TiO2 nanoparticles prepared by hydrochloric acid leaching of mechanically activated and carbothermic reduced ilmenite
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2012���5��
�������ߣ����� ����Ԫ ����Ƽ ������ ��Ӣ
����ҳ�룺1232 - 1238
�ؼ��ʣ�TiO2����������ĥ��̼�Ȼ�ԭ��������������
Key words��TiO2; ilmenite; ball milling; carbothermic reduction; leaching; nanoparticles
ժ Ҫ������һ���¹�����������Ȼ�������Ʊ���Ʒλ��������ʯ���������ù�����Ҫ������ĥ��̼�Ȼ�ԭ������������衣��Ȼ������ͨ����ĥ���������ߴ�����ȼ�С��������̼�Ȼ�ԭ�ᵼ�¸��������γɡ���������������У��ص㿼�������ʱ�䡢�¶Ⱥ����Ũ�ȶԽ������̵�Ӱ�졣ͨ���Ż�����������������õ��IJ�ƷΪ���ȳ���98.0%�������ߴ�Ϊ10~200 nm��TiO2��������
Abstract:
A new combination method consisting of ball milling, carbothermic reduction and hydrochloric acid leaching was proposed for the preparation of nanosized synthetic rutile from natural ilmenite. The ball milling was employed to grind ilmenite into small particles. The carbothermic reduction was carried out to yield a high titanium slag, which would be easily purified by subsequent leaching procedure. Factors affecting the hydrochloric acid process, namely the leaching time, temperature, and acid concentration, were studied. After leaching and calcining the milled and annealed mixture of FeTiO3/C under the optimal conditions, the TiO2 nanoparticles with size of 10-200 nm and purity>98.0% were obtained.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 1232-1238
TAO Tao1, CHEN Qi-yuan1, HU Hui-ping1, YIN Zhou-lan1, CHEN Ying2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. ARC Centre of Excellence for Functional Nanomaterials, Institute for Technology Research and Innovation,
Deakin University, Waurn Ponds, Victoria 3217, Australia
Received 2 April 2011; accepted 11 July 2011
Abstract: A new combination method consisting of ball milling, carbothermic reduction and hydrochloric acid leaching was proposed for the preparation of nanosized synthetic rutile from natural ilmenite. The ball milling was employed to grind ilmenite into small particles. The carbothermic reduction was carried out to yield a high titanium slag, which would be easily purified by subsequent leaching procedure. Factors affecting the hydrochloric acid process, namely the leaching time, temperature, and acid concentration, were studied. After leaching and calcining the milled and annealed mixture of FeTiO3/C under the optimal conditions, the TiO2 nanoparticles with size of 10-200 nm and purity>98.0% were obtained.
Key words: TiO2; ilmenite; ball milling; carbothermic reduction; leaching; nanoparticles
1 Introduction
Titanium dioxide has been widely used as a pigment for the manufacture of paints and paper. Ilmenite (FeTiO3), iron titanate naturally occurring, is the major source material of TiO2 pigment. The commercial technologies for the manufacture of pigment grade titanium dioxide from ilmenite are the sulphate route and the chloride route [1-4]. Each of these two routes requires different feedstocks. In the sulfate route, many kinds of low-titanium-content feedstocks are digested in concentrated sulfuric acid, and increased amounts of waste acidic iron (II) solutions are produced in this process. To limit waste generation, the chloride method is preferred, and is regarded as a clean process to yield titanium white. However, the high-titanium- content feedstock including natural rutile mineral (94%-98% TiO2) required in the chloride route is running short. Thus high titanium slag or synthetic rutile has encouragingly been converted from ilmenite to actually represent the major feedstock for the chlorination process.
Several main methods including thermal reduction, chemical leaching and selective chlorination have been developed for the production of high titanium slag or synthetic rutile from ilmenite [5-8]. Some new combined processes, such as reduction chloridization [9], mechanical alkaline treatment [10], and soda ash roasting [11,12], have also been proposed to produce high titanium slag or synthetic rutile from ilmenite, but selective reduction of iron oxides and leaching, such as the Becher process [13], is still one of the most effective technologies to produce synthetic rutile.
Mechanical activation has been demonstrated to significantly improve the efficiency of carbothermic reduction and accelerate dissolution process due to several fundamental reasons: extension of crystal defects and lattice distortions, decrease of particle size and increase of specific surface area [14-16]. In addition, the hydrochloric acid leaching process has some advantages, such as fast leaching, excellent impurity removal and acid regeneration technology [17], which can efficiently remove residual iron and other impurities from ilmenite to form synthetic rutile [18].
