWCNTs) and the ending combustion temperature is 20 °C of decrease from PLA to PLA/c-MWCNTs. The above TG and DTG curves demonstrate the speeding up of pyrolysis of PLA as a result of addition of carboxylated MWCNTs, suggesting that excellent heat conduction of carbon nano-tubes and carboxyl of MWCNTs present large contribution to the ester bond rupture of PLA to accelerate the pyrolysis of PLA.
D,L-PLA was successfully applied to coating carboxylated MWCNTs via in-situ polymerization. SEM images of pure MWCNTs, carboxylated MWCNTs, and PLA/c-MWCNTs are shown in Fig. 6. Pure MWCNTs assume hollow cylinder with long caliber and random state, and agglomerate severely (as low magnification image in Fig. 6(a) and high magnification image in Fig. 6(b). After modification of mixed acids, carboxylated MWCNTs present as short segments with open port, and disperse well (as low magnification image in Fig. 7(c) and high magnification image in Fig. 7(d), suggesting oxidization and carboxylation of MWCNTs. The low magnification image shown in Fig. 6(e) reveals that there is an even dispersion of MWCNTs on the surface of PLA, from the image with a high magnification (Fig. 6(f)). It can be found a little bigger caliber of PLA/c-MWCNTs than pure MWCNTs and carboxylated MWCNTs, and there is not any hollow structure at the port of MWCNTs, suggesting the successfully coating of PLA on the surface of carboxylated MWCNTs via chemical bonding and achieving a strong interface interaction.
Table 1 Content of MWCNTs and volume resistivity of composite material with lactide mass of 5 g

Table 2 Content of MWCNTs and surface resistivity of composite material with lactide mass of 5 g
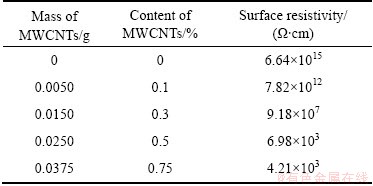

Fig. 6 SEM images of pure MWCNTs (a, b), modified MWNTs (c, d) and PLA/c-MWNTs (e, f)

Fig. 7 Correlation of volume resistivity and MWNTs content for PLA/MWCNTs (a) and of surface resistivity and MWNTs content of for PLA/c-MWCNTs (b)
Recordation of digital high resistance meter can be conversed to electrical conductivity to embody electrical feature of material. The volume resistivity of PLA/c-MWCNTs composites and the content of MWNTs are recorded in Table 1 and Fig. 7(a). Compared with pure PLA, the introduction of MWNTs brings an obvious nonlinear decrease of volume resistivity with the increase of MWNTs content in PLA/c-MWCNTs composites, and a sudden decrease from 2.75×1015 Ω・cm to 4.45×102 Ω・cm, implying the contribution of MWNTs to the electrical conductivity of the composite. The optimal MWNTs content is about 0.5%. The surface resistivity of PLA/c-MWCNTs composite and the content of MWNTs are recorded in Table 2 and Fig. 7(b), and there is a similar tendency of electric resistance for the surface resistivity of composite materials and is a sharp decrease of surface resistivity from 6.64×1015 to 6.98×103 Ω・cm. The introduction of c-MWNTs improves the electrical conductivity of PLA and broadens the application field of PLA.
4 Conclusions
1) Carboxyl was introduced to MWCNTs treated by mixed acids, and compatibility of MWCNTs and polymer was improved.
2) PLA/c-MWCNTs composites were achieved via in-situ polymerization. There is an interaction force between PLA and MWCNTs, and even certain PLA segments are grafted on MWCNTs.
3) Introduction of a small quantity of MWCNTs brings PLA/c-MWCNTs composite a glass transition temperature increase and thermal decomposition temperature decrease as a result of carboxyl introduction to PLA.
4) Scanning electron microscopic analysis demonstrates that carboxylated MWCNTs present an even dispersion in PLA matrix, and PLA is coated on the surface of carboxylated MWCNTs.
