Synthesis of aluminum tri-polyphosphate anticorrosion pigment from bauxite tailings
来源期刊:中国有色金属学报(英文版)2012年第2期
论文作者:胡岳华 卢清华
文章页码:483 - 488
关键词:三聚磷酸铝;废物处理;电化学阻抗谱
Key words:aluminum tri-polyphosphate; waste processing; electrochemical impedance spectroscopy
摘 要:
将铝土矿选矿尾矿与磷酸以P与Al 摩尔比等于3的比例混合,制备了三聚磷酸铝,并用氧化锌对所制备的三聚磷酸铝进行改性处理,得到三聚磷酸铝防腐颜料。改性的三聚磷酸铝的酸度、白度及粒度均得到了改善。运用电化学阻抗方法测试了所制备的三聚磷酸铝防腐颜料的防腐性能。结果表明:含有改性三聚磷酸铝防腐颜料的涂层在电解质中浸泡40 d后的阻抗为7×107 Ω,含商品三聚磷酸铝防腐颜料APW?2的涂层浸泡40 d后的阻抗为5.7×107 Ω,这表明所制备的三聚磷酸铝防腐颜料具有优于或相当于商品防腐颜料的防腐性能。
Abstract: Aluminum tri-polyphosphate was synthesized from bauxite tailings and phosphoric acid with a P/Al molar ratio of 3. This is highly advantageous from a waste recycling perspective as bauxite tailings are generally regarded as unusable waste. The acidity, whiteness and mean particle size of prepared aluminum tri-polyphosphate are all improved after modification. The possibility of using modified aluminum tri-polyphosphate as an anticorrosive pigment was also investigated by electrochemical impedance spectroscopy. The resistance of the coatings with modified aluminum tri-polyphosphate is 7×107 Ω for 40 d of immersion, which is superior or at least comparable to that of coatings containing APW?2, which exhibits a resistance of 5.7×107 Ω.
基金信息:the Major State Basic Research Development Program of China

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 483-488
LU Qing-hua1, HU Yue-hua2
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 18 January 2011; accepted 21 July 2011
Abstract: Aluminum tri-polyphosphate was synthesized from bauxite tailings and phosphoric acid with a P/Al molar ratio of 3. This is highly advantageous from a waste recycling perspective as bauxite tailings are generally regarded as unusable waste. The acidity, whiteness and mean particle size of prepared aluminum tri-polyphosphate are all improved after modification. The possibility of using modified aluminum tri-polyphosphate as an anticorrosive pigment was also investigated by electrochemical impedance spectroscopy. The resistance of the coatings with modified aluminum tri-polyphosphate is 7×107 Ω for 40 d of immersion, which is superior or at least comparable to that of coatings containing APW-2, which exhibits a resistance of 5.7×107 Ω.
Key words: aluminum tri-polyphosphate; waste processing; electrochemical impedance spectroscopy
1 Introduction
Classical anticorrosive paints normally contain inhibitors based on hexavalent chromium or lead compounds. The incorporation of these compounds is being restricted because they contaminate the environment and cause health problems to humans. Regulations in various countries led to an intense research effort to find alternative pigments of low toxicity without compromising performance. A new generation of pigment, layer-structured aluminum tri-polyphosphate (AlH2P3O10・2H2O: abbreviated as ALP) has been developed and used widely as an anticorrosion pigment due to its low toxicity to humans and the environment [1].
The synthesis, structure, thermal changes and ion-exchange properties of acidic aluminum tri-polyphosphate were first reported in 1960 before its industrial production methods were established in 1970. The industrial production method was based on their patent of producing ALP by means of the substance containing aluminum and phosphoric acid or phosphate [2]. This maintains the current industrial preparation method of ALP [2]. The development and application of ALP in the paint industry was achieved by the Imperial Japanese Corporation until the 1970s. Since then, many studies have been carried out on its synthesis and application. TSUHAKO et al [3] found that the formation of ALP is greatly dependent on the water content of the primary product and on the vapor pressure in the secondary treatment. They also showed that AlH2P3O10・2H2O dehydrates at 150 °C to form AlH2P3O10 which decomposes to form Al4(P4O12)3 at 500 °C. SHI and LIN [4] showed that the optimum parameters for maximizing ALP yield was when a P to Al molar ratio of 3:1 was used, coupled with a condensation temperature in the range of 200-320 °C and condensation time of about 10 h.
