���������оֲ���ƽ�����ʽ����ķ���ģ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2012���11��
�������ߣ�S. L. SOBOLEV
����ҳ�룺2749 - 2755
�ؼ��ʣ���Ԫ�Ͻ����ʽ������������̣��ֲ���ƽ����ɢ��˫����ɢ���̣�����Ũ�ȣ�����ͨ������
Key words��binary alloys; solute trapping; rapid solidification; local-nonequilibrium diffusion; hyperbolic diffusion equation; solute concentration; solute flux fields
ժ Ҫ���Զ�Ԫ�Ͻ�������̹����еľֲ���ƽ����ɢģ��(LNDM)���иĽ����Ľ���ģ�Ϳ���������������Ũ�Ⱥ�����ͨ��������ֲ�ƽ���ƫ�����˫��������ɢ�������������������Ũ�Ⱥ�ͨ����ȷ�⡣������������κι�-Һ����Ķ���ѧ������Ч��ɢϵ�� ����v��vDb������ȫ���ʽ���KLNDM(v)��1ʱ�����̹��̽�����ɢ����ת��Ϊ��ȫ���ȿ��ơ�����ɢ���̺���ȫ���ʽ������ٽ����Ϊ�������е���ɢ�ٶ�vDb�������˲�ͬ���涯��ѧ;�������ʽ���ģ�͡�
Abstract: Updated version of local non-equilibrium diffusion model (LNDM) for rapid solidification of binary alloys was considered. The LNDM takes into account deviation from local equilibrium of solute concentration and solute flux fields in bulk liquid. The exact solutions for solute concentration and flux in bulk liquid were obtained using hyperbolic diffusion equations. The results show the transition from diffusion-limited to purely thermally controlled solidification with effective diffusion coefficient ��0 and complete solute trapping KLNDM(v)��1 at v��vDb for any kind of solid-liquid interface kinetics. Critical parameter for diffusionless solidification and complete solute trapping is the diffusion speed in bulk liquid vDb. Different models for solute trapping at the interface with different interface kinetic approaches were considered.
Trans. Nonferrous Met. Soc. China 22(2012) 2749-2755
S. L. SOBOLEV
Institute of Problems of Chemical Physics, Academy of Sciences of Russia, Chernogolovka, Moscow Region, 142432, Russia
Received 6 December 2011; accepted 29 March 2012
Abstract: Updated version of local non-equilibrium diffusion model (LNDM) for rapid solidification of binary alloys was considered. The LNDM takes into account deviation from local equilibrium of solute concentration and solute flux fields in bulk liquid. The exact solutions for solute concentration and flux in bulk liquid were obtained using hyperbolic diffusion equations. The results show the transition from diffusion-limited to purely thermally controlled solidification with effective diffusion coefficient 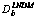 ��0 and complete solute trapping KLNDM(v)��1 at v��vDb for any kind of solid-liquid interface kinetics. Critical parameter for diffusionless solidification and complete solute trapping is the diffusion speed in bulk liquid vDb. Different models for solute trapping at the interface with different interface kinetic approaches were considered.
��0 and complete solute trapping KLNDM(v)��1 at v��vDb for any kind of solid-liquid interface kinetics. Critical parameter for diffusionless solidification and complete solute trapping is the diffusion speed in bulk liquid vDb. Different models for solute trapping at the interface with different interface kinetic approaches were considered.
Key words: binary alloys; solute trapping; rapid solidification; local-nonequilibrium diffusion; hyperbolic diffusion equation; solute concentration; solute flux fields
1 Introduction
Rapid solidification phenomena occur under conditions that are far from equilibrium [1-22]. The most obvious manifestations of the deviations from local equilibrium is solute trapping which increases the solute concentra tion in solid phase and reduces the segregations in the liquid side of interface. The degree of solute trapping is usually quantified by the partition coefficient K, defined as a ratio of the concentration of solute in the solid to that in the liquid at the interface. This process is useful to obtain very fine structure with uniform properties. Examples of rapid solidification products accompanied by solute trapping are powders, wires and foils which can be used in powder metallurgy or in producing higher performance composite materials. As an emerging rapid solidification technology, direct strip casting is a continuous casting process for producing as cast metallic sheet of carbon and stainless steels, aluminum, magnesium, titanium and other alloys without any further thermo-mechanical processing. Therefore, the development of capability in predicting solute trap ping phenomena is an important task in designing new materials and new processes.
