REE behavior and effect factors in AMD-type acidic groundwater at sulfide tailings pond, BS nickel mine, W.A.
ņī‘ī∆ŕŅĮ£ļ÷–Ļķ”–…ęĹū Ű—ßĪ®(”Ęőńįś)2008ńÍĶŕ4∆ŕ
¬Řőń◊ų’Ŗ£ļņ◊Ńľ∆ś ňőī»į≤ –ĽŌśņž ņÓ—řļž
őń’¬“≥¬Ž£ļ955 - 955
Key words£ļREE pattern; acid mine drainage; sulfide tailings; groundwater contamination
Abstract: AMD(Acid Mine Drainage)-type acidic groundwater (pH£ľ4) from oxidizing sulfide tailings in BS nickel mine (Western Australia) is of higher total rare earth element(REE) contents and Ce enrichment (PAAS normalization), different from setting groundwater (pH£ĺ6.5, with lower total REE contents, Ce depletion). While the AMD contaminated groundwater (pH=4.0-6.5) around tailings pond is characterized by transition from acidic to setting groundwater in total REE content, and associated with Ce depletion (like setting groundwater). The light REE in all type groundwater shows up depletion, but its depleted extent in acidic groundwater is more remarkable. This work indicates that REE behavior in AMD-type acidic groundwater is controlled mainly by pH value and metal (Al, Mn and Fe) contents. And the critical pH value that affects REE behavior in ground acidic water would be 4, lower than the previous value (pH=5) that has been believed prevalently in surface acidic waters. The pH could affect REE behavior in groundwater by controlling the solubility of metal (Al, Mn and Fe) hydroxides and the valence of cerium. Finally, light REE depletion in acidic groundwater may due to element affinity. High content Al (affinity with heavy REE) and low content Fe (affinity with light REE) may lead to heavy REE enrichment while light REE relative depletion in water.

LEI Liang-qi(ņ◊Ńľ∆ś)1, 2, SONG Ci-an (ňőī»į≤)1, XIE Xiang-li(–ĽŌśņž)1, LI Yan-hong(ņÓ—řļž)1
1. Department of Resources and Environment Engineering, Guilin University of Technology,
Guilin 541004, China;
2. Key Laboratory of Geological Engineering Centre of Guangxi Province,
Guilin University of Technology, Guilin 541004, China
Received 25 October 2007; accepted 10 March 2008
Abstract: AMD(Acid Mine Drainage)-type acidic groundwater (pH£ľ4) from oxidizing sulfide tailings in BS nickel mine (Western Australia) is of higher total rare earth element(REE) contents and Ce enrichment (PAAS normalization), different from setting groundwater (pH£ĺ6.5, with lower total REE contents, Ce depletion). While the AMD contaminated groundwater (pH=4.0-6.5) around tailings pond is characterized by transition from acidic to setting groundwater in total REE content, and associated with Ce depletion (like setting groundwater). The light REE in all type groundwater shows up depletion, but its depleted extent in acidic groundwater is more remarkable. This work indicates that REE behavior in AMD-type acidic groundwater is controlled mainly by pH value and metal (Al, Mn and Fe) contents. And the critical pH value that affects REE behavior in ground acidic water would be 4, lower than the previous value (pH=5) that has been believed prevalently in surface acidic waters. The pH could affect REE behavior in groundwater by controlling the solubility of metal (Al, Mn and Fe) hydroxides and the valence of cerium. Finally, light REE depletion in acidic groundwater may due to element affinity. High content Al (affinity with heavy REE) and low content Fe (affinity with light REE) may lead to heavy REE enrichment while light REE relative depletion in water.
Key words: REE pattern; acid mine drainage; sulfide tailings; groundwater contamination
1 Introduction
Rare earth element(REE) can record subtle geochemical processes in natural systems, and has been extensively applied to earth science field. However, REE geochemical behavior in aquatic systems is a relatively new field of study. In recent years, many studies have focused on REE concentration, differentiation and effect factors in surface acidic waters, e.g. river, lake and mine surface water[1-5], but few papers published have been involved in ground acidic waters[6-7], especially AMD(Acid mine drainage)-type acidic groundwater.
