Characterization of acylated homoserine lactone derivatives and their influence on biofilms of Acidithiobacillus ferrooxidans BY-3 under arsenic stress
来源期刊:中南大学学报(英文版)2020年第1期
论文作者:李红玉 高启禹 唐德平 宋鹏 周剑平
文章页码:52 - 63
Key words:acetylated homoserine lactone; chemical synthesis; Acidithiobacillus ferrooxidans; Quorum sensing; arsenic contamination
Abstract: Quorum sensing is one kind of cell-to-cell signalling system among microorganisms that works in response to their population density via autoinducers exemplified by AHL and oligopeptides. In this study, fourteen AHL derivatives were synthesised by a chemical synthesis method, and two types of AHL derivatives were measured and screened by crystal violet staining assay, which have more obvious inhibitory effects on A. ferrooxidans biofilms under arsenic environment. Their structures were verified through IR and NMR identification. The morphological changes of A. ferrooxidans under the influence of the AHL derivatives were compared. In addition, the effects of AHL derivatives (0.1 μg/mL and 1 μg/mL) on membrane formation of A. ferrooxidans under high concentration of arsenic resistance (1,600 mg/L) were explored. Solid experimental data firstly showed that a portion of logarithmic microorganisms were ruptured under the effect of high arsenic concentration. Secondly, the volume of the cell shrank and the number of extracellular polymeric substances decreased after the addition of the AHL derivatives at high concentrations. Therefore, we found here that two derivatives used at concentrations of 0.1 μg/mL and 1 μg/mL accompanied with high concentration of arsenic can both effectively restrict biofilms formation by A. ferrooxidans.
Cite this article as: GAO Qi-yu, TANG De-ping, SONG Peng, ZHOU Jian-ping, LI Hong-yu. Characterisation of acylated homoserine lactone derivatives and their influence on biofilms of Acidithiobacillus ferrooxidans BY-3 under arsenic stress [J]. Journal of Central South University, 2020, 27(1): 52-63. DOI: https://doi.org/10.1007/s11771-020- 4277-2.

J. Cent. South Univ. (2020) 27: 52-63
DOI: https://doi.org/10.1007/s11771-020-4277-2
GAO Qi-yu(高启禹)1, 2, TANG De-ping(唐德平)1, SONG Peng(宋鹏)1,ZHOU Jian-ping(周剑平)3, LI Hong-yu(李红玉)1, 4
1. Institute of Microbiology, School of Life Sciences, Lanzhou University, Lanzhou 730000, China;
2. Department of Life Science and Technology, Xinxiang Medical University, Xinxiang 453003, China;
3. Institute of Biology, Gansu Provincial Academy of Sciences, Lanzhou 730000, China;
4. Institute of Microbiology and Biochemical Pharmacy, School of Pharmaceutics, Lanzhou University, Lanzhou 730000, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: Quorum sensing is one kind of cell-to-cell signalling system among microorganisms that works in response to their population density via autoinducers exemplified by AHL and oligopeptides. In this study, fourteen AHL derivatives were synthesised by a chemical synthesis method, and two types of AHL derivatives were measured and screened by crystal violet staining assay, which have more obvious inhibitory effects on A. ferrooxidans biofilms under arsenic environment. Their structures were verified through IR and NMR identification. The morphological changes of A. ferrooxidans under the influence of the AHL derivatives were compared. In addition, the effects of AHL derivatives (0.1 μg/mL and 1 μg/mL) on membrane formation of A. ferrooxidans under high concentration of arsenic resistance (1,600 mg/L) were explored. Solid experimental data firstly showed that a portion of logarithmic microorganisms were ruptured under the effect of high arsenic concentration. Secondly, the volume of the cell shrank and the number of extracellular polymeric substances decreased after the addition of the AHL derivatives at high concentrations. Therefore, we found here that two derivatives used at concentrations of 0.1 μg/mL and 1 μg/mL accompanied with high concentration of arsenic can both effectively restrict biofilms formation by A. ferrooxidans.
Key words: acetylated homoserine lactone; chemical synthesis; Acidithiobacillus ferrooxidans; Quorum sensing; arsenic contamination
Cite this article as: GAO Qi-yu, TANG De-ping, SONG Peng, ZHOU Jian-ping, LI Hong-yu. Characterisation of acylated homoserine lactone derivatives and their influence on biofilms of Acidithiobacillus ferrooxidans BY-3 under arsenic stress [J]. Journal of Central South University, 2020, 27(1): 52-63. DOI: https://doi.org/10.1007/s11771-020- 4277-2.
1 Introduction
Quorum sensing (QS) is a kind of signal communication mechanism found in microorganisms [1, 2]. Through secreting and releasing specific signal molecules, autoinducers (AIs), accompanying the change in specific gene expression, the density of floras is regulated when the concentration of AIs reaches a certain threshold [3, 4]. With the help of this unique mechanism, microorganisms can adapt to the changes around their ecological environment. Since the QS system was first described in Vibrio fischeri in the 1960 s [5], this kind of information transmission mechanism has been widely found among other species, such as different bacteria and fungi. However, the types of signal molecules have been found different in different microorgnisms. For instance, gram positive bacteria such as Staphylococcus aureus, Bacillussp and Streptococcus pneumoniae utilise modified cyclised oligopeptides as signal molecules [6], while acylated homoserine lactones (AHLs) generally serve as the AIs of gram negative bacteria including Escherichia coli, Pseudomonas aeruginosa and Deinococcus radiodurans [7]. In addition, quorum sensing phenomena with organic materials as pheromones such as farnesol and tyrosol, have also been discovered in fungi [8, 9]. The AHL signal molecules of Gram-negative bacteria have many similarities in their structures. So,artificial AHLs was synthesized. GESKE et al [10] and SANCHEZSANZ et al [11] found that 4-Br-PHL, an AHL molecular analogue, is an active leading molecule having inhibition effects on the QS systems of A. tumefaciens, P. aeruginosa, and V. fischeri. In addition, the artificial AHLs shows favorable inhibitory activity on biofilm formation by P. aeruginosa, Acidithiobacillus ferrooxidans (A. ferrooxidans), as the earliest discovered and most thoroughly studied eosinophilic Gram-negative bacterium, also has QS system which was initially discovered in A. ferrooxidans ATCC23270 [12, 13], and AI molecules of bacterium are a series of AHL molecules synthesised by LuxI-type enzymes [14]. These kinds through molecule constitute the QS system of A. ferrooxidans together with LuxR receptor proteins. Typical AHL molecule, contains an amine chain and an AHL ring, while different AHL derivatives are generated owing to the different numbers of carbon atoms contained in the amine chain of this kind of compound. Moreover, there are mainly three kinds of substituent groups, namely, hydrogen, hydroxyl, and carbonyl, on the third position of the amine chain. The number of carbon atoms and the type of substituent groups in the third position of the amine chain are together responsible for different regulatory functions of these compounds on the QS system of bacteria [15, 16].