5) The introduction of MWCNTs improves the electrical conductivity of PLA (with a optimal content of 0.5% for MWCNTs), and will resolve problems of high dielectric constant and electrostatic problem for PLA materials.
References
[1] KULKARNI R K, MOORE E E G, HEGYELI A F, LEONARD F. Biodegradable poly (lactic acid) polymers [J]. Journal of Biomedical Materials Research, 1971, 5(3): 169-181.
[2] NDRZEJ K B, ADAM J, DIETRICH S. Mechanical properties of PLA composites with manmade cellulose and abaca fibers [J]. Composites: Part A, 2009, 40(4): 404-412.
[3] CHENG Yan-ling, DENG Shao-bo, CHEN P, RUAN R. Polylactic acid (PLA) synthesis and modifications: A review [J]. Frontiers of Chemistry in China, 2009, 4(3): 259-264.
[4] KIM S Y, SHIN K S, LEE S H, KIM K W, YOUN J R. Unique crystallization behavior of multi-walled carbon nanotube filled poly (lactic acid) [J]. Fibers and Polymers, 2010, 11(7): 1018-1023.
[5] CHRISSAFIS K, PARASKEVOPOULOS K M, JANNAKOUDAKIS A, BESLIKAS T, BIKIARIS D. Oxidized multiwalled carbon nanotubes as effective reinforcement and thermal stability agents of poly (lactic acid) ligaments [J]. Journal of Applied Polymer Science, 2010, 118(5): 2712-2721.
[6] WU C S, LIAO H T. Study on the preparation and characterization of biodegradable polylactide/multi-walled carbon nanotubes nanocomposites [J]. Polymer, 2007, 48: 4449-4458.
[7] WU De-feng, ZHANG Yi-sheng, ZHANG Ming, YU Wei. Selective localization of multi-walled carbon nanotube in PLA/PCL blend [J]. Biomacromolecules, 2009, 10(2): 417-424.
[8] KO S W, HONG M K, PARK B J, GUPTA  R K, CHOI H J, BHATTACHARYA S N. Morphological and rheological characterization of multi-walled carbon nanotube/PLA/PBAT blend nanocomposites [J]. Polymer Bulletin, 2009, 63: 125-134.
R K, CHOI H J, BHATTACHARYA S N. Morphological and rheological characterization of multi-walled carbon nanotube/PLA/PBAT blend nanocomposites [J]. Polymer Bulletin, 2009, 63: 125-134.
[9] ZOU J, ZHANG Y C, HUANG J N, WU H Y, QIU Y P. Preparation and properties of PP/PLA/multiwall carbon nnaotube composites filaments obtained by melt compounding [J]. Materials Science Forum, 2009, 620-622: 465-468.
[10] PANG Ya, ZENG Guang-ming, TANG Lin, ZHANG Yi, LI Zhen, CHEN Li-juan. Laccase biosensor using magnetic multiwalled carbon nanotubes and chitosan/silica hybrid membrane modified magnetic carbon paste electrode [J]. Journal of Central South University of Technology, 2011, 42(6): 1849-1856. (in Chinese)
[11] LAHIRI D, ROUZAUD F, NAMIN S, KESHRI A K, VALDE’S J J, KOS L, TSOUKIAS N, AGARWAL A. Carbon nanotube reinforced polylactide-caprolactone copolymer: Mechanical strengthening and interaction with human osteoblasts in vitro [J]. Applied Materials Interfaces, 2009, 1(11): 2470-2476.
[12] LEE S, KIM C H, PARK J K. Improvement of processability of clay/polylactide nanocomposites by a combinational method: In situ polymerization of L-lactide and melt compounding of polylactide [J]. Journal of Applied Polymer Science, 2006, 101: 1664-1669.