There are a volume of solid wastes (including mill tailings, waste rock, etc.) produced during the period of metal sulfide mining activity. When the sulfide minerals remaining in the solid wastes expose to earth surface, they react with H2O and O2 in atmosphere, resulting in the generation of sulfuric acid. The acid drainage/AMD will happen only if the neutral/buffer capacities of the solid waste itself and environment were overwhelmed by its acid generation potentials. In contrast with the surface acidic water rich in oxygen, the ground acidic water which was formed by acid drainage infiltrating downward may be almost under anaerobic condition, meaning that biological and biochemical action may be of no consideration relatively in groundwater. Hence, there should be a difference in REE distribution patterns between surface and ground acidic water. In this study, REE behavior and effect factors in AMD-type acidic groundwater are studied through sampling and testing groundwater from drilling holes, which are placed nearby and beyond tailing bond in BS nickel sulfide mine, Western Australia.
2 Setting
BS nickel sulfide mine is near to Kalgoorlie, Western Australia. In BS mine, milling wastes, massive sulfide tailings (approximately 58 000 t/a) and disseminated sulfide tailings (approximately 325 000 t/a) are stored in MST and DST pond, respectively (Fig.1). Acidified groundwater has been recognized around the tailings pond three years after its establishment. A recent assessment of the tailings disclosed that the massive sulfide tailings were totally classified as potentially acid forming, and approximately 16% by volume of the disseminated sulfide tailings held a potential to be acid generating[8].
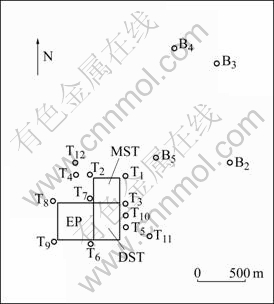
Fig.1 Location map showing sampling sites: MST°™Massive sulfide tailings pond; DST°™Disseminated sulfide tailings pond; EP°™Evaporation pond; T1-12°™Site and number of monitoring bores; B2-5°™Site and number of active pump bores
In the vicinity of tailings pond, there is a clayey ferricrete cover (4-20 m deep), which is underlain by saprolitic clay with minor ferruginous bands (62-79 m deep). Weathered depth (approached to Archaean mafic and ultramafic rock base) is up to 62-126 m. Surface runoff is to Southwest then South into a poorly defined channel outside the mine site. Groundwater is also interpreted to flow to South under low hydraulic gradients, and natural water table was 25-29 m below ground level.
3 Sampling and test results
3.1 Sampling
Groundwater samples were collected from monitoring bores (T1-12) nearby the pond and active pump bores (B2-5) beyond the pond (Fig.1). Groundwater pH (and electrical conductivity, static water level and temperature) was measured on site. A portion of each water sample (filtered through 0.45 ?£Ū nitrocellulose filter, then acidified with ultra pure nitric acid) was analyzed for REE and trace elements by ICP-MS and ICP-AES in department of Applied Chemistry, Curtin University of Technology (Western Australia). REE test values of groundwater samples were normalized using Post-Archaean Australian Shale (PAAS), and Cerium anomaly (Ce/Ce*) was calculated using the formula:
A(Ce/Ce*)=N(Ce)/[N(La)°ŃN(Pr)]0.5
where N denotes PAAS-normalized value.
3.2 Results
Based on pH values, the groundwater samples can be divided into three types (Table 1): typeĘŮ, pH£ľ4, acidic groundwater (including sample T2, T3, T5, T7, T10), collected in the vicinity of pond; type ĘÚ, pH=4.0°™6.5, acid contaminated groundwater (including T4, T6, T8, T12), also collected in the vicinity of pond; type Ęů, pH£ĺ6.5, setting groundwater, no acid contamination. Type Ęů samples include B2, B3, B4, B5 (sampling sites away from the tailing pond), and T1, T9, T11 (sampling sites near to the pond, but their water samples are of pH£ĺ6.5, hence these samples are classified as type Ęů).
