Mild and highly efficient transfer hydrogenation of aldehyde and ketone catalyzed by rubidium phosphate
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2016���7��
�������ߣ������� ���ƾ� ������ �����
����ҳ�룺1603 - 1610
Key words��rubidium phosphate; transfer hydrogenation; heterogeneous catalysis; cyclic transition state
Abstract: Rubidium phosphate can be more conveniently obtained by extracting trace Rb+ from the salt lake brine. Rb3PO4 was found to be an excellent heterogeneous catalyst for transfer hydrogenation. Rb3PO4 lost 70% of its active sites after adsorbing water, but the remaining was not affected. The reductions of aldehydes and ketones, when promoted by Rb3PO4, were allowed at room temperature. The activities of substrates at room temperature followed a descending order of 2,6-dichlorobenzaldehyde> 4-bromobenzaldehyde>benzaldehyde>acetophenone>anisaldehyde>butanone. A new catalytic cycle postulating a six-membered cyclic transition state for the reductions of aldehydes and ketones was proposed. These results exploited the catalytic usage of Rb3PO4 and worth in industrial application.
J. Cent. South Univ. (2016) 23: 1603-1610
DOI: 10.1007/s11771-016-3214-x
HUANG Yun-jing(���ƾ�), YANG Wei-jun(������), QIN Ming-gao(������), ZHAO Hao-liang(�����)
College of Chemistry and Chemical Engineering, Hunan University, Changsha 410082, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: Rubidium phosphate can be more conveniently obtained by extracting trace Rb+ from the salt lake brine. Rb3PO4 was found to be an excellent heterogeneous catalyst for transfer hydrogenation. Rb3PO4 lost 70% of its active sites after adsorbing water, but the remaining was not affected. The reductions of aldehydes and ketones, when promoted by Rb3PO4, were allowed at room temperature. The activities of substrates at room temperature followed a descending order of 2,6-dichlorobenzaldehyde> 4-bromobenzaldehyde>benzaldehyde>acetophenone>anisaldehyde>butanone. A new catalytic cycle postulating a six-membered cyclic transition state for the reductions of aldehydes and ketones was proposed. These results exploited the catalytic usage of Rb3PO4 and worth in industrial application.
Key words: rubidium phosphate; transfer hydrogenation; heterogeneous catalysis; cyclic transition state
1 Introduction
Hydrogenation of carbonyl compounds, which is important in both organic synthesis and industrial application [1-4], was performed with hazardous molecular H2 and specifically treated catalysts under very rigorous conditions before the Meerwein-Ponndorf- Verley (MPV) reaction was reported [5]. Although MPV reaction is carried out at mild temperature by using secondary alcohols such as 2-propanol as H donors, large amounts of Al alkoxides are required to catalyze the reduction with high yields [6]. Nickel phthalocyanine [7], ruthenium [8], iridium [9], rhodium [10], and gold [11] have also been reported to promote H transfer efficiently. However, these precious metals are costly and toxic, and the ligands are insufficiently active [12].
Alkali carbonates are often used as additives in catalytic transfer hydrogenation. BACKVALL [13] reported that with base additives RuCl2(PPh3)2 could form active dihydride catalyst RuH2(PPh)3. Base additives have accelerated the reaction greatly. The importance of the base is illustrated by KOH and NaOH used alone without any transition metal or ligands for the reduction of some aromatic aldehydes and ketones [14-15]. It is known to all that KOH and NaOH could react with alcohols which is the H donator in this reaction. So, when the reaction is finished, KOH and NaOH dissolved in the solvent are hard to be separated, not to mention their reusability. To find a more efficient and environmental friendly catalyst for transfer hydrogenation, mild basic Rb3PO4 was used to promote the reduction (Scheme 1) in this work. Now, Rb3PO4 can be more conveniently obtained by our new process of extracting trace Rb+ from the salt lake brine [16]. With the similar structure as Rb3PO4 [17], anhydrous K3PO4 was also used as catalyst to go comparing with Rb3PO4. Surprisingly, the experiment results indicated that the catalytic property of rubidium phosphate is very high. The reaction can be generated at room temperature. A new catalytic cycle postulating a six-membered cyclic transition state for the reductions of aldehydes and ketones is proposed.
2 Experimental
All chemicals were analytically pure. Rb3PO4 and K3PO4 were obtained from Shanghai Energy Lithium Industrial Co., Ltd. They were calcined at 600 ��C before use and their structures after calcined had been confirmed as Rb3PO4/K3PO4 by XRD. Catalytic transfer hydrogenation was typically carried out using 6.0 mmol carbonyl substrate and 50.0 mL of 2-propanol as reductant and solvent. The mixture was placed in a two-neck round-bottom flask and heated in air with a reflux device whereupon 0.4 mmol calcined Rb3PO4 or 4.0 mmol calcined K3PO4 was added. Liquids were sampled (0.1 mL each time) at regular time intervals and analyzed by gas chromatography. Gas chromatography analysis was performed on a Shimadzu GC-2014 equipped with a 0.5 mm i.d. _25 m PEG20000 capillary column and flame ionization detector.
