���±�ţ�1004-0609(2008)06-1135-08
ZnCl2-EMIC����Һ���е����п
�����£��� ��������⣬�� ��
(����������ѧ ��Դ�뻷������ѧԺ���Ϻ� 200237)
ժ Ҫ������ZnCl2-EMIC����Һ�壬�õ绯ѧ��������ϵ�����缫���ٵ缫�ϵķ�Ӧ�������̽����о�������п�������ƣ�����ֱ���������Դ���е����ʵ�飬�о������ܶȡ��¶Ⱥ�������������ضԶƲ��Ӱ�졣���������������ӦΪ���淴Ӧ��п���������缫���ٵ缫���κ�����ά˲ʱ�κ˰�������ɢ������̣�ֱ�������ܶ�Ϊ4mA/cm2���¶�Ϊ80 ��ʱ���Ʋ�������ã����������õ��ĶƲ�Ҫ����ֱ��������õ��ĶƲ㣬�������������Ϊ2ms��������Ϊ8ms�������ܶ�Ϊ8mA/cm2ʱ���õ��ĶƲ�⻬�����ȣ�������С�ӽ�0.3��m ��
�ؼ��ʣ�Zn��ZnCl2������Һ�壻�����
��ͼ����ţ�TQ 153 ���ױ�ʶ�룺A
Electrodeposition of zinc from zinc chloride-1-ethyl-3-methylimidazolium chloride molten salt
MA Jun-de, LI Bing, YAN Ling-guang, CHEN Yan
(School of Resource and Environmental Engineering, East China University of Science and Technology,
Shanghai 200237, China)
Abstract: The electrodeposition of zinc from zinc chloride-1-ethyl-3-methylimidazolium chloride molten salt was adopt to study the process of the cathode reaction. The electrodeposition was carried out with direct current and impulse current. The factors influencing the coating were studied. The result indicate that: the electrodeposition of zinc on nickel and tungsten substrates involves instantaneous three-dimensional nucleation. The coating is the best when the current density is 4mA/cm2 and the temperature is 80 ��. The coating by the method of the impulse electrodeposition is better than the direct current electrodeposition. Especially when f is 100Hz, ton/toff is 2/8 and J is 8mA/cm2, the coating is clean and uniformity. The crystal size is nearly 0.3�� m .
Key words: Zn; ZnCl2; ionic liquids; electrodeposition;
����ڻ�ø����������Ʋ��ͬʱ���������Һ�Ի�����Σ���ѳ�Ϊ��ɫ�绯ѧ�ͻ�����ҵؽ����������⣬������Һ��ij���ʹ֮��Ϊ���ܣ�����Һ���������ѹ����Ϊ�㣬���ӷ�������Ⱦ�����Ե��ص�[1-2]�������¼��ɵõ��ڸ��������в��ܵ�����õ��Ľ����ͺϽ𣬵�û�и�������������ǿ��ʴ�ԣ�����Һ�������ʹ֮��Ϊ������е�ո��Һ��[3-4]��
PITNER ��[5]���û���AlCl3 ����������Һ���о���п�ڽ𡢲����ٺͲ�̼�缫�ϵĵ�������̣������ڽ����缫����Ƿ��λ�����ڲ�̼�缫��ֻ����Ԫ�ɺ˹��̣��õ�����п�����ĺϽ�SUN��[6-8]��EMIC��ZnCl2 ��ϵõ��ܹ��ڿ������ȶ����ڵ�����Һ�壬�����ڵ����п��ѭ���������Ա�������ZnCl2��EMIC��Ħ����Ϊ1?3ʱ����ϵ��Lewis���ԣ��绯ѧ���ڴ�ԼΪ3.0V������Ե�EMIC-A1C13��ͬ����ZnCl2��EMIC��Ħ����Ϊ0.5?1ʱ����ϵ��Lewis���ԣ��绯ѧ����ԼΪ2V�����ʵ����¶Ⱥ͵�ѹ�£�����������Һ�����ܹ��õ�п�Ʋ㡣
Ŀǰ�����������й�������Һ��ı���Ҳ�϶�
[9-12]������û�й�������Һ���ڵ��������Ӧ�õı�������������Һ������ԣ����Կ˷��������κ�ˮ��Һ�ڵ�����������������ȱ���絼�²���������ࡢ����Ч�ʵ͡���ˮ�������⡢�����¶ȸߡ��������
[13]����ˣ��������߶�������Һ��ZnCl
2-EMIC�е����п�������о���������������ƣ�����Ӱ��Ʋ�ĸ������ؽ����о���Ϊ�������͵��Һ�ڵ���������Ӧ���ṩ�������ݡ�
1 ʵ��
1.1 ��Ҫʵ���������Լ�
�Ȼ�-1-��-3-�һ�����(EMIC)(���ȴ���97%���ձ�����������ʽ����)�� �Ȼ�п(������)����ͪ(������)�������Դ(SMD-30���������Ƶ�Դ�������д�˴����豸��)��CHI1140�绯ѧ������(�Ϻ�������������˾)��JMS-2���������(�Ͼ������Կؼ�������˾)���絼�ǣ�ͭƬ(1cm��1cm)(����Ϊ99%)����˿(����Ϊ99.95%)����˿(����Ϊ99%)��пƬ(1cm��1cm)( ����Ϊ99%); п˿(����Ϊ99%)��
1.2 ʵ�����
��������Һ�弫����ˮ���������̶�ˮ�������У�����ˮ�Ȼ�п���к�ǿ����ˮ�ԣ��ڿ����м���ˮ�⡣��ˣ����۷�������ˮ�Ȼ�п�����ZnCl2��Zn(OH)Cl�Ļ������ԣ���ʵ��֮ǰҪ�Է�������ˮ�Ȼ�п������ˮ��
������ZnCl2(������)װ��ֱ���ͳ����ʵ���ʯӢ�������У�ͨ�����ĸߴ�������������150�沢ʹ�����Դ�һ�㣬ʹ���ܹ���ˮ������ȫ������ֹͣ���£���ˮ��ȫ�������£����н��Գ�ˮ��Լ��550��ʱ��������ˮ����������С�����������¶���770~800�棬Լ750��ʱ��ʼ�а�ɫZnCl2����������ʯӢ�ܱ��ϣ�������Ϻ����������䣬�ڵ��������£���ZnCl2��ĩ�γ�����[14]��
����Һ��������ڵ��������µ��������н��У�������ˮP2O5���������������������ZnCl2��EMIC��1?1��Ħ�������ƣ����ȵ�90�汣��2h��
���������У��õ絼�Ƕ�����Һ���ڲ�ͬ�¶��½��е絼���ԡ�
Ϊ���˽�����������Zn�ĵ�������̣���Ҫ���е绯ѧʵ�飬��Ϊ��ͨ�IJαȵ缫�����������в����ã���ˣ��Ʊ�Zn/Zn2���αȵ缫��ѡȡ��Ϊ90mm��ֱ��Ϊ8mm�IJ����ܣ��ڲ����ܵ�һ���ս�һ�鲣��ɰо������2mL���ҵ�ZnCl2-EMIC����Һ��(Ħ����1?1)����5mL��ҽ�ò�����Ͳ����������Һ��ע�벣�����ڣ���Ƕ�иߴ�п˿�����������ڲ������ϣ������ܷ����ܷ�̶���
�ڵ��ʵ���У���������ͭƬ(2 cm��1 cm��0.01 cm)����������пƬ(2 cm��1 cm��0.01 cm)���缫�ڽ���ʵ��֮ǰ��Ҫ���д�3��ɰֽ��5��ɰֽ����ĥ�⣬֮���ñ�ͪȥ��������H3PO4��H2SO4��HNO3�Ļ�ϻ�Һ(70%?25%?5%)�⣬Ȼ����ȥ����ˮϴ�ӣ���ɱ��á���п�ĵ����ʵ���У��ֱ�ֱ�������ܶȡ�����������¶ȶԶƲ��Ӱ�졣�ڵ绯ѧ�����У������缫�ֱ������˿����˿(��Ч���0.251 2 cm2)���Ե缫Ϊп˿(��Ч���0.251 2 cm2)���αȵ缫ΪZn/Zn2��(��Ч���0.251 2 cm2)��
2 ���������
2.1 ����Һ��ĵ絼��
�����������õ絼�ǶԵ��Һ���е絼���ԣ����ȵ��Һ��80 �棬Ȼ�������¶������½������½��Ĺ��̼�¼��ͬ�¶ȵĵ絼�ʡ�Ħ����Ϊ1?1��ZnCl2-EMIC�ڲ�ͬ�¶��µĵ絼�����1���С��ɱ���֪���絼�����¶����߶�����
��1 ZnCl2-EMIC����Һ���ڲ�ͬ�¶��µĵ絼��
Table 1 Electrical conductivity of ZnCl2-EMIC ionic liquids at various temperatures

2.2 п�ĵ��������
2.2.1 ѭ��������
ʵ���в������缫��ϵ��1?1 ZnCl2-EMIC����Һ����е绯ѧ���ԡ�ͼ1��ʾΪ80��ʱ����˿(��Ч���0.251 2cm2)����˿(��Ч���0.251 2cm2)Ϊ�����缫��п˿Ϊ�Ե缫(��Ч���0.251 2cm2)���αȵ缫ΪZn/Zn2��(��Ч���0.251 2cm2)��ѭ���������ߡ������缫�ϴӿ�ʼ����ɨ�赽-0.5VΪֹ������������ɨ�赽2V����ͼ��֪��������ɨ������2����ԭ��(a��b)����Ӧ����3��������(c��d��e)����������֪�����ڲαȵ缫ΪZn/Zn2��������п��ԭ��λӦ����0V����������ԭ��(b)����������(c)Ϊ����п�ij������ܽ⣬����0.06Vʱ����п�Ļ�ԭ��1V�����������Ե�2��С������(d��e), ����Kolb-Gerischer��Ƿ��λ��ϵʽ[15-16]��

��ͼ1��֪��1V��������������(d)�����Ƕ�ӦǷ��λ������п���ܽ⣬����(e)���������缫�����������йأ���0.4V������������Ϊ����п����������0.06Vʱ����п�Ļ�ԭ������Ƿ��λ�������岻�Ǻ����ԡ�
��ʵ�����ò��缫���������Һ�е�ˮ�֣��ڲ��缫��ֻ��һ�Ի�ԭ���������(��ͼ1(b))����п�Ļ�ԭ���������ڲ��缫��п�κ˳�����Ҫһ���Ĺ���λ����-0.025V��п��ʼ��ԭ��ͼ�в�û�г��������ӵĻ�ԭ�壬�ɴ˿���֪�������Һ��ˮ�ּ��͡�
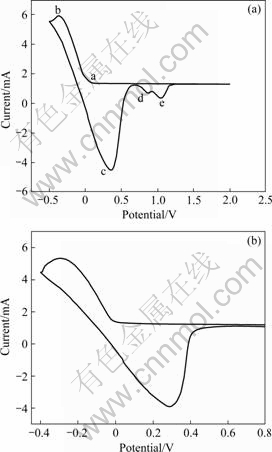
ͼ1 80 ��ʱĦ����Ϊ1?1��ZnCl2�CEMIC����Һ���ѭ����������
Fig.1 Cyclic voltage current characteristic curves of ZnCl2-EMIC at 80�� and 100mV/s: (a) Nickel electrodes; (b) Platinum electrodes
ͼ2(a) ��ʾΪ�����缫Ϊ��˿(��Ч���0.251 2cm2)��Ħ����Ϊ1?1��ZnCl2-EMIC����������80�����Բ�ͬɨ�����ʵ�ѭ���������ߡ���ͼ��֪������ɨ�����ʵ����ӣ������ ����
���� �����ƶ���
�����ƶ��� �����ƶ�������ȷ����ӦΪ����ġ�ͼ2(b)��ʾΪ�����������ɨ������ƽ�����Ĺ�ϵ���ߡ���ͼ��֪�������
�����ƶ�������ȷ����ӦΪ����ġ�ͼ2(b)��ʾΪ�����������ɨ������ƽ�����Ĺ�ϵ���ߡ���ͼ��֪������� ��ɨ������ƽ����v1/2�������ι�ϵ���ڸ���ɨ�������ƫ��ֱ�ߣ��ɴ˿���ȷ����ӦΪ���淴Ӧ��
��ɨ������ƽ����v1/2�������ι�ϵ���ڸ���ɨ�������ƫ��ֱ�ߣ��ɴ˿���ȷ����ӦΪ���淴Ӧ��

ͼ2 80 �治ͬɨ������ʱĦ����Ϊ1?1 ZnCl2-EMIC���ε�ѭ���������������������ɨ������ƽ�����Ĺ�ϵ
Fig.2 Cyclic voltage current curves of ZnCl2�CEMIC at 80�� and different scanning rates (a) and dependence of Ipc on v1/2(b)
2.2.2 ��ʱ������
��λ��Ծ�����о��ɺ˺������ij��÷������������缫��ϵ������Һ����е�����ʱ����̬������ͼ3��ʾΪ�ֱ�����˿(��Ч���0.251 2 cm2)����˿(��Ч���0.251 2 cm2)Ϊ�����缫��п˿(��Ч���0.251 2 cm2)Ϊ�Ե缫��Zn/Zn2��(��Ч���0.251 2cm2)Ϊ�αȵ缫��80��ʱ��ͬ��λ��Ħ����Ϊ1?1��ZnCl2-EMIC�������εļ�ʱ�������ߡ�

ͼ3 80��ʱ�����ڲ�ͬ�缫�ϵ�I��t����
Fig.3 I��t curves of melt on different electrodes at 80��: (a) Tungsten electrodes; (b) Nickel electrodes
��ͼ3���Կ�����ʵ�鿪ʼʱ�����ܴ�������Ϊ�缫˫���ij����̼����˵����������¡�˫��������Ծ��������Ȼ�������������������Ϊп����ԭ���ɺ˲�����ᾧ�������ھ��˵��γɺ���������ɣ�����Ѹ���������ڵ����ﵽ���ֵ����ֵ���˥������ʱ��п��ᾧ���̾������������ĵĽ���������������ͬʱ�����������ĵ���ʧ�����������ĵ��γɣ������������½�����������ɢ���ƣ�����˫�������������½�������һ����������Ϊ�ٵ缫��п�ij�����Ҫ���ߵ��κ˹���λ����˫������������Ƹ����ԡ�����п�������缫�Ϸ���һ����Ƿ��λ�������κ˵Ĺ���λ��С������ͬ������λ�¸��Ʋ����ԡ���������Ϊ����״�����������ٶ�����Һ�е�������ӵ���ɢ����ʱ����ͬ�ɺ˻�������̬������ʽΪ[17]

ͨ����ʽ(2)��(3)�����ֱ任���ɷֱ�õ�tm��Im��I2/ �ı���ʽ��������ڱ�2��
�ı���ʽ��������ڱ�2��
��2 ��ʽ(2)��(3)�����Ĺ�ϵ
Table 2 Expressions derived from Eqns. (2) and (3)

�ɱ�2��֪�����ڲ�ͬ�ijɺ˷�ʽ�������������I2/ ��t/tm����״��ͬ����ͼ3�����ݽ��д�����ʱ��͵����ֱ��ȥ˫�������̵�t0��I0�ó���t��=t-t0��
��t/tm����״��ͬ����ͼ3�����ݽ��д�����ʱ��͵����ֱ��ȥ˫�������̵�t0��I0�ó���t��=t-t0�� =tm-t0��I��=I-I0��
=tm-t0��I��=I-I0�� =Im-I0��Ȼ��ͼ3��ʽ(2)��(3)ת��
=Im-I0��Ȼ��ͼ3��ʽ(2)��(3)ת�� ��
�� ����ι�ϵ�������ͼ4��ʾ��
����ι�ϵ�������ͼ4��ʾ��
�Ƚ�˲ʱ�ɺ˺������ɺ˵���������ߣ����֣������ɺ˵���������߳���һ���ϼ���ķ壬��˲ʱ�ɺ���һ���Ͽ��ķ塣��ͼ4���Կ�������ʵ��ĵ�λ��Χ�ڣ���õ����ݵ������˲ʱ�ɺ��������Ǻϣ����缫��˫���ij����̽��ٵ缫ʱ��Ҫ���������κ���ʼ�����Խϴ�Щ����������˲ʱ�ɺ��������Ǻϣ���ˣ���Ϊп�������ٵ缫�ϵ�ᾧ�ǰ���ά˲ʱ�κ˰����ε���ɢ������̡�

ͼ4 ��Ӧͼ3�����������
Fig.4 Non-dimensional plots of (I/Im)2 vs t��tm data corresponding to potential steps in Fig.3: (a) Tungsten electrodes; (b) Nickel electrodes
2.3 п�ĵ����
2.3.1 п��ֱ�������
����ͭƬ(1cm��1cm)���������ϣ�пƬ(1cm��1cm)Ϊ�������ϣ���ZnCl2-EMIC��ϵ�н��е����ʵ�飬�о���ͬ�¶��¡���ͬ�����ܶȶԶƲ��Ӱ�졣�ڵ�ƵĹ����У��¶��ڽϵ�ʱ����50��ʱ���Ʋ�������ڣ�����ѻ���һ���ɫ�����ʣ���XRD�����ó�����ɫ�ѻ�����ȻΪп��ͼ5��ʾΪ����ɫ�ɷֵĶƲ��XRD�ס�

ͼ5 ����ɫ�ɷֶƲ��XRD��
Fig.5 XRD pattern of coating containing gray composition
