
Synthesis and crystal structure and nonlinear optical properties of polymeric W (Mo)-Cu-S cluster
ZHOU Jian-liang(周建良), CHEN Qi-yuan(陈启元), GU Ying-ying(古映莹), ZHONG Shi-an(钟世安)
School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The polymeric chalcogenide [W2O2S6Cu4(NCMe)4]n (compound 1) was synthesized by the self-assembly reaction of (NH4)2(WOS3) with CuBr in MeCN in the presence of tricyclohexylphosphane (PCy3) under a purified nitrogen atmosphere using standard Schlenk techniques. It gives rise to a novel 1D polymeric compound 1 with solvent MeCN coordinated to the copper atom. This situation is unprecedented in the W(Mo)/Cu/S system. The crystals were characterized by elemental analysis, IR and single-crystal X-ray crystallography. The configuration of the polymeric compound can be viewed as a helical chain which is propagated along the crystallographic c axis. The excited state absorption and refraction of compound 1 in CH3CN solution were studied by using the Z-scan technique with laser pulses of 40 ps pulse-width at a wavelength of 532 nm. The polymeric compound possesses an optical self-focusing performance. The positive nonlinear refraction is attributed to population transitions between singlet states. Compound 1 displays a strong excited-state absorption.
Key words: W(Mo)-Cu-S cluster; chalcogenide; self-assembly; Schlenk technique; Z-scan; excited-state
1 Introduction
Coordination chemistry of the thiometalates [MOnS4-n]2- (M=Mo, W; n=0-2) and their related compounds has been well-documented owing to their relevance to certain biological process, catalysis reactions. Recently, the heterometallic polymeric clusters have received much attention due to their fascinating properties such as catalysis, conductivity and strong nonlinear optical characteristics[1-7]. Mo(W)/Cu/S polymeric clusters are scarce and only a few structures have been reported. For example, the linear one-dimensional chains in {[WOS3Cu3(CN)(py)4]n (M = Mo, W), [8,9] the two-dimensional framework in {[NEt4][Mo2O2S6Cu6I3(4,4′-bipy)5] MeOH?H2O}n [10]. Herein, the synthesis and structure of a novel 1D polymeric cluster [W2O2S6Cu4(NCMe)4]n, with a novel zigzag chain configuration along the c axis and solvent as a ligand, are reported for the first time. Additionally, the excited state nonlinear refraction and absorption of the compound 1 were investigated by using the Z-scan technique with picosecond pulses a steady-state model of singlet excited state nonlinear refraction was proposed, which agrees well with experimental results of the excited-states refraction and absorption.
2 Experimental
2.1 General
The reagents [NH4]2[WOS3] was prepared accord-
ing to the results reported by ZHANG et al[11]. Other chemicals were of AR grade and used without further purification. Infrared spectra were recorded with a Fourier transform Nicolet FT-170SX spectrophotome-
ter. The electronic spectrum was taken on a Shimadzu UV-240 spectrophotometer and elemental analyses were performed on a PE-240C elemental analyzer.
2.2 Preparation of compound 1
The synthesis was performed under a purified nitrogen atmosphere using standard Schlenk techniques. To a mixture of [NH4]2[WOS3] (332 mg, 1 mmol) and CuBr (287 mg, 2 mmol) in MeCN (40 mL), PCy3 (560 mg, 2 mmol) in MeCN (10 ml) was added. The resulting mixture was stirred for 24 h and then filtered. The red filtrate was layered with hexane (20 mL) and allowed to stand at room temperature for several weeks. Deep-red crystals were obtained, yield: 65%. Characteristic infrared absorptions were found at 420 cm-1 and 440 cm-1 for n(W―S), 910 cm-1 for n(W―O), and 2100 cm-1 for n(C??N). Anal: Calcd. for C4H6Cu2N2OS3W∶C, 9.51; H, 1.20; N, 5.54; Found: C, 9.21; H, 1.23; N, 5.32%.
2.3 Crystal structure and determination and refinement
The structure of the compound was determined by a single crystal X-ray diffraction study. A suitable crystal was mounted on a glass fiber. Cell dimension measurements and data collection were performed on a Siemens Smart CCD diffractometer with graphite-mono-
chromatized MoKa radiation at room temperature. The structure was solved by direct methods and refined by full-matrix least-square methods on F2 using SHELXTL software. Intensity data for the crystal was obtained in the range 4.56?<2θ<50? using ω scan technique. All H atoms were geometrically fixed and allowed to ride on their attached atoms. Details of data collection and structure refinement are summarized in Table 1. Selected bond lengths and angles are listed in Table 2. Atomic coordinates, thermal parameters have been deposited at the Cambridge Crystallographic Data Centre (CCDC: 223470).