The products were identified by comparison with an authentic sample through gas chromatography and mass spectrometry. To test the reusability of Rb3PO4, the used sample was recovered by filtration after reaction and washed with 2-propanol. It was reused for another reaction after drying.
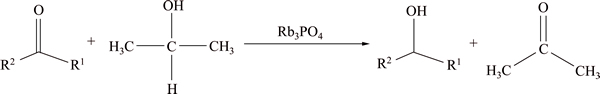
Scheme 1 Catalytic transfer hydrogenation of carbonyl compounds
3 Results and discussion
3.1 Operational conditions for Rb3PO4
Oxygen and H2O in the air, which may be adsorbed on the surface cations, poison the catalyst. Although nitrogen environment can reduce the content of the interaction between oxygen and H2O in the air and the catalysts, it also rendered the operation more complicated. Therefore, comparative experiments were conducted (Fig. 1) in order to find out whether nitrogen protection was necessary.
One system was opened to the air with a reflux device and the other one was protected by N2(Fig. 1). Nitrogen protection plays little effect in the reaction as shown.
The influences of water on the catalytic properties of moisture-vulnerable anhydrous alkali metal compounds with extremely high hygroscopicity were also evaluated (Fig. 2).
Anhydrous Rb3PO4, which was exposed in air for 20 min on purpose, became wet and viscous, suggesting that water adsorption may significantly affect its catalytic activity. As a result, Rb3PO4 lost nearly 70% of active sites (Fig. 2). Then, the exposure time was prolonged to 2.0 h, during which Rb3PO4 totally turned into a concentrated solution because of copious water adsorption. The Rb3PO4 solution was used to promote H transformation, the catalytic activity of which remained partially with even more water adsorbed. After 1.0 h, 27% of 2,6-dichlorobenzaldehyde was converted into corresponding alcohols.
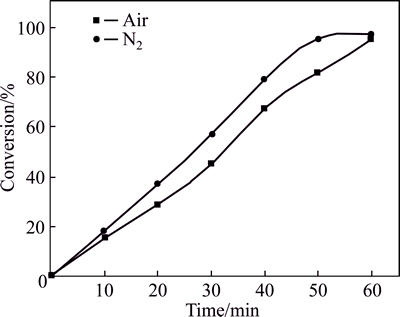
Fig. 1 Conversion of 2,6-dichlorobenzaldehyde in air and N2 (Reaction conditions: 6.0 mmol substrate, 50.0 mL of 2-propanol as solvent, 0.4 mmol Rb3PO4, 70 ��C)
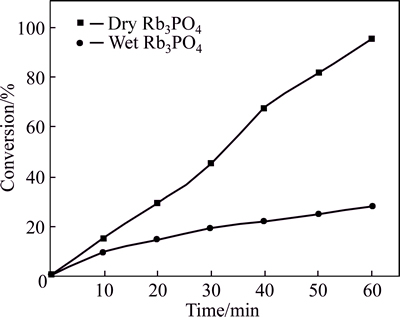
Fig. 2 Conversion of 2,6-dichlorobenzaldehyde promoted by dry Rb3PO4 and wet Rb3PO4 (Reaction conditions: 6.0 mmol substrate, 50.0 mL of 2-propanol as solvent, 0.4 mmol Rb3PO4, 70 ��C, air)
Equimolar anhydrous K3PO4 was used as catalyst to go comparing with Rb3PO4. After 1.0 h, only 14% of 2,6-dichlorobenzaldehyde was converted. So, even Rb3PO4 was fully saturated in air, its catalytic activity was higher than that of anhydrous K3PO4. Then,4.0 mmol K3PO4 (ten times of Rb3PO4) was used to promote the reaction, with other conditions being the same as those shown in Fig. 2. After 1.0 h, 98% conversion was observed. K3PO4 that had been exposed in air for 2.0 h and turned into concentrated solution was then used to catalyze the reaction. After another 1.0 h, 33% of 2,6-dichlorobenzaldehyde turned into corresponding alcohols. Water exerted similar effect on the activity of K3PO4 to that on Rb3PO4.
3.2 Reductions of aldehydes and ketones
To demonstrate the efficiency of Rb3PO4, comparative experiments were carried out on every substrate by using K3PO4 as hydrogenation catalyst (Table 1). Different amounts of catalyst were used to promote the hydrogenation of aldehydes and ketones to shorten the reaction time.