The effect of solute trapping has been investigated theoretically using analytical models [1,2,8-13,16-18, 21,22], phase-field models [14], molecular dynamics simulations [15, 23], Monte Carlo computer simulations [19] as well as experimental investigations [1 �C 7,9]. The most outstanding question in all these studies is transition to complete solute trapping K(v)=1, where v is the interface velocity. The continuous growth model (CGM) [8], the stepwise growth model (SGM) [9], the aperiodic stepwise growth model (ASGM) [9], and the JACKSON et al model [18,19] predict complete solute trapping K(v)=1 only at v����. By contrast, the local nonequilibrium diffusion model (LNDM) [10-13], based on the assumption of absence of local-equilibrium during solute diffusion in bulk liquid, leads to diffusionless solidification with complete solute trapping K(v)=1 at a finite interface velocity v=vDb, where vDb is the diffusive velocity in the bulk liquid, i.e. the speed of atomic diffusion in liquid phase. The complete solute trapping was also observed in the experiment [3-7, 9], phase-field models [14], and molecular dynamics simulations [15].
The classical theoretical treatments of rapid solidification [1,8,9,18,19] take into account only the deviation from equilibrium at the interface introducing the velocity dependent partition coefficient K(v). Such a modeling is valid only for relatively low interface velocities v<
2 Local nonequilibrium diffusion model (LNDM)
According to extended irreversible thermodynamics (EIT) [24] and some other local-nonequlibrium approaches [25-28], the simplest generalization of the classical Fick law for mass transport, which includes the relaxation to local equilibrium of the diffusion field, is given as
 (1)
(1)
where J is the solute flux, C is the solute concentration, Db is the diffusion coefficient in bulk liquid, �� is the relaxation time of J. In contrast to the Fick law, which is based on local-equilibrium approach, the evolutional Eq. (1) takes into account deviation of diffusion field from local equilibrium. Thus, local nonequilibrium diffusion model (LNDM) for solute concentration during rapid solidification takes the form [10-13, 25]:
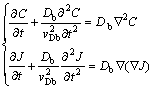 (2)
(2)
The hyperbolic Eq. (2) predict the finite speed of the diffusive wave, i.e. maximum speed with which the diffusional perturbations can propagate in the liquid [10-13,24-28]:
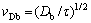 (3)
(3)
It should be noted that vDb limits only the speed of diffusive perturbations (diffusive signal), but the interface velocity v can be greater than vDb. To derive the interface condition, we integrate the balance low over an infinitesimal zone that includes the interface between liquid and solid phases. The interface condition is
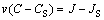 (4)
(4)
where CS and JS are the solute concentration and solute flux in the solid at the interface. Assuming JS=0 and using Eq. (1), we can rewrite the interface condition as
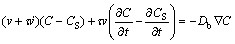 (5)
(5)
where the superscript dot implies differentiation with respect to time. Note that condition Eq. (5) includes not only the interface velocity, but also the interface acceleration.
Now let us consider the solute concentration field ahead of the interface moving with constant velocity v=const. Following the usual steady-state approach, we view the solidification from a reference frame attached to a planar liquid-solid interface. In such a case, a one-dimensional version of Eqs. (2) and (4) takes the form:
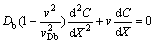 (6)
(6)
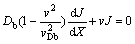 (7)
(7)
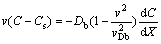 (8)
(8)
Equations (5) and (6) result in the solute concentration and solute flux distributions in the liquid X>0 (the origin of the reference frame is fixed on the beginning of interface X=0) [10-13,25]:
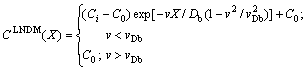 (9)
(9)
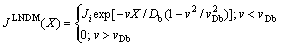 (10)
(10)
where C0 and Ci are the solute concentration in the liquid far from (X����) and at the interface (X=0), respectively.
