以PEG为分散剂从废绿矾中回收Fe3O4
来源期刊:中国有色金属学报(英文版)2018年第7期
论文作者:彭映林 余旺 郑雅杰
文章页码:1465 - 1474
关键词:绿矾;Fe3O4;PEG;共沉淀;磁分离;湿磨
Key words:ferrous sulfate; Fe3O4; PEG; co-precipitation; magnetic separation; wet milling
摘 要:以CaOH为沉淀剂通过共沉淀从绿矾溶液中回收Fe3O4,研究分散剂PEG对Fe3O4磁分离的影响。结果表明,分散剂PEG对Fe3O4的回收有显著影响。在Fe3O4制备过程中添加PEG不利于Fe3O4的回收。当PEG-6000浓度从0增加到8 g/L时,Fe3O4产品的铁品位从65.58%降至57.79%,中值粒径从2.35 μm 降至1.35 μm。然而,在混合产物的湿磨过程中添加PEG可促进随后的Fe3O4回收。当PEG-200用量从粉末质量的0%增加到4%时,Fe3O4产物中铁的品位从65.58% 升高到68.32%。当PEG-200用量为4%时,改变PEG相对分子质量从200增加到20000,Fe3O4产物中铁品位从68.32%降至66.70%。
Abstract: The effect of PEG dispersant on the magnetic separation of magnetite (Fe3O4) synthesized from ferrous sulfate solution via co-precipitation method with calcium hydroxide as the precipitant was investigated. The results indicated that a PEG dispersant could significantly affect Fe3O4 recovery. Adding PEG during the preparation of Fe3O4 was unfavorable for Fe3O4 recovery. When the PEG-6000 concentration was increased from 0 to 8 g/L, the iron grade and median particle size of the Fe3O4 product decreased from 65.58% and 2.35 μm to 57.79% and 1.35 μm, respectively. However, adding PEG during the wet milling of the mixed product promoted the subsequent recovery of Fe3O4. When the amount of PEG-200 increased from 0% to 4% of the powder mass, the grade of iron in the Fe3O4 product increased from 65.58% to 68.32%. While the relative molecular mass of PEG at an amount of 4% of the powder mass increased from 200 to 20000, the grade of iron was reduced from 68.32% to 66.70%.
Trans. Nonferrous Met. Soc. China 28(2018) 1465-1474
Ying-lin PENG1, Wang YU2, Ya-jie ZHENG2
1. School of Materials and Chemical Engineering, Hunan City University, Yiyang 413000, China;
2. School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 14 March 2017; accepted 25 July 2017
Abstract: The effect of PEG dispersant on the magnetic separation of magnetite (Fe3O4) synthesized from ferrous sulfate solution via co-precipitation method with calcium hydroxide as the precipitant was investigated. The results indicated that a PEG dispersant could significantly affect Fe3O4 recovery. Adding PEG during the preparation of Fe3O4 was unfavorable for Fe3O4 recovery. When the PEG-6000 concentration was increased from 0 to 8 g/L, the iron grade and median particle size of the Fe3O4 product decreased from 65.58% and 2.35 μm to 57.79% and 1.35 μm, respectively. However, adding PEG during the wet milling of the mixed product promoted the subsequent recovery of Fe3O4. When the amount of PEG-200 increased from 0% to 4% of the powder mass, the grade of iron in the Fe3O4 product increased from 65.58% to 68.32%. While the relative molecular mass of PEG at an amount of 4% of the powder mass increased from 200 to 20000, the grade of iron was reduced from 68.32% to 66.70%.
Key words: ferrous sulfate; Fe3O4; PEG; co-precipitation; magnetic separation; wet milling
1 Introduction
Ferrous sulfate (FeSO4・7H2O) is a by-product of titanium dioxide manufacture via the sulfate method. In this method, 3-4 t ferrous sulfate is generated to obtain 1 t titanium dioxide. In China, about 98% of the titanium dioxide is manufactured by the sulfate method, thereby yielding more than 7×106 t ferrous sulfate waste each year, and the annual growth rate in waste emissions exceeds 10% as titanium dioxide production expands [1,2]. Unfortunately, this waste is less marketable and difficult to be utilized due to its high impurity contents (i.e., Mg, Mn, Ti, Zn and Al) [3], which causes not only severe environmental problems but also the waste of iron resource.