Table 1 Crystal data and standard refinement details for compound 1

2.4 Nonlinear optical measurements
The nonlinear optical properties of the metal cluster solution were measured by the Z-scan technique at 532 nm. A continuum ns/ps Nd∶YAG laser system with a pulse width of 40 ps was used. The metal complex 1 in CH3CN solution was filled into a 2 mm-thick cuvette, which had a linear transmittance of 92%. The input and the output energies of beams were measured with energy meters (Laser Precision Corporation Rjp-735). The focal length of the lens was 90 mm. The laser beam waist was about 36 mm at focus point, and the corresponding Rayleigh length was thus much larger than the thickness of sample. The aperture diameter was 2 mm and this corresponds to a far filed transmittance of 9.8 %. The experimental data were collected utilizing a single shot at a rate of 1 pulse per minute to avoid thermal effects. The input pulse energies were 700 mJ for picosecond pulses. 3 Results and discussion 3.1 Synthesis of compound 1
The reaction of [NH4]2[WOS3] and CuBr in the presence of PCy3 (molar ratio of 1∶2∶2) in MeCN solution led to deep-red crystals of compound 1 in 65% yield, after allowing the orange-red filtrate to stand in anaerobically for several days. The ligand PCy3 was not implicated, but the solvent MeCN molecules behaved additionally as a ligand in compound 1. This differs from our former result, when an analogous ligand, PPh3, was used in place of PCy3. Reaction of [WOS3]2- with CuBr and PPh3 produced a single cubic-like compound [WOS3Cu3(PPh3)3Br] [13].
3.2 Single-crystal X-ray diffraction analysis
Compound 1 crystallizes in the monoclinic system with space group P2(1)/c. An ORTEP drawing is shown in Fig.1. The configuration of the polymeric compound can be viewed as a helical chain which is propagated along the crystallographic c axis. For clarity, a simplified diagram is shown in Fig.2. It will be noted that the chain turns at each W atom. Each approximately tetrahedral W atom is coordinated by one terminal O and three m3-S atoms with angles subtended at W of 108.61(10)?- 110.8(3)?. However, the coordination tetrahedron around the Cu(1) atom, formed by two m3-S and two N atoms from MeCN, is much more distorted, the angles of Cu ranging from 105.14(11)? to 116.0(3)?. The average W―Cu―W and Cu―W―Cu angles are 177.05(6)? and 85.80(2)?, respectively. The W==O band length of 0.171 8(8) nm is typical of a double bond. The bond W―m3-S lengths are 0.224 4(2), 0.224 5(3), and 0.223 8(3) nm.
There are two types of copper atoms in the W2O2S6Cu4 core. The Cu(1) atom is tetrahedrally coord-
Table 2 Selected bond lengths and angles of compound

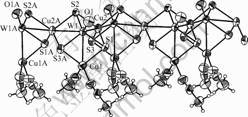
Fig.1 ORTEP drawing of portion of anion of compound 1 with atomic labeling (30 % displacement ellipsoids)
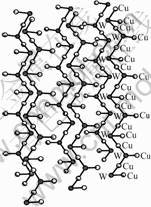
Fig.2 Simplified diagram of anionic helical chain of compound 1 for clarify(Sulfur atoms are ommited)
inated by two m3-S atoms and two nitrogen atoms of the MeCN ligands, resulting in a (m3-S)2Cu(NCMe)2 arrangement, while the Cu(2) atom is tetrahedrally coordinated by the four m3-S atoms resulting in a (m3-S)4Cu arrangement. Owing to the different coordination modes of the copper atoms, the W-Cu lengths are slightly different, for example, the W1―Cu1 bond length [0.269 99(16) nm] is shorter than those for the W1―Cu2 bonds [av. 0.274 89(15) nm]; on the other hand, the Cu-S distances also reflect the different coordination modes of the copper atoms, the Cu(2)―S bonds [av. 0.232 64(3) nm] are longer than those for Cu(1) [av. 0.229 2(3) nm].
