Ammonia nitrogen removal from aqueous solution using zeolite modified by microwave-sodium acetate
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2016���6��
�������ߣ��ֺ� ��ӱ��
����ҳ�룺1345 - 1352
Key words��modified zeolite; microwave; sodium acetate; ammonia-nitrogen; sorption characterization
Abstract: The characteristics of the zeolite modified by microwave and sodium acetate and its sorption of ammonia-nitrogen from simulated water sample were investigated. The results show that the modified zeolite by microwave-sodium acetate (SMMZ) has a high sorption efficiency and removal performance. The ammonia-nitrogen removal rate of SMMZ reaches 92.90%. The surface of SMMZ becomes loose and some pores appear, the specific surface area, total pore volume and average pore diameter increase after modification. Compared to the natural zeolite, SMMZ has a more concentrated pore size distribution in the range of 0-10 nm. The cation exchange capacity (CEC) of SMMZ is higher than that of the natural zeolite. And the ammonia nitrogen removal rate is consistent with the change of CEC. The SMMZ possesses rapid sorption and slow balance characteristics and ammonia-nitrogen sorption is consistent with both Langmuir adsorption isotherm model and Freundlich adsorption isotherm model. The adsorption kinetics of ammonia-nitrogen follows the pseudo-second order kinetic model.
J. Cent. South Univ. (2016) 23: 1345-1352
DOI: 10.1007/s11771-016-3186-x

DONG Ying-bo(��ӱ��)1, 2, LIN Hai(�ֺ�)1, 2
1. Department of Environmental Engineering, University of Science and Technology Beijing, Beijing 100083, China;
2. Beijing Key Laboratory on Resource-oriented Treatment of Industrial Pollutants, Beijing 100083, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: The characteristics of the zeolite modified by microwave and sodium acetate and its sorption of ammonia-nitrogen from simulated water sample were investigated. The results show that the modified zeolite by microwave-sodium acetate (SMMZ) has a high sorption efficiency and removal performance. The ammonia-nitrogen removal rate of SMMZ reaches 92.90%. The surface of SMMZ becomes loose and some pores appear, the specific surface area, total pore volume and average pore diameter increase after modification. Compared to the natural zeolite, SMMZ has a more concentrated pore size distribution in the range of 0-10 nm. The cation exchange capacity (CEC) of SMMZ is higher than that of the natural zeolite. And the ammonia nitrogen removal rate is consistent with the change of CEC. The SMMZ possesses rapid sorption and slow balance characteristics and ammonia-nitrogen sorption is consistent with both Langmuir adsorption isotherm model and Freundlich adsorption isotherm model. The adsorption kinetics of ammonia-nitrogen follows the pseudo-second order kinetic model.
Key words: modified zeolite; microwave; sodium acetate; ammonia-nitrogen; sorption characterization
1 Introduction
Ammonia nitrogen is one of the main pollutants, bringing the water eutrophication and environmental pollution problem, which could inhibit the nitration reaction, consume the oxygen in water, make fish poisoning, and reduce the self-purification ability of water [1]. At present, more than 85% of the lakes present eutrophication phenomenon in China [2-3]. This problem is as well as global. Therefore, controlling and removing the ammonia nitrogen in water through economic and effective methods is very important for the use of reclaimed water [4].
Adsorption method is widely used to remove the ammonia nitrogen in water, due to many advantages, such as low cost, low investment, fast processing, high efficiency, wide range of applications, easy management and environment friendly [5-6]. Thus, finding new efficient materials for adsorption has become a research highlight [7-9]. The natural zeolite has large adsorption ratio surface area comparing to other mineral materials. Therefore, researchers focus on sorbents based on zeolites in recent years. The natural zeolite is usually modified to get more efficient adsorption effects. At present, the main modification methods of natural zeolite include roasting modification and chemical modification [10-16]. But the research of the microwave modification and microwave-chemical modification is still few [17]. Meanwhile, the most research of zeolite focuses on removing high concentration of ammonia nitrogen in wastewater, but there is few research on the removing the low concentration (less than 10 mg/L) of ammonia nitrogen in water, which is more difficult to treat than high concentration of ammonia nitrogen [18].