Using ferrous sulfate to produce iron oxides is an effective method for ferrous sulfate re-utilization because of the increasing demand for iron oxides driven by the increasing consumption in industry. Fe3O4, with fascinating properties, including good biocompatibility, high surface area, low toxicity, super-paramagnetism, and high saturation field [4], has attracted much attention from both academic and industrial interests in many application fields, such as magnetic storage media, solar energy transformation, catalysis, biological and chemical sensing, ferrofluids and environmental remediation [5-9]. In environmental engineering, however, the utilization of Fe3O4 particles prepared from reagent-grade chemicals could not be cost-effective in the full-scale process, due to the characteristics of the water or wastewater, such as high flow rate, and the presence of various pollutants and associated high consumption and loss of particles [10]. In this work, we proposed a novel and low cost method to synthesize Fe3O4 particles from waste ferrous sulfate.
Numerous methods have been developed for preparing Fe3O4 particles, such as co-precipitation [11], microemulsions [12], sol-gel syntheses [13], and hydrothermal or solvothermal process [14]. However, the most common method for producing synthetic Fe3O4 particles is the co-precipitation of Fe2+/Fe3+ ions (molar ratio 1:2) by sodium hydroxide or ammonia solution [15]. In our approach, Fe3O4 was recovered from ferrous sulfate solution by co-precipitation and magnetic separation with calcium hydroxide as the precipitant [16,17]. We used lime instead of other alkali reagents due to its availability and low cost. Although caustic soda (NaOH) can be used for the pH adjustment, its price is 5 times higher than that of lime [18].
In the magnetic separation process, the Fe3O4 particles tend to heterocoagulate, so a good dispersion is required to improve their separation efficiency. The application of a polymeric dispersant in the magnetic separation of Fe3O4, however, has not drawn considerable attention. Since PEG is a water soluble polymer together with the other properties, such as bio- compatibility, low immunogenicity, and ease of use [19], we investigated the effect of a PEG dispersant on the magnetic separation of Fe3O4 synthesized from waste ferrous sulfate. The aim of this work is to investigate the effect of adding PEG during the preparation of Fe3O4 or during the wet milling of the mixed product on magnetite recovery.
2 Experimental
2.1 Materials
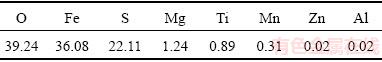
Reagent-grade NaOH, CaO, and PEG (Guangdong Xilong Chemical Co., Ltd., China) were used directly without further purification. The dried ferrous sulfate was obtained from Guangdong Hui Yun Titanium Industry Corporation Limited, China. The dried ferrous sulfate sample was further dried in a vacuum atmosphere at 80 °C for 12 h, and then its chemical composition was analyzed by X-ray fluorescence (XRF), as given in Table 1. The sample was composed of O (39.24%), Fe (36.08%), S (22.11%) and other impurities.
Table 1 Chemical composition of dried ferrous sulfate (mass fraction, %)
The ferrous sulfate solution was purified with the addition of NaOH solution to precipitate impurities [16]. The solution pH was adjusted to 3.5-4.0 by 5 mol/L NaOH solution, followed by continuous stirring for 30 min at room temperature. After filtration, the solution was diluted with distilled water. The diluted solution was used as an iron source for the Fe3O4 preparation and its composition is given in Table 2.