Table 1 Analysis results of groundwater in drill holes, BS mine site*
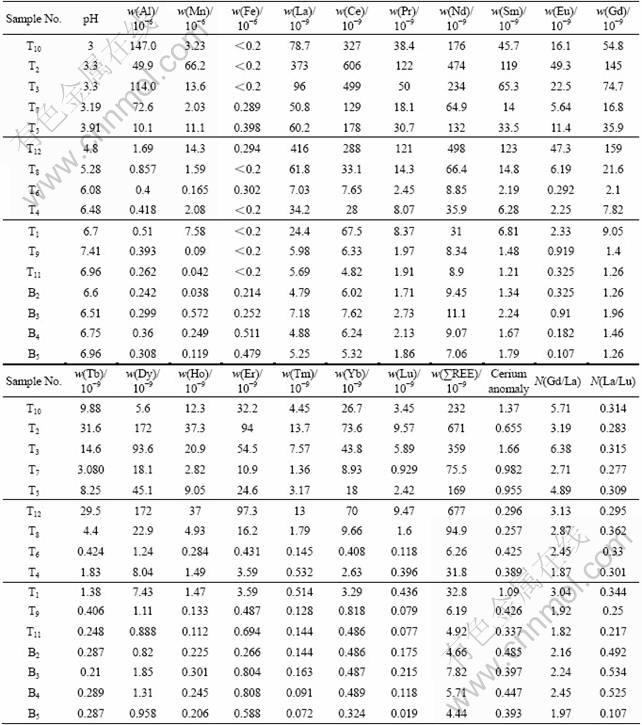
T1-12°™Drilling holes around tailing pond and sample number; B2-5°™Drilling holes away from pond and sample number.
Three-type groundwaters display different characteristics in solved REE distribution patterns (PAAS normalization) (Fig.2 and Table 1): 1) REE concentration in typeĘŮ((75.5-671)°Ń10-9, average 301.3°Ń10-9) is of 1-2 orders of magnitude higher than that in type Ęů ((4.44-32.8)°Ń10-9, average 9.51°Ń10-9); while REE concentration in type ĘÚ is of greater variation ((6.26-677)°Ń10-9, average 202.49°Ń10-9), and transition from typeĘŮto type Ęů. 2) cerium anomaly in typeĘŮhas a vaster variation (Ce/Ce*=0.655-1.66, average 1.12), but most of the samples (except T2) show weak-middle Ce positive anomaly; while types ĘÚ and Ęů (except T1) display marked Ce negative anomaly (type ĘÚ: Ce/Ce*=0.257-0.425, average 0.342; type Ęů: Ce/Ce*=0.337-1.09, average 0.511). 3) Light REE shows depletion in all three types, but light REE depleted extent in type ĘŮ is more remarkable than that in types ĘÚ and Ęů [typeĘŮ: (Gd/La)N=2.71-6.38, average 4.34; type ĘÚ: (Gd/La)N=1.87-2.87, average 2.06; type Ęů: (Gd/La)N= 1.82-3.04, average 2.23].
Grossly speaking, the acidic groundwater (type ĘŮ) in the mine is characterized by REE high concentration, Ce enrichment, and light REE depletion. In addition, it has a weak-middle REE enrichment (Fig.2).