Bauxite tailings are the aluminosilicate-waste generated during the process of improving the molar ratio of aluminum to silicon in bauxite by beneficiation methods. It was reported that 0.2 t of aluminosilicate tailings were generated from every ton of bauxite ores processed by flotation. In China, a large quantity of bauxite tailings are stored in the tailing dam after flotation [5]. The main minerals in bauxite tailings are diaspore, kaolinite, illite and pyrophyllite. Some investigations and trials for the utilization of bauxite tailings in building materials [6], fillers [7] and refractory materials [8] have been carried out in recent years. However, because of the high iron content in the waste,its large-scale application has been rendered infeasible. In order to expand the application volume of bauxite tailings, researchers attempted to remove iron from bauxite tailings by magnetic separation, acid pickling, reduction and other methods, but failed. This is due to the iron in the bauxite tailings distributed between numerous minerals that are mutually embed with complicated relationships and fine particles.
With industrial development, low-cost production for a variety of materials is required. Much industrial waste has been studied and recycled [9-11]. ALP was generally prepared using chemicals. Till now, there are no reports on preparation of ALP from industrial waste. Aluminum oxide content in bauxite tailings is generally more than 50%. Therefore, we attempted to prepare ALP using bauxite tailings and phosphoric acid. In addition, we believe that it is possible to prepare a white anti-corrosive pigment by means of the reactions between iron and phosphoric acid, which not only reduces the production cost of ALP, but also recycles the bauxite tailings.
2 Experimental
2.1 Bauxite tailings
The bauxite tailings were obtained from Zhongzhou bauxite flotation plant in China. The chemical compositions were 57.6% Al2O3, 19.8% SiO2, 2.18% TiO2, 1.6% MgO, 1.08% CaO, 4.84% TFe, 0.69% K2O and 0.82% Na2O. Diaspore, kaolinite, illite, anatase, hematite and quartz were determined as the main minerals in bauxite tailings by X-ray powder diffractometry. The bauxite tailings were milled for 5 h with a planet-ball mill. The average diameter of milled tailings measured with a laser diffraction analyzer was 3.8 μm.
2.2 Synthesis of white anti-corrosion pigments
The milled bauxite tailings were calcined at 600 °C for 2 h in order to eliminate organic matter and to increase its reactivity. The aluminum tri-polyphosphate was prepared via two heat treatments. The specific process for preparation is as follows.
First, tailings and phosphoric acid were mixed in a porcelain crucible with P to Al molar ratios of 2.0, 2.5 and 3.0. The quantity of used phosphoric acid is equal to the sum of the stoichiometric phosphoric acid of the following each reaction due to the fact that the calcium, magnesium, iron and other metal elements except silicon for tailings likely react with the phosphoric acid.
Al2O3+H3PO4→AlH2P3O10・2H2O
Fe2O3+H3PO4→Fe (PO3)3+H2O
CaO+H3PO4→Ca3 (PO4)2+H2O
MgO+H3PO4→Mg3 (PO4)2+H2O
TiO2+H3PO4→Ti3 (PO4)4+H2O
Second, the mixture was then dehydrated by heating over a weak flame with vigorous agitation to obtain a white, highly viscous product in each case. This is designated as the primary heat treatment, giving the primary products.
Third, the primary product was heated at 300 °C for 10 h in a sealed tube. This is designated as the secondary heat treatment, giving the secondary products. The secondary product was washed, milled and dried, eventually a white powder was obtained.