Table 1 Reductions of aldehydes and ketones with 2-propanol
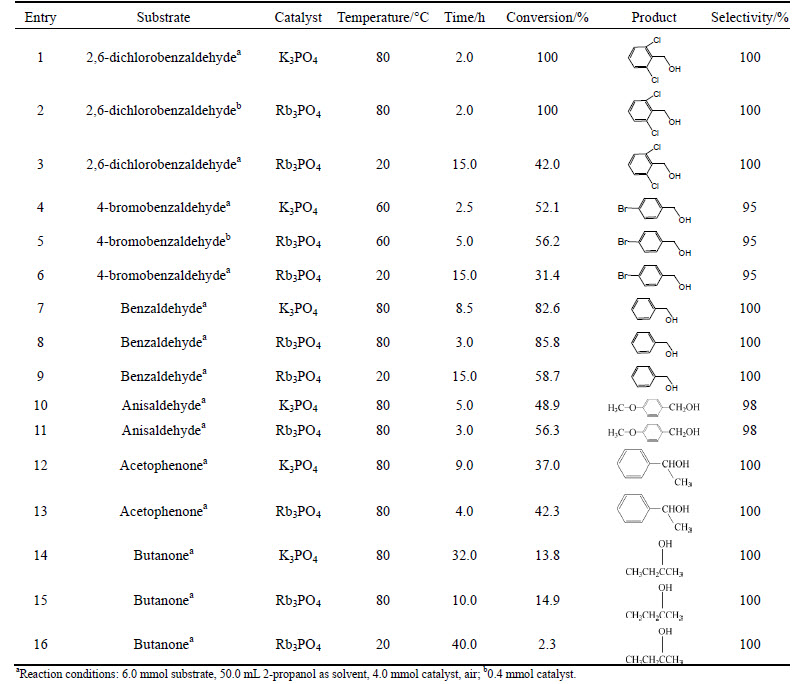
The rate of reaction was increased by electron- withdrawing groups on the benzene ring (Entries 1-6, Table 1) and decreased by electron-donating groups (Entries 10 and 11, Table 1). The reduction of butanone was much slower due to reversible H transformation. The reduction did happen at room temperature very slowly, when promoted by Rb3PO4, which has never been reported hitherto. Under the same conditions (Rb3PO4 as catalyst), 2,6-dichlorobenzaldehyde, benzaldehyde and 4-bromobenzaldehyde were converted into corresponding alcohols slowly, whereas acetophenone and anisaldehyde remained stable after 10.0 h, but not after 30 h. According to the conversion rates of all substrates after 30 h, the activities at room temperature followed the descending order of 2,6-dichloroben-zaldehyde>4-bromobenzaldehyde>benzaldehyde>aceto-phenone>anisaldehyde>butanone.
3.3 High efficiency of Rb3PO4
IVANOV et al [5] reported that alcohols could be adsorbed on base catalysts via their hydroxyl hydrogen bound to the oxygen of base catalysts and their hydroxyl oxygen bound to the surface cation of base catalysts, and that aldehydes and ketones could be absorbed through their oxygen atoms bound to the surface cation of base catalysts. It is well-established that rubidium atom adsorbs oxygen atom in the substrates stronger than potassium atom does [18].
To test the postulation that adsorption was a key step of the whole reduction process promoted by Rb3PO4 or K3PO4, an experiment was set as Entry 2 in Table 1 and liquids were sampled at regular time intervals (0.1 mL each time). Then, the liquids were cooled to room temperature. The solid catalyst Rb3PO4 swelled dozens of times (about 1.0 mmol Rb3PO4 absorbed 2.0 mL reaction solution) that of the original volume in the early stage. After 2,6-dichlorobenzaldehyde was fully converted, Rb3PO4 shrank to its initial volume. A comparative experiment catalyzed by K3PO4 was set as Entry 1 in Table 1, and K3PO4 barely swelled and also recovered to its original volume after complete conversion of 2,6-dichlorobenzaldehyde. The swollen solids may be the intermediate products of Rb3PO4 or K3PO4 adsorbing 2,6-dichlorobenzaldehyde. Therefore, by adsorbing aldehydes and ketones more rapidly and efficiently, Rb3PO4 promoted H transformation faster than K3PO4 did.
VORONIN et al [17] reported that Rb3PO4 has a higher electroconductivity than K3PO4. In our experiment, alcohols, aldehydes and ketones could be bound to the exposed alkali-metal atoms and the oxygen atoms in [PO4] tetrahedra. The high conductivity of Rb3PO4 is conducive to electron transfer, and electron transformation is the key step of transfer hydrogenation (Scheme 2). Thus, with a higher conductivity than K3PO4, Rb3PO4 could promote H transformation faster.
