CaO/SiO2和Fe/SiO2比对空气中PbO-ZnO-CaO-SiO2-“Fe2O3”系相平衡的影响
来源期刊:中国有色金属学报(英文版)2012年第3期
论文作者:M. PEREZ-LABRA A. ROMERO-SERRANO A. HERNANDEZ-RAMIREZ ALMAGUER-GUZMAN R. BENAVIDES-PEREZ
文章页码:665 - 674
关键词:铅渣;相平衡;液相线温度
Key words:lead slags; phase equilibria; liquidus temperature
摘 要:
实验测定了CaO/SiO2和Fe/SiO2质量比分别为1~1.6和1.3~1.7、含40% PbO和8% ZnO的PbO-ZnO-CaO- SiO2-“Fe2O3”渣系的相平衡和液相线温度。将该渣系在1273~1573 K达到平衡,然后快速淬火冷却。XRD和SEM-EDS分析结果表明,该渣的成分位于锌铁尖晶石相的初始成相区,在1273~1473 K生成硅酸钙/铅,部分硅酸钙/铅和磁铁铅矿相是通过转熔反应生成的。实验结果和热力学计算结果表明,液相线温度随着渣中CaO/SiO2比的增加和Fe/SiO2比的降低而增加。
Abstract:
Experimental studies on phase equilibria and liquidus temperature in the PbO-ZnO-CaO-SiO2-“Fe2O3” system, with the mass ratios of CaO/SiO2=1-1.6 and Fe/SiO2= 1.3-1.7, and 40% PbO and 8% ZnO, were carried out between 1273 and 1573 K. Slags were equilibrated at 1273 to 1573 K and cooled rapidly by quenching. The XRD and SEM-EDS results showed that the slag compositions are in the franklinite primary phase field. Calcium and lead silicates are formed between 1373 and 1473 K. The Ca/Pb silicate and magnetoplumbite phases are partially formed by an incongruent reaction. The experimental and thermodynamical results showed that the liquidus increased by increasing CaO/SiO2 mass ratio and decreasing Fe/SiO2 mass ratio.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 665-674
M. PEREZ-LABRA1, A. ROMERO-SERRANO1, A. HERNANDEZ-RAMIREZ1,
I. ALMAGUER-GUZMAN2, R. BENAVIDES-PEREZ2
1. Metallurgy and Materials Department, IPN-ESIQIE, UPALM, Lindavista, C.P. 07738, Mexico D.F.;
2. Servicios Administrativos Pe?oles S. A de C. V. Prol. Comonfort Sur 2050,
Col. L. Echeverría, Torreón, C. P. 27300 Coahuila, Mexico
Received 23 September 2011; accepted 8 December 2011
Abstract: Experimental studies on phase equilibria and liquidus temperature in the PbO-ZnO-CaO-SiO2-“Fe2O3” system, with the mass ratios of CaO/SiO2=1-1.6 and Fe/SiO2= 1.3-1.7, and 40% PbO and 8% ZnO, were carried out between 1273 and 1573 K. Slags were equilibrated at 1273 to 1573 K and cooled rapidly by quenching. The XRD and SEM-EDS results showed that the slag compositions are in the franklinite primary phase field. Calcium and lead silicates are formed between 1373 and 1473 K. The Ca/Pb silicate and magnetoplumbite phases are partially formed by an incongruent reaction. The experimental and thermodynamical results showed that the liquidus increased by increasing CaO/SiO2 mass ratio and decreasing Fe/SiO2 mass ratio.
Key words: lead slags; phase equilibria; liquidus temperature
1 Introduction
The primary production of lead and zinc metal is mainly undertaken in blast furnaces, which operate in a wide range of temperature. HABASHI [1] showed that the reduction of lead oxide in this furnace produces slags in the multicomponent system PbO-ZnO-CaO-SiO2- “Fe2O3”. This system represents the major components of lead/zinc smelting slags in oxidizing conditions. The oxidation roasting of lead/zinc sulfide concentrates requires the fluxes addition of SiO2 and CaO to form a mixture metallurgically balanced. The CaO/SiO2 and Fe/SiO2 ratios must be used according to the composition of lead concentrates. The information of the liquidus temperatures and the mineralogical species formed is useful to optimize the industrial practice.