Fourth, the aluminum tri-polyphosphate prepared and zinc oxide (ZnO) were mixed with the mass ratio of 17:3. The resultant mixture was milled for 8 h by wet process, then reacted for 3 h at 120 °C in a reactor, dried, pulverize, a modified aluminum tri-polyphosphate was obtained.
2.3 Test and characterization
X-ray diffraction (XRD) patterns were recorded on a RIGAKU D/max-2550VB+18 kW powder diffractometer with Cu Kα-radiation (λ=1.541806 ?). The thermal stability of the samples was measured in air with a heating rate of 10 °C/min in the range of 35-800 °C on a STA449C and simultaneously on a DSC-TG instrument. The morphology of the sample was examined by scanning electron microscopy (SEM, Sirion 200 field emission, FEI Company). The whiteness samples were determined according to the criterion of GB/T 5950―2008 of the National Standards of China. The pH value of aqueous suspension of sample was determined by the criterion of GB-T 1717―1986. The average diameter of the particle was measured on a MS2000 laser diffraction size analyzer.
2.4 Evaluation of anticorrosive properties of pigments
2.4.1 Method
Electrochemical impedance spectroscopy (EIS) was employed to assess the anticorrosive properties of the pigments. EIS measurements were performed using a PAR 2273 potentiostat. The coated samples were immersed in a 3.5% NaCl solution. A three-electrode system comprising of a saturated calomel electrode as a reference electrode, the coated sample as a working electrode and platinum as a counter electrode was employed (Fig. 1). A sine wave of 10 mV was applied across the cell. The measurements were made in the frequency range of 50 kHz-0.01 Hz.
2.4.2 Preparation of coated samples
The pigment powder was added to an epoxy resin solution (Epikote1001 solution 70% in toluene) and mechanically dispersed. The liquid substance with dispersed pigment powder was mixed with a curing agent (polyamide, Crayamid 115) at stoichiometric ratio. The pigment content in the paint was 15%. The paint was coated on one surface of steel panels with dimensions of 140 mm×100 mm×4 mm by brushing to a thickness of (80±5) μm, and cured at room temperature for 7 d. The surfaces of panels were polished, degreased with acetone and ethanol, and dried in air before painting. In addition, the coatings with no pigment (blank coating) and with zinc aluminium phosphate (ZnAlPO4) under the trade mark APW-2 were employed as references.
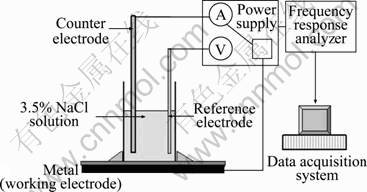
Fig. 1 Sketch of EIS experimental set-up
3 Results and discussion
3.1 XRD analysis
The phases present in each prepared sample were determined via XRD analysis. Figure 2 shows the XRD patterns of the samples. There are some peaks that cannot be discerned, in addition to aluminum phosphate and aluminum tri-polyphosphate peaks in patterns of n(P)/n(Al)=2.0 and n(P)/n(Al)=3.0. We denoted the unidentifiable diffraction peaks as “Unknown”. It is apparent that phases of samples depend on the amount of phosphoric acid. Sample of n(P)/n(Al)=2.0 contains aluminum phosphate, a small amount of aluminum tri-polyphosphate and other unidentifiable crystalline phases. Compared with sample of n(P)/n(Al)=2.0, sample of n(P)/n(Al)=3.0 showed an increase of aluminum tri-polyphosphate, a decrease in unidentifiable crystalline phases, and a disappearance of aluminum phosphate. Among all samples, sample of n(P)/n(Al)=2.5 exhibited the highest intensity peak of aluminum tri-polyphosphate, which was the only crystalline phase present in it, and the pattern of sample of n(P)/n(Al)=2.5 also exhibited the minimum baseline noise, which indicates that sample of n(P)/n(Al)=2.5 is more crystalline with a maximum yield.
