三价铬的电化学沉积
杨余芳1,龚竹青2,李强国1
(1. 湘南学院 化学与生命科学系,湖南 郴州,423000;
2. 中南大学 冶金科学与工程学院,湖南 长沙,410083)
摘 要:通过稳态极化曲线、循环伏安、恒电流阶跃、交流阻抗等方法,研究柠檬酸水溶液体系中Cr3+的电沉积还原反应机理,确定Cr3+电沉积的动力学参数和动力学方程。研究结果表明,Cr3+的电沉积为电化学步骤控制过程,该电化学反应分2步进行:第1步是Cr3+获得2个电子还原为Cr+,为控制步骤和不可逆过程;第2步是Cr+ 获得1个电子还原为金属Cr,为准可逆过程;Cr3+在电沉积过程中无前置转化反应存在,但有电活性的中间产物吸附在电极表面;阴极和阳极表观传递系数分别为0.490和2.455;化学计量数为1;阴极反应级数为1,阳极反应级数为0;扩散系数为1.606 10-5 cm2/s。
10-5 cm2/s。
关键词:三价铬;电沉积;恒电流阶跃;循环伏安;交流阻抗
中图分类号:O646.54 文献标识码:A 文章编号:1672-7207(2008)01-0112-06
Electrochemical deposition of trivalent chromium
YANG Yu-fang1, GONG Zhu-qing2, LI Qiang-guo1
(1. Department of Chemistry and Life Science, Xiangnan University, Chenzhou 423000, China;
2. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: The electrodeposition reduction mechanism of Cr3+ in the system of citric acid aqueous solution was investigated by using steady polarization curve, cyclic voltammetry, chronopotentiometry, and AC impedance. The kinetic parameters and equation of Cr3+ electrodeposition were decided. The results show that the electrodeposition of Cr3+ is an electrochemistry determinative step, and the electrochemical reaction is finished in two steps. The first step is a rate determinative step and an irreversible process, in which Cr3+ obtains two electrons and is reduced to Cr+. The second step is a quasi-reversible process, in which Cr+ obtains one electron and is reduced to metal Cr. Cr3+ ion does not undergo prepositive chemical transformation reaction while it is reduced, but there are electrochemistry active middle products adsorbed on the cathodic surface. The apparent transfer coefficients of cathode and anode are 0.490 and 2.455, respectively. The chemical metric number of Cr3+ electrodeposition is 1, the order of reaction of cathode and anode is 1 and 0, respectively, and the apparent diffusion coefficient is 1.606×10-5 cm2/s.
Key words: trivalent chromium; electrodeposition; chronopotentiometry; cyclic voltammetry; AC impedance