Experimental studies on phase equilibria in the PbO-ZnO-CaO-SiO2-“Fe2O3” system in air using different CaO/SiO2 ratios were carried out by JAK et al [2]. They studied a slag system with constant basicity CaO/SiO2 ratio 0.933 and two different PbO/(CaO+SiO2) ratios (2.0 and 3.2). They used a small quantity of synthetic oxide mixture, pelletized and equilibrated at temperature below the liquidus so that two or more phases were formed. Then the samples were cooled rapidly by quenching. The result was that the phases presented at high temperature and their compositions were retained to room temperature. Their results showed that the liquidus in the pseudoternary ZnO-“Fe2O3”- (PbO+CaO+SiO2) system contained primary phase fields of spinel (ZnxFe3-xO4+y), zincite (ZnxFe1-xO), hematite (Fe2O3), melilite (PbvCa2-vZnwFe1-wSi2O7), magneto- plumbite (PbFe12O19) and calcium and lead silicates.
ETTLER et al [3,4] studied the lead and zinc slags produced 100-150 years ago and found that the major constituents were clinopyroxene, melilite, olivine and spinel. ZHAO et al [5] carried out experimental studies in the ZnO-“FeO”-Al2O3-CaO-SiO2-MgO system in equilibrium with metallic iron. Their results showed that wustite (Fe,Zn)O and spinel (Fe,Zn)O・(Al,Fe)2O3 were the major phases. JAK and HAYES [6] carried out experimental studies on the PbO-ZnO-“Fe2O3”-CaO- SiO2 system in air to characterize the phase relations and reported the liquidus pseudoternary section for slags with mass ratio of CaO/SiO2 being 0.35 and PbO/(CaO+SiO2) being 5.0. JAK and HAYES [7] studied the same system with the ratios of CaO/SiO2 being 0.1 and 0.6, and PbO/(CaO+SiO2) being 4.3 and 6.2.
The mineralogical species formed in the lead blast furnace depend on the compositions of the original ores and the plant practice adopted. Thus, there exist significant variations in the microstructures in sintered materials and the solid phases formed from the slag. Systems with high concentrations of PbO-CaO-SiO2 produce a glassy slag phase with a low melting point. The presence of ZnO promotes the formation of melilite phase with platelike crystals which support the sintering and prevent from collapse.
The present study is conducted to provide information on the liquidus temperature and the mineralogical species formed in the slags of the PbO-ZnO-CaO-SiO2-“Fe2O3” system with different CaO/SiO2 and Fe/SiO2 mass ratios at temperature range from 1273 to 1573 K. A thermodynamic analysis is carried out with the software FACTSage developed by THOMPSON et al [8], to estimate the liquidus temperature of the slags as well as the primary solid phases formed in this system.
2 Experimental
The experimental design was based on the sinter compositions used in Mexican industrial blast furnaces, which includes the PbO-ZnO-CaO-SiO2-“Fe2O3” slag system with CaO/SiO2 mass ratio from 1.0 to 1.6, Fe/SiO2 mass ratio from 1.3 to 1.7 and 40% (mass fraction) PbO and 8% ZnO. Pure oxide powders (99.5% purity) were used as starting materials to prepare the synthetic slags.
The major difficulty in the experimental procedure was the high vapor pressure of lead species. In order to reduce this problem, master slags with the required amounts of PbO and SiO2 were prepared. The master slags were then mixed with appropriate addition of the other pure oxide powders (ZnO, CaO and Fe2O3) to prepare the final mixtures compositions given in Table 1.
20 g of each slag sample was homogenized and equilibrated in platinum crucible in air in two steps. In the first step the samples were melted at 1573 K for 4 h. In the second step the samples were cooled down from 1573 K to a given temperature (1473, 1373 and 1273 K) and kept at high temperature for 4 h in order to reach the equilibrium; then, the samples were quenched into ice water. The furnace temperature variation was controlled within ±3 K with an R-type thermocouple (Pt-Pt, 13%Rh). The estimated maximum lead oxide and zinc oxide losses during equilibration, using the X-ray Fluorescence technique, were 2.5% and 1.1%, respectively.