Although, the process of mechanically activated and carbothermic reduction for the refinement of ilmenite has been discussed previously [14,19], the leaching process of the mechanically activated and carbothermic reduced ilmenite using hydrochloric acid is seldom investigated. In the present work, a high grade of TiO2 nanoparticles can be prepared by a combination method as follows: firstly milling the mixtures of ilmenite and active carbon, and subsequently annealing in argon flow. After annealing, the reduced samples are leached with hydrochloric acid, and calcined at 600 �� for 2 h in air. The effects of ball milling and annealing on the dissolution of ilmenite in hydrochloric acid solution are investigated, and structure and morphology of the products are characterized.
2 Experimental
2.1 Materials
Ilmenite was provided by Consolidated Rutile Limited located in Australia. All the other reagents used for annealing, leaching and chemical analysis were analytical, and deionized water was used in the corresponding procedures during the experiment.
2.2 Ball-milling experiments
A specially designed ball mill consisted of a horizontal rotating stainless steel cell with four hardened steel balls (diameter of 25.4 mm). The ball milling was used in the mechanical pre-treatment of the mixtures (6 g) of ilmenite and active carbon (mass ratio of 4:1). The milling with a rotation speed of 165 r/min was carried out for 50 h at room temperature in argon atmosphere of 100 kPa [20].
2.3 Carbothermic reducing experiments
Heating 4 g of milled mixtures of ilmenite and active carbon was carried out in the center of a horizontal tube furnace at 1000 �� for 1 h, then mixtures were naturally cooled down to room temperature. The whole experiment was conducted under a flow of Ar gas.
2.4 Leaching and calcining experiments
A 250 mL single-necked glass reactor with a magnetic stirring bar (length of 14 mm) was filled with a preheated HCl (50 mL, 4 mol/L). 2 g of reduced samples were added to the acid solution. The stirring speed was kept at a constant of 800 r/min throughout all the experiments. The leaching experiments were carried out isothermally at 90 �� for 4 h in separate batch experiments. At the end of each leaching experiment, the sampled slurry was filtered and washed, and the residual solid was calcined at 600 �� for 2 h in air to remove residual carbon for products which were characterized.
The obtained products were analyzed for iron and titanium to calculate their leaching efficiencies. Total iron and titanium contents of the samples were also determined using redox titration technique, in which ferric ions were firstly reduced to Fe2+ using stannous chloride. After excess stannous chloride was oxidized by mercury chloride, the ferrous ions were titrated using a K2Cr2O7 standard solution. For determination of the titanium and iron in the prepared synthetic rutile, a weighed sample was melted with sodium peroxide and then leached. The resulting solution was analyzed by the same method mentioned above.
2.5 Characterization
The structure and morphology of the samples were characterized by XRD (Philips 3020, Co target, 40 kV, 30 mA, ��=0.1789 nm) and field emission scanning electron microscopy (FESEM, Supra55vp). The relative element contents of the products were determined with the energy-dispersive X-ray spectrometry (EDS).
3 Results and discussion
3.1 Preparation of TiO2 nanoparticles
A survey SEM image of the material obtained after acid leaching of the annealed sample and subsequent calcining in air is shown in Fig. 1(a), and a more-detailed higher magnification image is presented in Fig. 1(b). The obtained material consists of particles with a typical size of 10-30 ��m. Many of these particles are aggregates of smaller nanoparticles (with size of 10-200 nm).
The XRD pattern (Fig. 2) of the final product shows the presence of the rutile TiO2 phase (JCPDS file No. 01��077��0440) only. Its diffraction peaks are strong and sharp, which confirms the formation of the highly crystalline phase. Based on the above results, it is concluded that the procedure consisting of ball milling of carbon/ilmenite mixtures, annealing, acid leaching and calcining is a viable way to produce rutile TiO2 nanoparticles.

Fig. 1 SEM image of material obtained from milled and annealed mixture of FeTiO3/C after acid leaching and calcining (a) and high-magnification SEM image (b)

Fig. 2 XRD pattern of nanosized synthetic rutile obtained after annealing, acid leaching and calcining of milled mixture
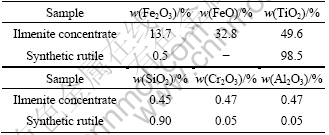
The chemical compositions of ilmenite and the obtained TiO2 nanoparticles, analyzed by EDS, are shown in Table 1. The nanoparticles contain 98.5% TiO2, 0.5% Fe2O3, 0.90% SiO2, 0.05% Cr2O3 and 0.05% Al2O3. The SiO2 is slightly higher about 0.90%, a generally accepted value in our experiments. The sum of impurities is not above 1.5%, which meets the requirement for industrial production. It is believed that a higher quality synthetic rutile can be obtained from ilmenite by the combination method of ball milling, annealing, and hydrochloric acid leaching.