三价铬电镀具有低毒低污染、镀层颜色白亮、硬度高且力学性能优良等特点;电沉积过程中电流效率高,能耗小,分散能力和覆盖能力好,可在常温下操作,废水处理简单,具有六价铬电镀无法比拟的优点,因此,对三价铬的电沉积研究引起了许多研究者的重视[1-10]。但是,从不同的反应体系中所得到的Cr3+的电沉积反应机理是不同的,到目前为止,对Cr3+的电沉积反应机理有3种看法。邓姝皓等[11]认为Cr3+在DMF体系中分2步放电:第1步Cr3+得到1个电子变成Cr2+,第2步是Cr2+得到2个电子变成Cr原子;Szynkarczuk等[12]认为当SCN-作配位体时Cr3+分3步放电,每步得到1个电子;Hsieh等[13]认为当CH3COO-作配位体时Cr3+直接还原为Cr,不经过中间过程,由此说明Cr3+的电沉积与反应体系有关。在此,本文作者利用线性扫描、循环伏安、恒电流阶跃、交流阻抗等方法,对Cr3+在柠檬酸体系中的电化学反应机理进行研究。
1 实 验
采用带有砂芯玻璃膜的三电极“H”形电解槽体系,研究电极为密封于玻璃管中的直径为1 mm且露出端面的铜电极,辅助电极为大面积Pt片,参比电极为饱和甘汞电极(SCE)。采用带有Luggin毛细管的盐桥以消除不同液体之间的接界电位。实验前将Cu电极用细金相砂纸打磨抛光,用酒精、丙酮等有机溶剂清洗,以稀盐酸浸泡活化,再用蒸馏水洗涤。溶液用分析纯试剂和蒸馏水配制,溶液组成为:50 g/L H3BO3,25 g/L CrCl3・6H2O,20 g/L NaBr,0.1 g/L十二烷基硫酸钠和49 g/L柠檬酸。溶液经过陈化处理,通入N2 15 min以除去溶液中的溶解氧;温度为23~45 ℃,pH值为2.3。采用线性扫描、循环伏安、恒电流阶跃及交流阻抗等方法,利用CHI660A电化学工作站进行分析测试。
2 结果与讨论
2.1 Cr3+电沉积活化能的测定
测量不同温度下Cr3+电沉积的稳态极化曲线,根据不同温度下的Tafel曲线,可得到在某一过电位下电流密度的对数lg J与温度的倒数1/T的关系曲线。图1所示为过电位为0.9 V时Cr3+ 电沉积的lg J~1/T关系曲线,可见lg J与1/T呈线性关系,根据Arrhenius公式和直线的斜率,可求出在该过电位下Cr3+的表观活化能为60.72 kJ/mol。

图1 lg J~1/T曲线
Fig.1 lg J―1/T curve
图2所示为Cr3+电沉积的表观活化能与过电位的关系曲线。根据电化学理论可知,当电极反应受扩散步骤控制时,反应速度的温度系数较小,电极反应的活化能较低,一般为12~16 kJ/mol;当电化学反应为控制步骤时,反应速度的温度系数较大,电极反应的活化能较大,一般在40 kJ/mol以上。从图2可以发现,Cr3+电沉积的表观活化能较大,当过电位为0.6~ 1.2 V时,表观活化能为37.19~78.37 kJ/mol,所以,该值说明Cr3+的电沉积由电化学步骤控制。同时,随着过电位的增大,Cr3+电沉积的表观活化能不断下降,说明较负的电极电位有利于Cr3+的电沉积。根据Tafel公式
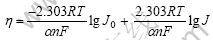
可计算出交换电流密度J0。通过实验测定了23~45 ℃范围内的交换电流密度J0,均在10-5 A/cm2的数量级。将lg J0与1/T作图,根据直线的斜率得到平衡电位下的表观活化能为70.04 kJ/mol。
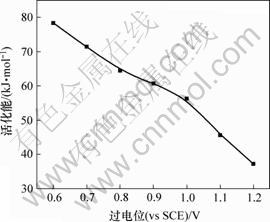
图2 过电位对Cr3+ 表观活化能的影响
Fig.2 Influence of overpotential on apparent activating energy of Cr3+
2.2 Cr3+电沉积前置反应的判断
用恒电流阶跃法可以判断Cr3+离子在电沉积过程中是否经历了前置化学转化反应,根据Sand方程 式[14-15]

可知,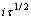 与i无关。用
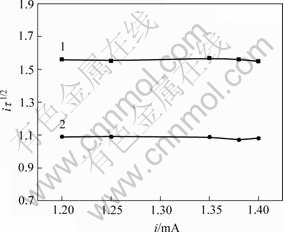