Eosinophilic A. ferrooxidans is generally association with the acid mine drainage (AMD) generated after the mining of sulphide ores. Various heavy metal ions, such as Pb2+, As3+, As5+, Cu2+, Ni2+, Hg2+, Zn2+, etc. are released in AMD [17, 18]. AMD is the result of the oxidative dissolution of sulphide-rich metalliferous minerals under the combined action of water and oxygen, and especially in the presence of acidophilic microorganisms [19]. In this process, the bacteria existing in biofilms, instead of free bacteria, are the first to participate in the aerobic decomposition of ores [20]. Through observation under the TEM, early researchers found that there are plenty of corrosion pits distributed regularly on the surface of ores in the presence of A. ferrooxidans [21]. The phenomenon indicates that the bacterium tightly adheres onto the surface of ores and acquires energy by dissolving the ores, which supports the adhesion of bioleaching bacteria. After steadily adhering onto the surface of ores, the bacterium begins to grow and reproduce, and then generates signal substances. When the density of the bacterium reaches the threshold, the QS system of the bacterium is activated. In this environment, the bacterium secretes large amounts of EPS which is then inserted onto the surface of the bacterium, and biofilms form through a series of programmed processes including cell attachment, EPS production, microcolony formation, differentiation of mature biofilms and detachment. For this reason, reducing the formation of AMD by means of inhibiting the secretion of EPS by A. ferrooxidans using the AHL signal molecular analogues is an interesting approach. As we all know, high concentration of metal ions will cause more serious environmental pollution, including As3+ and As5+, etc [22]. Therefore, the treatment of arsenious AMD, has attracted significant research attention. In addition, owing to the existence of inorganic As3+ and As5+, which is of super-strong toxicity in arsenic pollution [23], at present, activated sludge adsorption, chemical reagents, and ion exchange are most commonly used to weaken and relieve the toxicity of arsenic in the treatment of arsenic pollution. In recent years, the discovery of various arsenic resistant microorganisms and related mechanisms provides a new scientific idea for the treatment of arsenic pollution. Synthesizing and screening compounds able to inhibit the QS system of A. ferrooxidans could reduce the amount of arsenic released in AMD and therefore reduce the environmental pollution of arsenic. In this way, the solution lies in the improvement of the ecological management and ecological protection.
In this study, 2-(4-bromophenyl)-N- (2-oxotetrahydrofuran-3-yl) acetamide (4-Br-PHL), which is an analogue of acyl-homoserine lactones (AHLs), has been widely studied in pathogenic bacteria as an efficient biofilm inhibitor [24]. There are nine natural AHL signaling molecules in the A. ferrooxidans QS system, which are structurally characterized by a homoserine lactone ring linked to an acyl side chain. The acyl side chain is an unsaturated fatty chain composed of 8-16 C atoms, on which the H atom is easily substituted by a functional group such as C=O and ―OH, so that the AHL signal molecule can be structurally modified by a substitution reaction [25]. Among the 14 compounds such as C-1, C-2, C-3 to C-14 synthesized in this way, C-1 and C-2 have more obvious inhibitory effects on A. ferrooxidans biomembranes under arsenic environment. The structures of C-1 and C-2 compounds were characterized by infrared spectroscopy (IR) and nuclear magnetic resonance (1H NMR and 13C NMR). Meanwhile, by using the synthesised C-1 and C-2 as QS signal molecule analogues, the influence of these two derivatives on the morphology of the A. ferrooxidans BY-3 under the stress of high arsenic concentration was explored using a transmission electron microscope (TEM). Moreover, the effects of C-1 and C-2 with different concentrations on the formation of the biofilms of the A. ferrooxidans BY-3 were compared by scanning using a microplate spectrophotometer and confocal laser microscopy. This study provides a potential application for the treatment of arsenic pollution using bioremediation.
2 Experimental
2.1 Reagents and equipment
Cyclopentane, 4-bromophenylacetic acid, and crystal violet were purchased from Beijing J&K Scientific Ltd, China. The (R)-(+)-α-amino-γ- butyrolactone hydrochloride, ethyl acetate, petroleum ether, and dichloromethane were bought from Shanghai Meryer Chemical Technology Co., Ltd. In addition, 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC・HCl) and 4-(dimethylamino) pyridine (DMAP) prepared by Sun Chemical Technology Co., Ltd, China were used. The sodium arsenite used was purchased from Chengdu Xiya Regent Co., Ltd, while L13152 fluorescence staining kits (LIVE/DEAD Bac Light Bacterial Viability Kits) and the colouring agent Alexa Fluor
Bac Light Bacterial Viability Kits) and the colouring agent Alexa Fluor 594 for extracellular polymer substances (EPS) were bought from USA Invitrogen Company. All the above regents were analytically pure and were used without further treatment in the experiment. The A. ferrooxidans BY-3 used was provided by the Institute of Microbiology of Lanzhou University, China.
594 for extracellular polymer substances (EPS) were bought from USA Invitrogen Company. All the above regents were analytically pure and were used without further treatment in the experiment. The A. ferrooxidans BY-3 used was provided by the Institute of Microbiology of Lanzhou University, China.
The NEXUS 670 infrared spectrometer and the Multiskan Spectrum microplate spectrophotometer were made by Thermo Nicolet Corporation and Thermo Fisher Scientific of USA. The PB -10 precise pH meter and the Esquire 6000 mass spectrometer were purchased from Sartorius Stedim Biotech and Bruker Scientific Technology Co.,Ltd, Germany, respectively. Furthermore, an upright FV1000MPE two-photon laser scanning confocal microscope (Olympus Optical Co., Ltd, Japan), and a Tecnai G2 TF20 TEM (FEI, The Netherlands) were used. Microorganisms were cultured in a 9K culture medium according to the preparation and culture methods proposed in a previous study [26].