In this work, removal of low concentration ammonium from aqueous solution using microwave modified zeolite (MMZ for short) and microwave- sodium acetate modified zeolite (SMMZ for short) was investigated. The characteristics of SMMZ and its adsorption mechanism of ammonia nitrogen are the main factors studied.
2 Materials and methods
2.1 Materials
The zeolite material with particle size ranging from 100 ��m to 150 ��m was supplied by a company in Shenyang, China. The X-ray diffraction (XRD) pattern of the material is shown in Fig. 1. The main component of the natural zeolite was clinoptilolite associated with cristobalite and sanidine. The main chemical composition of zeolite is given in Table 1.
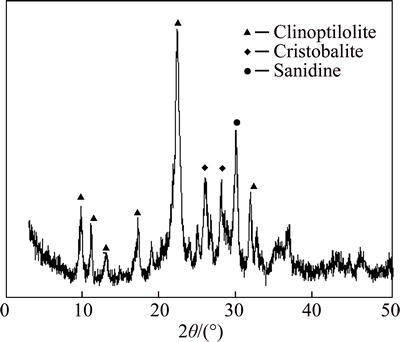
Fig. 1 XRD pattern of natural zeolite
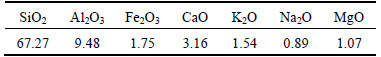
Table 1 Main chemical compositions of natural zeolite (mass fraction, %)
2.2 Water sample
According to the condition of the reclaimed water, the simulated wastewater sample was prepared containing ammonia-nitrogen of 6 mg/L. The reagents used in this work were ammonium chloride. The pH value of water sample was adjusted to 6.0.
2.3 Modification of natural zeolite
Modification experiments were carried out in a microwave oven. The microwave modified zeolite (MMZ) was obtained as follows. A crucible containing 8 g natural zeolite was placed in the microwave oven to irradiate at 343 W and 2450 MHz for 4 min. Secondly, the zeolite modified by microwave was cooled for 12 h in closed container. Then the MMZ was kept in dry place.
The zeolite modified by microwave-sodium acetate was obtained as follows. 8 g natural zeolite and 200 mL 2% CH3COONa solution were placed in a 400 mL flask. The flask was maintained at 25��C and shaken at 300 r/min for 30 min before irradiation in a microwave oven at 343 W and 2450 MHz for 10 min. The modified zeolite was washed with distilled water three times and dried at 85��C. Then the microwave-sodium acetate modified zeolite (SMMZ) was kept in dry place.
2.4 Sorption experiment
Water samples containing 6 mg/L ammonia- nitrogen was prepared at 25 ��C. The 400 mL flasks containing the water samples and 0.4 g modified zeolite were rotated for some time in a thermo stated rotator (30 ��C, 300 r/min) in order to achieve sorption equilibrium.
2.5 Cation exchange capacity test
0.5 g of adsorbing material and 25 mL NH4Cl solution of 1 mol/L were mixed in a 150 mL conical flask. The suspension was stirred at 100��C for 30 min. Subsequently, the adsorbing material was separated using a quantitative filter paper and washed with distilled water until no chloride was present in the supernatant. Then, the filter paper and adsorbing material were washed into a 250 mL flask with 40 mL KCl solution of 1 mol/L. After that, 8 mL formaldehyde was added in the flask and kept rest for 5 min. Finally, 2-4 drops of phenolphthalein indicator were added in the flaks before the titration with 0.1 mol/L NaOH standard solution.
The cation exchange capacity (CEC, Cce) of the adsorbing materials was calculated using the following equation:
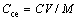
where C is the concentration of the NaOH standard solution (mol/L); V is the consumed volume of the NaOH standard solution (mL); and M is the mass of adsorbing materials (g).