与i无关。用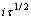 对i作图,若得到平行电流坐标的直线,则表明电极反应不伴随有表面转化反应;否则表示有表面转化反应。当温度为25 ℃、阶跃电流i为1.2 mA时所得到的电位-时间关系如图3所示。由图3可知,曲线上有2个阶梯,对应着2个过渡时间τ,分别为τ1=1.69 s,τ2=0.83 s,表明在阴极大量析出氢气之前发生了2个平行的电荷传递反应,Cr3+的还原分2步进行。随着阶跃电流的增大,τ逐渐减小。图4所示为在不同阶跃电流下所得到的i~
对i作图,若得到平行电流坐标的直线,则表明电极反应不伴随有表面转化反应;否则表示有表面转化反应。当温度为25 ℃、阶跃电流i为1.2 mA时所得到的电位-时间关系如图3所示。由图3可知,曲线上有2个阶梯,对应着2个过渡时间τ,分别为τ1=1.69 s,τ2=0.83 s,表明在阴极大量析出氢气之前发生了2个平行的电荷传递反应,Cr3+的还原分2步进行。随着阶跃电流的增大,τ逐渐减小。图4所示为在不同阶跃电流下所得到的i~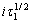 和i~
和i~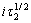 的关系曲线,均为平行于电流坐标的直线,说明Cr3+在还原时无前置化学反应存在。
的关系曲线,均为平行于电流坐标的直线,说明Cr3+在还原时无前置化学反应存在。
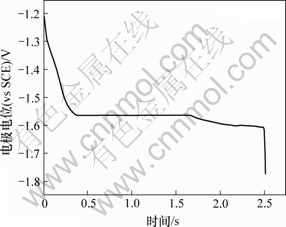
图3 恒电流阶跃电位-时间曲线
Fig.3 Potential-time curve of Cr3+ by chronopotentiometry
1―i- ;2―i-
;2―i-
图4 Cr3+恒电流阶跃的i-iτ1/2关系曲线
Fig.4 i-iτ1/2 curves of Cr3+ by chronopotentiometry
2.3 Cr3+电沉积的循环伏安曲线
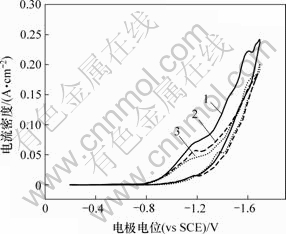
图5所示为25 ℃时在不同扫描速度下所得到的Cr3+的循环伏安图。可见,曲线上有2个还原峰,其峰电位分别在-1.19 V和-1.60 V附近,进一步说明Cr3+的还原是分2步进行的。第1个还原峰出现在 -1.19 V附近,在-0.2~-1.4 V之间对Cr3+溶液进行电位扫描,所得到的循环伏安曲线反映的是Cr3+的第1步还原反应情况。
扫描速度/(V?s-1): 1―0.1; 2―0.07; 3―0.03
图5 Cr3+电沉积的循环伏安曲线
Fig.5 Cyclic voltammogram curves of Cr3+ electrodeposition
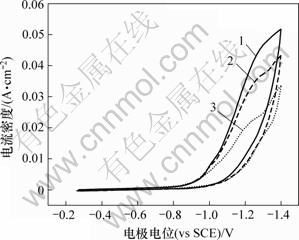
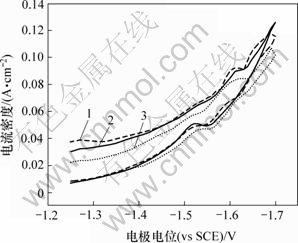
图6所示为第1个峰的循环伏安图。可见,峰电流随着正向扫描速度的增大而增大;在反向扫描中没有出现对应的氧化峰,表明Cr3+的第1步还原反应是不可逆的。从图5可知,扫描速度较大时,反向曲线上没有出现氧化峰;扫描速度较小时,反向曲线上却出现了氧化峰,可以初步确定第2步反应是准可逆的。图7所示为第2个峰的循环伏安曲线。可见,在不同扫描速度下,所得到的循环伏安曲线在-1.6 V附近出现了还原峰,反向扫描时在-1.55 V附近出现了氧化峰;还原峰和氧化峰的峰电位随扫描速度的变化很小,表明Cr3+的第2步还原反应接近于可逆过程[16]。图中还显示Cr3+的还原电位较高,表明在Cr3+沉积过程中不可避免地有大量H2析出,因此,阴极电流效率不会很高。
扫描速度/(V?s-1): 1―0.1; 2―0.05; 3―0.02
图6 第1个还原峰的循环伏安曲线
Fig.6 Cyclic voltammogram curves of the first reduction peak
扫描速度/(V?s-1): 1―0.07; 2―0.05; 3―0.03
图7 第2个还原峰的循环伏安曲线
Fig.7 Cyclic voltammogram curves of the second reduction peak
表1所示为Cr3+的第2个还原峰的特征参量,其中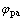 和
和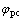 分别为氧化峰和还原峰的峰电位。根据可逆电极过程的特征,氧化峰和还原峰的电位差为
分别为氧化峰和还原峰的峰电位。根据可逆电极过程的特征,氧化峰和还原峰的电位差为 mV(25 ℃),可计算出Cr3+第2步还原反应的电子数n为1,则第1步还原反应的电子数应为2。