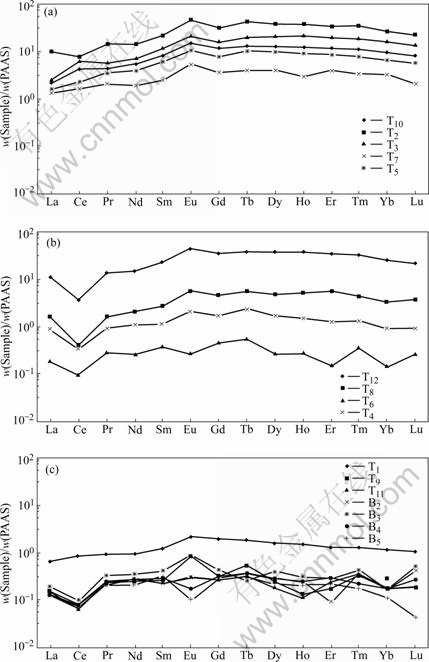
Fig.2 PAAS normalization REE patterns in groundwater, BS mine: (a) pH£ľ4; (b) pH=4.0-6.5; (c) pH£ĺ6.5
4 Discussion
4.1 REE contents
Many previous studies indicated that REE concentration is higher in acidic water than in normal water, and that pH plays an important role in controlling the concentration of solved REE in water, and its critica value is pH=5. Below a pH of 5 in acidic water, REE concentrates in water facies. The reason is that REE behaves conservatively in acidic water, and the elements concentrate in water facies as liberal ion or complex with SO42- (F-). While at pH£ĺ5, REE tends to deposit though completing with CO32+ or (CO2)-, and be adsorbed by the metal (e.g. Al, Fe, Mn) hydroxide colloid, leading REE contents in waters to reduce[9-17]. And there is a negative correlation between REE content and pH value[18-19].
This paper got similar results to the previous studies. As pH value is below 5, especially below 4, REE contents in groundwater increase greatly (Fig.3(a)). And Fig.4 displays a negative correlation between REE and pH in the groundwater (correlation coefficient r=-0.82), and a positive correlation between Al (Mn) and pH (r=0.92 and 0.87, respectively), with the exception of Fe (there is not remarkable correlation between Fe and pH). This reveals that REE contents in groundwater are controlled not only by pH, but also by Al, Mn contents. pH may control REE concentration through regulating adsorption-desorption of colloids (Al, Mn-hydroxides), meaning that REE adsorbed on the colloids in water can be released into water with pH decreasing (which leads colloid electronegativity to decrease), and vice versa[16]. These imply that pH is a dominant factor in controlling REE contents in groundwater.
Fig.3 Plots of REE content, Ce/Ce* and (La/Gd)N against pH
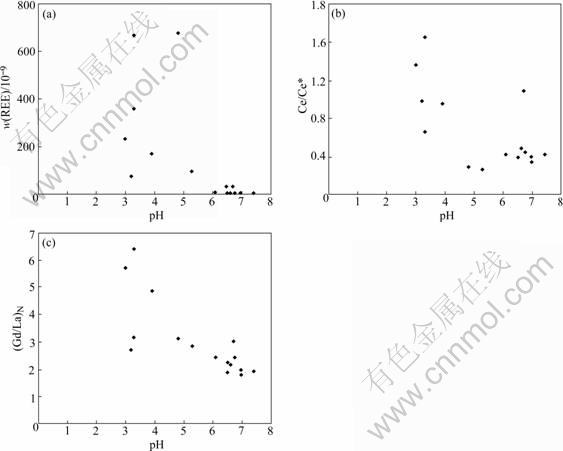
Fig.3 Plots of REE content, Ce/Ce* and (La/Gd)N against pH

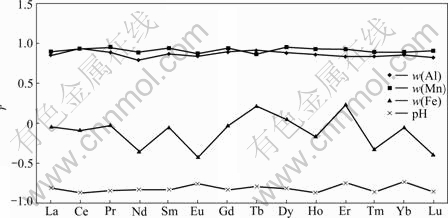
Fig.4 Plots of correlation coefficient (r) of w(REE) with pH, w(Al), w(Mn) and w(Fe)
In addition, the previous studies also revealed that REE contents in AMD-type acidic water, e.g. Odiel acid river (pH=3.25, REE 42.62°Ń10-9), Osamu Utsmi mine groundwater (pH=4.43, REE 1.046°Ń10-6), Metalliferous Hills Cu-Pb-Zn mine stream (pH=3.1, REE 929°Ń10-9), and Chinese Sitai coal drainage (pH=3.52-3.75, REE (54.37-68.80)°Ń10-9)) have 2-3 orders of magnitude higher than those in normal water[1-2, 5-6]. In BS mine, the acidic groundwater (ĘŮtype, pH=3-3.91) should be classified as AMD-type acidic water, because its REE contents ((75.5-671)°Ń10-9) are in the REE range ((42.62-1 046)°Ń10-9) above AMD-type water and much higher than that in the normal setting groundwater.