3.4 Preliminary consideration of Rb3PO4/K3PO4 catalytic cycle
There is no report about the catalytic cycle of Rb3PO4 promoting transfer hydrogenation so far. RADHAKRISHAN et al [19] added benzaldehyde and 2,6-dichlorobenzaldehyde simultaneously in the reaction mixture (K3PO4 promoted transfer hydrogenation), and reported that the rate of reaction for each substrate was unaffected by the other substrate [19]. They then concluded that benzaldehyde and 2,6-dichloro- benzaldehyde were transformed into alcohols at different sites on K3PO4. Thus, benzaldehyde and 2,6-dichloro- benzaldehyde will only compete for 2-propanol.
VORONIN et al [17] reported that the crystal structure of K3PO4 consisted of [KO6] octahedra and [PO4] tetrahedra, with layers of potassium chains in between. Then, they emphasized that Rb3PO4 had the same crystal structures as that of K3PO4 [20]. Since Rb3PO4 and K3PO4 have the similar structure, it is reasonable to investigate their catalytic cycle together.

Scheme 2 Possible catalytic cycle of forming six-membered cyclic transition states for reductions of aldehydes and ketones catalyzed by M3PO4 (M=K, Rb):
RADHAKRISHAN et al [19] designed all the reactions with excessive 2-propanol and a mixture of equimolar benzaldehyde and 2,6-dichlorobenzaldehyde. However, in this case, weak interactions between the two substrates could not be easily observed.
Therefore, to amplify interactions between the two substrates, we increase the use of benzaldehyde to 40 mL and reduce the use of 2-propanol to 10.0 mL. The reaction conditions herein were set as follows: 40 mL of benzaldehyde as solvent, 6.0 mmol 2,6-dichlorobenzal- dehyde, 10.0 mL of 2-propanol, 4.0 mmol K3PO4 as catalyst, 80 ��C and air. After 9.0 h, only 7.0% of 2,6-dichlorobenzaldehyde was converted into 2,6-dichlorobenzyl alcohol. The result of 2 h reaction of this competitive experiment is shown in Fig. 3.
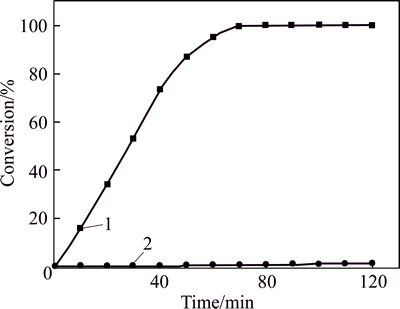
Fig. 3 Conversion of 2,6-dichlorobenzaldehyde (Reaction conditions: 1-6.0 mmol 2,6-dichlorobenzaldehyde, 50.0 mL of 2-propanol as solvent, 4.0 mmol K3PO4, 80 ��C and air; 2-40 mL of benzaldehyde as solvent, 6.0 mmol 2,6-dichlorobenzaldehyde, 10.0 mL of 2-propanol, 4.0 mmol K3PO4 as catalyst, 80 ��C and air)
2,6-dichlorobenzaldehyde was reduced much faster than benzaldehyde (Table 1), and the transfer hydrogenation of 2,6-dichlorobenzaldehyde finished in 2.0 h when used alone. Very little 2-propanol (about 3.0%) was observed to be consumed after 9.0 h (in 40 mL of benzaldehyde as solvent, and 6.0 mmol 2,6-dichlorobenzaldehyde reaction mixture). Thus, 2-propanol is always adequate for the reduction of 2,6-dichlorobenzaldehyde. CHUAH et al held that benzaldehyde and 2,6-dichloro- benzaldehyde only competed for 2-propanol. Therefore, 2,6-dichlorobenzal- dehyde will be fully converted into 2,6-dichlorobenzyl alcohol after 9.0 h as they postulated. But in fact, only 7.0% of 2,6-dichlorobenzaldehyde turned into 2,6-dichlorobenzyl alcohol in this work. Given the distinctive difference, there must be other competitive relationships between 2,6-dichloro- benzaldehyde and benzaldehyde.
Based on our experimental results and the crystal structures of K3PO4 and Rb3PO4, we postulated that the hydrogenations of benzaldehyde (or acetophenone) and benzaldehydes (or acetophenone) with substituent groups on the benzene ring went through different six- membered cyclic transition states (Scheme 2). (CHA [6] also postulated a similar six-membered cyclic transition state of solid base catalyst system)
In Scheme 2(a), benzaldehyde (or acetophenone), when used alone, is adsorbed on the same alkali atom with 2-propanol, forming a six-membered cyclic transition state. Subsequently, acetone is yielded owing to the disappearance of the state, while the �� carbon of benzaldehyde (or acetophenone) obtains a hydrogen from that of 2-propanol. The carbon-oxygen double bond of benzaldehyde (or acetophenone) breaks, but oxygen thereof is still linked with the alkali atom. During the next step, the oxygen of benzaldehyde (or acetophenone) obtains a hydrogen from 2-propanol (its hydroxyl hydrogen), forming the corresponding alcohol as a result. After desorption, the catalyst is ready for another cycle.