2.6 Sorption isotherm experiment
Water samples containing 3, 6, 9, 12, 15, 20, 30 and 60 mg/L of ammonia-nitrogen were prepared. First, the water samples and 0.4 g of the raw natural zeolite and SMMZ were placed in several 400 mL flasks, respectively. The flasks were then placed in a constant temperature oscillator for some time at 30 ��C and 300 r/min rotational speed to achieve sorption equilibrium. Then the solution was filtered using a 0.45 ��m filter membrane and the concentrations of ammonia-nitrogen in the filtered solution were measured. Total amount of adsorbed ammonia-nitrogen was divided by the dry mass of zeolite to find out the average amount adsorbed, which was plotted against effluent ammonia-nitrogen concentrations to show isotherm test.
2.7 Adsorption kinetics experiment
Approximately 0.4 g MMZ and SMMZ was weighed, respectively. The sample was placed in a 200 mL solution with 6.0 mg/L ammonia-nitrogen. The solution was stirred with speed of 300 r/min. The solution was then filtered using a 0.45 ��m filter membrane and the concentrations of ammonia-nitrogen in the filtered solution were measured.
2.8 Analytical methods
The ammonia-nitrogen content of the water samples was determined using the Nessler reagent spectrophotometric methods. The morphological features of the natural and different modified zeolites were analyzed by SEM. The nitrogen adsorption analyzer was used for surface structure analysis of the natural and different modified zeolites.
3 Results and discussion
3.1 Results of sorption experiment
The ammonia-nitrogen removal rates of natural zeolite, MMZ and SMMZ are shown in Fig. 2. The results show that after sorption by natural zeolite, MMZ and SMMZ at equilibrium, the residual ammonia- nitrogen contents in the water sample are 2.36 mg/L, 1.41 mg/L and 0.43 mg/L, respectively. And the ammonia-nitrogen removal rates are 60.7%, 76.5% and 92.9%, respectively. Compared with natural zeolite, the ammonia-nitrogen removal rates of MMZ and SMMZ are higher by 32.2% and 16.4%, respectively. The observation indicates that the adsorption capacity of zeolites modified by microwave or microwave-sodium acetate has been improved. Moreover, the SMMZ has the best ammonia-nitrogen removal efficiency.
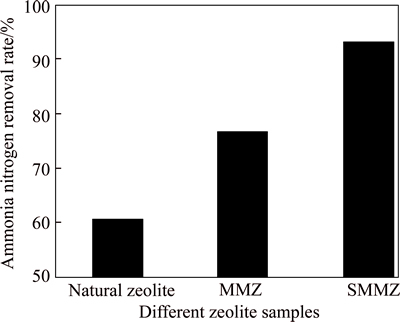
Fig. 2 Ammonia-nitrogen removal rate of different zeolite samples
3.2 SEM analysis
The SEM results of natural zeolite, MMZ and SMMZ are compared, as shown in Figs. 3(a), (b) and (c), respectively. The figures show that the surface of raw natural zeolite is much smoother than those of MMZ and SMMZ. Furthermore, the surface of SMMZ is looser than that of MMZ. In addition, the surface of the SMMZ is dispersed and pores are present.
3.3 Surface structure analysis
The specific surface area, pore size distribution and pore volume are very important physical constants of porous materials [19]. The specific surface area, total pore volume and average pore diameter of natural zeolite, MMZ and SMMZ are compared. The results are listed in Table 2. The results show that the specific surface area, total pore volume and average pore diameter of zeolite all increase after the modification. And the SMMZ has the largest specific surface area, total pore volume and average pore diameter, which may improve the adsorption performance of zeolite and verify the total pore volume. The specific surface area of zeolite is positively related to the zeolite adsorption capacity of ammonia-nitrogen.
