
Thermal solidification of stainless steelmaking dust
TANG Mo-tang(唐谟堂)1, PENG Ji(彭 及)1, PENG Bing(彭 兵)1, YU Di(余 笛)2, TANG Chao-bo(唐朝波)1
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Information Science and Engineering, Central South University, Changsha 410083, China
Received 4 January 2006; accepted 12 June 2007
Abstract: Stainless steelmaking dust makes an environmental problem in the disposal or landfills and has been assigned as a hazardous waste by various government regulatory agencies because it leaches heavy metals to the groundwater or rainwater in the concentrations exceeding the environmental guidelines for solid waste disposal. Solidification of the dust is to stabilize the hazardous components into amorphous silica-alumina-based clays. Various mixtures of stainless steelmaking dust and clay were investigated and the softening temperatures of these mixtures were measured. The results indicate that the mixture of stainless steelmaking dust and clay additive with 1?1 ratio has the lowest softening temperature of 1 100 ℃. The clinkers can pass the TCLP leaching test after being thermally treated at the softening temperature for 15 min. A thermal process for the solidification of stainless steelmaking dust with typical clay is developed and the product is desirable for the production of bricks or disposal and landfill.
Key words: solidification; stainless steelmaking dust; environmental protection
1 Introduction
Stainless steelmaking dust is a by-product of steelmaking operation. Approximately 1%-2% (mass fraction) of scrap charged in smelting furnaces enters into the exhaust gases and then is converted into the dust[1]. The disposal or landfill of this waste becomes a serious environmental problem in recent years due to the fact that the production of stainless steels increases in China. The dust contains elements such as Cr, Ni, Pb, Zn, Cd and their solubility in leaching media exceeds the environmental regulatory limits[2].
Several stainless steelmaking dust treatment technologies have been developed and they can be divided into two main groups. One group is to recover the metals present in the dust[3-5] and the other is to stabilize hazardous components of the dust[6-7]. Some high temperature metal recovery processes have presently been applied to treat most part of stainless steelmaking dust. But they are only suitable for the dust with high contents of nickel, chromium and other valuable metals[8]. As for the dust with low metal content or a small amount of dust, solidification is an appropriate choice before disposal or landfills.
Solidification technology has been widely used for the treatment of inorganic wastes and contaminated soils before final disposal. CONNER and HOEFFNER[9] summarized the solidification technologies and indicated that fly ash was one of the most common binders in waste stabilization. HASSETT and PFLUGHOEFT- HASSETT[10] made use of coal combustion by-products for the solidification of hazardous wastes. Fluidized bed combustion coal ash was used as a binder for the solidification and stabilization of metal-bearing sludges from a hazardous waste treatment facility[11]. RODRIGUEZ-PI?ERO et al[12] described the chromium behavior in the solidification of a steel industry waste using a common fly ash from a pulverized coal power station as the binder and indicated that the stabilization of Cr(Ⅵ) present in the waste required a reducing pretreatment with ferrous sulfate to attain toxicity characteristics leaching procedure(TCLP) leachates within the limits. PELINO et al[13] developed a vitrification process to immobilize the hazardous elements in electric arc furnace dust by mixing the dust with cullet and sand, and pointed out that the stability of the product was influenced by the glass structure which depended on the Si/O ratio. The thermal behavior of the electric arc furnace dust from stainless steel and carbon steel operations was determined and a thermal process for the stabilization of the dust by vitrifying with clay was developed[14].
The objectives of this study are to find a low cost additive for the solidification of stainless steelmaking dust, to optimize the compositions of the dust-additive mixture and process conditions and to ensure that the clinkers of solidification passes TCLP test. In order to achieve these, the solidification temperature is a key point. It was found that stainless steelmaking dust softened above 1 600 ℃. This temperature is too high to process the dust clinkers economically. Therefore, it is necessary to reduce the melting temperature of the dust by adding certain modifying ingredients. Silica and alumina are additives that readily modify the melting temperatures of ceramic ware. They are also effective glass forming substances which help to immobilize the heavy metals present in the stainless steelmaking dust. The mixture of the dust and additive is investigated and the softening temperature is measured in this study. Consequently, the thermal characteristics and leachability of the clinkers are investigated.