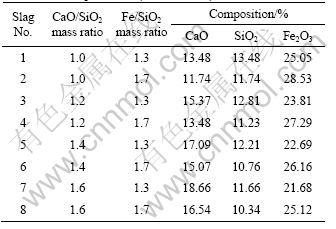
Table 1 Mixture compositions containing 40% PbO and 8% ZnO used for experiments (mass fraction)
Samples of each slag were crushed into fine powders and characterized by X-ray diffraction (XRD Bruker D8 Focus) to determine the mineralogical species. The microstructural analysis was carried out by mounting and polishing the samples, then by examination using scanning electron microscopy coupled with an energy-dispersive spectra analyzer, FEI Quanta 600, EDAX EDS, with software GENESIS-MLA (Mineral liberation analysis) to determine the compositions of each phase. The mineral reference standards for EDS analysis were gold, silver, platinum, copper, quartz, galena, sphalerite, chalcopyrite, pyrite and wollastonite. The advantage of this experimental technique is that each experiment provides information on the compositions of the liquid at high temperature and the solid phases formed. The experimental procedure is shown in Fig. 1.
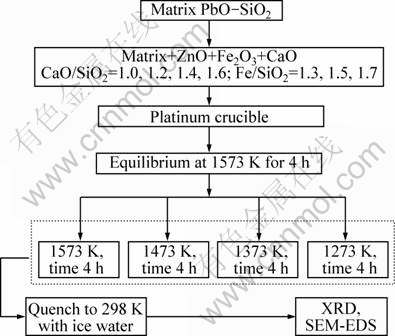
Fig. 1 Experimental procedure
The experimental conditions for the 8 slags were thermodynamically simulated using the software FACTSage (THOMPSON et al [8]) to estimate the liquidus temperature and the phases in equilibrium at high temperature.
3 Results and discussion
3.1 SEM-EDS microanalysis
The results of 32 quenching experiments are given in Table 2. Each phase composition given in this table is the average of up to three compositions measured at various locations within that phase. The results of the EDS microanalysis were compared with the EDS database of minerals, reported by REED [9], and the mineral phases were identified. The main solid phases obtained in the slags can be classified in to three types of compounds: 1) Spinel: ZnxFe3-xO4+y; 2) Calcium and lead silicates: Ca2SiO4, Pb5SiO7, Ca2PbSi3O9; 3) Magneto- plumbite: PbFe12O19.
No measurements of the ferric/ferrous ratio were performed in this study, thus iron is represented as ferric oxide for the purposes of describing the composition data.
Table 2 shows that the liquidus temperature is between 1473 K and 1573 K in all the slags and the spinel is the first phase to form during crystallization.
Table 2 Experimental result on samples with CaO/SiO2 mass ratio of 1.0-1.6 and Fe/SiO2 of 1.3-1.7


During cooling further calcium/lead silicates start to precipitate. Measurements of the spinel composition indicate that in air and in the range of temperatures investigated, this phase is close to franklinite composition (ZnFe2O4: 33.76% ZnO in mass fraction and 66.23% Fe2O3). The concentration results show that the liquid composition becomes rich in PbO as temperature is diminished from 1573 to 1273 K. There are also crystals observed in a combination of franklinite, calcium/lead silicates and magnetoplumbite. It is possible that in such crystals calcium/lead silicates and magnetoplumbite are formed as a result of an incongruent reaction between liquid and franklinite.
3.2 Microstructure
The microstructures of slags quenched at different temperatures for slag number 2 (CaO/SiO2=1.0 and Fe/SiO2=1.7) are shown in Fig. 2. It is observed that at 1573 K the slag is completely melted and the rapid cooling to room temperature produces a dendritic structure. The microanalysis carried out in the arms of dendrites showed zinc and iron segregation which produced precipitation of spinel phase at low temperature.