The pore size distributions of natural zeolite, MMZ and SMMZ are shown in Fig. 4. The pore diameters of zeolites samples are all mainly distributed between 0 and 30 nm. However, the MMZ and SMMZ have a more concentrated pore size distribution, particularly the SMMZ, compared with natural zeolite. The channels distribution of MMZ in the range from 0 to 10 nm accounts for 76.1% of the total channels number, whereas that of SMMZ accounts for 84.6%, exhibiting increase of 24.3% compared with the natural zeolite. The reasons are as follows. The channels of natural zeolite contain open pore and closed pore. The closed pores are opened and the pore size distributions are more even after microwave treatment [20], which improves the adsorption performance of zeolite. Moreover, sodium acetate as the additive can distinctly accelerate the diffusion effect during the microwave treatment, and pore size distribution changes more even after microwave-sodium acetate treatment. The adsorption sites of zeolite are mainly ascribed to the mesoporous and microporous channels, so the SMMZ has better adsorption capacity of ammonia-nitrogen than the natural zeolite.

Fig. 3 SEM images of natural zeolites (a), MMZ (b) and SMMZ (c)
Table 2 Specific surface area, total pore volume and average pore diameter of different zeolite samples
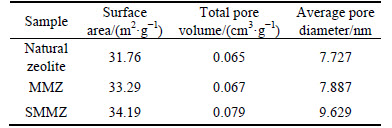
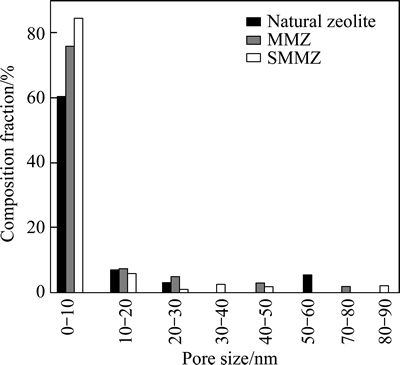
Fig. 4 Pore size distribution of natural zeolite, MMZ and SMMZ
3.4 Results of CEC test
Metal cations, such as potassium ion, calcium ion and magnesium ion, can be separated from the zeolite crystal skeleton through exchange effect without changing the structure of zeolite. CEC is one of the important indicators of adsorption capacity of zeolite, and it partly reflects the maximum ion exchange capacity of adsorption material [21]. In order to compare ammonia-nitrogen adsorption capacity of natural zeolite, MMZ and SMMZ, the CEC of them are tested. The CEC of the raw natural zeolite is 1.62 mmol/g.
The effects of microwave treatment time on the CEC and ammonia-nitrogen removal rate of MMZ and SMMZ at microwave power of 343 W are shown in Fig. 5. The results show that MMZ has the largest CEC when the microwave treatment time is 4 min, whereas SMMZ has the largest CEC when the microwave treatment time is 10 min. The ammonia nitrogen removal rates of MMZ and SMMZ are consistent with the change of CEC, which reaches the maximum at the microwave treatment time of 4 min and 10 min, respectively.
Figure 6 shows the effects of microwave power on the CEC and ammonia-nitrogen removal rate of MMZ and SMMZ at microwave treatment time of 4 min and 10 min, respectively. The observation indicates that the ammonia-nitrogen removal rate of MMZ and SMMZ is consistent with the change of CEC once again. Under a microwave power of 343 W for 4 min, the largest CEC and ammonia-nitrogen removal rate of MMZ are 1.75 mmol/g and 76.5%, respectively. Whereas under a microwave power of 343 W for 10 min, the largest CEC and ammonia-nitrogen removal rate of SMMZ are 1.90 mmol/g and 92.9%, respectively. The CECs of MMZ and SMMZ are 0.13 and 0.28 mmol/g higher than that of raw natural zeolite (1.62 mmol/g), respectively. The reasons are as follows. The microwave treatment can increase the porosity and the channel number of zeolite, which brings the increase of CEC. Ca2+ and Mg2+ could be replaced by Na-ions during the microwave-sodium acetate treatment. The ionic radius of Na+ is shorter than that of Ca2+ and Mg2+, so the effective aperture and exchange capacity of the modified zeolite increase [22]. The ammonia-nitrogen removal effects of SMMZ are improved. In a sense, the CEC test results reveal the reason why the ammonia-nitrogen removal rate of MMZ and SMMZ are higher than that of the natural zeolite.