The results of LNDM Eqs. (9) and (10) clearly demonstrate that the diffusion speed vDb decisively affects the diffusion field in the bulk liquid. When v
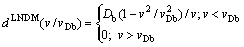 (11)
(11)
approaches zero at v=vDb.
When v>vDb, LNDM Eqs. (9) and (10) imply that C(X)=C0, J(X)=0, and d(v)=0. This means that the solute concentration field ahead of the interface is undisturbed. The result has a clear physical meaning: a source of perturbations (i.e. the interface) moving with a velocity greater than the maximum speed of perturbations cannot disturb the medium ahead of itself [24,25]. In this case, the solute atoms do not have enough velocity to escape the solid-liquid interface. Thus, there is no diffusion of solute in the liquid at v>vDb and, consequently, the solidification cannot be controlled by diffusion. Hence, the solidification mechanism changes qualitatively when the interface velocity v passes through the critical point v=vDb. In this point, a sharp transition from diffusion controlled to purely thermally controlled growth occurs.
Now, let us compare the solute concentration and solute flux distributions according to LNDM, Eqs. (9)-(11), with classical local equilibrium ones:
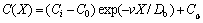 (9a)
(9a)
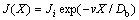 (10a)
(10a)
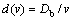 (11a)
(11a)
Equations (9a)-(11a) are the solutions to the solute concentration and solute flux fields obtained from local equilibrium diffusion equation of parabolic type. Thus, the comparison of exact solutions to LNDM, Eqs. (9)-(11), with classical solute concentration and solute flux fields, Eqs. (9a)-(11a), allows us to introduce the effective diffusion coefficient in bulk liquid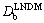 [10-13] as
[10-13] as
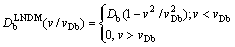 (12)
(12)
If v<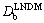 reduces to the classical diffusion coefficient Db for the local-equilibrium conditions. But when v is of the order of VDb,
reduces to the classical diffusion coefficient Db for the local-equilibrium conditions. But when v is of the order of VDb, 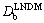 predicts less solute flux
predicts less solute flux 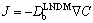 than expected from the classical Fick law
than expected from the classical Fick law 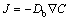 . If v��vDb, then
. If v��vDb, then 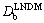 =0 that differs qualitatively from the prediction of the classical approach. It implies the absence of solute diffusion ahead of the interface. The effective diffusion coefficient Eq. (12) can be used to modify some results of classical rapid solidification theory based on Fick law by substituting
=0 that differs qualitatively from the prediction of the classical approach. It implies the absence of solute diffusion ahead of the interface. The effective diffusion coefficient Eq. (12) can be used to modify some results of classical rapid solidification theory based on Fick law by substituting 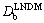 for Db at least for steady-state regimes. WALDER [5] took into account the effective diffusion coefficient Eq. (12) to modify the dendritic growth theory of TRIVEDI, LIPTON and KURZ and obtained better agreement with experiment.
for Db at least for steady-state regimes. WALDER [5] took into account the effective diffusion coefficient Eq. (12) to modify the dendritic growth theory of TRIVEDI, LIPTON and KURZ and obtained better agreement with experiment.
3 Models for solute partitioning
The partition coefficient K(v) (ratio of solid to liquid concentration of solute at the interface) plays an important part in rapid solidification phenomena. The results of LNDM Eqs. (9) and (10) clearly demonstrate that when vvDb there is no any diffusion of solute at the interface and therefore the partition coefficient KLNDM does not depend on v and KLNDM=1. It should be stressed that the result of LNDM, KLNDM=1 at vvDb, does not depend on kinetic effects at the solid-liquid interface. It is purely diffusive effects [9-12]. It means that for any interface kinetic model the limit KLNDM(v)��1 at v��vDb must be achieved as long as no other mass transport process apart from diffusion (for example, convective solute flow or something like that) comes into play. The fact will be illustrated here below.