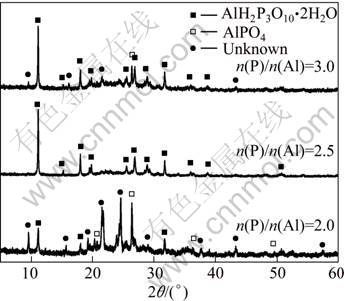
Fig. 2 XRD patterns of prepared samples of n(P)/n(Al)=2.0, 2.5 and 3.0
3.2 DSC-TG analysis
Figure 3 shows the DSC-TG curves of sample of n(P)/n(Al)=2.5. The curves reveal a two-stage dehydration process. The dehydration of the first stage occurred in the range of 120-260 °C, the mass loss of 6.48% was the loss of two molecules of water in the crystalline structure. The second stage occurred in the range of 430-570 °C, the mass loss of 4.21% was indicative of a further loss of one water molecule [12]. The mass losses in the two stages are different from the theoretical loss of 11.32% and 5.66%, which resulted from impurities in the sample. Simultaneously, formation of new phases also occurred in the course of dehydration. The process is expressed as follows:
![]()
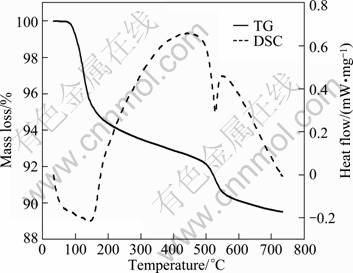
Fig. 3 DSC-TG curves of sample at n(P)/n(Al)=2.5
3.3 Morphology of sample
SEM morphology of sample of n(P)/n(Al)=2.5 is shown in Fig. 4. The particles have a layered structure and irregular shape, but most of the particles are flaky. The grain sizes range from 0.1 to 5 μm. There are a few agglomerations between grains. Flaky particles are better than granular particles in obstructing air and water whose penetration is the main cause of steel corrosion.

Fig. 4 SEM image of sample with n(P)/n(Al)=2.5
3.4 pH value and whiteness
The physical parameters for pigment of APW-2 and the samples of n(P)/n(Al)=2.5 before and after modification are shown in Table 1. The pH value for APW-2 is 6.8. The unmodified sample has only pH value of 5.8, which is too low to be anticorrosive, so it is indispensable to improve its pH via modification.
Table 1 Chemical compositions and physical parameters of samples of n(P)/n(Al)=2.5, modified samples of n(P)/n(Al)=2.5 and APW-2
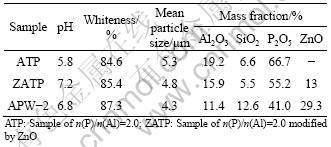
People think that protection depends more on the high pH provided by the pigment than on other of its properties [13, 14]. The pH value increases to 7.2, the whiteness and the mean particle size are all improved after the sample modification.
3.5 Electrochemical impedance spectroscopy
EIS is a well established quantitative method for examining protective properties, such as anticorrosion performance of organic coatings. This technique is widely used with good results for the electrochemical characterization of protective organic coatings on metal [15]. Impedance can reflect the degree of difficulty of an electrolyte passing through the coating, and is a valid method to extrapolate the long-term anti-corrosion effect of coatings. Greater impedance indicates a better anticorrosion effect [16].
In this study, the anticorrosive behaviors of coatings containing modified aluminum tri-polyphosphate pigment, APW-2 pigment, and no pigment were investigated by means of electrochemical impedance method. The coating resistance as a function of time during the immersion in 3.5% NaCl solution is shown in Fig. 5.
It is clear from Fig. 5(a) that the resistance of the coatings with modified aluminum tri-polyphosphate pigment at 0.01 Hz was higher than 108 Ω at the beginning of exposure and it decreased to 6.5×107 Ω after 20 d of exposure. After that it showed a small increase to 7×107 Ω until 40 d. The decrease in resistance at the initial stage of exposure may be due to the permeation of water and ions into the film. The increase after 40 d of exposure seems to be attributed to the formation of passive states, such as iron phosphate and Fe3(PO4)2・nH2O [17].