ͼ6��ʾΪ��ͬ�¶��µ��15 min�ĶƲ�����SEM����ͼ��֪����ͬһ�����ܶ��£�50��ʱ�ó��ĶƲ㾧��Ҫ��80��ʱ�ó��ĶƲ㾧��ϸС���Ҳ����ȣ��Ʋ�ܱ�����Ϊ��ͬһ�����ܶ��£�50��ʱ����Һ��絼��С����Ҫ����Ĺ���λ����ô�κ��ٶȽ�80��ʱ�����Ծ�����С�����ҵ����ܶ���������¶Ʋ㷢�ڡ�

ͼ6 ��ͬ�¶����Ʊ��Ʋ��SEM��
Fig.6 SEM images of coating prepared at different tempera- tures (Molar ratio of EMIC to ZnCl2 of 1?1, J=1.39 mA/cm2): (a) 50��; (b) 80��
��ͬһ�¶��£��ڵ����ܶ�Ϊ1mA/cm2ʱ��������С����С1.5��m���ң����������ܣ���ͼ7(a)��ʾ����ͼ���Կ������Ʋ��������ɫ���⻬�����ʵ���������ܶȵ�1.39mA/cm2ʱ����������2.5��m���ң���ͼ7(b) ��ʾ����ͼ��֪���������ܶȼ�������ʱ�����ŵ����ܶȼ���������Խ��Խϸ�£�������4mA/cm2ʱ��������СΪ1��m���ң���ͼ7(c)��ʾ����ͼ��֪���Ʋ�������ɫ���⻬�������ܶ���0~1.39mA/cm2ʱ�����ŵ����ܶ����������ľ�����֣��������ڵ����ܶȵͣ�����λ��С�����¾������ܶȼ�������ʱ������λ���ӣ������γɵ��ٶ��������ӣ�������ᾧϸ�¡������������ܶȷ�Χ�ڣ�������ᾧ����ϸ���������ܶȳ�����������5mA/cm2ʱ��������������ȱ�ٷŵ�������ӣ�һ������Ǻ�����λ�ŵ磬���ֽ�����֦״�ᾧ����ͼ7(d)��ʾ����ͼ��֪���Ʋ����ʻ�ɫ����50��ʱ�Ʊ��ĶƲ�һ������ɫ������ȻΪп��

ͼ 7 80�治ͬ�����ܶ��¶Ʋ��SEM��
Fig.7 SEM images of coating at 80 �� and different current densities: (a) J = 1 mA/cm2; (b) J = 1.39mA/cm2; (c) J = 4mA/cm2; (d) J = 6mA/cm2
2.3.2 ���������
���������Ľ�����3���С��õ��������Դ������Һ����е�������ɿصĵ����ܶȸ��ӵĿ����������ܶ�����10mA/cm2ʱ�����ѵõ��õĶƲ㣬����ѻ��˻�ɫ�����ʡ��Ʋ�����SEM����ͼ8��ʾ����ͼ��֪���������ܶ���8mA/cm2���������9ms��������1msʱ�õ��ĶƲ����ʰ�ɫ���⻬������ϸС��һ����Ե�о�֦��

ͼ8 80��ʱ�¶Ʋ�����SEM��
Fig.8 SEM image of coating at 80�� (f=100Hz, ton/toff =9/1, J=10mA/cm2 )
ʹ���������ܹ���ý�ֱ������ϸС�Ŀ�����������Ϊ�������ڵ�����ͨʱ���ӽ������Ľ��������ڸߵ����ܶ��³�����ϸС�Ŀ������������ض�ʱ����ɢ����ʧ��������Χ�ķŵ����������»ָ�����ʼŨ��[18]����������������ɢ���ڵĽ�������Ũ�Ⱦ͵õ��˼�ʱ���䣬��ɢ�����ڼ�϶ʽ�γɣ���������ɢ���ʵ�ʺ�ȣ��Ӷ������˶Ʋ�����ܡ�
��80���µ����ܶ�Ϊ8mA/cm2����ͬƵ�ʺ�ͨ�ϱ�ʱ�õ��ĶƲ�SEM����ͼ9��ʾ����ͼ���Կ�������f = 100Hz��ton/toff = 2/8�����¶Ʋ�������ã��õ��ĶƲ����⻬�Ҿ��ȣ�������С�ӽ�0.3��m ��f = 50 Hz��ton/toff = 2/8��f = 100Hz��ton/toff = 5/5������Ҳ�ܻ�ñȽϺõĶƲ㡣���������ֽ�����֦״�ᾧ����������Ի�ɫ����ˣ�������Ϊ�û�ɫ����ҲΪп��
��3 80 ����������������
Table 3 Experiment results of Zn electrodeposition by PCP at 80 ��


ͼ9 80��ʱ��ͬƵ�ʺ�ͨ�ϱ��µ��15min�Ʋ��SEM��
Fig.9 SEM images of coating at different impulse frequencies and different ratios of ton to toff at 80�� (J =8mA/cm2): (a) f = 100Hz, ton/toff =2/8; (b) f =50Hz, ton/toff = 2/8; (c) f =10Hz, ton/toff = 2/8; (d) f =500Hz, ton/toff = 2/8; (e) f =100Hz , ton/toff = 5/5; (f) f = 100Hz, ton/toff = 9/1
3 ����
1) ZnCl2-EMIC����Һ���е����п�ķ�ӦΪ���淴Ӧ��п�������ٵ缫���κ�����ά˲ʱ�κ˰�������ɢ������̡���ͬһ�����ܶ��£�50 ��ʱ�ó��ĶƲ㾧��Ҫ��80 ��ʱ�ó��ĶƲ㾧��ϸС���������ȡ�
2) ��80��ʱ���������ܶ���0~1.39mA/cm2�仯ʱ�����ŵ����ܶ���������ľ�����֣������ܶȼ�������ʱ������λ���ӣ������γɵ��ٶ��������ӣ�������ᾧϸ�¡��������ܶȳ�����������5.0 mA/cm2ʱ���õ��ĶƲ����ʻ�ɫ���������ֽ�����֦״�ᾧ��
3) ����������ֱ��������Ʋ�����������ã�����������ʱ�������ܶȴﵽ8mA/cm2���������2ms��������8ms, �����ܶ�Ϊ8mA/cm2ʱ���õ��ĶƲ����⻬�Ҿ��ȣ�������С�ӽ�0.3��m��
REFERENCES
[1] ��˫ƽ, ������, ���ؽ�, ������, �� ��. ����Һ������ʺ��Ʊ�����[J]. ��ϸ�����м���, 2004, 34(5): 10-12.
CHEN Shuang-ping, WANG Shou-wu, WANG Shou-jian, DAI Wei-dong, FENG Li. The character and preparation method of ionic liquids[J]. Fine Chemical Intermediates, 2004, 34(5): 10-12.
[2] ������, ������, �»�Ƽ, �� ��, ������. ����Һ������ʼ����ڴ���Ӧ�е�Ӧ��[J]. ���̹���ѧ��, 2003, 3(2): 177-185.
WANG Jun-feng, ZHANG Shuo-jiang, CHEN Hui-ping, LI Xian, ZHANG Mi-lin. The character of the ionic liquids and the appliance on catalysis reaction[J]. Chinese Journal of Process Engineering, 2003, 3(2): 177-185.
[3] ��Ӣ��, ���, ����ɽ, ������. ����Һ��ķ��ࡢ�ϳ���Ӧ��[J]. ��ѧ����, 2005(2): 7-12.
ZHANG Ying-feng, LI Chang-jiang, BAO Fu-shan, ZHANG Yong-an. The classify synthesize and application of the ionic liquids[J]. Chemistry Education, 2005(2): 7-12.
[4] ������, �� ��, �촺��, �����. ����Һ���ڵ���������Ͱ뵼������е�Ӧ��[J]. ��ѧͨ��, 2004, 67: 470-475.
LIU Yan-sheng, YAN Jun, XU Chun-ming, HU Yu-feng. The application of the ionic liquids electrodeposition of metal and semiconductor material[J]. Chemistry, 2004, 67: 470-475.
[5] PITNER W R, HUEESY C L. Electrodeposition of zinc from ionic liquids[J]. J Electrochem Soc, 1997, 144: 3095-3104.