mV(25 ℃),可计算出Cr3+第2步还原反应的电子数n为1,则第1步还原反应的电子数应为2。
表1 Cr3+ 第2个还原峰的特征参量
Table 1 Parameters of the second reduction peak of Cr3+

由以上分析可知,Cr3+在阴极电沉积受电化学步骤控制,分2步进行放电:第1步得到2个电子,第2步得到1个电子。Cr3+在阴极上的还原反应历程为:
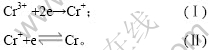
2.4 Cr3+电沉积的交流阻抗
当温度为25 ℃、正弦波的振幅为5 mV、频率为100 kHz至1 Hz时,Cr3+在不同电位下电沉积的交流阻抗复数平面图如图8所示。
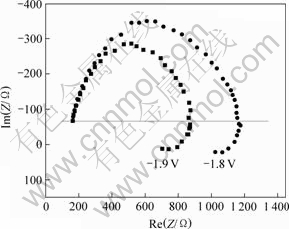
图8 Cr3+电沉积的交流阻抗复数平面图
Fig.8 AC impedance of Cr3+electrodeposition
由图8可见,Cr3+电沉积反应的交流阻抗复数平面图由位于第1象限和第4象限的2个半圆形圆弧组成,半圆形圆弧表明Cr3+的电沉积受电化学步骤控制;2个半圆形圆弧表明电极反应分2步进行。位于第4象限的半圆形圆弧表明Cr3+在电沉积过程中有电活性的中间产物吸附在电极表面,吸附状态对第1步反应(Ⅰ)的影响比对第2步反应(Ⅱ)的影响要大,并且吸附电阻是以电感的形式存在。位于第1象限的半圆形圆弧的直径远大于第4象限的半圆形圆弧的直径,说明第1步的电化学反应电阻较大,反应速度较慢,第1步反应为速度控制步骤[17]。从图8还发现,随着电极电位的负移,半圆形圆弧的直径缩小,电化学反应电阻减小,反应速度加快,说明较负的电极电位有利于Cr3+的电沉积。
2.5 Cr3+电沉积的动力学参数和动力学方程
2.5.1 阴极表观传递系数 和阳极表观传递系数
和阳极表观传递系数
25 ℃时测量Cr3+的稳态阴极极化曲线,其对应的Tafel曲线如图9所示,Tafel斜率为0.121,截距为0.568,由斜率求得阴极表观传递系数 =0.49,由截距求得交换电流密度J0=2.02×10-5 A/cm2。 同理,根据Cr3+溶液中Cr阳极溶解的稳态极化曲线和对应的阳极Tafel斜率(0.024),得到阳极过程的表观传递系数
=0.49,由截距求得交换电流密度J0=2.02×10-5 A/cm2。 同理,根据Cr3+溶液中Cr阳极溶解的稳态极化曲线和对应的阳极Tafel斜率(0.024),得到阳极过程的表观传递系数 =2.455。
=2.455。

图9 Cr3+的阴极Tafel曲线
Fig.9 Cathodic Tafel curve of Cr3+
2.5.2 控制步骤化学计量数
根据阴极表观传递系数 和阳极表观传递系数
和阳极表观传递系数 ,得到化学计量数
,得到化学计量数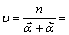
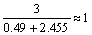 ,表明Cr3+ 在电沉积过程中,总反应进行1次,控制步骤的反应只进行1次。
,表明Cr3+ 在电沉积过程中,总反应进行1次,控制步骤的反应只进行1次。
2.5.3 Cr3+电沉积反应级数
25 ℃时改变溶液中Cr3+的活度,分别测量对应的阴极稳态极化曲线。在Tafel区的某一恒定电位下,得到Cr3+的活度 与电流密度J的对数关系如图10所示。图中直线斜率为1.14,所以,Cr3+电沉积的阴极反应级数为1.14≈1。同理,改变溶液中Cr3+的活度,分别测量阳极稳态极化曲线,在Tafel区的某一恒定电位下,得到Cr3+的活度与电流密度的对数关系,由直线的斜率求出Cr3+的阳极反应级数为0.045≈0。
与电流密度J的对数关系如图10所示。图中直线斜率为1.14,所以,Cr3+电沉积的阴极反应级数为1.14≈1。同理,改变溶液中Cr3+的活度,分别测量阳极稳态极化曲线,在Tafel区的某一恒定电位下,得到Cr3+的活度与电流密度的对数关系,由直线的斜率求出Cr3+的阳极反应级数为0.045≈0。
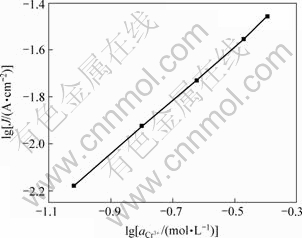
图10  ~lg J曲线
~lg J曲线
Fig.10  -lg J curve
-lg J curve
2.5.4 Cr3+的扩散系数D0
对于不可逆电极反应,循环伏安曲线的峰电流密度与扫描速度的关系为[13] : ?