3.1 Continuous growth model (CGM)��Dilute limit
The continuous growth model (CGM) of AZIZ and KAPLAN [6,8,9] treats the case when the interface is atomically rough enough that growth and redistribution occur simultaneously as strictly steady-state processes, even on the microscopic scale of the crystal lattice. It predicts a velocity dependence of the partition coefficient given by
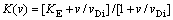 (13)
(13)
where KE is the equilibrium partition coefficient, VDi is the interface diffusion speed. At v=vDi, Eq. (13) describes mid-transition between KE and unity and predicts complete solute trapping KE=1 only at v����.
The diffusive speed in LNDM, vDb, and the interface diffusion speed, vDi, in Eq. (13) require further discussion. The CGM defines the vDi as the ratio of solute diffusivity through the interface Di to the atomic jump distance at the interface ��i. In other words, vDi is the average diffusive speed over the interface region and it has been called as the interface diffusive speed vDi. The interface diffusive speed is a kinetic rate parameter for solute redistribution at the interface for relatively low interface velocity v
Thus, introduction of 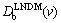 , Eq. (12), into the expression for K(v), Eq. (13), leads to the generalized partition coefficient KLNDM(v), which takes into account both interfacial kinetic effects according to CGM (the interface redistribution parameter vDi) and local- nonequilibrium diffusion effects according to LNDM (the bulk liquid diffusion speed vDb) [11,12]:
, Eq. (12), into the expression for K(v), Eq. (13), leads to the generalized partition coefficient KLNDM(v), which takes into account both interfacial kinetic effects according to CGM (the interface redistribution parameter vDi) and local- nonequilibrium diffusion effects according to LNDM (the bulk liquid diffusion speed vDb) [11,12]:
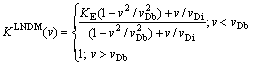 (14)
(14)
Expression (14) clearly demonstrates that the transition to complete solute trapping KLNDM(v)=1 occurs at a finite interface velocity v=vDb, while the CGM, Eq. (13) predicts the complete solute trapping only at v. The result of LNDM, Eq. (14), is in good agreement with the phase-field simulation [14], molecular dynamic simulation [15], and experimental data [6,7] (see also Refs. [11,12] and references therein).
3.2 Continuous growth model (CGM)��Non-dilute limit
According to CGM the partition coefficient in non-dilute limit takes the form:
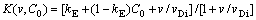 (15)
(15)
where kE is the equilibrium partition coefficient for the solute divided by the equilibrium partition coefficient for the solvent [7,8]. This equation can be modified to local nonequilibrium diffusion case by using concept of the effective diffusion coefficient 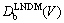 , Eq.(12). After some algebra we have
, Eq.(12). After some algebra we have
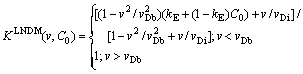 (16)
(16)
The partition coefficient KLNDM(v,C) takes into account both interfacial kinetic effects in non-dilute limit, according to CGM [7,8], and local nonequilibrium diffusion effects according to LNDM, Eqs. (6)-(12). As it was predicted earlier, the partition coefficient KLNDM(v,C0)��1 when v��vDb due to local nonequilibrium diffusion effects.