3.3 Nonlinear optical properties Compound 1 shows two absorption peaks with the first maximum located at 367 nm. The polymer has relatively low linear absorption in the visible and near-IR regions. Indeed, the solution is nearly transparent in the visible and near infrared range, which qualifies the cluster as a potential optical limiting material.
The open aperture Z-scan results are shown in Fig.3. The nonlinear transmittances of the sample are dependent only on the incident intensity for nanosecond and picosecond laser pulses, which indicates that the metal cluster possesses excited state absorption and not two-photon absorption. The nonlinear absorption of about 90% at the focus shows that the metal cluster possesses strong excited state absorption and a large optical limiting response. The normalized closed aperture Z-scan curve (Fig.4) has a pair of sharp peaks and valley and the occurrence of the peak indicates that the cluster possesses positive nonlinear refraction or is self-focusing. Both the peak and the valley are asymmetric, and the width of valley is widened. The asymmetry of peak and valley of the normalized Z-scan curve is caused by the strong excited state absorption. The excited state optical nonlinearity can be studied using excited-state theory. The NLO properties of 1 were determined by using Z-scan techniques. A Z-scan measurement of 1 is shown in Figs.3 and 4.

Fig.3 Open-aperture Z-scan results of the compound 1

Fig.4 Z-scan results of compound 1 observed under closed-aperture configuration divided by those obtained under open-aperture configuration.
The optical propagation equation for the pulsed light intensity is given by [14]
 (1)
(1)
where  is the non-linear absorption coefficient of the sample, which can be expressed as the function of the incident pulsed light intensity (I) from [14]
is the non-linear absorption coefficient of the sample, which can be expressed as the function of the incident pulsed light intensity (I) from [14]
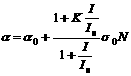 (2)
(2)
where α0 is the linear absorption coefficient of the sample; σ0 is the absorption cross-section of the ground-state molecular solution; N is the concentration of the sample solution, Is is the saturation intensity, with τe0 being the lifetime of the excited-state, is the ratio of the excited-state absorption cross-section to the ground-state cross-section.
The nonlinear absorption components were evaluated by the Z-scan method under an open aperture configuration and the NLO absorptive experimental data obtained under the conditions used in this study can be well described by Eqns.(3) and (4) [14], which are used to describe a third-order NLO absorptive process:
 (3)
(3)
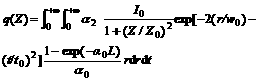 (4)
(4)
where T is a function of the sample’s z-position (against the focal point z=0); z is the distance of the sample from the focal point; L is the sample thickness; I0 is the peak irradiation intensity at focus; , where ω0 is the spot radius of the laser pulse at focus and λ is the laser wavelength; γ is the radial coordinate; t is the time; t0 is the pulse width.
, where ω0 is the spot radius of the laser pulse at focus and λ is the laser wavelength; γ is the radial coordinate; t is the time; t0 is the pulse width.
The nonlinear refractive properties of compound 1 were assessed by dividing the normalized Z-scan data obtained under the closed aperture configuration by the normalized Z-scan data obtained under the open aperture configuration. The valley/peak patterns of the corrected transmittance curves show characteristic self-focusing behaviors of the propagating light in the sample. An effective third-order nonlinear refractive index n2 of compounds 1 can be derived from the difference between normalized transmittance values at valley and peak position (ΔTv-p ) by [15]
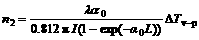 (5)
(5)
where I is the peak irradiation intensity at focus and λ is the wavelength of the laser. The filled boxes in Figs.3 and 4 are the optical limiting experimental data measured with linear transmittance. It can be seen that the ratio of refraction volumes of the singlet excited-state to the ground-state is greater than compound 1 for picosecond pulses, so it is self-focusing. The ratio of the singlet excited-state absorption cross-section to the ground-state is very large, so the compound possesses a strong excited state absorption, which induced asymmetry of peak and valley.
4 Conclusions
A novel 1D polymeric compound [W2O2S6Cu4-(NCMe)4]n with solvent MeCN coordinated to the copper atom was obtained through the self-assembly reaction of [NH4]2[WOS3] with CuBr in the presence of PCy3. The compound 1 exhibits a strong excited state absorption performance, the ratio of absorption cross-section of the singlet excited-state to that of the ground-state was about 18. Therefore compound 1 is considered to be a promising candidate for picosecond optical-limiting. The ratio of refrangibility of the singlet excited-state to that of the ground state is about 1.1. It possesses a self-focusing property with picosecond pulses. The asymmetry of the peak and valley of the Z-scan curve is caused by the excited-state absorption. The nonlinear refraction and absorption model of the excited-states is presented to simulate the experiment results.