Fig. 2 Morphologies of slag samples with CaO/SiO2 mass ratio of 1.0 and Fe/SiO2 mass ratio of 1.7 quenched from different temperatures: (a) 1573 K; (b) 1473 K; (c) 1373 K; (d) 1273 K
The micrographs of the samples equilibrated at 1373 K and 1473 K shows dark angular crystals of the spinel phase. The present experiments indicate that the composition of spinel in equilibrium with the liquid phase approaches the franklinite composition (ZnFe2O4) in air in the range of temperatures investigated. The sample equilibrated at 1273 K shows spinel crystal together with elongated crystals which, according to their microanalysis, correspond to calcium and lead silicates. Figure 2 also shows white regions that correspond to liquid slag in equilibrium with the solid phases at high temperature. The microstructures shown in Fig. 2 illustrate the crystallization sequences that can take in the real lead slags.
Figure 3 shows the X-ray mapping images of Pb, Ca, Si, Fe and Zn elements for the sample number 2 quenched from 1273 K. The X-ray mapping image shows a contrast due to element concentration gradient. For example, in the X-ray image for calcium, the bright areas correspond to calcium-rich zones and the dark areas correspond to calcium-poor zones. This figure shows that the irregular polygons mainly contain iron and zinc; thus, it is believed that these correspond to the spinel-type phase (ZnFe2O4). The X-ray mapping shows that the elongated crystals contain silicon and calcium and correspond to calcium silicates. It is also observed that the main amount of lead is present in the white zone in this sample which corresponds to the liquid phase at 1273 K.
Figure 4 shows the microstructures of sample 3 (mass ratio of CaO/SiO2=1.2 and Fe/SiO2=1.3). The dendritic structure in Fig. 4(a) shows again that at 1573 K the slag was completely liquid. The sample equilibrated at 1473 K shows that spinel is the first solid to form. There are crystals with combined phases, franklinite, silicates and magnetoplumbite, as shown in Fig. 4(d) and reported in Table 2.
Figure 5 shows the evidence that Ca/Pb silicates and magnetoplumbite are partially formed as a result of an incongruent reaction between liquid and franklinite. An incongruent reaction can be carried out when a pre-existing solid is consumed or resorbed in the process. Figure 5(a) shows franklinite crystals before the incongruent reaction, for a slag with mass ratio of CaO/SiO2=1.0 and Fe/SiO2=1.7, equilibrated at 1373 K. Figure 5(b) shows a franklinite crystal which has been partially transformed in a slag with the same global composition and equilibrated at 1273 K.

Fig. 3 X-ray mapping images of Pb, Ca, Si, Fe and Zn elements for slag with mass ratio of CaO/SiO2 being 1.0 and Fe/SiO2 being 1.7 quenched from 1273 K
Figure 6 shows the SEM-EDS analysis of a franklinite crystal which has been partially transformed for a slag with mass ratio of CaO/SiO2=1.0 and Fe/SiO2=1.7. These EDS results confirm that the former crystals correspond to franklinite ZnFe2O4 (white zones), while the gray zones mainly correspond to a combination of Ca/Pb silicate (obtained from CaO, SiO2 and PbO) and magnetoplumbite (obtained from Fe2O3 and PbO).
JAK et al [2] reported the liquidus surface for the ZnO-“Fe2O3”-(PbO+CaO+SiO2) system projected onto a pseudo-ternary diagram with the mass ratios of CaO/SiO2 being 0.933 and PbO/(CaO+SiO2) being 2.0. This pseudo-ternary diagram shows that there exists an incongruent reaction according to the following expression:
Liquid + Franklinite = Magnetoplumbite + Ca/Pb silicate
The slags shown in Figs. 5 and 6 have the CaO/SiO2 mass ratio of 1.0 and PbO/(CaO+SiO2) of 1.7, which are close to those values of the slags reported by JAK et al [2]. Then, it is possible that Ca/Pb silicates and magnetoplumbite are produced in the former franklinite crystals by the above mentioned incongruent reaction.