3.3 Stepwise growth model (SGM)
The SGM [9] treats the case in which an atomically smooth, sharp interface advances by the periodic lateral passage of monolayer, including any solute atoms in the layer. Solute diffuses back into the liquid during the period before the passage of the next step, at which point any remaining solute is assumed to be permanently trapped into the solid. The predicted velocity-dependence of K for this mechanism is
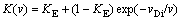 (17)
(17)
Note that in the SGM dependence K from v, Eq. (17), yields complete solute trapping K=1 only at v. Taking into account local-nonequilibrium diffusion effects in bulk liquid according to LNDM approach, Eq. (17) can be rewritten as
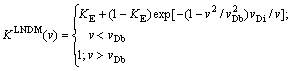 (18)
(18)
The expression for KLNDM(v), Eq. (18), takes into account both departure from local equilibrium of diffusion process in bulk liquid according to LNDM and interface kinetic conditions assumed by SGM. It leads to complete solute trapping KLNDM(v)=1 at v��vDb.
3.4 Aperiodic stepwise growth model (ASGM)
The ASGM [6,9] treats the same case as the SGM, except that the passage of steps is assumed to occur randomly, rather than periodically, in time. It predicts variation of K with interface orientation. The ASGM assumes that at any orientation the interface is broken into (111) terraces of single double-layer height and random width and that solidification proceeds by the lateral passage of these steps at random intervals. Some (not all, as in SGM) of the solute atoms in the monolayer of liquid adjacent to the interface are trapped as a step passes. The ASGM expression for K(v,��) is given by
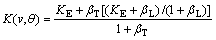 (19)
(19)
where ��T=v/(vDTcos ��), ��L=v/(vDLcos ��), vDL is diffusive speed at the ledge, vDTis diffusive speed at the terrace, and �� is the angle of inclination from (111). Using the effective diffusion coefficient Eq. (12) LNDM leads to
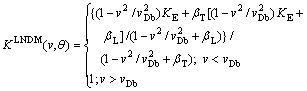 (20)
(20)
Equation (20) generalizes Equation (19) for local nonequilibrium case and predicts complete solute trapping at v��vDb in contrast to Eq. (19) which gives K(v,��)��1 only at v����.
3.5 JACKSON et al model
Another theory for solute trapping due to JACKSON et al [18,19] is derived using reaction rate theory to describe the rate of atom attachments to the active sites of a sharp solid-liquid interface. The model is based on the standard quasi-equilibrium model for crystallization, which has been extended to include the non-equilibrium effects found in the Monte Carlo simulations of alloy solidification. The parameter in the model, ��, is clearly identified as the important parameter for solute trapping. This parameter embodies the relationship between the growth and diffusion parameters found in the Monte Carlo simulations. This parameter is interpreted as the ratio of the distance that the interface advances to the distance that an atom can move by diffusion during the time in which it is at the interface. If �� is the average time it takes for an atom to join the crystal, the distance that the interface advances during this time is v��. The distance that an atom can move by diffusion during this time is (Db��)1/2, where Db is the diffusion coefficient of B atoms in the liquid. The ratio of these two distances is the dimensionless parameter �� [18,19]:
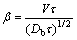 (21)
(21)
Taking into account Eq. (3) for diffusive velocity in bulk liquid and the fact that the relaxation time in LNDM corresponds to the average time it takes for an atom to join the crystal in JACKSON et al model, the parameter ��, Eq. (21), transforms to the ratio ��=v/vDb. The parameter v/vDb is also the critical parameter in LNDM, Eqs. (1)-(12), and in some other models [25], but in LNDM v/vDb governs solute diffusion in bulk liquid, and hence, solute partitioning at v��vDb, and in JACKSON et al model parameter �� governs non-equilibrium segregation at the solid-liquid interface. Solute partitioning is governed by both solute segregation process at the interface with governing parameter �� and local nonequilibrium diffusion effects with governing parameter v/vDb. But the parameter �� in JACKSON et al model [18,19] plays an important role at moderate interface velocity v
Thus, for rough interfaces and small concentration of solute, the theory of JACKSON et al yields [18,19]
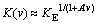 (22)
(22)
where A' depends on the square root of the diffusion coefficient, namely, A��=A��/(Db)1/2. From Eqs. (12) and (22) one can calculate
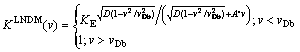 (23)
(23)
The partition coefficient Eq. (23) incorporates both interface kinetic model of JACKSON et al [17,18] and local nonequilibrium diffusion field according to LNDM. As it was expected one can observe partitionless solidification KLNDM(v)��1 when v��vDb in contrast to Eq. (22) that leads to complete solute trapping only at v����.
