Pyrolysis process of ferromanganese crust
CUI Ying-chun(īř”≠īļ), SHI Xue-fa( Į—ß∑®), LIU Ji-hua(ŃűľĺĽ®),
FANG Xi-sheng(∑ĹŌį…ķ), GAO Jing-jing(łŖĺßĺß), ZHU Ai-mei(÷žįģ√ņ)
(Key Laboratory of Marine Sedimentology and Environmental Geology of State Oceanic Administration,
Qingdao 266061, China)
Abstract: The pyrolysis characteristics and varieties of evolved gas of ferromanganese crusts were investigated using thermogravimetry coupled on-line with Fourier transform infrared spectroscopy. The pyrolysis of ferromanganese crust is a one-step process under nitrogen atmosphere. The major gaseous products identified are carbon dioxide and water. A new mineral phase (¶Ń-Fe2O3) occurs in the solid residue.
Keywords: ferromanganese crust; TG-FTIR; pyrolysis
CLC number: P744.3 Document code: A Article ID: 1672-7207(2011)S2-0089-03
1 Introduction
Ferromanganese crusts (hereafter called crusts) mostly occur on the surfaces of seamounts where sediments are free from strong currents. These crusts consist mainly of vernadite and X-ray amorphous Fe oxyhydroxide (FeOOH.xH2O)[1]. They have a high special surface area (average 300 m2/g) and slow growth rates[2-3], so that many trace metals, such as Co, Pt, REEs, and so on, are enriched. Hence, crusts are regarded not only as potential commercial seabed ore, but also as important adsorbents.
Certain physical properties of the crusts have to be measured because these are important factors in the exploration and exploitation of crust resources[1]. However, the thermal properties of crusts, which are also important for further utilization of the crusts, are always ignored. A technique that features thermogravimetry (TG) coupled on-line with Fourier transform infrared (FTIR) spectroscopy (TG-FTIR) has recently been widely used in the thermal analysis of matters[4]. This technique can provide not only mass change with temperature or time, but also the gas that evolves on-line. The aim of this work is to understand the thermal characteristics of the crusts and identify the evolved gas using the TG-FTIR technique.
2 Experimental
2.1 Sample preparation
The M42 crust on a basaltic substrate was collected from the Magellan seamounts at a water depth between 3 171 m and 3 284 m by chain-bag dredging during a cruise of the R.V. Haiyangsihao in 2003. The crust is approximately 1.7-2.2 cm thick, with a smooth and small botryoidal surface and a dark brown massive layer. The sample was first washed with double distilled water to remove contaminants, such as sediments and other debris, and then dried at room temperature and ground in an agate mortar. The grain size of the ground sample ranged from 0.25-125 ¶Őm, measured using a Mastersizer 2 000 (Malvern Instruments Ltd., UK).
2.2 Methods
2.2.1 Determination of chemical composition
Major and trace element concentrations of M42 were determined using an inductively coupled plasma optical emission spectrometer. The reference standard GSPN-1 was used to estimate the accuracy of the analyses.
2.2.2 XRD analysis
The X-ray diffraction (XRD) patterns obtained on a Rigaku X-ray diffractometer using Cu-K¶Ń radiation at a scan rate of 0.02 (°„)/s were used to identify the mineralogy of the samples. The accelerating voltage and applied current were 40 kV and 150 mA, respectively.
2.2.3 TG-FTIR analysis
Thermogravimetry was performed using a TG-DTA instrument (Netzsch TG 209 F1) under a highly pure N2 flow of 20 mL/min with a heating rate of 10 °„C/min at 40-850 °„C. The evolved products were analyzed using an on-line coupled Nicolet 6700 spectrometer (resolution 4 cm-1, gain 4 000-400 cm-1, 4 scans). The temperature of the transfer tube connecting TG with FTIR was kept at 200 °„C to prevent the evolved gas from condensing, as well as avoid secondary decomposition.