[13] YAN Xue-bin, GU Yong-hong, HUANG Dong, GAN Li, WU Li-xiang, HUANG Li-hua, CHEN Zhe-dong, HUANG Su-ping, ZHOU Ke-chao. Binding tendency with oligonucleotides and cell toxicity of cetyltrimethyl ammonium bromide-coated single-walled carbon nanotubes [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(5): 1085-1091.
[14] IIJIMA S. Helical microtubules of graphitic carbon [J]. Nature, 1991, 354: 56-58.
[15] YAKOBSON B I, BRABEC C J, BERNHOLC J. Nanomechanics of carbon tubes: Instabilities beyond linear response [J]. Physical Review Letters, 1996, 76: 2511-2514.
[16] HONE J. Phonons and thermal properties of carbon nanotubes [J]. Applied Physics, 2001, 80: 273-286.
[17] KHARE R, BOSE S. Carbon nanotube based composites―A review [J]. Journal of Minerals & Materials Characterization & Engineering, 2005, 4(1): 31-46.
[18] CAI Hui, YAN Feng-yuan, XUE Qun-ji. Investigation of tribological properties of polyimide/carbon nanotube nanocomposites [J]. Materials Science and Engineering A, 2004, 364: 94-100.
[19] WU De-feng, WU Liang, ZHOU Wei-dong, ZHANG Ming, YANG Tao. Crystallization and biodegradation of polylactide/ carbon nanotube composites [J]. Polymer Engineering & Science, 2010, 50: 1721-1733.
[20] SHIH Y F, WANG Y P, HSIEH C F. Preparation and properties of PLA/long alkyl chain modified multi-walled carbon nanotubes nanocomposites [J]. Journal of Polymer Engineering, 2012, 31(1): 13-19.
[21] KUAN C F, KUAN H C, MA C C, CHEN C H. Mechanical and electrical properties of multi-wall carbonnanotube/poly (lactic acid) composites [J]. Journal of Physics and Chemistry of Solids, 2008, 69(5-6): 1395-1398.
多壁碳纳米管-聚乳酸复合材料的原位聚合制备及热性能和导电性能
李清华1, 周勤华2, 邓 丹3, 俞巧珍3,谷 俐3, 龚科达3,徐科航3
1. 常州大学 材料科学与工程学院,常州 213164;
2. 嘉兴学院 医学院,嘉兴 314001;
3. 嘉兴学院 材料与纺织工程学院,嘉兴 314001
摘 要:采用原位聚合的方法,制备了多壁碳纳米管-聚乳酸复合材料。通过红外光谱、X射线衍射仪、差示扫描量热分析、热分析、扫描电镜和高阻计等对材料的结构性能进行分析。结果表明:多壁碳纳米管经混合酸处理,表面引入了有机基团,从而具有有机活性,与聚乳酸分子链之间的作用力加强,并可能存在部分聚乳酸分子链接枝于碳纳米管上;引入碳纳米管,使聚乳酸的玻璃化转变温度升高,但热降解温度降低。扫描电镜分析发现,多壁碳纳米管均匀分散于基体中,聚乳酸基体包覆在碳纳米管表面,界面作用增强。高阻计分析结果表明,随着碳纳米管含量的增加,材料的导电性能得到增强,从而克服了PLA材料本身存在的介电常数大、易带静电等不足。
关键词:原位聚合;多壁碳纳米管;聚乳酸;热性能;导电性能
(Edited by Hua YANG)
Foundation item: Projects (21107032, 51073072) supported by the National Natural Science Foundation of China; Projects (Y406469, Y4110555, Y4100745) supported by Natural Science Foundation of Zhejiang Province, China; Projects (2011AY1048-5, 2011AY1030) supported by the Science Foundation of Jiaxing Science and Technology Bureau, China
Corresponding author: Li GU; Tel: +86-573-83646195; E-mail: guli@mail.zjxu.edu.cn
DOI: 10.1016/S1003-6326(13)62612-6