4.2 Ce differentiation
pH value is one of the factors controlling Ce behavior in water, and the critical value is pH=5. At pH£ĺ5, Ce is oxidized and changes from solved Ce3+ to dissoluble Ce4+, then deposits as CeO2 from water (BROOKINGS had ever certified the capability of CeO2 sediment in nature waters)[20], resulting in a negative Ce anomaly in waters[21-23]. While in the pH£ľ5 acid water, Ce negative anomaly disappears and even it may show a small positive anomaly instead[11, 21].
Moreover, Ce behavior in water may relate to redox condition. LEYBOURNE et al[7] discovered that when they studied the difference of solved REE contents between surface and ground water in a mine site in Canada, Ce negative anomaly can be lowered down to 0.08 (NASC normalization) in surface water, while this Ce anomaly was not demonstrated in groundwater mostly. It may be due to the fact that, under surface aerobic condition, Ce is easier to be oxidized and deposited as CeO2, causing Ce depletion in surface water; however, Ce in anoxic groundwater occurs as reduced Ce3+ instead and concentrates in water.
In BS mine site, Ce anomaly in groundwater is controlled by pH, but the critical value is pH=4, relatively lower than the previous critical value (pH=5). In pH£ľ4 acidic groundwater, Ce shows weak-middle positive anomaly. In pH£ĺ4, the positive anomaly tends to disappear(Fig.3(b)).
4.3 REE differentiation
Light REE is more active than heavy REE in water [22, 24], causing REE differentiation, but pH effect is more significant. Under high pH alkaline condition, light REE tends to be divorced from waters and concentrates into secondary minerals[13, 15]. And with pH increasing, the sequence of REE adsorption to particles (in lake) is: light REE£ĺmiddle REE£ĺheavy REE; contrary to pH decreasing, REE release has similar sequence[9]. Therefore, it is not surprising that the REE model with light REE depletion and heavy REE enrichment often appears in the alkaline water[10, 25], while light REE concentrates in the acid water relatively[4, 13].
In this study, all groundwater samples demonstrate light REE depletion (Fig.2), and in the pH£ľ4 acidic groundwater, the extent of light REE depletion is vaster ((Gd/La)N values are higher in acidic water than in setting water, see Fig.3(c)). The unusual phenomenon of light REE depletion in acidic water may be due to the element affinity. MARMOLEJO-RODRIGUEZ et al[17] indicated that light REE tends to affiliate with Fe, while heavy REE prefers to do with Al in land water. In BS mine site, Al (and Mn) high content and Fe low content (Fe contents in many samples even are lower than test limitation, Table 1) in acidic groundwater may lead to heavy REE enrichment while light REE depletion relatively. Besides, WORRALL and PEARSON[26] had another explanation for light REE depletion in acidic water. It is due to the results of sulfide (e.g. pyrite) oxidizing, without involving in latter water-rock reaction. Therefore, the mechanism of light REE depletion in acidic water may be more complex, and it is necessary to do further study to get more persuasive explanation.
Besides, the middle REE enrichment, which exists in the general acidic water[1, 5], is also shown up in the AMD-type acidic groundwater in BS mine site, although its enrichment extent is markedly small (Fig.2).
5 Conclusions
1) REE behavior in AMD-type acidic groundwater is controlled by pH and metal (Al, Mn and Fe) contents.
2) The critical pH value that affects REE behavior in AMD-type acidic groundwater would be 4, lower than the critical value (pH=5) in general surface acidic water. The pH could affect REE behavior in groundwater by controlling the solubility of metal (Al, Mn and Fe) hydroxides and the valence of cerium. In pH£ľ4 acidic groundwater, metal hydroxides (with higher solubility), REE and Ce tend to concentrate in water, resulting in REE and Ce enrichment. However, in higher pH groundwater, the metal hydroxides deposit as colloid coagulation because of the solubility decreasing, leading to REE content decreasing. And Ce is easier to be oxidized and deposited as CeO2 (Ce separates from other lanthanide by this way) under this condition, causing Ce depletion in water.