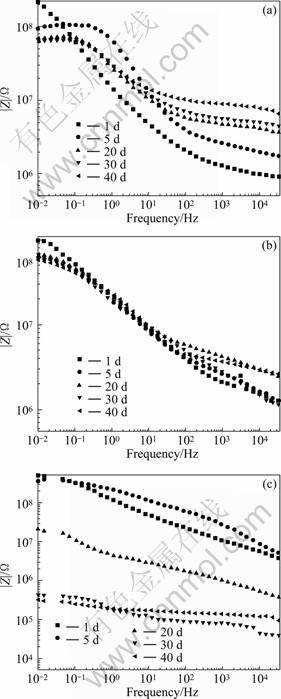
Fig. 5 Resistance of coatings with sample of n(P)/n(Al)=2.5 (a), with APW-2 (b) and blank coating (c) as function of time during immersion in 3.5% NaCl
Figure 5(b) presents the change of impedance with exposure time for an APW-2 pigmented film. At the beginning of exposure, the resistance at 0.01 Hz was around 9.8×107 Ω and it decreased continuously to 5.7×107 Ω until 40 d of exposure. The range of decrease also decreased after 5 d of exposure.
In the case of the blank coating, as shown in Fig. 5(c), the resistance at 0.01 Hz was higher than 108 Ω up to the first 5 d of exposure. However, it decreased quickly to 1×107 Ω at 20 d. After 40 d of exposure, a few blisters were found on the specimen and the resistance was 3×105 Ω. It is generally known that the coating films with a resistance higher than 108 Ω reveals good anticorrosive performance, while that with the resistance lower than 106 Ω has poor anticorrosive properties. So the blank coating lost its anticorrosive properties within the first 5 d of exposure.
From the comparison of results for the modified aluminum tri-polyphosphate, APW-2 and blank coating films, it can be seen that the anticorrosive performance of the modified aluminum tri-polyphosphate is superior or at least comparable to that of the APW-2, which has been used as anticorrosive pigments.
4 Conclusions
1) Aluminum tri-polyphosphate was successfully synthesized from bauxite tailings and phosphoric acid via two heat treatments, giving a molar ratio of P to Al of 2.5, a condensation temperature of 300 °C and condensation time of 10 h.
2) The acidity, whiteness and mean particle size for prepared aluminum tri-polyphosphate were all improved after modification. The possibility of using modified aluminum tri-polyphosphate as an anticorrosive pigment was investigated using electrochemical impedance spectroscopy.
3) The resistance of the coatings with modified aluminum tri-polyphosphate was 7×107 Ω, while that of coatings with APW-2 was 5.7×107 Ω after 40 d of immersion, which showed the superior anticorrosive performance of modified aluminum tri-polyphosphate, or at least comparable to that of APW-2. The whiteness of modified aluminum tri-polyphosphate is 85.4%, which meets the industrial requirements.
References
[1] NISHIKAWA H, OISO Y, ISHIHARA M. Photocatalytic behavior of layered aluminum dihydrogen triphosphate under UV irradiation [J]. Materials Letters, 2007, 61: 1820-1822.
[2] LUO Ming. Aluminum tripolyphosphate anticorrosive pigment and its modified synthesis methods [J]. Technology and Development of Chemical Industry, 2004, 33: 31-34. (in Chinese)
[3] TSUHAKO M, HASEGAWA K, MATSUO T, MOTOOKA I, KOBAYASHI M. Studies of the preparation and physical properties of multivalent metal condensed phosphates and their thermal transformation [J]. Bulletin of the Chemical Society of Japan, 1975, 48: 1830-1835.
[4] SHI Xiao-bo, LIN Bin-fa. The study of the resultant and heat-change in the Al203-H3P04 system [J]. Journal of Jiangxi Normal University, 1989, 13(2): 52-55. (in Chinese)
[5] LIU Wei-ping, YUAN Jian-xiong. The application of tailings in the non-organism nonmetal material [J]. China Mining, 2004, 13(11): 16-18. (in Chinese)
[6] YANG H Z, CHEN C P, PAN L J. Preparation of double-layer glass-ceramic/ceramic tile from bauxite tailings and red mud [J]. Journal of the European Ceramic Society, 2009, 29: 1887-1894.