3 Results and discussion
3.1 Characteristics of M42
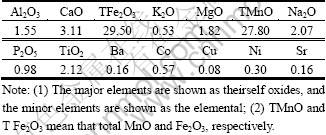
The bulk chemical composition of the M42 crust is shown in Table 1. The major components of the sample are Mn and Fe but the Ca, Na, and Ti contents are also high. All element contents determined are in good agreement with the published data[1]. The typical XRD patterns of the bulk mineralogy of M42 crust are shown in Fig. 1. Vernadite, which has two broad peaks at 24.5 and 14.2 nm, dominates in the M42 sample, with the X-ray amorphous FeOOH°§xH2O deduced from chemical composition as the second dominant mineral[5]. Other minerals detected include plagioclase and quartz, which have peaks at 3.34°Ń10-10 m and 3.21°Ń10-10 m, respectively. Carbonate fluorapatite was not identified, indicating that the M42 crust is not phosphatized. The infrared spectra of M42 crust is presented in Fig.2. The FTIR spectroscopy results show obvious absorbance peaks at 995.75, 1 634.08, and 3 224 cm-1, similar to those of nodules[6-8]. The first peak can be attributed to Si°™O or Si°™O°™Al, whereas the last two are assumed to be caused by the OH group stretching and bending vibrations. These results, together with the XRD findings, indicate that iron-manganese oxide, silicate minerals, and water are the major components of the M42 crust.
Table 1 Chemical composition (mass fraction, %) of cobalt-rich crust M42
3.2 Thermogravimetric analysis
The thermal stability of the M42 crust and thermal effects related to M42 crust pyrolysis were investigated. The TG and DTG curves of the M42 crust are presented in Fig.3. The desorption behavior of M42 shows a continuous weight loss as temperature rises, and no equilibrium occurs. Based on the TG-DTG curve and calculation, it is found that the M42 crust exhibits the following features: First, no initial separating temperature of volatiles is observed, indicating that the volatiles escape when temperature rises; Second, the crust has a maximum rate of mass loss of -1.093 65%/min at 94.707 °„C, and the mass loss at 850 °„C is about 20%. These figures are quite similar to those of nodules[6-8].
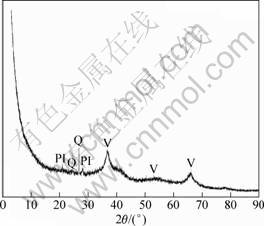
Fig.1 X-ray diffraction pattern of crust M42
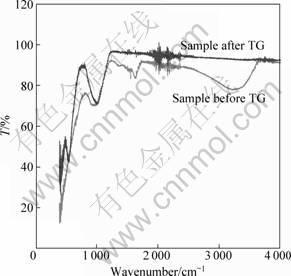
Fig.2 FTIR spectra of crust M42 and the TG residua of M42 (The blue line is the FTIR spectra of the solid residue of
the crust; the red line represents the FTIR spectra of the original M42)
3.3 Analyses of evolved gas by on-line coupled FTIR spectroscopy
Numerous simple organic and inorganic gaseous compounds, such as CO2, CH4, and H2O, formed and/or emitted during thermal treatment, were successfully detected. The on-line evolved gaseous products of the crust recorded by FTIR spectroscopy are presented in Fig.4. Compared with the data in Refs. [9-11], the main gaseous products of the crust are H2O and CO2. The water has typical absorption peaks at 3 900-3 500 cm-1 and 1 900-1 300 cm-1, whereas CO2 has absorption peaks at 2 360 cm-1 and 3 740 cm-1. The broad bands at 1 800-1 650 cm-1 may have been induced by the C=O stretching vibrations, and the broad bands at 3 470-3 100 cm-1 may be the mark of NH3.
Water may occur in hydroscopic, crystal, and constitutional forms. The hydroscopic water in the crust varies between 5.9% and 24.1%[12]. When the temperature reaches 110 °„C, the hydroscopic water escapes, indicating that the water discharged below 110 °„C is hydroscopic water. The water that escaped at a temperature higher than 110 °„C is crystal water. The major minerals of the crust contain high amounts of crystal water. For example, vernadite contains 15%-25% crystal water in weight[13]. Crystal water may remain in the mineral at temperatures between 100 and 200 °„C, or sometimes up to 600 °„C, thus, the water escaping at a higher temperature (200-850 °„C) is inferred as constitutional type. For instance, X-ray amorphous FeOOH°§xH2O, another major component in the crust, may contain the (OH)- ion. Under higher temperatures, the crystal lattice of this mineral is destroyed, thus, these ions escape in the form of water.
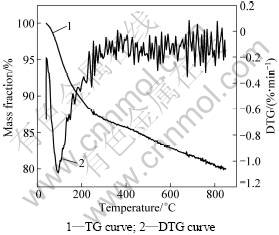
Fig.3 TG-DTG curves of crust M42 at a heating rate of 10 K/min
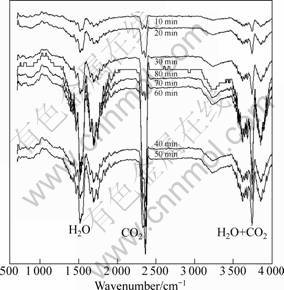
Fig.4 Evolved gases of ferromanganese crust with time
The destruction of the crystal lattice can be seen from the FTIR spectra of the heated sample. A new peak at around 540-560 cm-1 (Fig.2) appears, indicating the formation of ¶Ń-Fe2O3[6-8].