Table 2 Chemical composition of purified ferrous sulphate solution (g/L)

2.2 Experimental procedures
Fe3O4 was prepared from ferrous sulfate solution by co-precipitation method with calcium hydroxide as the precipitant. 4 L of purified ferrous sulfate solution (Table 2) was added to a 5 L, four-necked flask fitted with a reflux condenser and electric heater. Lime milk was obtained by adding distilled water to calcium oxide (CaO) at a liquid-to-solid mass ratio of 3:1 under vigorous stirring. Then, the lime milk with a 1.4:1 molar ratio of CaO/Fe2+ was added dropwise to the solution under sufficient stirring. The reactor was heated to 80 °C, and air was bubbled into the solution at 0.6 m3/h. When the mole ratio of Fe3+/Fe2+ in the reaction solution reached 1.9:1-2.1:1, the reaction was stopped. The obtained mixture of Fe3O4 and gypsum (CaSO4・2H2O) was then filtrated, dried, and ground for use in subsequent experiment. The wet-milling pretreatment was conducted at room temperature in a ball mill using a slurry with a mixed product-to-water mass ratio of 1:40 and a rotation speed of 130 r/min. The time of milling was 20 min, and the treated mixed product was directly subjected to magnetic separation. Magnetic separation was performed using a low-intensity magnetic separator (CRIMM DC CXGφ50, Changsha Research Institute of Mining and Metallurgy Co., Ltd., China), with a working electrical current of 1.75 A. The magnetic fractions were washed, filtered, dried, weighed and subjected to various analyses. Fe3O4 can be formed from ferrous salts according to the following reactions [20]:
CaO+H2O→Ca(OH)2 (1)
FeSO4+Ca(OH)2+2H2O→Fe(OH)2+CaSO4・2H2O (2)
4Fe(OH)2+O2+2H2O→4Fe(OH)3 (3)
2Fe(OH)3+Fe(OH)2→Fe3O4+4H2O (4)
In this work, the effect of PEG dispersant on the magnetic separation of Fe3O4 was investigated as follows: 1) During the preparation of Fe3O4, PEG-6000 with a concentration ranging from 0 to 8 g/L was added to the reaction solution; 2) During the wet milling of the mixed product, PEG-200, in amounts ranging from 0% to 16% of the powder mass, was added to the slurry; PEG, with a relative molecular mass ranging from 200 to 20000, was also studied at an amount under 4% of the powder mass.
The procedure used to recover Fe3O4 from ferrous sulfate is shown in Fig. 1.
2.3 Analysis
The samples were analyzed to examine the ferrous and ferric iron contents according to GB/T 1863-2008 (Chinese National Standard). The contents of trace elements in sample were determined by inductively coupled plasma optical emission spectrometry (ICP-OES, iCAP 7000 SERIES, Thermo Scientific) after the total dissolution of samples. Phase analysis of the products was conducted by a Rigaku D/max-TTR III X-ray diffractometer (XRD) with Cu Kα radiation (λ= 1.54056 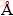 , voltage of 40 kV, and current of 250 mA) and at the scanning rate of 10 (°)/min from 10° to 70°. To identify the functional groups present in the product, Fourier transform infrared (FT-IR) spectrometer (WQF-510A, Beijing Beifen-Ruili Analytical Instrument (Group) Co., Ltd., China) was used to record the spectrum from 400 to 4000 cm-1. The raw material sample was measured by X-ray fluorescence (XRF) with a Bruker S4 Pioneer system equipped with two X-ray detectors. Surface morphology of the samples was observed by using a FEI Quanta 200 scanning electron microscope (SEM), coupled with an energy dispersive X-ray spectroscope (EDS). Laser particle size analyzer (LS-pop(6), Zhuhai OMEC Instrument Co., Ltd., China) was used to analyze the size distribution of samples. Magnetic properties of the samples were measured with a vibrating sample magnetometer (VSM, Quantum Design) at room temperature.
, voltage of 40 kV, and current of 250 mA) and at the scanning rate of 10 (°)/min from 10° to 70°. To identify the functional groups present in the product, Fourier transform infrared (FT-IR) spectrometer (WQF-510A, Beijing Beifen-Ruili Analytical Instrument (Group) Co., Ltd., China) was used to record the spectrum from 400 to 4000 cm-1. The raw material sample was measured by X-ray fluorescence (XRF) with a Bruker S4 Pioneer system equipped with two X-ray detectors. Surface morphology of the samples was observed by using a FEI Quanta 200 scanning electron microscope (SEM), coupled with an energy dispersive X-ray spectroscope (EDS). Laser particle size analyzer (LS-pop(6), Zhuhai OMEC Instrument Co., Ltd., China) was used to analyze the size distribution of samples. Magnetic properties of the samples were measured with a vibrating sample magnetometer (VSM, Quantum Design) at room temperature.
After the experiment, the recovery rate of iron in the Fe3O4 product (R) was calculated based on the mass of iron in the raw material (the purified ferrous sulfate solution) and defined as follows:
R=m1/m×100% (5)
where m1 is the mass of iron in the Fe3O4 product and m is the mass of iron in the raw material.