[7] HU Yue-hua, YANG Hua-ming, HU Xiao-dong. A kind of modified tailings powder and the powder packing method for Polymer: China, 200710034335 [P]. 2007-10-03. (in Chinese)
[8] YANG Dong, FENG Nai-xiaog, WANG Yao-wu. Preparation of primary Al-Si alloy from bauxite tailings by carbothermal reduction process [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(1): 147-152.
[9] NORLIZA I, MURTHY V, TENG W D. Ceramic membrane fabrication from industrial waste: Effect of particle size distribution on the porosity [J]. Journal of Applied Sciences, 2009, 9: 3136-3140.
[10] BHATNAGAR A, MINOCHA A K. Utilization of industrial waste for cadmium removal from water and immobilization in cement [J].Chemical Engineering Journal, 2009, 150: 145-151.
[11] OHM T I, CHAE J S, KIM J E, KIM H K. A study on the dewatering of industrial waste sludge by fry-drying technology [J]. Journal of Hazardous Materials, 2009, 168: 445-450.
[12] YUAN A Q, ZHOU Z G, BAI L J. One-step synthesis and thermal decomposition kinetic of aluminum dihydrogen tripolyphosphate dehydrate I [J]. Chemical Engineering, 2007, 35: 32-36.
[13] ROMAGNOLI R, DEY?M C, DEL AMO B. The mechanism of the anticorrosive action of calcium-exchanged silica [J]. Surface Coatings International, 2003, 86: 135-141.
[14] CHICO B, SIMANCAS J, VEGA J M, GRANIZO N. Anticorrosive behaviour of alkyd paints formulated with ion-exchange pigments [J]. Progress in Organic Coatings, 2008, 61(2-4): 283-290.
[15] SKALE S, DOLECEK V, SLEMNIK M. Electrochemical impedance studies of corrosion protected surfaces covered by epoxy polyamide coating systems [J]. Progress in Organic Coatings, 2008, 62: 387-392.
[16] ZHANG J Y, LI J, WANG X H. Corrosion prevention with electrically conductive polymers [J]. Journal of Functional Polymers, 1999, 12: 350-356.
[17] PARK J H, LEE G D, NISHIKATA A, TSURU T. Anticorrosive behavior of hydroxyapatite as an environmentally friendly pigment [J]. Corrosion Science, 2002, 44: 1087-1095.
卢清华1, 胡岳华2
1. 中南大学 冶金科学与工程学院,长沙 410083;
2. 中南大学 资源加工与生物工程学院,长沙 410083
摘 要:将铝土矿选矿尾矿与磷酸以P与Al 摩尔比等于3的比例混合,制备了三聚磷酸铝,并用氧化锌对所制备的三聚磷酸铝进行改性处理,得到三聚磷酸铝防腐颜料。改性的三聚磷酸铝的酸度、白度及粒度均得到了改善。运用电化学阻抗方法测试了所制备的三聚磷酸铝防腐颜料的防腐性能。结果表明:含有改性三聚磷酸铝防腐颜料的涂层在电解质中浸泡40 d后的阻抗为7×107 Ω,含商品三聚磷酸铝防腐颜料APW-2的涂层浸泡40 d后的阻抗为5.7×107 Ω,这表明所制备的三聚磷酸铝防腐颜料具有优于或相当于商品防腐颜料的防腐性能。
关键词:三聚磷酸铝;废物处理;电化学阻抗谱
(Edited by LI Xiang-qun)
Foundation item: Project (2005CB623701) supported by the National Basic Research Program of China
Corresponding author: HU Yue-hua; Tel: +86-731-88877203; Fax: +86-731-88710804; E-mail: luqinghua@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(11)61202-8