4 Hierarchy of deviation from local equilibrium
Solidification of undercooled melts occurs at wide range of velocities up to hundreds m/s and includes various physical phenomena such as solute diffusion, heat conduction, interface kinetics of solidification. Each of these processes has its own characteristic time and length scales (in a steady-state �C characteristic velocities). It implies that while interface velocity increases, the solute diffusion, interface kinetics, and heat conduction deviate from equilibrium at different values of interface velocity. This allows us to introduce a hierarchy of deviations from equilibrium which is followed by increasing solidification velocity [2,11,12]:
1) v=0. Full equilibrium. No chemical potential gradient (composition of phases are uniform), no temperature gradients, K=KE.
2) v����
3) v��VDb. Non-equilibrium interface kinetics. There is no local equilibrium at the interface and the partition coefficient depends on the interface velocity v. In this case (as well as in case 2), the solute concentration and temperature fields are governed by the classical (local equilibrium) transport equation of parabolic type.
4) v��vDb. Non-equilibrium diffusion field. In this case, there is no local equilibrium in the bulk liquid. Hence, according to LNDM, solute concentration and solute flux fields in bulk liquid are governed by the mass transport equations of hyperbolic type (1)�C(2) [10-13]. The temperature field is still at local equilibrium due to vvDb<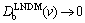 ) with complete solute trapping KLNDM(v)��1 at v��vDb for all interface kinetic models (see Eqs. (14), (16), (18), (20), and (23)).
) with complete solute trapping KLNDM(v)��1 at v��vDb for all interface kinetic models (see Eqs. (14), (16), (18), (20), and (23)).
5) v��vDb. Diffusionless solidification. According to LNDM, at this interface velocity KLNDM(v)��1 and 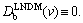 This implies thermally controlled diffusionless solidification with complete solute trapping. Ahead of the interface C(X)=C0 and J(X)=0. The result does not depend on the interface kinetics and is a consequence of deviation from local equilibrium in bulk liquid with vDb being the critical parameter [10-13].
This implies thermally controlled diffusionless solidification with complete solute trapping. Ahead of the interface C(X)=C0 and J(X)=0. The result does not depend on the interface kinetics and is a consequence of deviation from local equilibrium in bulk liquid with vDb being the critical parameter [10-13].
6) v~T. Local-nonequilibrium temperature field. At such high velocities heat transport occurs far from local equilibrium. The temperature field is governed by hyperbolic transport equations [20-23]. If solid-liquid interface propagates only due to undercooling effects, the interface velocity v is limited by vT-the speed of heat wave [25].
5 Conclusions
LNDM clearly demonstrates that a sharp transition from diffusion-controlled to purely thermally controlled solidification occurs due to local nonequilibrium solute diffusion in the bulk liquid. The critical parameter for the transition is vDb (the diffusive velocity in the bulk liquid), i.e. the speed of atomic diffusion in liquid phase. When v
Local nonequilibrium diffusion effects lead to the effective (velocity dependent) diffusion coefficient in bulk liquid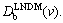 The effective diffusion coefficient has been used to modify a number of existing solute partitioning models. Due to local nonequilibrium diffusion effects all the models predict the transition to diffusionless solidification with complete solute trapping KLNDM(v)=1 when the interface velocity v passes through the critical point v=vDb. Thus, at high interface velocity v��vDb, the solute partitioning at the solid-liquid interface is mainly governed by solute diffusion in bulk liquid rather than by interface kinetics of solidification.