Fig. 5 Effect of microwave treatment time on CEC (a) and ammonia-nitrogen removal rate (b)

Fig. 6 Effects of microwave power on CEC (a) and ammonia- nitrogen removal rate (b)
The CEC of zeolite is closely related to the particle electronegativity. Meanwhile, the particle electro- negativity is affected by the internal silica-alumina ratio of zeolite. The CEC and Si/Al mass ratios of the raw natural zeolite, MMZ and SMMZ are summarized in Table 3. The results show that a negative correlation is observed between the CEC and silica�Calumina ratio. Lower silica�Calumina ratio results in higher CEC. It is because that the relatively high concentration of aluminum can choose to adsorb water, resulting in the increase of coordination cation, so the zeolite has larger CEC and adsorption performance [23].
Table 3 CEC and Si/Al mass ratio of different zeolite samples

3.5 Results of adsorption isotherm experiment
The ammonia-nitrogen adsorption quantities of natural zeolite and SMMZ under different initial ammonia-nitrogen concentration are shown in Fig. 7. The results indicate that the ammonia-nitrogen adsorption quantities of natural zeolite and SMMZ both increase with the increase of initial ammonia-nitrogen concentration. Meanwhile, the ammonia-nitrogen adsorption quantity of SMMZ is higher than that of natural zeolite at the same initial ammonia-nitrogen concentration.
Both Freundlich and Langmuir models are used for the evaluation of experimental results. The Langmuir adsorption isotherm model is expressed as [24]:
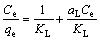 (1)
(1)

Fig. 7 Evolution of ammonia-nitrogen adsorption quantity vs initial ammonia-nitrogen concentration
On the other hand, the Freundlich adsorption isotherm model is expressed as:
 (2)
(2)
The Langmuir and Freundlich adsorption isotherms of natural zeolite and SMMZ absorbing ammonia- nitrogen are shown in Figs. 8 and 9, respectively. And the fitting parameters are shown in Table 4.
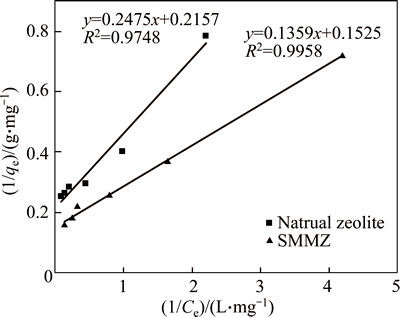
Fig. 8 Ammonia-nitrogen Langmuir adsorption curve of natural zeolite and SMMZ

Fig. 9 Ammonia-nitrogen Freundlich adsorption curve of natural zeolite and SMMZ
Table 4 Fitting parameters of Langmuir adsorption isotherm and Freundlich adsorption isotherm

The experiment results show that the correlation coefficient of Langmuir adsorption is higher than that of the Freundlich adsorption for the natural zeolite. The ammonia-nitrogen adsorption isotherms are well- reproduced to the Langmuir adsorption isotherm model, which means that the ammonia-nitrogen adsorption type of natural zeolite involves monolayer coating. Whereas the ammonia-nitrogen adsorption isotherms are both well-reproduced to the Langmuir adsorption isotherm model and Freundlich adsorption isotherm model for SMMZ, which means that the ammonia-nitrogen adsorption type of SMMZ involves monolayer and multilayer coating [25]. For Langmuir adsorption isotherm model, the monolayer adsorption saturation capacity of SMMZ is 6.56mg/L, which is improved by 38% compared to the natural zeolite. These indicate that SMMZ has better ammonia-nitrogen adsorption capacity than the raw natural zeolite.
3.6 Results of adsorption kinetics experiment
The ammonia nitrogen adsorption quantities of natural zeolite and SMMZ for different adsorption time are given in Fig. 10. The results show that the ammonia nitrogen adsorption characteristics of natural zeolite and SMMZ are both fast absorption and slow balance with increasing time. The adsorption reaction continues for 240 min, thus, the concentration of ammonia nitrogen in the effluent is lower than 0.4 mg/L. In the first 60 min of the adsorption reaction, the ammonia nitrogen adsorption rate increases rapidly, and ammonia adsorption quantitylargely increases. After the adsorption time of 90 min, the adsorption rate slows down, but the adsorbed content of ammonia nitrogen increases. After adsorption time of 3 h, the ammonia nitrogen adsorption content of natural zeolite and SMMZ both reach the adsorption equilibrium.