2.2 Experimental process
Based on the method presented elsewhere [27], each compound (0.1 μg/mL and 1 μg/mL) was added to the bacterial suspension. The culture without any compound was used as a control. Afterwards, 150 μL of each bacterial suspension (1×108 cells/mL) was transferred to 96-well plates (Costar, USA) and the plates were incubated at 30 °C for 36 h without agitation. The biofilms formed on the plates were stained with 0.5% crystal violet for 15 min. Subsequently, 150 μL 33% acetic acid was added to each well and the OD570 nm was measured using a microplate reader (1500, Thermo, USA). Meanwhile, the effects of compounds on pre-formed A. ferrooxidans biofilms were evaluated, among the effects of compounds on biofilm formation showed that AHL derivatives C-1 and C-2 had higher efficiency (Figures 1 and 2).
2.3 Structural modification and synthesis of AHL derivatives C-1 and C-2
Chemical modifications of 4-Br-PHL were selected for the homoserine lactone ring, acyl side chain, and benzene ring. Fourteen types of QS signallingmolecules were synthesised and the activity of them was determined by crystal violet staining, the concentration dependence for inhibition of A. ferrooxidans biofilm formation by C-1, C-2, C-3 to C-14. Results showed that C-1 and C-2 compounds exerted significant inhibitory effects on the total biomass of their biofilms but not affecting the bacterial growth.
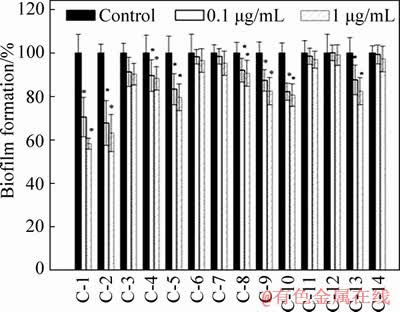
Figure 1 Concentration dependence for inhibition of A. ferrooxidans biofilm formation by C-1, C-2, C-3 to C-14 compounds. The total biomass of biofilms was determined by measuring the optical density at 570 nm. (Results are presented as the mean ± standard deviation (n=8))
The synthesis stage was started by adding 92 mg of EDC and 24 mg of DMAP, which were mixed to the stirred CH2Cl2 solution prepared using (R)-(+)-α-amino-γ-butyrolactone hydrochloride and 4-bromophenylacetic acid. Then, the mixture was stirred for 20 h at 25 °C and extracted using 100 mL of ethyl acetate to extract the mixture. Next, the mixture was washed using 1 mol/L of HCl solution and saturated NaCl solution, respectively. Meanwhile, eluent compounds was dried using MgSO4. Followed by ethyl acetate and petroleum ether with their volume ratio being 1.5:1 were used to elute the derivatives separated using SiO2 column chromatography, thus obtaining the synthesis of AHL derivatives C-1 and C-2.
2.4 Characterisation and analysis of compounds acquired
The structures of the synthesised compounds were analysed by Fourier transform infrared spectroscopy (FTIR) and NMR (1H NMR and 13C NMR). In addition, the physical properties, yields, and melting points of C-1 and C-2 were compared and analysed.
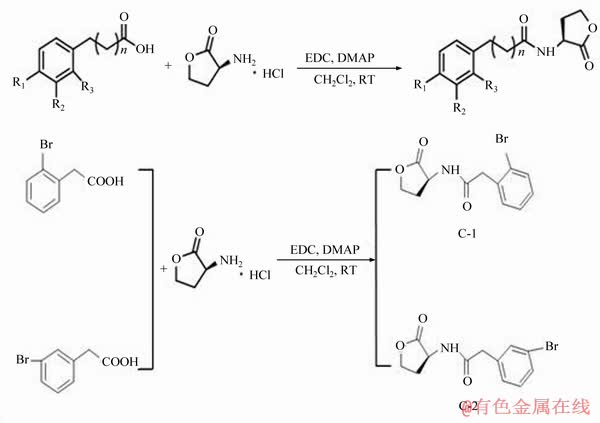
Figure 2 Synthesis method of No. 1-14 compounds and structural formula of C-1 and C-2
2.5 Culture and domestication of arsenic- resistant A. ferrooxidans BY-3
Arsenic-resistant A. ferrooxidans BY-3 was cultured and acclimated using the method described in previous study [28]. By continuously acclimating to the concentration of the sodium arsenite used in the culture medium, the final concentration of the sodium arsenite in culture medium reached 1600 mg/L.
2.6 Morphological changes in A. ferrooxidans BY-3
The A. ferrooxidans BY-3 samples were prepared using the method illustrated in relevant study [29]. A TEM was used to observe and compare bacterial morphologies. After the micro-organisms in the experimental groups grew to logarithmic phase, the culture solution of 500 mL was centrifuged (CR22G II, Japan) at 8500 r/min for 15 min. The supernatant was discarded and the pellet was transferred to a 10 mL centrifuge tube by suspending in 5 mL of distilled water and then further centrifuged at 1500 r/min for 5 min. The supernatant was collected and centrifuged at 10000 r/min for 10 min, and the bacteria were collected and prepared for TEM observation. The preparation steps are shown as follows: The specimen was first fixed with 2.5% glutaraldehyde in phosphate buffer (pH 7.0) for more than 4 h; washed three times in the phosphate buffer; then post fixed with 1% OsO4 in phosphate buffer (pH 7.0) for 1 h and washed three times in the phosphate buffer. Gradient dehydration was dehydrated by a graded series of ethanol (30%, 50%, 70%, 80%, 90%, 95% and 100%) for about 15 to 20 min at each step, transferred to the mixture of alcohol and isoamyl acetate (V:V=1:1) for about 30 min, then transferred to pure isoamyl acetate for about 1 h. In the end, the specimen was dehydrated in Hitachi (HCP-2, Japan). After being soaked and embedded, the samples were cut into less than 100 nm using a LK B-5 ultra-thin microtome. TEM observation was carried out after dyeing the samples with uranium acetate and lead for 30 and 10 min respectively.