In Scheme 2(b), when 2,6-dichlorobenzaldehyde is used alone, it cannot be adsorbed on the same alkali atom with 2-propanol due to steric effect. Instead, it is adsorbed on the adjacent alkali atoms, forming a different six-membered cyclic transition state. After the state disappears, 2,6-dichlorobenzaldehyde obtains the �� carbon hydrogen and hydroxyl hydrogen of 2-propanol, yielding 2,6-dichlorobenzyl alcohol and acetone. After desorption, the catalyst can be used for another cycle. 4-bromobenzaldehyde and anisaldehyde are reduced through the same way as 2,6-dichlorobenzaldehyde.
When benzaldehyde and 2,6-dichlorobenzaldehyde are used together in the reaction mixture, there are four possible situations. Situation 1: Benzaldehyde is adsorbed on the same alkali atom with 2-propanol and no 2,6-dichlorobenzaldehyde is adsorbed on the adjacent alkali atoms. Then, benzaldehyde goes through the steps in Scheme 2(a). Situation 2: 2,6-Dichlorobenzaldehyde is adsorbed on the adjacent alkali atoms with 2-propanol and no benzaldehyde is adsorbed on the same alkali atom with this 2-propanol, and then, 2,6-dichlorobenzaldehyde goes through the steps in Scheme 2(b). Situation 3: Benzaldehyde is adsorbed on the same alkali atom with 2-propanol, and after that, 2,6-dichlorobenzaldehyde is adsorbed on the interfacing alkali atom in Scheme 2(c). Benzaldehyde got the opportunity to form the six-membered cyclic transition state with 2-propanol because of early binding. The oxygen of 2,6-dichloro- benzaldehyde attracts the hydroxyl hydrogen of 2-propanol and biases the electronic doublet toward oxygen in O��H bond. Due to transmission effects, electronic doublet in the �� carbon hydrogen bond becomes more biased toward hydrogen. Thus, the six-membered cyclic transition state which is formed by benzaldehyde and 2-propanol vanishes more quickly, and 2,6-dichloro-benzaldehyde waits for another 2-propanol to be adsorbed on the interfacing alkali atom. In this way, the reduction of benzaldehyde is accelerated. Situation 4: 2,6-dichlorobenzaldehyde is adsorbed on the interfacing alkali atom with 2-propanol, and after that, benzaldehyde is adsorbed on the same alkali atom with 2-propanol. Or benzaldehyde and 2,6-dichlorobenzaldehyde are adsorbed at the same time, while the former is adsorbed on the same alkali atom with 2-propanol and the latter is adsorbed on the interfacing alkali atom. In the presence of electron-withdrawing groups on the benzene ring, 2,6-dichlorobenzaldehyde binds the �� carbon hydrogen (electronic doublet is biased toward hydrogen in this C��H bond) of 2-propanol more easily, forming a six- membered cyclic transition state with 2-propanol in the two kinds of conditions. The oxygen of benzaldehyde attracts the hydroxyl hydrogen of 2-propanol and biases the electronic doublet toward oxygen in O��H bond. Electronic doublet in the �� carbon hydrogen bond is biased more toward hydrogen owing to transmission effects. Thus, the six-membered cyclic transition state which is formed by 2,6-dichlorobenzaldehyde and 2-propanol disappears more quickly, and benzaldehyde waits for another 2-propanol to be adsorbed on the same alkali atom. As a result, the reduction of 2,6-dichloro- benzaldehyde is accelerated.
When benzaldehyde and 4-bromobenzaldehyde or benzaldehyde and anisaldehyde are used as reactant pairs, there are five possible situations. Situations 1-3 are the same as the outcomes of benzaldehyde and 2,6-dichloro- benzaldehyde. In Situation 4, when benzaldehyde and 4-bromobenzaldehyde are used as the reactant pairs, benzaldehyde is adsorbed on the same alkali atom with 2-propanol and 4-bromobenzaldehyde is adsorbed on the interfacing alkali atom at the same time. The presence of electron-withdrawing groups on the benzene ring of 4-bromobenzaldehyde and the distance between 4-bromobenzaldehyde and 2-propanol, the pairs bind 2-propanol resembling a tug-of-war, the winner of which undergoes reduction that is accelerated by the attraction of the other. When benzaldehyde and anisaldehyde are used as the reactant pairs, benzaldehyde is adsorbed on the same alkali atom with 2-propanol and anisaldehyde is adsorbed on the interfacing alkali atom at the same time. Due to the presence of electron-donating groups on the benzene ring of anisaldehyde and the distance between anisaldehyde and 2-propanol, benzaldehyde binds 2-propanol and forms a six-membered cyclic transition state. The reduction of benzaldehyde is accelerated by being attracted by anisaldehyde oxygen. In Situation 5, when benzaldehyde and 4-bromobenzaldehyde are used as the reactant pairs, 4-bromobenzaldehyde is adsorbed on the interfacing alkali atom with 2-propanol, and after that, benzaldehyde is adsorbed on the same alkali atom with 2-propanol. 4-bromobenzaldehyde forms a six- membered cyclic transition state. The reduction of 4-bromobenzaldehyde is accelerated by being attracted by benzaldehyde oxygen. When benzaldehyde and anisaldehyde are used as the reactant pairs, anisaldehyde is adsorbed on the interfacing alkali atom with 2-propanol, and after that, benzaldehyde is adsorbed on the same alkali atom with 2-propanol. Anisaldehyde binds 2-propanol and forms a six-membered cyclic transition state. The reduction of anisaldehyde is accelerated by being attracted by benzaldehyde oxygen.