3) Light REE depletion can appear in AMD-type acid groundwater. One of the reasons may be due to the element affinity. High content Al (affiliated with heavy REE) and low content Fe (affiliated with light REE), like the studied acidic groundwater in BS mine, may cause heavy REE enrichment and light REE depletion relatively.
Acknowledgements
The authors acknowledge very much the invaluable aids from A/Prof. Ron Watkins (director of EIGG£≠Environment Inorganic Geochemistry Group, Curtin University of Technology, W.A.) and colleagues, Dr. Dave Oldmeadow, Dr. Ryan Noble and Dr. Troy Cook, during the study period. Many thanks for help from Ms. Miranda Taylor, who provided some fundamental documents pertaining to the hydrogeology about the research area and gave assistance for field sampling.
References
[1] ELBAZ-PLULICHET F, DUPUY C. Behaviors of rare earth elements at the freshwater-seawater interface of two acid mine rivers: The tinto and odiel (Andalucia, Spain) [J]. Applied Geochemistry, 1999, 14: 1063-1072.
[2] PROTANO G, RICCOBONO F. High contents of rare earth elements (REEs) in stream water of a Cu-Pb-Zn mining area [J]. Environmental Pollution, 2002, 117: 499-514.
[3] GAMMONS C H, WOOD S A, JONAS J P, MADISON J P. Geochemistry of the rare-earth elements and uranium in the acidic Berkeley Pit Lake, Butte, Montana [J]. Chemical Geology, 2003, 198: 269-288.
[4] BOZAU E, LEBLANC M, SEIDEL J L, ST?RK H J. Light rare earth elements enrichment in an acidic mine lake (Lusatia, Germany) [J]. Applied Geochemistry, 2004, 19: 261-271.
[5] ZHAO Feng-hua, CONG Zi-yuan, SUN Hong-fu, REN De-yi. The geochemistry of rare earth elements(REE) in acid mine drainage from the Sitai coal mine, Shanxi Province, North China [J]. International Journal of Coal Geology, 2007, 70: 184-192.
[6] MIEKELEY N, COUTHINO DE JESUS H, PORTO DA SILVEIRA C L. Rare earth elements in groundwater from the Osamu Utsumi mine and Morro do Ferro analogue study sites, Plcos de Caldas, Brazil [J]. Journal of Geochemical Exploration, 1992, 45: 365-387.
[7] LEYBOURNE M I, GOODFELLOW W D, BOYLE D R, HALL G M. Rapid development of negative Ce anomalies in surface water and contrasting REE patterns in groundwater associated with Pb-Zn massive sulfide deposits [J]. Applied Geochemistry, 2000, 15: 695- 723.
[8] LEI Liang-qi, WATKINS R. Acid drainage re-assessment of mining tailings, Black Swan nickel mine, Kalgoorlie, Western Australia [J]. Applied Geochemistry, 2005, 20: 661-667.
[9] WOOD S A. The aqueous geochemistry of the rare earth elements and yttrium: Review of available low-temperature date for inorganic complexes and the inorganic REE speciation of natural waters [J]. Chemical Geology, 1990, 82: 159-168.
[10] JOHANNESSON H K, ZHOU X, GUO C. Origin of rare earth signatures in groudwaters of circumneutral pH from southern Nevada and eastern California, USA [J].Chemical Geology, 2000, 164: 239- 257.
[11] OHTA A, KAWABE I. REE(III) adsorption onto Mn dioxide (¶ń-MnO2) [J]. Geochimica et Cosmochimica, 2001, 65(5): 695-703.
[12] JANSSEN R P T, VERWEIJ W. Geochemistry of some rare earth elements in groundwater, Vierlingsbeek, The Netherlands [J]. Water Research, 2003, 37: 1320-1350.
[13] STILLE P, GAUTHIER-LAFAYE F, JENSEN A K, SALAH S, BRACKE G, EWING C R, LOUVAT D,MILLION D. REE mobility in groundwater proximate to the natural fission reactor at Bangomb®¶(Gabon) [J]. Chemical Geology, 2003, 198: 289-304.