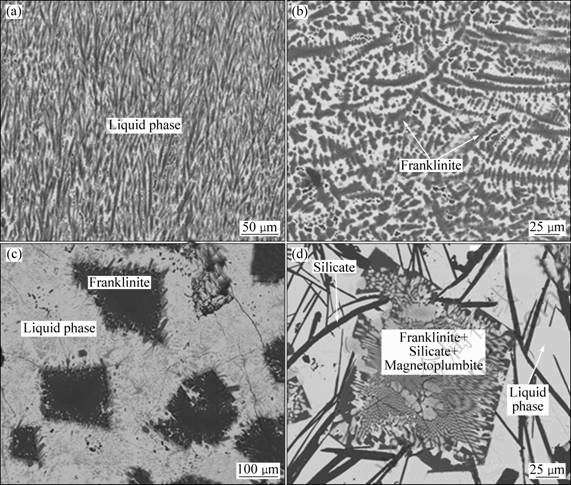
Fig. 4 Morphologies of slags with mass ratio of CaO/SiO2 being 1.2 and Fe/SiO2 being 1.3 quenched from different temperatures: (a) 1573 K; (b) 1473 K; (c) 1373 K; (d) 1273 K

Fig. 5 Franklinite crystals before incongruent reaction at 1373 K (a) and after reaction at 1273 K (b) for slag with mass ratio of CaO/SiO2 being 1.0 and Fe/SiO2 being 1.7
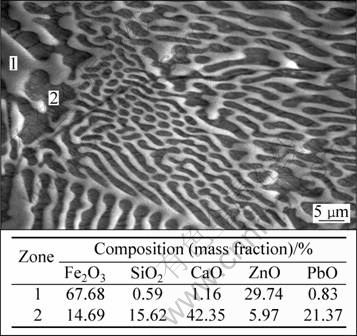
Fig. 6 SEM-EDS analysis of franklinite crystal partially transformed into magnetoplumbite and Ca/Pb silicate
3.3 X-ray diffraction results
The X-ray diffraction (XRD) analysis was used to confirm phase identification. Figure 7 shows the XRD patterns for slags 1 and 3 with mass ratio of CaO/SiO2 being 1.0 and Fe/SiO2 being 1.3 and 1.7, respectively, equilibrated at 1273 K and quenched to room temperature. Franklinite ZnFe2O4 (JCPD file 22-1012), margarosanite Ca2PbSi3O9 (JCPD file 20-0219) and hardystonite Ca2ZnSi2O7 (JCPD file 35-0745) are the main species present in both slags; however, the peaks of franklinite increase in intensity and those of hardystonite diminishes as the Fe/SiO2 is increased.
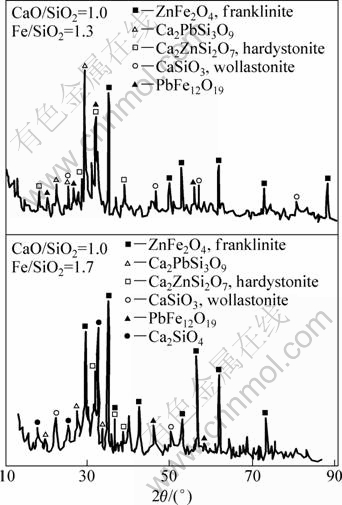
Fig. 7 XRD patterns for slags with mass ratio of CaO/SiO2 being 1.0 and Fe/SiO2 being 1.3 (a) and 1.7 (b), equilibrated at 1273 K and quenched to room temperature
The X-ray diffraction analysis also shows the presence of wollastonite (CaSiO3) (JCPD file 27―1064) and lead iron oxide whose composition is close to the stoichiometry of the magnetoplumbite compound PbFe12O19 (JCPD file 310686) which was reported by JAK et al [2]. It is worth noting that the presence of crystals of hardystonite (Ca2ZnSi2O7) and lead iron oxide (PbFe12O19) were not clearly observed in the SEM-EDS microanalysis.
3.4 Thermodynamic calculation
The thermodynamic modeling was carried out using the FACTSage computer system (THOMPSON et al [8]) which used a modified quasichemical model for the molten slag phase. A polynomial model was used for solid solutions with one sublattice. A compound energy model was used for more complex solid solutions with several sublattices. This software was used to estimate the phase distribution and liquidus temperatures for the slags in terms of slag basicity from 1.0 to 1.6 and Fe/SiO2 ratios from 1.3 to 1.7, and temperatures from 1273 K to 1673 K (1000 °C to 1400 °C).