In Scheme 2, when overdose of benzaldehyde (40 mL benzaldehyde as solvent) is used in the reactant pairs, the excess part occupies most of Rb3PO4/K3PO4 surface, and very little 2,6-dichlorobenzaldehyde is adsorbed and catalyzed into 2,6-dichlorobenzyl alcohol. When a mixture of equimolar benzaldehyde and 2,6-dichlorobenzaldehyde with excessive 2-propanol were added in the reaction mixture, benzaldehyde and 2,6-dichlorobenzaldehyde competed for the active surface sites of Rb3PO4/K3PO4, whereas the interactions between them accelerated each other��s reduction. Hence, there seems no macroscopic interactions between benzaldehyde and 2,6-dichlorobenzaldehyde because of the two opposite effects.
To verify our postulated catalytic cycle, another experiment was performed under the following conditions: 40 mL of benzaldehyde as solvent, 6.0 mmol 2,6-dichlorobenzaldehyde, 10.0 mL of 2-propanol, 0.4 mmol Rb3PO4 as catalyst, 80 ��C and air. Compared with the experiment above, only 4.0 mmol K3PO4 was replaced by 0.4 mmol Rb3PO4. After 5.0 h, 19.7% of 2,6-dichlorobenzaldehyde was reduced and 34.3% of it turned into 2,6-dichlorobenzyl alcohol after 9.0 h. Obviously, 2,6-dichlorobenzaldehyde, when promoted by Rb3PO4, became more competitive in the reaction mixture. The outcomes cannot be ascribed to different active sites indicated by CHUAH et al. The result of this competitive experiment is shown below in Fig. 4.
In our postulated catalytic cycle, trace 2,6-dichlorobenzaldehyde competes for the active sites with excess benzaldehyde. Compared with K3PO4, Rb3PO4 promoted the reduction of 2,6-dichlorobenzal- dehyde almost 10 times faster and that of benzaldehyde only 3 times faster (Table 1). Accordingly, when Rb3PO4 was used, 2,6-dichlorobenzaldehyde bound 2-propanol more competitively and became more prone to forming a six-membered cyclic transition state.
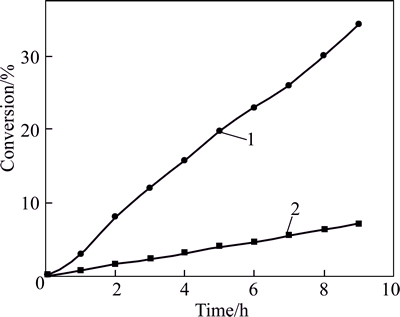
Fig. 4 Conversion of 2,6-dichlorobenzaldehyde(Reaction conditions: 1-40 mL of benzaldehyde as solvent, 6.0 mmol 2,6-dichlorobenzaldehyde, 10.0 mL of 2-propanol, 0.4 mmol Rb3PO4 as catalyst, 80 ��C and air; 2-40 mL of benzaldehyde as solvent, 6.0 mmol 2,6-dichlorobenzaldehyde, 10.0 mL of 2-propanol, 4.0 mmol K3PO4 as catalyst, 80 ��C and air)
Moreover, to test the synergist and competed effect of different substrates, interesting experiments were carried out under the following reaction conditions: 20.0 mL of 2-propanol as solvent, 2.0 mmol substrates, 1.0 mmol Rb3PO4, 80 ��C and air. When benzaldehyde, 4-bromobenzaldehyde and anisaldehyde were used alone, 62%, 6% and 49% of them were converted after 2.0 h, respectively. When benzaldehyde and 4-bromobenzal- dehyde or benzaldehyde and anisaldehyde were used as reactant pairs, their conversions were 73.5% and 3% or 83% and 24.6%, respectively. Surprisingly, the conversion of benzaldehyde increased when used in pairs. The results of these competitive experiments are shown in Figs. 5-7.