The grade of iron in the Fe3O4 product (G) was calculated based on the mass of the Fe3O4 product and defined as follows:
G=m1/m2×100% (6)
where m2 is the mass of the Fe3O4 product.
3 Results and discussion
3.1 Characterization of Fe3O4 product without adding PEG
In this work, a mixed product was first prepared from ferrous sulfate solution by co-precipitation with calcium hydroxide as the precipitant. Figure 2(a) shows the XRD pattern of the mixed product. XRD analysis indicated that Fe3O4 (JCPDS, No. 19-0629) and CaSO4・2H2O (JCPDS, No. 33-0311) were the major components. The SEM image of the mixed product is shown in Fig. 2(b). From Fig. 2(b), two distinct phases (zones A and B) were observed: the phase in zone A was irregularly shaped plates, while fine phase in zone B were apt to aggregate. The subsequent EDS analysis proved that the two phases were CaSO4・2H2O (zone A) and Fe3O4 (zone B), which is in agreement with the above XRD results.

Fig. 1 Flow sheet of Fe3O4 recovery from ferrous sulfate

Fig. 2 XRD pattern (a) and SEM image (b) of mixed product without adding PEG
After the co-precipitation stage, Fe3O4 was obtained from the mixed product with wet milling prior to the magnetic separation. The grade of iron in the mixed product without adding PEG was determined by titration to be 20.55%, while that in the Fe3O4 product was 65.58%. The XRD pattern and SEM image of the Fe3O4 product are shown in Figs. 3(a) and (b), respectively. Figures 3(a) and (b) show that a highly crystallized Fe3O4 product with a nearly spherical shape was obtained. However, a peak attributable to calcite (CaCO3) emerged in the product, as shown in Fig. 3(a). The reaction to form CaCO3 can be expressed by the following reaction equation:
Ca(OH)2+CO2→CaCO3+H2O (7)
In subsequent experiments, the PEG dispersant was added before the magnetic separation to decrease the content of calcite in the Fe3O4 product.

Fig. 3 XRD pattern (a) and SEM image (b) of Fe3O4 product without adding PEG
3.2 Adding PEG during preparation of Fe3O4
3.2.1 Influence of concentration of PEG on magnetic separation of Fe3O4
To investigate the influence of the PEG dispersant on the magnetic separation of Fe3O4, a series of experiments were performed with the PEG-6000 concentrations ranging from 0 to 8 g/L. PEG-6000 was added to the reaction solution before the co-precipitation stage, and the effects of PEG-6000 concentration on the grade and recovery rate of iron in the Fe3O4 product are shown in Fig. 4. It is revealed that both the grade and recovery rate of iron decreased rapidly with the increase of PEG-6000 concentration. When the PEG-6000 concentration fluctuated from 0 to 8 g/L, the grade and recovery rate of iron decreased from 65.58% to 57.79% and from 80.35% to 27.20%, respectively. These results indicate that the addition of the PEG dispersant during the preparation of Fe3O4 was not conducive to Fe3O4 recovery.
Fig. 4 Effects of PEG-6000 concentration on grade and recovery rate of iron in Fe3O4 product
It has been proposed that Fe3O4 is formed by air-oxidation of Fe(OH)2 via dissolution and recrystallization [21]. PEG, as a crystalline water-soluble polymer, can greatly increase the viscosity of the solution system, which restrains the hydrated iron and ferrous ions migrating from the solution to the surface of the Fe3O4 crystal nucleus. Hence, the size of the Fe3O4 particle is not too large because of the growth-restraint of the Fe3O4 microcrystals [4,22]. This inhibition is proportional to the concentration of PEG in the reaction system. The particle size distributions of the Fe3O4 products are presented in Fig. 5. As shown, the size of the Fe3O4 particles decreased gradually with an increase in the PEG-6000 concentration, and the median particle sizes of the Fe3O4 particles obtained at 0, 2, 4 and 8 g/L PEG-6000 were 2.35, 1.80, 1.66 and 1.35 μm, respectively.