2.7 Influences of C-1 and C-2 on formation of biofilms
In the 9K culture medium (containing 1600 mg/L NaAsO2), the logarithmic growth phase of A. ferrooxidans BY-3 was inoculated with 15% (V/V) volume. Then bacterial suspension was cultured at 30 °C and a rate of rotation of 150 r/min for 36 h, followed by the filtering separation of the bacterial fluid. Subsequently, the obtained cells were re-suspended, using a 2K (containing 9.96 g/L FeSO4) culture medium, until the concentration of bacterial suspension reached 1.0×108 cells/mL. Afterwards, C-1 and C-2 were added into the above bacterial suspension to make the final concentrations of C-1 and C-2 to 0.1 and 1.0 μg/mL, respectively. The culture without any AHL derivatives was used as a blank control. Afterwards, 150 μL of each bacterial solution was transferred to 96-well plates incubated at a constant temperature of 30 °C to determine the formations of the biofilms after 12, 24 and 48 h. After the liquid in the wells was pipetted into test tube, and 150 μL of 0.5% crystal violet was added to dye the obtained cells for 15 min at room temperature. Then, the liquid was removed and the dyed cells were thrice- washed using sterile water. Subsequently, 150 μL of 33% acetic acid was added to each well for 30 s, the additional liquid was rapidly removed and absorbed by filter papers. Furthermore, a microplate reader (1500, Thermo, USA) was used to compare the light absorption values of each well at the optical density (OD) of 570 nm.
2.8 Testing formed biofilms
According to the method mentioned previous section, 150 μL of corresponding bacterial solution was added to the 96-well tissue culture plate incubated for 36 h. Then, additional liquid was removed and the cells were thrice-washed using sterile water. Next, 150 μL of basal salt solution (3 g/L (NH4)2SO4, 0.50 g/L K2HPO4, 0.50 g/L MgSO4・7H2O, 0.10 g/L KCl and 0.01 g/L Ca(NO3)2) containing no Fe2+ and the final concentration of C-1 and C-2 reached 0, 0.1 and 1.0 μg/mL were added, respectively. Afterwards, the bacteria were cultured at a constant temperature of 30 °C to determine the formation conditions of the biofilms after 12, 24 and 48 h. The biofilms were dyed by the previous section.
2.9 Observation biofilm formation using confocal laser scanning microscopy
Sterilised circular glass sheets with a diameter of 6 mm were placed in each well of a 6-well tissue culture plate. Then 1 mL (containing 1600 mg/L NaAsO2) of bacterial suspension, at a concentration of 1×107cells/mL and C-1, C-2 were added to each well to be statically cultured at 30°C for 168 h, the final concentrations of C-1 and C-2 in the culture solution reached 0.1 and 1.0 μg/mL, respectively. Then 0.5 mL of sterile water was supplemented every 48 h. After culturing for 168 h, the obtained cells were washed and dyed using SYTO 9/Alexa Fluor 594 based on a double fluorescence staining method. After being sealed using glycerol with 90% (W/V), the variation of biofilms was compared using confocal laser scanning microscopy. The images were processed using the software Fluoview Ver. 3.1.1.9 (Olympus, Japan).
2.10 Data analysis and processing
The obtained data were analysed by the statistical software SPSS 21.0 and all data were presented as mean ± standard deviation (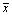 ±s). The laboratory data from tests were compared through single factor variance method.
±s). The laboratory data from tests were compared through single factor variance method.
3 Results and discussion
3.1 Characterization and analysis of target compounds
3.1.1 Physical properties of compounds
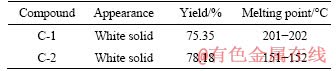
The physical properties of the compounds are listed in Table 1, which shows that the synthesised AHL derivatives were white solids and presented similar yields. However, their melting points were quite different. The melting point of C-1 was about 200 °C, while that of C-2 was 50 °C lower than the former.
Table 1 Appearance, yield, and melting point data of compounds C-1 and C-2
3.1.2 IR analysis of compounds
IR(KBr) vmax: C-1:3269(NH), 3057(C―H), 1781(C=O), 1473(C―C of benzene), 1147(C=O), 571(-Br of benzene); C-2:3282(NH), 3042(C―H), 1770(C=O),1470(C―C of benzene), 1167(C=O), 590(-Br of benzene) (Figure 3 and Table 2 ).
3.2 Morphological changes in A. ferrooxidans BY-3
The morphology of organisms, including environmental microorganisms, is supposed to correspondingly change under the impact of various ions. Studies confirmed that more glycocalyx was synthesized and preserved outside the outer membranes of cells, thus protecting cells against high potassium concentration stress through this thickened glycocalyx layer [30]. When A. ferrooxidans BY-3 was cultured in the 9K culture medium containing 1600 mg/L of sodium arsenite, its partial cells were ruptured owing to the toxic effect of As3+and therefore the cytoplasm diffused (Figure 4(d)). Nevertheless, with the addition of 1.0 μg/mL of C-1 and 1.0 μg/mL of C-2 to the culture solution, the cells shrank and therefore the extracellular polymer layer was thinned (Figure 4). The results indicated that the cell morphology changed from long rod-shaped to short rod-shaped after adding C-1 and C-2, to adapt to the environment with high concentration of As3+ (Figures 4(c) and (d)). Moreover, the amount of extracellular polymers decreased, which further weakened the adsorption of cells on sulphide ores. Thinning of extracellular polymers may affect the release of a large amount of other metal ions to the environment owing to the adsorption effect, and therefore reduced the likely environmental pollution [31, 32].