? 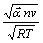 ,其中:A为电极面积,cm2;
,其中:A为电极面积,cm2; 为反应物的本体浓度,mol/cm3;D0为扩散系数,cm2/s;v为扫描速度,V/s。当扫描速度为0.1 V/s,温度为25 ℃时,Cr3+的循环伏安曲线上第1个阴极峰电流密度Jp为5.519×10-2 A/cm2,阴极表观传递系数
为反应物的本体浓度,mol/cm3;D0为扩散系数,cm2/s;v为扫描速度,V/s。当扫描速度为0.1 V/s,温度为25 ℃时,Cr3+的循环伏安曲线上第1个阴极峰电流密度Jp为5.519×10-2 A/cm2,阴极表观传递系数 为0.49,求得Cr3+的扩散系数D0为1.606
为0.49,求得Cr3+的扩散系数D0为1.606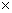 10-5 cm2/s。
10-5 cm2/s。
2.5.5 Cr3+的电化学反应动力学方程
根据上述反应历程,可推导出Cr3+的电化学动力学方程为

从中求得阴极Tafel斜率为0.118,表观传递系数 =0.5,阴极过程中Cr3+的反应级数为1;阳极Tafel斜率为0.024,表观传递系数
=0.5,阴极过程中Cr3+的反应级数为1;阳极Tafel斜率为0.024,表观传递系数 =2.5,阳极过程Cr3+的反应级数为0,理论值与实验值相吻合。
=2.5,阳极过程Cr3+的反应级数为0,理论值与实验值相吻合。
3 结 论
a. Cr3+在柠檬酸水溶液体系中的电沉积反应为电化学步骤控制过程,无前置转化反应存在。
b. Cr3+的电沉积分2步进行:第1步为速度控制步骤,得到2个电子,为不可逆过程;第2步得到1个电子,为准可逆过程。
c. 在Cr3+电沉积过程中有电活性的中间产物吸附在电极表面,吸附状态对第1步反应的影响比对第2步反应的影响大,吸附电阻以电感的形式存在。
d. Cr3+在电沉积过程中阴极过程和阳极过程的表观传递系数分别为0.49和2.455,化学计量数为1,阴极反应级数为1,阳极反应级数为0,扩散系数为1.606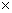 10-5 cm2/s,电化学动力学方程为
10-5 cm2/s,电化学动力学方程为
J=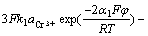
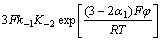 。
。
参考文献:
[1] 王先友, 蒋汉瀛. 甲酸盐型三价铬电镀液电化学研究[J].表面技术, 1995, 24(4): 17-20.
WANG Xian-you, JIANG Han-ying. Electrochemical study of trivalent chromium plating solution containging formate[J]. Surface Technology, 1995, 24(4): 17-20.
[2] 李惠东, 李惠琪, 段淑贞. Cr3+离子存在形态对其阴极过程的影响[J]. 中国有色金属学报, 1998, 8(1): 11-14.
LI Hui-dong, LI Hui-qi, DUAN Shu-zhen. Effect of species of Cr3+ ion on its cathodic reduction[J]. The Chinese Journal of Nonferrous Metals, 1998, 8(1): 11-14.
[3] 何湘柱, 龚竹青, 蒋汉瀛. 三价铬离子水溶液电沉积非晶态铬的电化学[J]. 中国有色金属学报, 2000, 10(1): 95-99.
HE Xiang-zhu, GONG Zhu-qing, JIANG Han-ying. Electrodeposition of amorphous chromium from Cr(Ⅲ) aqueous solution[J]. The Chinese Journal of Nonferrous Metals, 2000, 10(1): 95-99.
[4] Song Y B, Chin D T. Current efficiency and polarization behavior of trivalent chromium electrodeposition process[J]. Electrochimica Acta, 2002, 48: 349-356.