[14] VERPLANCK P L, NORDSTROM D K, TAYLOR H E, KIMBALL B A. Rare earth element partitioning between hydrous ferric oxides and acid mine water during iron oxidation [J]. Applied Geochemistry, 2004, 19: 1339-1354.
[15] K?HLER J S, HAROUIYA N, CHA?RAT, OELKERS H E. Experimental studies of REE fractionation during water-mineral interactions: REE release rates during apatite dissolution from pH 2.8 to 9.2 [J]. Chemical Geology, 2005, 222: 168-182.
[16] ZHU Zhao-zhou, LIU Cong-qiang, WANG Zhong-liang, LI Jun, ZHOU Zhi-hua. Inorganic speciation of rare earth elements in Chaohu Lake and Longganhu Lake, East China [J]. Journal of The Chinese Rare Earth Society, 2006, 24(1): 110-115.
[17] MARMOLEJO-RODRIGUEZ J A, PREGO R, MEYER- WILLERER A, AHUMILIN E, SAPOZHNIKOV D. Rare earth elements in iron oxy-hydroxide rich sediments from the Marabasco River-Estuary System(pacific coast of Mexico). REE affinity with iron and aluminium [J]. Journal of Geochemical Exploration, 2007, 94: 43-45.
[18] BANKS D, HALL G, REIMANN. Distribution of rare earth elements in crystalline bedrock groudwaters: Oslo and Bergen regions, Norway [J]. Applied Geochemistry, 1999, 14(1): 27-39.
[19] ZHU Zhao-zhou, WANG Zhong-liang, GAO Bo, WANG Shi-lu. Geochemical characteristics of rare earth elements in Lake Chaohu, East China [J]. Geochimica, 2006, 35(6): 639-644.
[20] BROOKINS D G. Aqueous geochemistry of rare earth elements. LIPIN B R, MCKAY G A, Ed. Geochemistry and mineralogy of rare earth elements. Reviews in mineralogy (Volume 21) [M]. Michigan, USA, Mineralogical Society of America, BookCrafters, 1989: 201- 225.
[21] TRICCA A, STILLE P, STEINMANN M. Rare earth elements and and Sr and Nd isotopic compositions of dissolved and suspended loads from small river systems in the Vosges mountains (France), the river Rhine and groundwater [J].Chemical Geology, 1999, 160(1/2): 139-158.
[22] NELSON J B, WOOD A S, OSIENSKY L J. Partitioning of REE between solution and particulate matter in natural waters: A filtration study [J]. Journal of Solid State Chemistry, 2003, 171: 51-56.
[23] HAN Gui-lin, LIU Cong-qiang. Controlling factors for variation in dissolved rare-earth elements in Karst Drainage basin [J]. Carsologica Sinica, 2004, 23(3): 177-186. (in Chinese)
[24] AUBERT D, PROBST A, STILLE P. Distribution and origin of major and trace elements (particularly REE, U and Th) into labile and residual phases in an acid soil profile (Vosges Mountains, Frence) [J]. Applied Geochemistry, 2004, 19: 899-916.
[25] YAN X P, KERRICH R, HENDRY M J. Sequential leachates of multiple grain size fraction from a clay-rich till, Saskatchewan, Canada: Implications for contrals on the rare earth element geochemistry of pore-water on an aquitard [J]. Chemical Geology, 1999, 158: 53-79.
[26] WORRALL F, PEARSON D G. Water-rock interaction in an acidic mine discharge as indicated by rare earth element patterns [J]. Geochimica et Cosmochimica, 2001, 65: 3027-3040.
Foundation item: Project supported by the Science Foundation of Guangxi Province, China; Project supported by the Director Fund Project of Key Laboratory of Geological Engineering Center of Guangxi Province, China
Corresponding author: LEI Liang-qi; Tel: +86-773-5896341; E-mail: LEILQ@glite.edu.cn; LLQ927@hotmail.com
(Edited by YANG Bing)