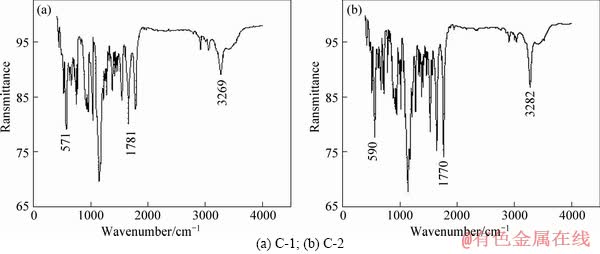
Figure 3 IR data of compounds C-1 and C-2:
Table 2 1H NMR and 13C NMR data of compounds C-1 and C-2
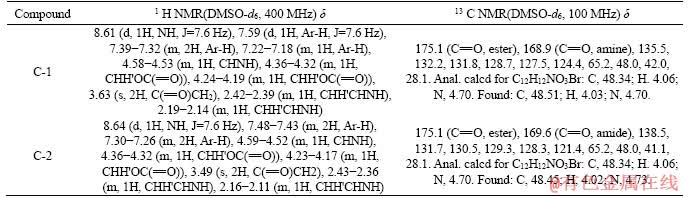
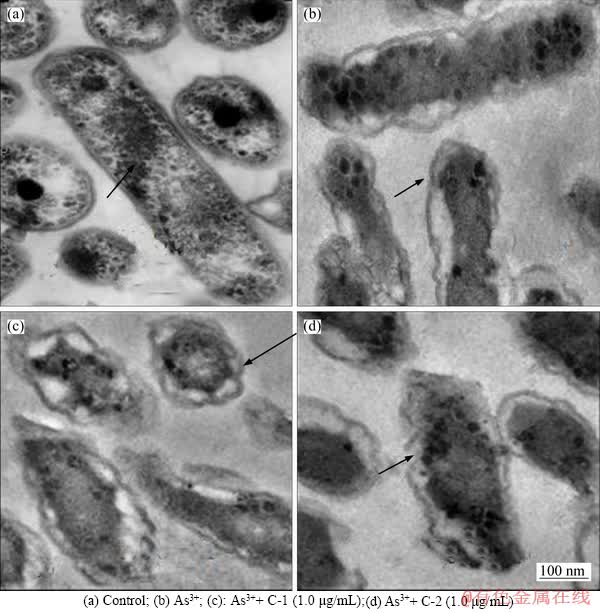
Figure 4 TEM photos of At. f-BY3 treated with different conditions:(Glycocalyx layer was indicated in arrow)
3.3 Influence of C-1 and C-2 on formation of biofilms
AMD presents an extreme condition which is not suitable for most forms of life, but many Fe- and S-oxidizing bacteria thrive in this environment and exist as biofilms [33]. Biofilm formation greatly promotes bacterial colonization on sulfide minerals, thus further enhances metal sulfide oxidation and plays a critical role in AMD production [34]. For this reason, the inhibition of biofilm development should be a promising strategy for controlling AMD at the source. In the culture process of microorganisms, 0.1 and 1.0 μg/mL of C-1 and C-2 were added to the 96-well plate, respectively. At the lag phase, since there were few bacteria, the AIs and the added analogues exerted no influence on the formation of biofilms; however, as the culture continued, the density of bacteria increased. It was found that after 36 h and 48 h of culture, compared with the blank control, the formation of biofilms in the C-1 and C-2 treated group was inhibited and showed statistical significance (p<0.05)(Figures 5(a) and (b)). This suggested that the two kinds of synthesised signal molecule analogues not only effectively restrain the QS of the A. ferrooxidans BY-3, but also play an important role in the treatment of heavy metal pollution.
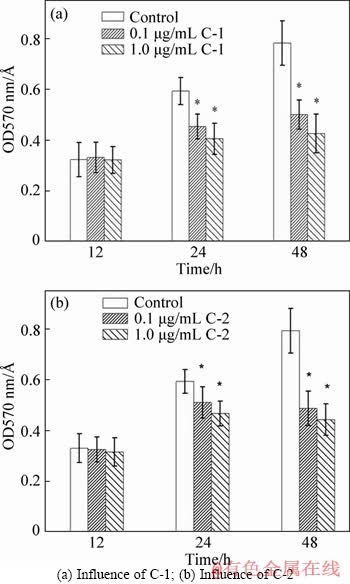
Figure 5 At. f-BY3 biofilm inhibition by C-1 and C-2 at different time:(Each group contains 1600 mg/L NaAsO2.*Significantly different (p<0.05) compared with the control)
3.4 Influence of C-1 and C-2 on formed biofilms of A. ferrooxidans BY-3
Arsenic (As) is ubiquitous in the natural environment, posing risks to human health in many parts of the world [35, 36]. Given the long-term presence of arsenic in mine waste water in the environment [37], the influence of the QS mechanism is possibly formed at an initial stage corresponding biofilms of A. ferrooxidans BY-3. Therefore, the effects of two tested concentrations (0.1 and 1.0 μg/mL) of C-1 and C-2 on biofilm formation at different times were studied (Figures 6(a) and (b)). The results showed that after 48 h of incubation, C-1 and C-2 significantly inhibited the formation of membranes (p<0.05) and also decomposed the biofilms formed at an initial stage. Therefore, the results showed that the inhibition and decomposition of biofilms are related to the concentrations of the AHL derivatives.
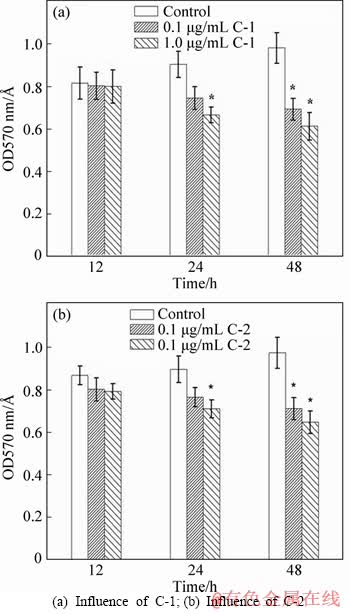
Figure 6 Pre-formed biofilms of At. f-BY3 inhibited by C-1 and C-2 at different time:(Each group contains 1600 mg/L NaAsO2.*Significantly different (p<0.05) compared with the control)
3.5 Examination of biofilms using CLSM
Biofilms, as a kind of complex three- dimensional colony structure, are generated owing to the coverage of microorganisms and other substances including water, protein, fat, amylum and extracellular polysaccharides (EPS) in the growth process of microbial populations [38, 39]. Observation and comparison of biofilms using the crystal violet staining method is a method of quantitative analysis used for total biofilms, while it fails to observe intuitively the spatial structure of biofilms. Therefore, using confocal laser scanning microscopy to compare the influence of C-1 and C-2 on the formation of the biofilms of A. ferrooxidans BY-3 from a microscopic perspective can further verify the inhibitory effect of target compounds on biofilms.
The green fluorescence signals for viable cells in the biofilms corresponding to the red fluorescence signals for polysaccharide (Figures 7 and 8). In the formation process of biofilms, the A. ferrooxidans BY-3 was treated using 0.1 and 1.0 μg/mL of C-1 and C-2, separately. Compared with the control group, the amount of EPS generated by the A. ferrooxidans BY-3 in the experimental groups decreased, so the results showed that the effects of C-1 and C-2 (1.0 μg/mL) on the formation of biofilms were more obvious. These results indicated that C-1 and C-2 can inhibit the biofilms formation and EPS of the A. ferrooxidans BY-3 on arsenic metabolism.