The phases considered in this calculation were the liquid slag phase, the spinel solid solution (ZnxFe3-xO4+y), melilite, which is a family of species with general formula (PbvCa2-vZnwFe1-wSi2O7), magnetoplumbite (PbFe12O19) and a solid solution of silicates (M2SiO4, where M = Ca, Pb, Fe and Zn).
In the calculations, we considered the initial slag composition given in Table 1. Figure 8 shows that these slags are in the spinel primary phase field and the liquidus temperatures, which were deduced from FACTSage model predictions, are between 1523 and 1613 K (1250 and 1340 °C). This information is of particular importance to lead blast furnace. It must be stressed that the experimental results showed that the liquidus temperatures were below 1573 K (1300 °C) for all the slags. The calculated liquidus temperature decreases as the slag basicity is increased at a given Fe/SiO2 ratio, whilst the liquidus increases when the Fe/SiO2 ratio is increased. The minimum liquidus temperature was predicted at about 1523 K (1250 °C) for the slag with mass ratio of CaO/SiO2 being 1.6 and Fe/SiO2 being 1.3.
Melilite was included in the calculations as a solid solution; however, the FACTSage results showed that the composition of this phase was close to hardystonite compound (Ca2ZnSi2O7). This phase was obtained at low mass values of CaO/SiO2 and low temperatures. Magnetoplumbite was also formed at low slag basicities.
4 Conclusions
1) The SEM results showed dendritic structures for all the slags equilibrated at 1573 K, which indicates that the slag is completely liquid. However, XRD analysis showed the presence of solid phases as franklinite. It is likely that this phase is formed within the “arms” of the dendrites during cooling.
2) The liquidus temperature is between 1473 K and 1573 K. Franklinite is the first phase to form on crystallization in all the slags. During cooling further, franklinite precipitation takes place and when the liquid composition reaches a given composition, Ca/Pb silicates start to precipitate.
3) Calcium and lead silicates and magnetoplumbite are partially formed as a result of an incongruent reaction between liquid and franklinite between 1273 K and 1373 K.
4) The calculated liquidus temperature for the franklinite primary field increases as the Fe/SiO2 mass ratio is increased at a given CaO/SiO2 mass ratio.
5) The highest value of the calculated liquidus temperature for the franklinite primary field is obtained for the slags with the lowest basicity (CaO/SiO2=1.0) at a given Fe/SiO2 mass ratio.
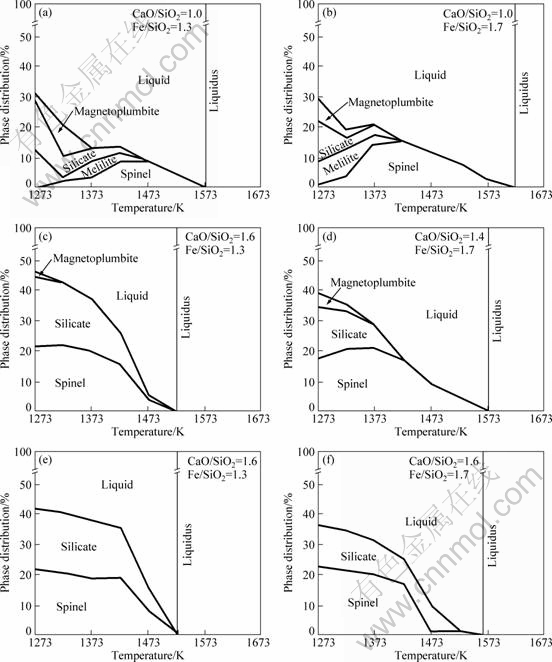
Fig. 8 Calculated phase distribution and liquidus temperature for slags in terms of CaO/SiO2 and Fe/SiO2 mass ratios
6) Hardystonite (Ca2ZnSi2O7) and magnetoplumbite (PbFe12O19) are calculated to form at a low CaO/SiO2 mass ratio and low temperature.
Acknowledgements
The authors thank Servicios Administrativos Pe?oles S.A. de C.V. for providing financial support for this project. The authors wish also to thank the Institutions CONACyT, COFAA and IPN for their permanent assistance to the Process Metallurgy Group at ESIQIE-Department of Metallurgical Eng.