Table 1 Chemical compositions of ilmenite and synthetic rutile produced
3.2 Effect of ball-milling and annealing
Figure 3(a) shows an SEM image of starting ilmenite, which consists of large particles with a typical size of 100-300 ��m and varied shapes. The leaching of the FeTiO3/C without the pre-ball milling and annealing treatment does not lead to noticeable changes in the material, as shown in Fig. 3(b). Only the surface of the material is partly disrupted due to the acid corrosion. As demonstrated below by XRD, TiO2 is hardly extracted by leaching ilmenite without the pre-ball milling and annealing treatment in dilute hydrochloric acid.

Fig. 3 SEM images of starting ilmenite (a) and sample obtained after acid leaching mixture of FeTiO3/C at 90 oC for 4 h without pre-ball milling and annealing treatment (b)
The XRD pattern (Fig. 4) indicates that the sample obtained after annealing of the mixture without the pre-ball milling and annealing treatment mainly consists of FeTiO3. Only small amounts of TiO2 are detected. It suggests that ilmenite is very resistant to dilute hydrochloric acid (4 mol/L) attacked at 90 �� for 4 h.
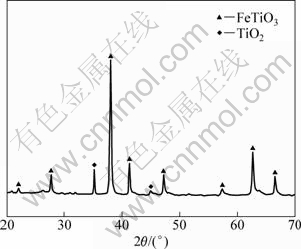
Fig. 4 XRD pattern of material obtained after annealing of mixture without pre-ball milling and annealing treatment
Figure 5(a) shows an SEM image of the materials prepared by leaching and calcining the milled mixture without annealing, which consists of large clusters with a typical size of 1-3 ��m. Actually, many of these clusters are aggregates of smaller nanoparticles. The XRD pattern (Fig. 5(b)) indicates that the phases in the material are TiO2 and FeTiO3. This suggests that iron cannot still be completely removed by acid leaching the milled FeTiO3/C without annealing treatment under the same conditions. The annealing enclosed in the combination method is an inevitable step for the preparation of higher quantity synthetic rutile. But the morphology and phase of mixture of FeTiO3/C after ball milling and acid leaching have significant changes. This can be attributed to mechanical activation effect.

Fig. 5 SEM image (a) and XRD pattern (b) of material prepared by leaching and calcining milled mixture without annealing
The ball milling of the mixture of FeTiO3/C leads to significant changes in both morphology and structure, as shown in Figs. 6(a) and (b). The SEM image in Fig. 6(a) shows the typical agglomerates of the mechanically milled mixtures that consist of particles of various sizes with irregular shapes. The diffraction peaks of the milled mixtures are broad (Fig. 6(b)). The broadening of the ilmenite��s XRD pattern after 50 h of ball milling is an indication of significant structural changes. The broadening of the peaks happens due to the nanocrystalline nature of the ball milled powders. The creation of nanocrystalline structure is a typical well known effect of ball milling [21].

Fig. 6 SEM image (a) and XRD pattern (b) of milled mixture
It is clear that the ball milling technique can conduce to a reduction in particle size and a decrease in the crystallite size of the ilmenite, which is beneficial to the dissolution in hydrochloric acid. The crystallite size of ilmenite is a function of logarithm milling time and determines the rate of dissolution [22]. Smaller crystallites dissolve much more rapidly than large ones because of the greater surface energy per unit volume. In addition, crystal defects and lattice distortions of material created by ball milling appear to be related to their dissolution.