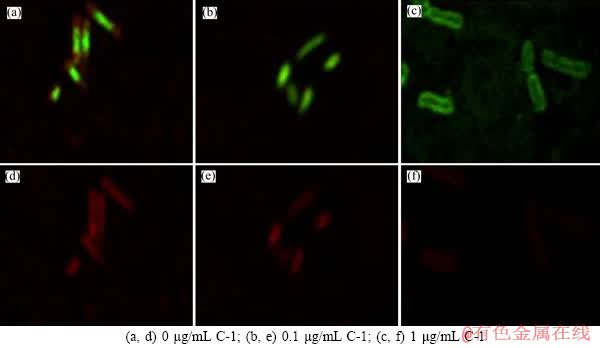
Figure 7 Biofilm images of At.f-BY3 (stained by SYTO 9 and EPS by Alexa Fluor 594 ) under CLSM grown under different C-1 concentrations after 168 h:((a, b, c) for attached cells, (d, e, f) for EPS, each group contains 1600 mg/L NaAsO2)
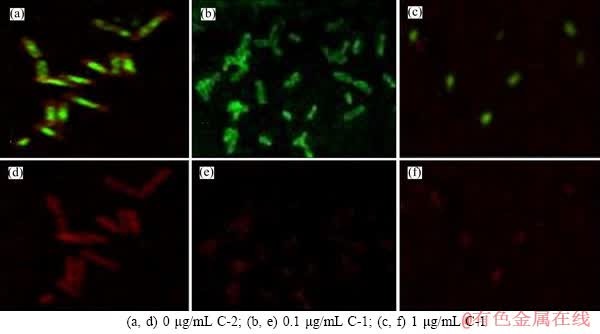
Figure 8 Biofilm images of At.f-BY3 (stained by SYTO 9 and EPS by Alexa Fluor 594 ) under CLSM grown under different C-2 concentrations after 168 h:((a, b, c) for attached cells, (d, e, f) for EPS, each group contains 1600 mg/L NaAsO2)
Electrolyte concentrations and medium compositions exert an important influence on biofilm formation [40, 41]. In the present study, divalent cations, such as Mg2+, Ca2+, Cu2+ and Zn2+, can both directly and indirectly, influence biofilm formation in various ways [42, 43]. In particular, the amount of extracellular polysaccharides decreased, which further weakened the adsorption of bacteria. Therefore, these results suggested that AHL derivatives accompanied with A. ferrooxidans BY-3 has potential influence on the release of a large amount of metal ions to the environment owing to the adsorption effect.
4 Conclusions
Based on the characterization and analysis of AHL signal molecule analogues, this study revealed that the AHL derivatives of C-1 and C-2 could not only inhibit biofilm formation and pre-formed biofilms, but also had the ability to decrease the EPS production. Meanwhile, the growth of the A. ferrooxidans BY-3 in the environment with high concentrations of arsenic was simulated. According to the results acquired, the variation in the morphology and biofilms of the A. ferrooxidans BY-3 under the effect of different concentrations of C-1 and C-2 was validated using two AHL derivatives. The experiment showed that under the concentrations of C-1 and C-2 (0.1 and 1.0 μg/mL), the volume of the A. ferrooxidans BY-3 shrank and therefore the extracellular polymer layer thinned. Meanwhile, the formation of biofilms was significantly inhibited at 36 h and 48 h (p<0.05). The results revealed that these AHL derivatives of C-1 and C-2 can interfere the biofilms formed. Synthesis of AHL derivatives accompanied with A. ferrooxidans provides a promising research prospect and far-reaching scientific significance for the bioremediation of arsenic pollution.
References
[1] ABISADO R G, BENOMAR S, KLAUS J R, DANDEKAR A A, CHANDLER J R. Bacterial quorum sensing and microbial community interactions [J]. Mbio, 2018, 9(3): e02331-17. DOI: 10.1128/mBio.02331-17.
[2] ALBERGHINIl S, POLONE E, CORICH V, CARLOT M, SENO F, TROVATO A. Consequences of relative cellular positioning on quorum sensing and bacterial cell-to-cell communication [J]. Fems Microbiology Letters, 2009, 292: 149-161. DOI: 10.1111/j.1574-6968.2008.01478.x.
[3] GALLOWAY W R, HODGKINSON J T, BOWDEN S D, WELCH M, SPRING D R. Quorum sensing in gram-negative bacteria: Small-molecule modulation of AHL and AI-2 quorum sensing pathways [J]. Chemical Reviews, 2011, 111(1): 28-67. DOI: 10.1021/cr100109t.
[4] SHOJIMA A, NAKAYAMA J. Quorum sensing in gram-positive bacteria: Assay protocols for Staphylococcal agr and Enterococcal fsr systems [J].Methods in Molecular Biology, 2014, 1147: 33-41. DOI: 10.1007/978-1-4939- 0467-9_3.
[5] NEALSON K H, PLATT T, HASTINGS J W. Cellular control of the synthesis and activity of the bacterial luminescent system [J]. Journal of Bacteriology, 1970, 104: 313-322. DOI: http://dx.doi.org/.
[6] LIAQAT I, BACHMANN R T, EDYVEAN R G. Type 2 quorum sensing monitoring, inhibition and biofilm formation in marine microrganisms [J].Current Microbiology, 2014, 68: 342-351. DOI: 10.1007/s00284-013-0484-5.
[7] LIN L, DAI S, TIAN B, LI T, YU J, LIU C. Dqsir quorum sensing-mediated gene regulation of the extremophilic bacteriumeinococcus radiodurans in response to oxidative stress [J].Molecular Microbiology, 2016, 100(3): 527-541. DOI: 10.1111/mmi.13331.
[8] NICKERSON K W, ATKIN A L, HORNBY J M. Quorum sensing in dimorphic fungi: Farnesol and beyond [J].Applied & Environmental Microbiology, 2006, 72(6): 3805-3813. DOI: 10.1128/AEM.02765-05.