References
[1] HABASHI F. Handbook of extractive metallurgy. Primary metals, secondary metals, light metals II [M]. New York: Wiley-VCH, 1997: 581-641.
[2] JAK E, ZHAO B, HARVEY I, HAYES P C. Experimental study of phase equilibria in the PbO-ZnO-“Fe2O3”-(CaO+SiO2) system in air for the lead and zinc blast furnace sinters (CaO/SiO2 weight ratio of 0.933 and PbO/(CaO+SiO2) ratios of 2.0 and 3.2) [J]. Metallurgical and Materials Transactions B, 2003, 34: 383-397.
[3] ETTLER V, JOHAN Z, TOURAY J C, JELINEK E. Zinc partitioning between glass and silicate phases in historical and modern lead-zinc metallurgical slags from the Pribram district, Czech Republic [J]. Comptes Rendus de l’Académie des Sciences, Paris: Série II, 2000, 331: 245-250.
[4] ETTLER V, LEGENDRE O, BODENAN F, TOURAY J C. Primary phases and natural weathering of old lead-zinc pyrometallurgical slag from Príbram, Czech Republic [J]. The Canadian Mineralogist, 2001, 39(3): 873-888.
[5] ZHAO B, HAYES P C, JAK E. Phase equilibria studies in zinc-containing systems and applications to lead and zinc blast furnace slags [C]//Lead-Zinc 2010 Conference. Vancouver, Canada, 2010: 603-614.
[6] JAK E, HAYES P C. Experimental study of phase equilibria in the PbO-ZnO-“Fe2O3”-(CaO+SiO2) system in air for high lead smelting slags (CaO/SiO2 = 0.35 and PbO/(CaO+SiO2)=5.0 by weight) [J]. Metallurgical and Materials Transactions B, 2002, 33: 817-825.
[7] JAK E, HAYES P C. The effect of the CaO/SiO2 ratio on the phase equilibria in the ZnO-“Fe2O3”-(PbO+CaO+SiO2) system in air: CaO/SiO2 = 0.1, PbO/(CaO+SiO2) = 6.2, and CaO/SiO2 = 0.6, PbO/(CaO+SiO2) = 4.3 [J]. Metallurgical and Materials Transactions B, 2003, 34: 369-382.
[8] THOMPSON W T, BALE C W, PELTON A D. Facility for the analysis of chemical thermodynamics (FACTSage) [EB/OL]. Montreal: Ecole Polytechnique, 2010. http://www.crct.polymtl.ca.
[9] REED S J B. Electron microprobe analysis and scanning electron microscopy in geology [M]. London: Cambridge, 2005: 56-72.
M. PEREZ-LABRA1, A. ROMERO-SERRANO1, A. HERNANDEZ-RAMIREZ1,
I. ALMAGUER-GUZMAN2, R. BENAVIDES-PEREZ2
1. Metallurgy and Materials Department, IPN-ESIQIE, UPALM, Lindavista, C.P. 07738, Mexico D.F.;
2. Servicios Administrativos Pe?oles S.A de C.V. Prol. Comonfort Sur 2050,
Col. L. Echeverría, Torreón, C.P. 27300 Coahuila, Mexico
摘 要:实验测定了CaO/SiO2和Fe/SiO2质量比分别为1~1.6和1.3~1.7、含40% PbO和8% ZnO的PbO-ZnO-CaO- SiO2-“Fe2O3”渣系的相平衡和液相线温度。将该渣系在1273~1573 K达到平衡,然后快速淬火冷却。XRD和SEM-EDS分析结果表明,该渣的成分位于锌铁尖晶石相的初始成相区,在1273~1473 K生成硅酸钙/铅,部分硅酸钙/铅和磁铁铅矿相是通过转熔反应生成的。实验结果和热力学计算结果表明,液相线温度随着渣中CaO/SiO2比的增加和Fe/SiO2比的降低而增加。
关键词:铅渣;相平衡;液相线温度
(Edited by YUAN Sai-qian)
Corresponding author: A. ROMERO-SERRANO; Tel/Fax: +55-57296000-55270; E-mail: romeroipn@hotmail.com
DOI: 10.1016/S1003-6326(11)61230-2