[6] HSIU S I, HUANG J F, SUN I W. Lewis acidity dependency of the electrochemical window of zinc chloride-1-ethy1-3- methylimidazolium chloride ionic liquids[J]. Electrochimica Acta, 2002, 47: 4367-4372.
[7] HUANG J F, SUN I W. Electmdeposition of Pt-Zn in a Lewis acidic ZnC12-1-ethyl-3-methylmidazolium chloride ionic liquid[J]. Electrochimica Acta, 2004, 49: 3251-3258.
[8] CHEN P Y, SUN I W. Electrodeposition of cobalt and Zinc-cobalt alloys from a lewis acidic zinc chloride-1-ethyl-3- methylimidazolium chloride molten salt[J]. Electrochimica Acta, 2001, 46: 1169-1177.
[9] �� ��, �չ���, �� Ԫ. ���ܻ�������Һ���ڵ绯ѧ�е�Ӧ��[J]. ��ѧͨ��, 2004(11): 795-801.
LIU Hui, TAO Guo-hong, KOU Yuan. The application of the ionic liquids on electrochemistry[J]. Chemistry, 2004(11): 795-801.
[10] �ž���, ������, ������. ����Һ�弰������ȡ�е�Ӧ���о���չ[J]. ������չ, 2001, 20(12): 16-19.
ZHANG Jing-tao, PU Xiang-lan, ZHU Shen-lin. The research of application on extraction of ionic liquids[J]. Chemical Industry and Engineering Progress, 2001, 20(12): 16-19.
[11] ������, ¦ ˧, ����ԥ, ���ı�. ����Һ��Ӧ���о���չ[J]. ��ϸ����, 2005, 22(5): 324-327.
ZHANG Pu-yu, LOU Shuai, JIN Lin-yu, LI Wen-bin. The research of the application of the ionic liquids[J]. Fine Chemical Intermediates, 2005, 22(5): 324-327.
[12] �Դ��, �캣��, �� ��, �� ��, �����. ��������Һ���еľۺϷ�Ӧ[J]. ��ѧ��չ, 2005, 17(4): 700-705.
ZHAO Da-cheng, XU Hai-tao, GAO Ge, XU Peng, LIU Feng-qi. Polymerization reaction of the ionic liquids[J]. Chemical Industry and Engineering Progress, 2005, 17(4): 700-705.
[13] ��ï��. ���п��п�Ͻ�չ��״[J]. �����Ϳ��, 2003, 22(6): 35-40.
AN Mao-zong. The status of electroplate zinc and alloy[J]. Electroplating and Coating, 2003, 22(6): 35-40.
[14] �ֻ���, �� Ƽ, �� ��, ������. �ߴ�����ˮ�Ȼ�п��һ��ʵ�����Ʊ�����[J]. ʯ��ׯѧԺѧ��, 2005(5): 22-25.
JIA Hui-zhen, ZHANG Ping, LI Yuan, HAN Ming-hui. A sort of laboratory preparation of high pure zinc chloride[J]. Journal of Shijiazhuang University, 2005(5): 22-25.
[15] GERISCHER H, KOLB D M, PRZASNYSKI M. Tunneling processes at highly doped zinc oxide electrodes in contact with aqueous electrolytes (��): Electron exchange with the conduction band[J]. Surf Sci, 1974, 45: 662-666.
[16] LI L F, TOTIR G G, TOTIR D O, CHOTTINER G S. Underpotential deposition of lithium on aluminum in ultrahigh- vacuum environments[J]. Phys Chem, 1999, 103: 164-168.
[17] SCHARIFLKER B, HILLS G. Theoretical and experimental studies of multiple nucleation[J]. Electrochemical Acta, 1983, 28(7): 45-48.
[18] �� ��, �����, κ�Ӷ�. �����Ƶ��о���״[J]. ����뾫��, 2005, 27(5): 25-29.
LIU Yong, LUO Yi-hui, WEI Zi-dong. The study status of impulse electroplate[J]. Electroplating and Coating, 2005, 27(5): 25-29.
������Ŀ��������Ȼ��ѧ����������Ŀ(50474027)
�ո����ڣ�2007-09-30�������ڣ�2008-01-16
ͨѶ���ߣ��� �������ڣ���ʿ���绰��021-64252170��E-mail: drlibing@163.com
(�༭ ������)