[9] CHEN Y F, GAO K, LIN H, DONG Y B, WANG H, LI Y Z, HUO H X,CAO L X. Adsorption properties of microbe resistant to lead and zinc on Zn(2+) and Pb(2+) in acid mine drainage (AMD) [J]. Journal of Central South University: Science and Technology, 2013, 44(4): 1741-1746. (in Chinese)
[10] GESKE G D, O'NEILL J C, MILLER D M, MATTMANN M E, BLACKWELL H E. Modulation of bacterial quorum sensing with synthetic ligands: Systematic evaluation of N-acylated homoserine lactones in multiple species and new insights into their mechanisms of action [J]. Journal of the American Chemical Society, 2007, 129(44): 13613-13625. DOI: 10.1021/ja074135h.
[11] SANCHEZSANZ G, CROWE D, NICHOLSON A, FLEMING A, CAREY E, KELLEHER F. Conformational studies of Gram-negative bacterial quorum sensing acyl homoserine lactone (AHL) molecules: The importance of the n→π* interaction [J]. Biophysical Chemistry, 2018, 238: 16-21. DOI: 10.1016/j.bpc.2018.04.002.
[12] FARAH C, VERA M, MORIN D, JEREZ C A, GUILIANI N. Evidence for a functional quorum-sensing type ai-1 system in the extremophilic bacterium Acidithiobacillus ferrooxidans [J]. Applied & Environmental Microbiology, 2005, 71: 7033-7040. DOI: 10.1128/AEM.71.11.7033- 7040.2005.
[13] FENG Y L, WANG H J, LI H R, CHEN X P, DU Z W, KANG J X. Effect of iron transformation on acidithiobacillus ferrooxidans bio-leaching of clay vanadium residue [J].Journal of Central South University, 2019, 26: 796-805. DOI: https://doi.org/10.1007/s 11771-019-4049-z.
[14] VAN H R, MOONS P, AERTSEN A, JANSEN A, VANOIRBEEK K, DAYKIN M. Characterization of a luxI/luxR-type quorum sensing system and N-acyl- homoserine lactone-dependent regulation of exo-enzyme and antibacterial component production in Serratia plymuthica RVH1 [J]. Research in Microbiology, 2007, 158(2): 150-158. DOI: 10.1016/j.resmic.2006.11.008.
[15] KAUR A, CAPALASH N, SHARMA P. Quorum sensing in thermophiles: Prevalence of autoinducer-2 system [J].BMC Microbiology, 2018, 18(1): 62-71. DOI: 10.1186/s12866- 018-1204-x.
[16] MANGANO S, CARUSO C, MICHAUD L, GIUDICE A L. First evidence of quorum sensing activity in bacteria associated with antarctic sponges [J].Polar Biology, 2018, 5: 1-11. DOI: 10.1007/s00300-018-2296-3.
[17] GAO Q, TANG D, SONG P, ZHOU J, LI H. Bio-adsorption and bio-transformation of arsenic by acidithiobacillus ferrooxidans by3 [J].International Microbiology,2018, 21(4): 207-214. DOI: 10.1007/s10123-018-0017-y.
[18] TANG D, DUAN J, GAO Q, ZHAO Y, LI Y, CHEN P. Strand-specific rna-seq analysis of the Acidithiobacillus Ferrooxidans transcriptome in response to magnesium stress [J].Archives of Microbiology, 2018, 200(7): 1025-1035. DOI: 10.1007/s00203-018-1503-5.
[19] KUANG J L, HUANG L N, CHEN L X, HUA Z S, LI S J, HU M. Contemporary environmental variation determines microbial diversity patterns in acid mine drainage [J].ISME Journal, 2013, 7(5): 1038-1050. DOI: 10.1038/ismej.2012. 139.
[20] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review part A: Progress in bioleaching: Fundamentals and mechanisms of bacterial metal sulfide oxidation [J]. Applied Microbiology & Biotechnology, 2003, 63(3): 239-248. DOI: 10.1007/s00253-003-1448-7.
[21] TRIBUTSCH H. Direct versus indirect bioleaching [J]. Hydrometallurgy, 2001, 59(2): 177-185. DOI: 10.1016/ S0304-386X(00)00181-X.
[22] GHOSH S, MOITRA M, WOOLVERTON C J, LEFF L G. Effects of remediation on the bacterial community of an acid mine drainage impacted stream [J]. Canadian Journal of Microbiology, 2012, 58(11): 1316-1326. DOI: 10.1139/ w2012-110.
[23] MAILLOT F, MORIN G, JUILLOT F, BRUNEEL O, CASIOT C, ONA-NGUEMA. Structure and reactivity of As(III)- and As(V)-rich schwertmannites and amorphous ferric arsenate sulfate from the Carnoulès acid mine drainage, France: Comparison with biotic and abiotic model compounds and implications for As remediation [J]. Geochimica Et Cosmochimica Acta, 2013, 104: 310-329. DOI: 10.1016/j.gca.2012.11.016.
[24] O'BRIEN K T, NOTO J G, NEILL L, PEREZ L J. Potent irreversible inhibitors of LasR quorum sensing in pseudomonas aeruginosa [J]. ACS Medicinal Chemistry Letters, 2014, 6: 162-167. DOI: 10.1021/ml500459f.
[25] ZHAO Y, CHEN P, NAN W, ZHI D, LIU R, LI H. The use of (5z)-4-bromo-5-(bromomethylene)-2(5h)-furanone for controlling acid mine drainage through the inhibition of acidithiobacillus ferrooxidans biofilm formation [J].Bioresource Technology, 2015, 186, 52-57. DOI: 10.1016/j.biortech.2015.02.017.
[26] NAN W B, ZHI D J, LENG F F, YAN L, CHEN P, LI H. Quorum-sensing system in Acidithiobacillus ferrooxidans involved in its resistance to Cu2+ [J]. Letters in Applied Microbiology, 2011, 53: 84-91. DOI: 10.1111/j.1472- 765X.2011.03066.x.
[27] MORKUNAS B, GALLOWAY W R, WRIGHT M, IBBESON B M, HODGKINSON J T, O’CONNELLK M G. Inhibition of the production of the pseudomonas aeruginosa virulence factor pyocyanin in wild-type cells by quorum sensing autoinducer-mimics [J].Organic & Biomolecular Chemistry,2012, 10(42): 8452. DOI: 10.1039/c2ob26501j.