The effective diffusion coefficient has been used to modify a number of existing solute partitioning models. Due to local nonequilibrium diffusion effects all the models predict the transition to diffusionless solidification with complete solute trapping KLNDM(v)=1 when the interface velocity v passes through the critical point v=vDb. Thus, at high interface velocity v��vDb, the solute partitioning at the solid-liquid interface is mainly governed by solute diffusion in bulk liquid rather than by interface kinetics of solidification.
References
[1] HERLACH D M. Non-equilibrium solidification of undercooled metallic melts [J]. Materials Science and Engineering A, 1994, 12: 177-272.
[2] LIU F, YANG G C. Rapid solidification of highly undercooled bulk liquid superalloy: recent developments, future directions [J]. International Materials Reviews, 2006, 51(3): 145-170.
[3] ECKLER K, COCHRANE R F, HERLACH D M, FEUERBACHER B, JURISCH M. Evidence for a transition from diffusion-controlled to thermally controlled solidification in metallic alloys [J]. Physical Review B, 1992, 45: 5019-5022.
[4] WALDER S, RYDER P L. Rapid dendritic growth in undercooled Ag-Cu melts [J]. Acta Metallurgica et Materialia, 1995, 43(11): 4007-4013.
[5] WALDER S. Dendritic growth rate in undercooled, dilute Ti-Ni melts [J]. Materials Science and Engineering A, 1997, 229(1-2): 156-162.
[6] KITTL J A, AZIZ M J, BRUNCO D P, THOMPSON M O. Nonequilibrium partitioning during rapid solidification of Si-As alloys [J]. Journal of Crystal Growth, 1995, 148: 172-182.
[7] BRUNCO D P, THOMPSON M O, HOGLUNG D E, AZIZ M J, GOSSMANN H J. Germanium partitioning I silicon during rapid solidification [J]. Journal of Applied Physics, 1995, 78(3): 1575-1582.
[8] AZIZ M J, KAPLAN T. Continuous growth model for interface motion during alloy solidification [J]. Acta Metallurgica, 1988, 36: 2335-2347.
[9] REITANO R, SMITH P M, AZIZ M J. Solute trapping of group III, IV and V elements in silicon by an aperiodic stepwise growth mechanism [J]. Journal of Applied Physics, 1994, 76(3): 1518-1529.
[10] SOBOLEV S L. Local-nonequilibrium model for rapid solidification of undercooled melts [J]. Physics Letters A, 1995, 199: 383-386.
[11] SOBOLEV S L. Effects of local non-equilibrium solute diffusion on rapid solidification of alloys [J]. Physica Status Solidi A, 1996, 156: 293-303.
[12] SOBOLEV S L. Rapid solidification under local nonequilibrium condition [J]. Physical Review E, 1997, 55: 6845-6854.
[13] SOBOLEV S L. Local non-equilibrium diffusion effects on the kinetic phase boundaries in solidification [J]. International Journal of Non-equilibrium Processing, 1997, 10: 49-58.
[14] FURTADO H S, BERNARDES A T, MACHADO R F, SILVAC C A. Numerical simulation of solute trapping phenomena using phase-field solidification model for dilute binary alloys [J]. Materials Research, 2009, 12(3): 345-351.
[15] YANG Y, HUMADI H, BUTA D, LAIRD B B, SUN D, HOYT J J, ASTA M. Atomistic simulations of nonequilibrium crystal-growth kinetics from alloy melts [J]. Physical Review Letters, 2011, 107: 025505.
[16] CHEN Z, WANG H, LIU F, YANG W. Effect of nonlinear liquidus and solidus on dendrite growth in bulk undercooled melts [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 490-494.
[17] WANG H, LIU F, CHEN Z, YANG G, ZHOU Y. Analysis of non-equilibrium dendrite growth in a bulk undercooled alloy melt: Model and application [J]. Acta Materialia, 2007, 55: 497-506.