[5] Mohanty U S, Tripathy B C, Singh P, et al. Effect of Cr3+ on the electrodeposition of nickel from acidic sulfate solutions[J]. Minerals Engineering, 2002, 15: 531-537.
[6] Dolati A G, Ghorbani M, Afshar A. The electrodeposition of quaternary Fe-Cr-Ni-Mo alloys from the chloride-complexing agents electrolyte. Part Ⅰ. Processing[J]. Surface and Coatings Technology, 2003, 166: 105-110.
[7] WANG Feng, Watanabe T. Preparation and characterization of the electrodeposited Fe-Cr alloy film[J]. Materials Science and Engineering A, 2003, 349: 183-190.
[8] 何湘柱, 曾振欧, 彭荣华, 等. 三价铬电沉积非晶态合金的研究[J]. 华南理工大学学报, 2003, 31(3): 15-20.
HE Xiang-zhu, ZENG Zhen-ou, PENG Rong-hua, et al. Research on the electrodeposition of amorphous Fe-Ni-Cr alloy in trivalent chromium aqueous[J]. Journal of South China University of Technology, 2003, 31(3): 15-20.
[9] 吴慧敏, 康健强, 左正忠, 等. 全硫酸盐体系三价铬电镀铬的研究[J]. 武汉大学学报: 理学版, 2004, 50(2): 187-191.
WU Hui-min, KANG Jian-qiang, ZUO Zheng-zhong, et al. Study of electroplating with trivalent chromium in sulfate system[J]. J Wuhan Univ: Nat Sci Ed, 2004, 50(2): 187-191.
[10] 杨余芳, 龚竹青, 邓丽元, 等. 三价铬镀液中电沉积纳米晶体Fe-Ni-Cr合金箔[J]. 中南大学学报: 自然科学版, 2006, 37(3): 509-515.
YANG Yu-fang, GONG Zhu-qing, DENG Li-yuan, et al. Electrodeosition of nanocrystalline Fe-Ni-Cr alloy foil in trivalent chromium bath[J]. J Cent South Univ: Science and Technology, 2006, 37(3): 509-515.
[11] 邓姝皓, 龚竹青, 易丹青, 等. 三价铬还原电沉积机理[J]. 中南大学学报: 自然科学版, 2005, 36(2): 213-218.
DENG Shu-hao, GONG Zhu-qing, YI Dan-qing, et al. Electrochemical mechanism of trivalent chromium electrodeposition[J]. J Cent South Univ: Science and Technology, 2005, 36(2): 213-218.
[12] Szynkarczuk J, Drela I, Kubicki J. Electrochemical behaviour of chromium (Ⅲ) in the presence of formic acid[J]. Electrochimica Acta, 1989, 34(3): 399-403.
[13] Hsieh A K, EE Y H, Chen K N. Electrochemistry of chromium deposition from thiocyanato trivalent system[J]. Metal Finishing, 1993, 91(4): 53-57.
[14] 龚竹青. 理论电化学导论[M]. 长沙: 中南工业大学出版社, 1997: 226-391.
GONG Zhu-qing. Introduction to theoretical electro- chemistry[M]. Changsha: Central South University of Technology Press, 1997: 226-391.
[15] 舒余德, 陈白珍. 冶金电化学研究方法[M]. 长沙: 中南工业大学出版社, 1990: 152.
SHU Yu-de, CHEN Bai-zhen. Methods of metallurgical electrochemistry[M]. Changsha: Central South University of Technology Press, 1990: 152.
[16] 田昭武. 电化学研究方法[M]. 北京: 科学出版社, 1984: 234-240.
TIAN Zhao-wu. Study methods of electrochemistry[M]. Beijing: Science Press, 1984: 234-240.
[17] 曹楚南, 张鉴清. 电化学阻抗谱导论[M]. 北京: 科学出版社, 2002: 45-52.
CAO Chu-nan, ZHANG Jian-qing. An introduction to electrochemical impedance spectroscopy[M]. Beijing: Science Press, 2002: 45-52.
收稿日期:2007-05-08;修回日期:2007-06-26
基金项目:湖南省教育厅资助科研项目(07C698);湘南学院资助课题(2007Z012)
作者简介:杨余芳(1967-),女,湖南邵东人,副教授,博士,从事材料与电化学研究
通信作者:杨余芳,女,副教授,博士;电话:0735-2653128;E-mail: zzyufang@163.com