2 Experimental
2.1 Raw materials
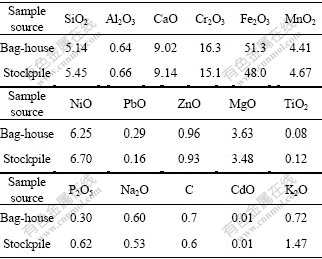
Two different kinds of stainless steelmaking dust samples used in this study were obtained from stainless steel industries. One was a newly collected dust acquired from the bag-house, which was a dry fine powder. The other was taken from a stockpile located in the open air, which contained a large amount of water. The stockpile dust was too wet to be processed in the experiments. Therefore it was dried for 24 h at 100 ℃ and broken down by passing a rod-roller. Since the dusts were formed in air at high temperature, all the metal elements were oxidized. Elemental analysis of two dust samples was performed using X-ray fluorescence(XRF) and inductively coupled plasma(ICP). The compositions of the samples are given in Table 1.
Table 1 Compositions of stainless steelmaking dust used (mass fraction, %)
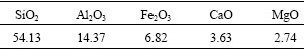
A low cost local clay was used as the additive for the solidification of stainless steelmaking dust. The main chemical compositions of the clay are given in Table 2. The clay was spread on the floor and dried for a few days, and then was split using a sample splitter before processing.
Table 2 Main compositions of additive (mass fraction, %)
2.2 Experimental procedure
The stainless steelmaking dust and the additive were mixed in different dust-to-additive ratio and then molded into the brick and dried at room temperature for 3 d. A muffle furnace was used to heat the bricks in the rate of 4 ℃/min to a given temperature and then kept for 15 min. A K-type thermocouple was used to detect the tempera-temperature of the bricks and to control the process. The softening temperatures of mixtures were measured by checking the volume changes of the clinkers in the thermal processing. The ratio of stainless steelmaking dust and additive clay in the mixture was determined according to the lowest softening temperature[15-16] in the consideration of economical solidification. The leachability and thermal characteristics of the clinkers were analyzed after thermal treatment.
Thermogravimetry analysis(TGA) was used to monitor the mass change of the mixtures as a function of temperature and time in thermal processes of the solidification and the thermal behavior of the clinker. Differential thermal analysis(DTA) was used to detect the temperature changes caused by chemical or physical reactions as a function of temperature and time in the solidified samples and clinkers. Fourier transform infrared spectroscopy(FTIR) was used to identify gaseous species evolution during the thermal processes of the mixtures and clinkers.
The clinkers produced through thermal solidification were subjected to leaching tests as specified in the US environmental protection agency (EPA) regulations for solid waste. The tests included breaking down the clinkers and selecting some small particles to make samples, with size less than 10 mm, followed by leaching at room temperature for 24 h (longer than 18 h, so stricter than TCLP) in a buffered solution of acetic acid and acetate (pH 5.0). Inductively coupled plasma(ICP) was used for the elemental analysis of the leachate solution to compare the results with EPA regulations.
3 Results and discussion
3.1 Determination of mixture composition and softening temperature
It was found in our previous investigations[2, 17] that the stainless steelmaking dust sintered without melting at high temperature over 1 300 ℃ and softened above 1 600 ℃. These temperatures were too high to process the dust clinkers economically. Therefore, the melting temperatures of the dust had to be lowered down by addition of additives. The temperature of solidification depended on the softening temperature of the dust and additive mixture.
The densities of clinkers changed in the thermal process. They got small at low temperature in the beginning of thermal process, but would get large after being heated over softening temperatures. In other words, the volume of thermally treated products increased under softening temperature. When being treated above the softening temperature, the volume of the clinkers would decrease quickly. In this consideration the softening temperatures could be determined by measuring the sharp volume changes after the clinkers cooled down to determine the composition of the mixtures. The results of volume changes with the temperature are given in Figs.1 and 2.

Fig.1 Volume changes of bag-house dust-clay mixture in thermal process
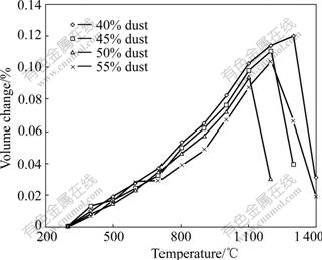
Fig.2 Volume changes of stockpile dust-clay mixture in thermal process
It can be seen from Figs.1 and 2 that the mixtures with two kinds of dusts and clay have the same thermal characteristics. The softening temperatures of the mixtures with 40%, 45%, 50% and 55% dust are 1 280, 1 200, 1 100 and 1 180 ℃, respectively.