Previous studies have also demonstrated that the mechanical activation can enhance the dissolution of ilmenite. For example, CHEN et al [23] have observed that no dissolution takes place in the unmilled ilmenite in the sulphuric acid solution. However, the products milled under vacuum for 200 h can be dissolved completely. LI et al [16,24,25] have investigated the dissolution of mechanically activated Panzhihua ilmenite in hydrochloric acid and sulfuric acid for the preparation of synthetic rutile. A highly quality synthetic rutile with a purity of 92.3% TiO2 (mass fraction) can be prepared from Panzhihua ilmenite milled for a short time of only 15 min in air by leaching it in a 20% HCl aqueous solution [24]. In the other case, simultaneous milling can greatly lead to a high leaching conversion of above 80% in a 50% dilute sulfuric acid [25]. Additionally, similar enhancement of dissolution has also been observed by ZHANG et al [26], and SASIKUMAR et al [3], respectively. All of these improved behaviours are attributed to mechanical activation effect. Several investigators have studied the leaching mechanism of ilmenite with hydrochloric acid or sulphuric acid [27,28]. A dissolution-hydrolysis precipitation mechanism has been proposed to explain the formation of synthetic rutile prepared by acid leaching the milled ilmenite [29].
The acid leaching mechanism of the milled and annealed mixture reported here is different from that of the milled ilmenite without carbothermic reduction because the ilmenite can be reduced to iron and TiO2, when the milled mixture of FeTiO3/C is annealed in Ar flow at 1000 �� for 1 h. The XRD pattern of the milled mixture of FeTiO3/C after annealing is shown in Fig. 7, which indicates the material obtained by annealing the milled mixture of FeTiO3/C mainly consists of TiO2. Only small amounts of FeTiO3 are detected by X-ray diffraction. A more detailed discussion of carbothermic reduction processes of the milled ilmenite can be found elsewhere [14,19]. In addition, it is well known that TiO2 is hardly dissolved with dilute acid. Thus we believe the nanosized synthetic rutile with a purity of 98.5% TiO2 mainly forms via an attack of acid on iron formed by the reduction of FeTiO3 into TiO2 and Fe. The dominant processes which are likely to happen during the annealing and leaching process can be summarized as follows:
FeTiO3+C��Fe+TiO2+CO (g) (1)
Fe+TiO2+2HCl��TiO2+FeCl2+H2(g) (2)
FeTiO3+2HCl��TiO2+FeCl2+H2O (3)
Carbothermic reduction (1) plays an important role in iron separation from ilmenite. The improvement of iron removal efficiency is obvious when the mineral is reduced. After leaching, the iron removing efficiency almost reaches 100% because no elemental iron could be detected by XRD (Fig. 2). Further investigation is needed to understand the detail of reaction mechanism during the leaching process of the milled and annealed mixture of FeTiO3/C. Of course, the interaction of diluted HCl solutions with ilmenite depends on a number of factors such as structural features of a ball milled FeTiO3, milling mode and atmosphere, presence of carbon, temperature of leaching and acid concentration.

Fig. 7 XRD pattern of milled mixture after annealing at 1000 �� for 1 h
3.3 Effect of concentration of HCl solution
In order to investigate the influences of the acid concentration on the leaching behavior of Fe and Ti from the milled and annealed mixtures of FeTiO3/C, a series of leaching experiments were performed at 90 �� for 4 h agitation with an S/L ratio of 1:25 (g/mL). The concentration of hydrochloric acid varies between 2 and 6 mol/L. The results are shown in Fig. 8, which indicates that the increase in acid concentration can greatly enhance the solubility of iron from the milled and annealed mixture. The iron removing efficiency reaches 98.5% after 4 h of leaching with 4 mol/L HCl. However, further increasing HCl concentration does not cause any significant change in the leaching efficiency of iron. On the other hand, with the increase of the acid concentration, a slight increase in the solubility of titanium dioxide from 0.21 to 1.32 can be observed only, which is negligible.

Fig. 8 Effect of acid concentration on leaching efficiency of milled and annealed mixtures of ilmenite and carbon
3.4 Effect of leaching temperature and time
The influences of temperature and time on the leaching behavior of Fe were also investigated. The results are presented in Fig. 9. As expected, the increase of temperature and time can improve the leaching efficiency of iron in hydrochloric acid, especially during the initial stage. 60 min-Fe leaching efficiencies are 60.3%, 69.3%, 83.5% and 85.6 %, respectively, at 70 ��, 80 ��, 90 �� and 100 ��. After 240 min of leaching, the Fe leaching efficiency can reach 98.5% with 4 mol/L acid at 90 ��. Since further increasing temperature will result in the volatile loss of HCl due to evaporation, this investigation is limited to 100 ��.