As we expected, 4-bromobenzaldehyde or anisaldehyde competed with benzaldehyde for the active surface sites of Rb3PO4, and the interactions between them also accelerated the reduction of benzaldehyde. As a result, the conversion of 4-bromobenzaldehyde or anisaldehyde was decreased and that of benzaldehyde was elevated.
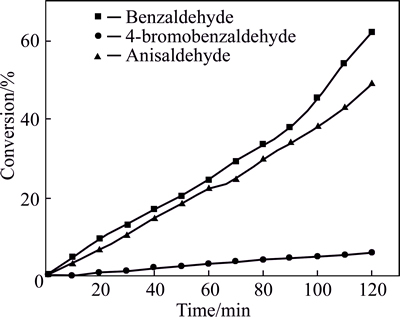
Fig. 5 Conversion of benzaldehyde, 4-bromobenzaldehyde and anisaldehyde (Reaction conditions: 20.0 mL of 2-propanol,2.0 mmol substrates, 1.0 mmol Rb3PO4, 80 ��C and air)

Fig. 6 Conversion of benzaldehyde and 4-bromobenzaldehyde (Reaction conditions: 20.0 mL of 2-propanol, each of substrates are 1.0 mmol, 1.0 mmol Rb3PO4, 80 ��C and air)
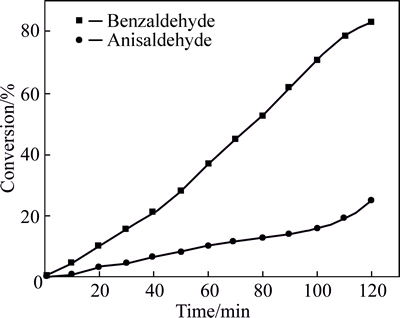
Fig. 7 Conversion of benzaldehyde and anisaldehyde (Reaction conditions: 20.0 mL of 2-propanol, each of substrates are 1.0 mmol, 1.0 mmol Rb3PO4, 80 ��C and air)
3.5 Reuse of Rb3PO4
The possibility of reusing Rb3PO4 was tested (Table 2). The solid base was recovered after reduction of 2,6-dichlorobenzaldehyde, washed with 2-propanol, dried, and thereafter used for another reduction experiment. After being reused 5 times, its catalytic activity was almost identical to that of fresh Rb3PO4. Reusing catalysts can reduce waste and save resources, so Rb3PO4 is a green, potentially eligible solid base catalyst for the catalytic transfer hydrogenation of aldehydes and ketones.
Table 2 Reuse of Rb3PO4 to reduce 2,6-dichlorobenzaldehyde

4 Conclusions
Rb3PO4 promoted transfer hydrogenation more efficiently than K3PO4 did, which can be attributed to easier adsorptions of aldehydes and ketones as well as higher conductivity. Water adsorption poisoned 70% of the active sites on Rb3PO4. All the tested substrates were successfully reduced at room temperature by Rb3PO4, with the activities following a descending sequence of 2,6-dichlorobenzaldehyde > 4-bromobenzaldehyde > benzaldehyde > acetophenone > anisaldehyde > butanone. As evidenced by the studies with reactant pairs, there were two different cyclic transition states. When 4-bromobenzaldehyde and anisaldehyde were used as reactant pairs, the reduction of benzaldehyde was accelerated. A possible catalytic cycle that postulated a six-membered cyclic transition state for the M3PO4 (M=K, Rb) catalyzed reductions of aldehydes and ketones was proposed, by which the experimental results were well explained. This research results expand the catalytic usage of Rb3PO4 which can be more conveniently obtained by extracting trace Rb+ from the salt lake brine, and worth in industrial application.
References
[1] STASTNA E, CERNY I, POUZAR V, CHODOUNSKA H. Stereoselectivity of sodium borohydride reduction of saturated steroidal ketones utilizing conditions of luche reduction [J]. Steroids, 2010, 75(10): 721-725.
[2] WU Xiao-feng, XIAO Jian-liang. Aqueous-phase asymmetric transferhydrogenationofketones-A greener approach to chiral alcohols [J]. Cheminform, 2007, 38(42): 2449-2466.
[3] NOYORI R, OHKUMA T. Asymmetric catalysis by architectural and functional molecular engineering: Practical chemo- and stereoselective hydrogenation of ketones [J]. Angewandte Chemie International Edition, 2001, 40(1): 40-73.
[4] LOGES B, BODDIEN A, JUNGE H, BELLER M. Controlled generation of hydrogen from formic acid amine adducts at room temperature and application in H2/O2 fuel cells [J]. Angewandte Chemie International Edition, 2008, 47(21): 3962-3965.
[5] IVANOV V A, BACHELIER J, AUDRY F, LAVALLEY J C. Study of the Meerwein��Pondorff��Verley reaction between ethanol and acetone on various metal oxides [J]. Journal of Molecular Catalysis, 1994, 91(1): 45-59.