Fig. 10 Evolution of ammonia-nitrogen adsorption quantity vs adsorption time
Meanwhile, regressions of the experimental results are conducted. The pseudo-first-order model, the pseudo- second-order model and the intra-particle diffusion model are used. The kinetic equations of different models are expressed as Eqs. (3), (4), and (5) [26-28].
The pseudo-first-order model equation:
 (3)
(3)
The pseudo-second-order model equation:
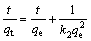 (4)
(4)
The intra-particle diffusion model equation:
 (5)
(5)
where k1 is the rate constant of the pseudo-first-order model (h-1); k2 is the rate constant of the pseudo-second- order model (g/(mg��h)); k3 is the rate constant of the intra-particle diffusion model (mg/(g��h0.5)); and C is the empirical constant.
The three kinds of models fitted with the experimental kinetic data for natural zeolite and SMMZ are shown in Figs. 11, 12, and 13.
The regression parameters and correlation coefficient (R2) in ammonia nitrogen adsorption kinetics equations of natural zeolite and SMMZ are shown in Table 5.
Based on the R2 of the adsorption power function and the pseudo-second-order model adsorption kinetics, the pseudo-second-order model adsorption kinetics equation is better than other models in depicting the SMMZ adsorption kinetics of ammonia nitrogen. Based on the pseudo-second-order model adsorption kinetics equation, the adsorption rate constant of SMMZ is higher than that of natural zeolite, indicating that the adsorption capacity of SMMZ is superior to that of natural zeolite.

Fig. 11 Pseudo-first order model of ammonia nitrogen adsorption
Table 5 Regression parameters of natural zeolite and SMMZ on ammonia nitrogen adsorption kinetics

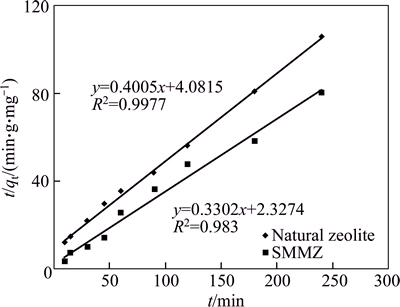
Fig. 12 Pseudo-second order model of ammonia nitrogen adsorption

Fig. 13 Intra-particle diffusion model of ammonia nitrogen adsorption
4 Conclusions
1) The microwave modified zeolite and the microwave-sodium acetate modified zeolite (SMMZ) have more efficient sorption performance and higher removal effect of ammonia-nitrogen than the raw natural zeolite, particularly the SMMZ.
2) The characteristics of modified zeolites and its sorption mechanism are revealed by some testing means. The SEM analyses show that the surfaces of MMZ and SMMZ are looser than that of the raw natural zeolite. The specific surface area, total pore volume and average pore diameter of zeolite increase after modification. And SMMZ has the largest index data. Compared to the natural zeolite, MMZ and SMMZ have more homogeneous and continuous pore size distribution in the range of 0-10 nm, particularly the SMMZ. The ammonia nitrogen removal rate of zeolite is consistent with the change of CEC. The CECs of MMZ and SMMZ are 0.13 and 0.28 mmol/g higher than that of the raw natural zeolite, respectively.
3) The ammonia-nitrogen adsorption data of SMMZ could be both well fitted by the Langmuir isotherm model and Freundlich adsorption isotherm model. The experimental data fit the pseudo-second order kinetic model well, and the sorption of SMMZ to ammonia- nitrogen process involves rapid adsorption and slow equilibrium.
References
[1] YENIG N O, DEMIREL B. Ammonia inhibition in anaerobic digestion: A review [J]. Process Biochemistry, 2013, 48(5/6): 901-911.