The main concern for thermal solidification of stainless steelmaking dust is the energy consume. The temperature of thermal solidification is dependent on the softening temperature because only by thermally treating over this temperature can the hazardous materials present in the dust be immobilized. The mixture of 50% dust and 50% clay showed the lowest softening temperature for the thermal solidification of two kinds of dusts. Therefore, the composition of the mixture could be determined in this way and 1 100 ℃ was confirmed as the thermal solidification temperature.
3.2 Analysis of thermal process
Dry mixtures were prepared by weighing and combining equal amounts of stainless steelmaking dust and clay. The thermal process of solidification was analyzed with TGA, DTA and FTIR. Fig.3 gives the diagram of TGA/DTA/FTIR of the mixture in the thermal process. From Fig.3, TGA/DTA up to 1 200 ℃ highlighted that the mass loss of the mixture was 6.5% (mass fraction). 2% mass loss in the temperature range of 580-800 ℃ was caused by the decomposition of CaCO3. Above this temperature range, the mass losses were also observed due to the volatilization of zinc and lead, accompanied by an endothermic effect in the DTA trace. FTIR analysis showed that the water evaporated at about 120 ℃. Small amounts of HCl, SO2 and HF were detected at high temperature over 950 ℃, and SiF was also detected, which resulted from the reaction of HF and SiO2 at high temperature.

Fig.3 TGA/DTA/FTIR curves of thermal process
3.3 Characterization of clinkers
The major crystalline phases of clinkers identified by XRD were (Fe, Cr)3O4, (Fe, Cr)2O3, Fe2O3 with minor amounts of anorthite and quartz (Fig.4). Most (Fe,Cr)3O4 present in the dust was oxidized into (Fe, Cr)2O3 and Fe2O3. The clinker appeared visually porous and had a sinter-like texture. SEM examinations for bag-house dust mixture (Fig.5) and stockpile dust mixture (Fig.6) clinkers indicated that extensive liquefaction and evolu- tion of gaseous phases occurred during thermal treatment. The formation of large vesicles in the clinker resulted from gases evolution from the liquefied regions of sample. Although the quartz particles retained in original angular morphology, the liquefaction and vitrification occurred and calcite totally disappeared. The heavy metals in the dust were packed in this way and distributed in the (Fe, Cr)3O4 and (Fe, Cr)2O3 phases, while some Mn, Zn, Al, Mg and Ti distributed in silicate phase.
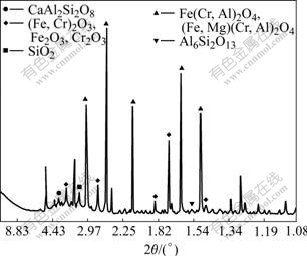
Fig.4 XRD pattern of clinker

Fig.5 SEM image of bag-house dust clinker

Fig.6 SEM image of stockpile dust clinker
Clinkers were also subjected to thermal analysis. Fig.7 shows the TGA/DTA/FTIR results for the clinker. It can be seen that the clinker was very stable thermally. The mass loss of the clinker was less than 0.5% up to 1 200 ℃. There were no significant reactions appeared on the DTA curves and only traces of moisture and carbon dioxide were detected by FTIR.

Fig.7 TGA/DTA/FTIR curves of clinker
3.4 Leaching test of clinkers
In order to assess the stabilization of the hazardous elements present in the stainless steelmaking dust after thermal solidification, two kinds of clinkers with bag-house and stockpile dusts were subjected to US EPA TCLP test. Table 3 lists the leaching results of the tests for the clinkers, which were thermally solidified at 1 100℃ for 15 min. It is observed that the heavy metals released to the solution are in the concentration limits of the regulatory for the solid waste disposal. But it should be noted that the amount of Hg and Se in the solutions is below the detection limit of the instrumentation (0.002 4 mg/L).
Table 3 TCLP results of clinkers (μg・L-1)

References
[1] ZUNKEL D. What to do with your EAF dust [J]. Steel Times International, 1996, 20(4): 46-50.