[6] CHA J S. Reactions for the reduction of organic functional groups using aluminum, boron, and other metal reagents: A review [J]. Organic Process Research & Development Journal, 2006, 10(5): 1032-1053.
[7] VERMA P K, SHARMA U, KUMAR N, BALA M, KUMAR V, SINGH B. Nickel phthalocyanine assisted highly efficient and selective carbonyl reduction in polyethylene glycol-400 [J]. Catalysis Letters, 2012, 142(7): 907-913.
[8] BLUM J, PRI-BAR I. Selective transfer hydrogenation of various unsaturated functions by polymethylhydrosiloxane in the presence of a recyclable rhodium catalyst [J]. Journal of Molecular Catalysis 1986, 27(2/3): 359-367.
[9] MOECZNIKOWSKI J R, CRABTREE R H. Transfer hydrogenation reduction of ketones, aldehydes and imines using chelated iridium(III) N-heterocyclic bis-carbene complexes [J]. Polyhedron, 2004, 23(17): 2857-2872.
[10] JIMENEZ S, LOPEZ J A, CIRIANO M A, TEJEL C, MARTINEZ A, SANCHEZ-DELGADO R A. Selective hydrogenation of cinnamaldehyde and other ��,��-unsaturated substrates catalyzed by rhodium and ruthenium complexes [J]. Organometallics, 2009, 28(11): 3193-3202.
[11] HE Lin, NI Ji, WANG Lu-cun, YU Feng-jiao, CAO Yong, HE He-yong, FAN Kang-nian. Aqueous room-temperature gold- catalyzed chemoselective transfer hydrogenation of aldehydes [J]. Chemistry-A European Journal, 2009, 15(44): 11833-11836.
[12] BACCHI A, BALORDI M, CAMMI R, ELVIRI L, PELIZZI C, PICHHIONO F, VERDOLINO V, GOUBITZ K, PE-SCHAR R, PELAGATTI P. Mechanistic insights into acetophenone transfer hydrogenation catalyzed by half-sandwich ruthenium(II) complexes containing 2-(Diphenylphosphanyl)aniline�CA combined experimental and theoretical study [J]. European Journal of Inorganic Chemistry, 2008, 2008(28): 4462-4473.
[13] BACKVALL J E. Transition metal hydrides as active intermediates in hydrogen transfer reactions [J]. Journal of Organometallic Chemistry, 2002, 652(1/2): 105-111.
[14] POLSHETTIWAR V, VARMA R S. Revisiting the Meerwein�C Ponndorf�CVerley reduction: A sustainable protocol for transfer hydrogenation ofaldehydesandketones [J]. Green Chemistry, 2009, 11(9): 1313-1316.
[15] OUALI A, MAJORAL J P, CAMINADE A M, TAILLEFER M. NaOH-promoted hydrogen transfer: Does NaOH or traces of transition metals catalyze the reaction? [J]. ChemCatChem, 2009, 1(4): 504-509.
[16] LIU Shi-ming, LIU He-hui, HUANG Yun-jing, YANG We-jun. Solvent extraction of rubidium and cesium from salt lake brine with t-BAMBP�Ckerosene solution [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(1): 329-334.
[17] VORONIN V I, BERGER I F, PROSKURNINA N V, SHEPTYAKOV D V, GOSHCHITSKII B N, BURMAKIND E I, STROEV S S, SHEKHTMAN G S. Crystal structure of the low-temperature forms of cesium and rubidium orthophosphates [J]. Inorganic Materials, 2008, 44(6): 646-652.
[18] ADAMS D L. New phenomena in the adsorption of alkali metals on Al surfaces [J]. Applied Physics A: Materials Science & Processing, 1996, 62(2): 123-142.
[19] RADHAKRISHAN R, DO D M, JAENICKE S, SASSON Y, CHUAH G K. Potassium phosphate as a solid base catalyst for the catalytic transfer hydrogenation of aldehydes and ketones [J]. ACS Catalysis, 2011, 1(11): 1631-1636.
[20] VORONIN V I, PONOSOV Y S, BERGER I F, PROSKURNINA N V, ZUBKOV V G, TYUTYUNNIK A P, BUSHMELEVA S N, BALAGUROV A M, SHEPTYAKOV D V, BURMAKIN E I,. SHEKHTMAN G S, VOVKOTRUB E G. Crystal structure of the low-temperature form of K3PO4 [J]. Inorganic Materials, 2006, 42(8): 908-913.
(Edited by YANG Bing)
Foundation item: Project(21576074) supported by the National Natural Science Foundation of China
Received date: 2015-05-26; Accepted date: 2015-08-30
Corresponding author: YANG Wei-jun, Professor, PhD; Tel: +86-731-88821449; E-mail: wjyang@hnu.edu.cn