[28] YAN L, YIN H H, ZHANG S A, LENG F F, NAN W B, LI H Y. Biosorption of inorganic and organic arsenic from aqueous solution by Acidithiobacillus ferrooxidans BY-3 [J].Journal of Hazardous Materials, 2010, 178(1): 209-217. DOI: 10.1016/j.jhazmat.2010.01.065.
[29] ROJAS-CHAPANA J A, GIERSIG M, TRIBUTSCH H. The path of sulfur during the bio-oxidation of pyrite by Thiobacillus ferrooxidans [J].Fuel, 1996, 75: 923-930. DOI: 10.1016/0016-2361(96)00057-9.
[30] OUYANG J, GUO W, LI B, GU L, ZHANG H, CHEN X. Proteomic analysis of differential protein expression in Acidithiobacillus ferrooxidans cultivated in high potassium concentration [J].Microbiological Research, 2013, 168(7): 455-460. DOI: 10.1016/j.micres.2013.01.007.
[31] CHAKRAVARTY R, MANNA S, GHOSH A K, BANERJEE P C. Morphological changes in an strain in response to heavy metal stress [J]. Research Journal of Microbiology, 2005, 2(10): 742-748. DOI: 10.3923/jm.2007.742.748.
[32] SONG B, LEFF L G. Influence of magnesium ions on biofilm formation by Pseudomonas fluorescens [J]. Microbiological Research, 2006, 161: 355-361. DOI: 10.1016/j.micres. 2006.01.004.
[33] SANCHEZ-ANDREA I, SANZ J L, BIJMANS M F, STAMS A J. Sulfate reduction at low pH to remediate acid mine drainage [J]. J Hazard Mater, 2014, 269: 98-109. DOI: 10.1016/j.jhazmat.2013.12.032.
[34] GOLTSMAN D S, COMOLLI L R, THOMAS B C, BANFILED J F. Community transcriptomics reveals unexpected high microbial diversity in acidophilic biofilm communities [J]. The ISME Journal, 2014, 9(4): 1014-1023. DOI: 10.1038/ismej.2014.200.
[35] HETTICK B E, CANAS-CARRELL J E, FRENCH A D, KLEIN D M. Arsenic: A review of the element’s toxicity, plant interactions, and potential methods of remediation [J].Journal of Agricultural & Food Chemistry, 2015, 63: 7097-7107. DOI: 10.1021/acs.jafc.5b02487.
[36] AKIHIDE K, MASAHIKO K. Arsenic release from marine sedimentary rock after excavation from urbanized coastal areas: Oxidation of framboidal pyrite and subsequent natural suppression of arsenic release [J].Science of the Total Environment, 2019, 670: 7520759. DOI: https://doi.org/10. 1016/j.scitotenv.2019.03.217.
[37] NOOKONGBUT P, KANTACHOTE D, MEGHARAJ M. Arsenic contamination in areas surrounding mines and selection of potential as-resistant Purple nonsulfur bacteria for use in bioremediation based on their detoxification mechanisms [J].Annals of Microbiology, 2016, 1: 1-11. DOI: 10.1007/s13213-016-1229-z.
[38] ANNOUS B A, FRATAMICO P M, SMITH J L. Quorum sensing in biofilms: Why bacteria behave the way they do [J].Journal of Food Science, 2009, 74: 24-37. DOI: 10.1111/j.1750-3841.2008.01022.x.
[39] LI Y H, TIAN X. Quorum sensing and bacterial social interactions in biofilms [J]. Sensors, 2012, 12(12): 2519-2538. DOI: 10.3390/s120302519.
[40] STOODLEY P, DODDS I, BOYLE J D. Influence of hydrodynamics and nutrients on biofilm structure [J]. Journal of Applied Microbiology, 1998, 85: 19S-28S. DOI: 10.1111/ j.1365-2672.1998.tb05279.x.
[41] HAORAN P, VALERIE L, ETIENNE P, GILLE S H. The influence of porous structure and biofilm on the hydrodynamics of two types of trickle filters [J]. Chemical Engineering Journal, 2013, 231: 163-171. DOI: 10.1016/ j.cej.2013.06.115.
[42] REHMAN Y, RIZVI F Z, FAISAL M, HASNAIN S. Arsenic and chromium reduction in co-cultures of bacteria isolated from industrial sites in Pakistan [J]. Microbiology, 2013, 82(4): 428-433. DOI: 10.1134/S0026261713040188.
[43] AKHMETOVA G R, MARTYKANOVA D S, RUDAKOVA N L. Influence of divalent metal ions on biofilm formation byBacillus subtilis [J]. BioNano Science, 2019(8): 1-7. DOI: 10.1007/s12668-019-00621-3.
(Edited by HE Yun-bin)
中文导读
高丝氨酸内酯衍生物的合成表征及在砷胁迫下对氧化亚铁硫杆菌BY-3生物膜形成的影响
摘要:群体感应作为微生物间的信号传递系统,通常借助微生物产生的诱导分子高丝氨酸内酯(AHL)或寡肽来响应种群密度。本研究通过化学合成法合成了14种AHL衍生物,采用结晶紫染色法筛选证实两种衍生物在砷环境下对氧化亚铁硫杆菌的生物膜抑制效应更加明显,并通过红外光谱(IR)及核磁共振(1H NMR、13C NMR)对其进行了结构表征,借助透射电镜分析了菌种形态的变化,同时探讨了两种衍生物在0.1 μg/mL及1 μg/mL浓度下对高砷(1600 mg/L)胁迫下的氧化亚铁硫杆菌生物膜形成的影响。研究结果表明在高砷环境下培养至对数期的氧化亚铁硫杆菌菌体出现结构改变,且加入高浓度高丝氨酸内酯衍生物后,菌体体积变小,胞外聚合物降低,同时0.1 μg/mL及1 μg/mL浓度的两种AHL衍生物皆能有效抑制氧化亚铁硫杆菌生物膜的产生。
关键词:高丝氨酸内酯;化学合成;氧化亚铁硫杆菌;群体感应;砷污染
Foundation item: Project(31660026) supported by the National Natural Science Foundation of China; Project(lzujbky-2016-152) supported by the National Basic Research Program of China
Received date: 2018-11-14; Accepted date: 2019-08-22
Corresponding author: LI Hong-yu, PhD, Professor; Tel: +86-18993168234; E-mail: lihy@lzu.edu.cn; ORCID: 0000-0001-5521-8005