[18] JACKSON K A, KIRK M. BEATTY K M, GUDGEL K A. An analytical model for non-equilibrium segregation during crystallization [J]. Journal of Crystal Growth, 2004, 271: 481-494.
[19] BEATTY K M, JACKSON K A. Monte Carlo modeling of dopant segregation [J]. Journal of Crystal Growth, 2004, 271: 495-512.
[20] LI S, ZHANG J, WU P. Numerical solution and comparison to experiment of solute drag models for binary alloy solidification with a planar phase interface [J]. Scripta Materialia, 2010, 62: 716-719.
[21] LI S, ZHANG J, WU P. A comparative study on migration of a planar interface during solidification of non-dilute alloys [J]. Journal of Crystal Growth, 2010, 312: 982-988.
[22] WANG H, LIU F, CHEN Z, YANG W. Solute trapping model based on solute drag treatment [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 887-881.
[23] COOK S, CLANCY P. Impurity segregation in Lennard-Jones A/AB heterostructures. [J]. Journal of Chemical Physics, 1993, 99: 2175.
[24] JOU D, CASAS-VAZQUEZ J, LEBON G. Extended irreversible thermodynamics [M]. Berlin: Springer, 1996: 383.
[25] SOBOLEV S L. Transport processes and travelling waves in systems with local nonequilibrium [J]. Soviet Physics-Uspekhi, 1991, 34(3): 217-229.
[26] NETTLETON R E, SOBOLEV S L. Application of extended thermodynamics to chemical, rheological, and transport processes: A special survey. Part I. Approaches and scalar rate processes. [J]. Journal of Non-equilibrium Thermodynamics, 1995, 20: 205-229.
[27] NETTLETON R E, SOBOLEV S L. Application of extended thermodynamics to chemical, rheological, and transport processes: A special survey. Part II. Vector transport processes, shear relaxation and rheology [J]. Journal of Non-equilibrium Thermodynamics, 1995, 20: 297-331.
[28] NETTLETON R E, SOBOLEV S L. Application of extended thermodynamics to chemical, rheological, and transport processes: A special survey. Part III. Wave phenomena [J]. Journal of Non-equilibrium Thermodynamics, 1995, 21: 1-16.
S. L. SOBOLEV
Institute of Problems of Chemical Physics, Academy of Sciences of Russia, Chernogolovka, Moscow Region, 142432, Russia
ժ Ҫ���Զ�Ԫ�Ͻ�������̹����еľֲ���ƽ����ɢģ��(LNDM)���иĽ����Ľ���ģ�Ϳ���������������Ũ�Ⱥ�����ͨ��������ֲ�ƽ���ƫ�����˫��������ɢ�������������������Ũ�Ⱥ�ͨ����ȷ�⡣������������κι�-Һ����Ķ���ѧ������Ч��ɢϵ��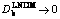 ����v��vDb������ȫ���ʽ���KLNDM(v)��1ʱ�����̹��̽�����ɢ����ת��Ϊ��ȫ���ȿ��ơ�����ɢ���̺���ȫ���ʽ������ٽ����Ϊ�������е���ɢ�ٶ�vDb�������˲�ͬ���涯��ѧ;�������ʽ���ģ�͡�
����v��vDb������ȫ���ʽ���KLNDM(v)��1ʱ�����̹��̽�����ɢ����ת��Ϊ��ȫ���ȿ��ơ�����ɢ���̺���ȫ���ʽ������ٽ����Ϊ�������е���ɢ�ٶ�vDb�������˲�ͬ���涯��ѧ;�������ʽ���ģ�͡�
�ؼ��ʣ���Ԫ�Ͻ����ʽ������������̣��ֲ���ƽ����ɢ��˫����ɢ���̣�����Ũ�ȣ�����ͨ������
(Edited by YUAN Sai-qian)
Corresponding author: S. L. SOBOLEV; E-mail: sobolev@icp.ac.ru; SSL55@yandex.ru
DOI: 10.1016/S1003-6326(11)61528-8