N O, DEMIREL B. Ammonia inhibition in anaerobic digestion: A review [J]. Process Biochemistry, 2013, 48(5/6): 901-911.
[2] HERBECK L S, UNGER D, WU Y, JENNERJAHN T C. Effluent, nutrient and organic matter export from shrimp and fish ponds causing eutrophication in coastal and back-reef waters of NE Hainan, tropical China [J]. Continental Shelf Research, 2013, 57(1): 92-104.
[3] YI L L, JIAO W T, CHEN X N, CHEN W P. An overview of reclaimed water reuse in China [J]. Journal of Environmental Sciences, 2011, 23(10): 1585-1593.
[4] MENA-DURAN C J, SUN M R S, LOPEZ T, AZAMAR-BARRIOS J A, AGUILAR D H,  M I, ODRIOZOLA J A, QUINTANA P. Nitrate removal using natural clays modified by acid thermoactivation [J]. Applied Surface Science, 2007, 253(13): 5762-5766.
M I, ODRIOZOLA J A, QUINTANA P. Nitrate removal using natural clays modified by acid thermoactivation [J]. Applied Surface Science, 2007, 253(13): 5762-5766.
[5] PITAKPOOLSIL W, HUNSOM M. Treatment of biodieselwastewaterbyadsorptionwith commercial chitosan flakes: Parameter optimization and process kinetics [J]. Journal Environmental Management,2014, 133(15): 284-292.
[6] CYRUS J S, REDDY G B. Sorption and desorption of ammonium by zeolite: Batch and column studies [J]. Journal of Environmental Science & Health Part A, 2011, 46(4): 408-414.
[7] HUANG H, XIAO X, YAN B, YANG L. Ammonium removal from aqueous solutions by using natural Chinese (Chende) zeolite as adsorbent [J]. Journal of Hazardous Materials, 2010, 175(1/2/3): 247-252.
[8] WANG S B, PENG Y L. Natural zeolites as effective adsorbents in water and wastewater treatment [J]. Chemical Engineering Journal, 2010, 156(1): 11-24��
[9] KRISHNANI K K,ZHANG Y,XIONG L,YAN Y,BOOPATHY R,MULCHANDANI A. Bactericidal and ammonia removal activity of silver ion-exchanged zeolite [J]. Bioresource Technology, 2012, 117: 86-91.
[10]  M,
M,  A,
A,  M,
M,  J,GATTA G D, ROTIROTI N. Characterization of lead sorption by the natural and Fe(III)-modified zeolite [J]. Applied Surface Science, 2013, 283(11): 764-774.
J,GATTA G D, ROTIROTI N. Characterization of lead sorption by the natural and Fe(III)-modified zeolite [J]. Applied Surface Science, 2013, 283(11): 764-774.
[11] ZHAN Y H, LIN J W, ZHU Z L. Removal of nitrate from aqueous solution using cetylpyridinium bromide (CPB)modified zeoliteas adsorbent [J]. Journal of Hazardous Materials, 2011, 186(2/3): 1972-1978.
[12] PANNEERSELVAM P, THINAKARAN N, THIRUVENKATARAVI K V, PALANICHAMY M, SIVANESAN S. Phosphoric acidmodified-Yzeolites: A novel, efficient and versatile ion exchanger [J]. Journal of Hazardous Materials, 2008, 159(2/3): 427-434.
[13] YAN C F, GRACE J R, LIM C J. Effects of rapid calcination on properties of calcium-based sorbents [J]. Fuel Processing Technology, 2010, 91(11): 1678-1686.
[14] KURAMA H, ZIMMER A, RESCHETILOWSKI W. Chemical modification effect on the sorption capacities of natural clinoptilolite [J]. Chemical Engineering & Technology, 2002, 25(3): 301-305.
[15] GUO J Y, YANG C P, ZENG G M. Treatment of swine wastewater using chemicallymodified zeoliteand bioflocculant from activated sludge [J]. Bioresource Technology, 2013, 143(1): 289-297.