Fig. 9 Effect of leaching temperature and time on leaching efficiency of milled and annealed mixtures of ilmenite and carbon
Based on these results, we suggest that the hydrochloric acid concentration of 4 mol/L, temperature of 90 �� and time of 240 min are the optimum acid concentration, temperature and time, respectively. In addition, the samples (titanium dioxide and active carbon) obtained after leaching the milled and annealed mixture with dilute hydrochloric acid could have a good filtering performance. One possible reason is that TiO2 can be immobilized on active carbon during the acid leaching process because of the higher adsorption of active carbon [30]. The spent hydrochloric acid solutions could be recycled after separation although some iron impurity is in the acid solution.
4 Conclusions
1) A high quality nanosized synthetic rutile has been prepared by a combination method consisting of ball milling, annealing, leaching, and calcining.
2) Ball milling not only leads to the formation of nanostructures, but also improves the iron removal efficiency from ilmenite as well as annealing. After annealing, it becomes easy to remove iron from high titanium slag through hydrochloric acid leaching because ilmenite has been reduced to TiO2 and Fe.
3) Factors affecting the hydrochloric acid leaching process, such as acid concentration, leaching temperature and time, are investigated. The hydrochloric acid concentration of 4 mol/L, temperature of 90 �� and time of 240 min are suggested to be the optimum acid concentration, temperature and time, respectively. Under the optimal conditions, the purity of the final TiO2 nanoparticles (with size of 10-200 nm) is more than 98.0%.
References
[1] MACKEY T S. Acid leaching of ilmenite into synthetic rutile [J]. Ind Eng Chem, 1974, 13(1): 9-18.
[2] CHEMET T. Applied mineralogical studies on Australian sand ilmenite concentrate with special reference to its behavior in the sulphate process [J]. Miner Eng, 1999, 12(5): 485-495.
[3] SASIKUMAR C, RAO D S, SRIKANTH S, RAVIKUMAR B, MUKHOPADHVAY N K, MEHROTRA S P. Effect of mechanical activation on the kinetics of sulfuric acid leaching of beach sand ilmenite from Orissa, India [J]. Hydrometallurgy, 2004, 75(1-4): 189-204.
[4] JAB?O?SKI M, PRZEPIERA A. Kinetic model for the reaction of ilmenite with sulphuric acid [J]. J Therm Anal Calorim, 2001, 65(2): 583-590.
[5] KAHN J A. Non-rutile feedstocks for the production of titanium [J]. J Met, 1984, 36: 33-38.
[6] FARROW J B, RITCHIE I M. The reaction between reduced ilmenite and oxygen in ammonium chloride solutions [J]. Hydrometallurgy, 1987, 18(1): 21-38.
[7] SINHA H N, Hydrochloric acid leaching of ilmenite [C]//Proceeding of the Symposium on Extractive Metallurgy. Melbourne: The Australian Institute of Mining and Metallurgy, 1984: 163-168.
[8] LASHEEN T A. Sulfate digestion process for high purity TiO2 from titania slag [J]. Front Chem Eng China, 2009, 3(2): 155-160.
[9] YUAN Z, WANG X, XU C, LI W, KWAUK M. A new process for comprehensive utilization of complex titania ore [J]. Miner Eng, 2006, 19(9): 975-978.
[10] AMER A. Alkaline pressure leaching of mechanically activated Rosetta ilmenite concentrate [J]. Hydrometallurgy, 2002, 67(1-3): 125-133.
[11] LASHEEN T. Soda ash roasting of titania slag product from Rosetta ilmenite [J]. Hydrometallurgy, 2008, 93(3-4): 124-128.
[12] ZHANG Y J, QI T, ZHANG Y. A novel preparation of titanium dioxide from titanium slag [J]. Hydrometallurgy, 2009, 96(1-2): 52-56.
[13] BECHER R, CANNING R G, GOODHEART B A, UUSNA S. A new process for upgrading ilmenite mineral sands [J]. Australian IMM Proceedings, 1965, 214: 21-44.
[14] CHEN Y, HWANG T, MARSH M, WILLIAMA J. Mechanically activated carbothermic reduction of Ilmenite [J]. Metall Mater Trans A, 1997, 28(5): 1115-1121.
[15] WEI L P, HU H P, CHEN Q Y, TAN J. Effects of mechanical activation on the HCl leaching behavior of plagioclase, ilmenite and their mixtures [J]. Hydrometallurgy, 2009, 99 (1-2): 39-44.
[16] LI C, LIANG B, CHEN S P. Combined milling�Cdissolution of Panzhihua ilmenite in sulfuric acid [J]. Hydrometallurgy, 2006, 82(1/2): 93-99.