References
[1] LANG J P, XU Q F, CHEN Z N, ABRAHAMS B F. Assembly of a supramolecular cube, (CPWS3Cu3)8Cl8(CN)12Li from a preformed incomplete cubane-like compound PPh4CPWS3(CuCN)3 [J]. J Am ChemSoc, 2003, 125: 12682-12683.
[2] KAWAGUCHI H, TATSUMI K. Reactions of tetrathiotungstate and tetrathiomolybdate with substituted haloalkanes[J]. Dalton Transactions, 2002, 2(12): 2573-2580.
[3] LIANG K, ZHENG H G, SONG Y L, LAPPERT M F, LII Y Z, XIN X Q, HUANG Z X. Self-assembly of interpenetrating coordination nets formed from interpenetrating cationic and anionic three-dimensional diamondoid cluster coordination polymers[J]. Angew Chem Int Ed Engl, 2004, 43: 5776-5779.
[4] POOJARY D M, CROHOL D, CLEARFIELD A. Stacking interactions between phenyl groups[J]. Angew Chem Int Ed Engl, 1995, 34: 1508-1512.
[5] ALBERTI G, MARMOTTINI F, MURCIA-MASCAROS S, VIVANI R. Computer-generated structural model[J]. Angew Chem Int Ed Engl, 1994, 33: 1594-1597.
[6] LOW K M, HOU H W, ZHENG H G, WONG W T, JIN G X, XIN X Q. Large optical limiting properties of the pentanuclear ‘open’ structural cluster compound [WS4Cu4(SCN)2(py)6] [J]. Chem Commun, 1998, 998(4): 505-506.
[7] LI B L, ZHU L M, WANG S W, ZHANG Y. Syntheses and crystal structures of two one-dimensional chain copper benzoylacetone complexes bridged through azobispyridine ligands[J]. J Coord Chem, 2003, 56: 933-941.
[8] TATSUMI K, KAWAGUCHI H, LU J M, JI W, SHI S. Microwave irradiation synthesis of Mo(W)/Tl/S linear chains and their nonlinear optical properties in solution[J]. Inorganic Chemistry, 1996, 35: 7924-7927.
[9] LANG J P, XU Q F, ELIM H I, TATSUMI K. Syntheses, crystal structures and optical limiting properties of three novel organometallic tungsten-copper-sulfur clusters: PPh4(eta(5)-C5Me5)-
[10] ZHANG Q F, NIU Y Y, LEUNG W H, SONG Y L, WILLIAMS I D, XIN X Q. Synthesis of a 2D polymeric cluster {[NEt4][Mo2O2S6Cu6I3(4,4′-bipy)5]?MeOH?H2O}n with a significant improvement of optical limiting effect[J]. Chem Commun, 2001, 2(8): 1126-1127.
[11] MCDONALD J W, FRIESON G D, ROSENHEIN C D, NEWTON W E. Syntheses and characterization of ammonium and tetraalkylammonium chiomolybdates and thiotungstates[J]. Inorg Chim Acta, 1983, 72: 205-210.
[12] SHELXTL Version 5.1 Reference Manual, Siemens Analytical X-ray Systems[M]. WI: Inc Madison, 1996.
[13] YU H, XU Q F, SUN Z R, JI S J, CHEN J X, LIU Q, LANG J P. Unique formation of two different W/Ag/S clusters from the same components via a low heating temperature solid state reaction and a solution reaction and their third-order nlo properties in solution[J]. Chemical Communications, 2001, 1(24): 2614-2615.
[14] SONG Y L, YANG M, WANG R B, LI C F. Measurement of excited state absorption cross sections in C60 by using steady state reverse saturable absorption method[J]. Chin J Lasers 1994, A21: 106-109.
[15] SONG Y L, ZHANG C, JIN G, LIU S, XIN X Q. Excited state optical nonlinearity of cubane-like shaped clusters[J]. Opt Commun. 2000, 186: 105-110.
(Edited by YANG Hua)
Foundation item: Project (2005038557) supported by the Post-Doctor Programs Foundation of Ministry of Education of China
Corresponding author: ZHOU Jian-liang; Tel: +86-731-8877679; Fax: +86-731-8879616;E-mail: zhoujl@mail.csu.edu.cn