The CO2 escaping at low temperature can be hydroscopic, but can also originate from the decomposition of the materials containing CO32-, such as calcite and other accessory minerals undetected by XRD in this crust sample.
4 Conclusions
The TG-FTIR technique is a useful tool for analyzing the thermal decomposition of crust. The TG curves of the crust show a one-step decomposition. The FTIR spectra proves that the major gaseous products evolved are CO2 and H2O, and ¶Ń-Fe2O3 forms in the solid residue.
Acknowledgment: We thank Professor HE Qi-xiang and other anonymous reviewers for improving the language of the manuscript.
References
[1] Hein J R, Cobalt-rich ferromanganese crusts: global distribution, origin and research activities[C]//ISA. Minerals Other than Polymetallic Nodules of the International Seabed Area, Workshop Report. Kingston, Jamaica, 2004: 188-256.
[2] Segl M, Mangini A, Bonani G., et al. 10Be-dating of a manganese crust from central North Pacific and implications for ocean paleocirculation[J]. Nature, 1984, 309: 540-543.
[3] Klemm V, Levasseur S, Frank M, et al. Osmium isotope stratigraphy of a marine ferromanganese crust[J]. Earth Planet Sci Lett, 2005, 238(1/2): 42-48.
[4] Akinade K A, Campbell R M, Compton D A C. The use of a simultaneous TGA/DSC/FT-IR system as a problem-solving tool[J]. J Mater Res, 1994, 29(14): 3802-3812.
[5] Hein J R, Schwab W C, Davis A. Cobalt-and platinum-rich ferromanganese crusts and associated substrate rocks from the Marshall Islands[J]. Mar Geol, 1988, 78(3/4): 255-283.
[6] Parida K M, Satapathy P K, Sahoo A K, et al. Studies on indian ocean manganese nodules I. Physicochemical characteristics and catalytic activity of ferromanganese nodules[J]. J Colloid Interface Sci, 1995, 173(1): 112-118.
[7] Parida K M, Satapathy P K, Das N N. Studies on Indian Ocean Manganese Nodules PART 2. Physico-chemical Characteristics and Catalytic Activity of Heat-Treated Marine Manganese Nodules[J]. J Colloid Interface Sci, 1996, 179(1): 241-248.
[8] Parida K, Samal A, Das D, et al. Effect of calcination temperature on Indian Ocean manganese nodules[J]. M?ssbauer, XRD, FT-IR and TG-DTA studies Thermochim Acta, 1999, 325(1): 69-76.
[9] Sikorska-Iwan M, Mrozek-?yszczek R. Application of coupled TG-FTIR system in studies of thermal stability of manganese(II) complexes with amino acids[J]. J Therm Anal Calorim, 2004, 78(2): 487-500.
[10] Madar®Ęsz J, Pokol G. Comparative evolved gas analyses on thermal degradation of thiourea by coupled TG-FTIR and TG/DTA-MS instruments[J]. J. Therm Anal Calorim, 2007, 88(2): 329-336.
[11] Gomez C, Velo E, Barontini F, et al. Influence of secondary reactions on the heat of pyrolysis of biomass[J]. Ind Eng Chem Res, 2009, 48(23): 10222-10233.
[12] Frank M, O°ĮNion R K, Hein J R, et al. 60 Ma records of major elements and Pb-Nd isotopes from hydrogenous ferromanganese crusts: Reconstruction of seawater paleochemistry[J]. Geochim. Cosmochim Acta, 1999, 63(11/12): 1689-1708.
[13] Post J E. Manganese oxide minerals: Crystal structures and economic and environmental significance[J]. PNAS, 1999, 96(7): 3447-3454.
(Edited by HE Yun-bin)
Received date: 2011-06-15; Accepted date: 2011-07-15
Foundation item: Project (40704029) supported by the National Science Foundation of China; Project (MGE2008KG01) supported by the Open Fund of the Key Laboratory of Marine Geology and Environment, China Academy of Sciencess; Project (DY115-01-2-1, DY115-01-1-03) supported by the China Ocean Mineral Resources R & D Association; Project (2006DFB21620) supported by the International Scientific and Technological Cooperation
Corresponding author: CUI Ying-chun, Associate professor; Tel: +86-532-88961651; E-mail: cuiyingchun@fio.org.cn