[17] DEMOPOULOS G P, LI Z B, BECZE L, MOLDOVEANU G, CHENG T C, HARRIS B. New technologies for HCl regeneration in chloride hydrometallurgy [J]. World Metall Erz, 2008, 61(2): 89-98.
[18] MAHMOUD M, AFIFI A, IBRAHIM I. Reductive leaching of ilmenite ore in hydrochloric acid for preparation of synthetic rutile [J]. Hydrometallurgy, 2004, 73(1-2): 99-109.
[19] WELHAM N J. A parametric study of the mechanically activated carbothermic reduction of ilmenite [J]. Miner Eng, 1996, 9(12): 1189-1200.
[20] TAO T, GLUSHENKOV A, CHEN Q Y, CHEN Y. Air-assisted growth of tin dioxide nanoribbons [J]. J Nanosci Nanotechnol, 2010, 10(8): 5015-5019.
[21] CHEN Y, BIBOE M, HAZIF R L, MARTIN G. Ball-milling induced amorphization in NixZry compounds: A parametric study [J]. Phys Rev B, 1993, 48(1): 14-21.
[22] WELHAM N J, LLEWELLYN D J. Mechanical enhancement of the dissolution of ilmenite [J]. Miner Eng, 1998, 11(9): 827-841.
[23] CHEN Y, WILLIAMS J S, CAMPBELL S J, WANG G M. Increased dissolution of ilmenite induced by high-energy ball milling [J]. Mater Sci Eng A, 1999, 271(1-2): 485-490.
[24] LI C, LIANG B, WANG H Y. Preparation of synthetic rutile by hydrochloric acid leaching of mechanically activated Panzhihua ilmenite [J]. Hydrometallurgy, 2008, 91(1-4): 121-129.
[25] LI C, LIANG B, GUO L H. Dissolution of mechanically activated Panzhihua ilmenites in dilute solutions of sulphuric acid [J]. Hydrometallurgy, 2007, 89 (1-2): 1-10.
[26] ZHANG L, HU H P, WEI L P, CHEN Q Y, TAN J. Effects of mechanical activation on the HCl Leaching behavior of titanaugite, ilmenite, and their mixtures [J]. Metall Mater Trans B, 2010, 41(6): 1158-1165.
[27] MACKEY T S. Acid leaching of ilmenite into synthetic rutile [J]. Ind Eng Chem Prod Res Dev, 1974, 13(1): 9-18.
[28] HAN K N, RUBCUMINTARA T, FUERSTENAU M C. Leaching behavior of ilmenite with sulfuric acid [J]. Metall Mater Trans B, 1987, 18(2): 325-330.
[29] SASIKUMAR C, SRIKANTH D S R, MUKHOPADHYAY N K, MEHROTRA S P. Dissolution studies of mechanically activated Manavalakurichi ilmenite with HCl and H2SO4 [J]. Hydrometallurgy, 2007, 88(1-4): 154-169.
[30] CARPIO E, ZUNIGA P, PONCE S, SOLISA J, RODRIGUEZA J, ESTRADA W R. Photocatalytic degradation of phenol using TiO2 nanocrystals supported on activated carbon [J]. J Mol Catal A, 2005, 228(1-2): 293-298.
�� ��1������Ԫ1������Ƽ1��������1���� Ӣ2
1. ���ϴ�ѧ ��ѧ����ѧԺ����ɳ 410083��
2. �Ĵ����ǵϿϴ�ѧ �о��봴�¼������ģ�ά������ 3217
ժ Ҫ������һ���¹�����������Ȼ�������Ʊ���Ʒλ��������ʯ���������ù�����Ҫ������ĥ��̼�Ȼ�ԭ������������衣��Ȼ������ͨ����ĥ���������ߴ�����ȼ�С��������̼�Ȼ�ԭ�ᵼ�¸��������γɡ���������������У��ص㿼�������ʱ�䡢�¶Ⱥ����Ũ�ȶԽ������̵�Ӱ�졣ͨ���Ż�����������������õ��IJ�ƷΪ���ȳ���98.0%�������ߴ�Ϊ10~200 nm��TiO2��������
�ؼ��ʣ�TiO2����������ĥ��̼�Ȼ�ԭ��������������
(Edited by YANG Hua)
Foundation item: Project (2007CB613601) supported by the National Basic Research Program of China; Project supported by the Postdoctoral Science Foundation of Central South University, China
Corresponding author: TAO Tao; Tel: +86-731-88877364; Fax: +86-731-88879602; E-mail: yunqing5252@sina.com
DOI: 10.1016/S1003-6326(11)61310-1