[16] KUCHERENKO I S, SOLDATKIN �� ��, SOY E, KIRDECILER K,  ZT
ZT RK S, AKATA B, JAFFREZIC-RENAULT N, SOLDATKIN A P, DZYADEVYCH S V. Effect of differentmodificationsof BEA-zeoliteson operational characteristics of conductometric biosensor [J]. Materials Science & Engineering C, 2012, 32(6): 1648-1653.
RK S, AKATA B, JAFFREZIC-RENAULT N, SOLDATKIN A P, DZYADEVYCH S V. Effect of differentmodificationsof BEA-zeoliteson operational characteristics of conductometric biosensor [J]. Materials Science & Engineering C, 2012, 32(6): 1648-1653.
[17] LEI L C, LI X J, ZHANG X W. Ammonium removal from aqueous solutions using microwave-treated natural Chinese zeolite [J]. Separation & Purification Technology, 2008, 58(3): 359-366.
[18] WANG Y, KMIYA Y, OKUHARA T. Removal of low- concentration ammonia in water by ion-exchange using Na-mordenite [J]. Water Research, 2007, 41(2): 269-276.
[19] FREIDING J, PATCAS F C, KRAUSHAAR-CZARNETZKI B. Extrusion of zeolites: Properties of catalysts with a novel aluminium phosphate sintermatrix [J]. Applied Catalysis A: General, 2007, 328(2): 210-218.
[20] MONTI M, SANTORO A. Natural zeolite reactivity towards ozone: The role of compensating cations [J]. Journal of Hazardous Materials, 2012, 227-228(5): 34-40.
[21] YENER N,  C,
C,  NAL M, SARIKAYA Y. Simultaneous determination of cation exchange capacity and surface area of acid activated bentonite powders by methylene blue sorption [J]. Applied Surface Science, 2012, 258(7): 2534-2539.
NAL M, SARIKAYA Y. Simultaneous determination of cation exchange capacity and surface area of acid activated bentonite powders by methylene blue sorption [J]. Applied Surface Science, 2012, 258(7): 2534-2539.
[22] CHENG X W, ZHONG Y, WANG J, GUO J, HUANG Q, LONG Y C. Studies on modification and structural ultra-stabilization of natural STI zeolite [J]. Microporous & Mesoporous Materials, 2005, 83(1): 233-243.
[23] KARIMI L, SALEM A. The role of bentonite particle size distribution on kinetic of cation exchange capacity [J]. Journal of Industrial & Engineering Chemistry, 2011, 17(1): 90-95��
[24] HUO H X, LIN H, DONG Y B, CHENG H, WANG H, CAO L X. Ammonia-nitrogenand phosphates sorption from simulated reclaimed waters by modified clinoptilolite [J]. Journal of Hazardous Materials, 2010, 229(5): 292-297.
[25] WANG Ming, FANG Chun-sheng, YAN Chang-zhou, WEI Qun-shan, WANG Zao-sheng. Modification of natural zeolites and their ammonia adsorption characteristics [J]. Research of Environmental Science, 2012, 25(9): 1024-1029. (in Chinese)
[26] ANNADURAI G, KRISHNAN M R V. Adsorption of acid dye from aqueous solution by chitin: Equilibrium studies [J]. Indian Journal of Chemical Technology, 1997, 4(5): 217-222.
[27] HO Y S, MCKAY G. The sorption of lead (II) ions on peat [J]. Water Research, 1999, 33(2): 578-584.
[28] ZHANG X, BAI R. Mechanisms and kinetics of humic acid adsorption onto chitosan-coated granules [J].Journal of Colloid & Interface Science, 2003, 264(1): 30-38.
(Edited by FANG Jing-hua)
Foundation item: Project(51174017) supported by the National Natural Science Foundation of China
Received date: 2014-10-20; Accepted date: 2015-01-30
Corresponding author: LIN Hai, Professor, PhD; Tel: +86-10-62332526; E-mail: linhai@ces.ustb.edu.cn