[2] PENG Bing, PENG Ji, KOZINSKI J A, LOBEL J, CHAI Li-yuan, ZHANG Chuan-fu, CHENG Wei-liang. Thermodynamic calculation on the smelting slag of direct recycling of electric arc furnace stainless steelmaking dust [J]. Journal of Central South University of Technology: Science and Technology, 2003, 10(1): 20-26. (in Chinese)
[3] GELDENHUIS J M A. Recovery of valuables from flue dust fines [J]. Minerals Engineering, 2002, 15: 95-98.
[4] PENG B, LOBEL J, KOZINSKI J A, BOURASSA M. Non- isothermal reduction kinetics of EAF dust-based pellets [J]. CIM Bulletin, 2001, 94: 64-70.
[5] PENG Ji, PENG Bing, YU Di, TANG Mo-tang, SONG Hai-chen, LOBEL J, KOZINSKI J A. Kinetics of isothermal reduction of stainless steelmaking dust pellets [J]. Trans Nonferrous Met Soc China, 2004, 14(3): 593-598.
[6] PEREIRA C F, RODRIGUEZ-PINERO M, VALE J. Solidification/ stabilization of electric arc furnace dust using coal fly ash [J]. Journal of Hazardous Materials, 2001, B82: 183-195.
[7] IONESCU D, MEADOWCROFT T R, BARR P V. Glassification of EAF dust: The limits for Fe2O3 and ZnO content and an assessment of leach performance [J]. Canadian Metallurgical Quarterly, 1997, 36(4): 269-281.
[8] PENG Bing, PENG Ji, ZHANG Chuan-fu, LOBEL J, KOZINSKI J A. Thermodynamics calculation on the oxidation and sulfur removal abilities of slag in EAF dust pellet reduction process [J]. Journal of Central South University of Technology: Science and Technology, 2001, 8(1): 64-68. (in Chinese)
[9] CONNER J R, HOEFFNER S L. Critical review of stabilization/ solidification technology [J]. Environmental Science and Technology, 1998, 28(4): 397-462.
[10] HASSETT D J, PFLUGHOEFT-HASSETT D F. Use of coal combustion by-products for solidification/stabilization of hazardous wastes [C]// Proceedings of the Annual Meeting of the North Dakota Academy of Science, Washington DC, 1997: 24-25.
[11] KNOLL K L, BEHR-ANDRES C. Fluidized-bed-combustion ash for the solidification and stabilization of a metal-hydroxide sludge [J]. Journal of Air Waste Management, 1998, 48(1): 35-43.
[12] RODRIGUEZ-PI?ERO M, FERNANDEZ PEREIRA C, RUIZ DE ELVIRA FRANCOY C, VALE PARAPAR J F. Stabilization of a chromium-containing solid waste: Immobilization of hexavalent chromium [J]. Journal of Air Waste Management, 1998, 48(11): 1093-1099.
[13] PELINO M, KARAMANOV A, PISCIELLA P, CRISUCCI S, ZONETTI D. Vitrification of electric arc furnace dusts [J]. Waste Management, 2002, 22: 945-949.
[14] MIKHAIL S A, TURCOTTE A M, AOTA J. Thermoanalytical study of EAF dust and its vitrification product [J]. Thermochimica Acta, 1996, 287: 71-79.
[15] SIEGMANN A, NARKIS M, ROSENZWEIG N. Softening temperature of glassy polymers as affected by residual stresses [J]. Polymer Engineering and Science, 1979, 19(3): 223-225.
[16] PENG Bing, CHAI Li-yuan, ZHANG Chuan-fu, PENG Ji, MIN Xiao-bo, WANG Yun-yan, HUANG Rui, HE De-wen. Study on the zinc reduction of stainless steelmaking dust [C]// KONGALI F, REDDY R G. International Symposium on Advanced Processing of Metals and Materials, Iron and Steel and Recycling and Waste Treatment. San Antonio: TMS, 2006: 497-505.
Foundation item: Project(50274073) supported by the National Natural Science Foundation of China
Corresponding author: PENG Ji; Tel: +86-731-8821505; E-mail: jipeng2003@126.com
(Edited by YANG Bing)