Fig. 5 Size distributions of Fe3O4 products obtained at different PEG-6000 concentrations
In this work, Fe3O4 was obtained from the mixed product by magnetic separation. However, magnetic, gravity and hydrodynamic drag forces are the major forces that govern the overall behavior of mineral particles in a magnetic separator [23]. While magnetic forces attract magnetic particles, gravity and drag forces work against magnetic forces. Particle size of minerals has a pronounced influence on the magnitude of these forces. It has been reported that the hydrodynamic drag forces are dominant for fine particles, the gravitational forces are dominant for the coarse particles, and the magnetic forces are dominant for particles in the intermediate size range [24]. Thus, adding PEG dispersant during the preparation of Fe3O4 would be unfavorable for Fe3O4 recovery because of the size reduction of the Fe3O4 particles.
3.2.2 XRD patterns and FT-IR spectra
Figure 6(a) gives the XRD patterns of the Fe3O4 products obtained at different PEG-6000 concentrations. All the XRD patterns were in good agreement with the standard pattern for Fe3O4. These peaks were very intensive, indicating a high crystallinity. However, a peak attributable to CaCO3 emerged in all the XRD patterns.

Fig. 6 XRD patterns (a) and FT-IR spectra (b) of Fe3O4 products obtained at different PEG-6000 concentrations
More intense diffraction peaks indicate more extensive crystallization of the particles [25]. Figure 6(a) shows that the peak intensity of Fe3O4 decreased with the increase of PEG-6000 concentration, indicating that the obtained product contained less Fe3O4 phase with a lower degree of crystallization. These results are consistent with the results shown in Fig. 4.
The FT-IR spectra of the Fe3O4 products obtained at different PEG-6000 concentrations are shown in Fig. 6(b). All the products showed characteristic absorption bands at wave numbers around 579, 876, and 1429 cm-1. The IR bands at 577-579 cm-1 could be indexed as the Fe―O bond of Fe3O4 [26,27], whereas the IR bands at 876 cm-1 and 1429-1436 cm-1 could be assigned to the bending vibrations of the  group in the calcite structure [28,29]. These results are in agreement with the above XRD analysis.
group in the calcite structure [28,29]. These results are in agreement with the above XRD analysis.
3.3 Adding PEG during wet milling of mixed product
3.3.1 Influence of amount of PEG on magnetic separation of Fe3O4
Generally, in many industries handling fine powders, small quantities of dispersants can remarkably improve the grinding efficiency, so they have been widely used in ultrafine wet grinding processes [30]. In this work, Fe3O4 was recovered from the mixed product by wet milling prior to magnetic separation, and the PEG dispersant was added to the mixed product slurry at the beginning of the milling process. To study the effect of the amount of PEG on the magnetic separation of Fe3O4, a series of experiments were performed with the amount of PEG-200 ranging from 0% to 16% of the powder mass. Figure 7 shows the effects of the PEG-200 amount on the grade and recovery rate of iron in the Fe3O4 product. It can be seen that both the grade and recovery rate increased significantly with the amount of PEG-200 ranging from 0% to 4% and achieved their maximum (68.32% and 83.14%) at 4%. A slight downward trend was observed with a further increase of the PEG-200 amount. It is clearly demonstrated by the obtained results that adding the PEG dispersant during the wet milling of the mixed product was favorable for Fe3O4 recovery and the optimal amount of PEG-200 was 4% of the powder mass.

Fig. 7 Effects of PEG-200 amount on grade and recovery rate of iron in Fe3O4 product
In the magnetic separation process, fine particles are attracted to each another by van der Waals attractive forces to form aggregates or flocs; hence, a good dispersion is required to improve the separation efficiency [31]. However, by the addition of a small amount of a water-soluble surface-active polymer, it is possible to change the nature of the interparticle forces to be entirely repulsive. The fine particles will therefore not flocculate, but remain as discrete entities [32,33]. For small PEG dosages, the newly-created fracture surface generated in the mixed product grinding process could not adsorb enough PEG to form a stable dispersion system. However, when an excessive amount of PEG was used, it led to the polymer bridging among the particles [34], which had an adverse effect on the stability of the mixed product slurry. Therefore, the optimal PEG-200 dosage for further experiments was chosen to be 4% of the powder mass. A potential mechanism of the PEG-200 dispersant action in milling prior to the magnetic separation process is schematized in Fig. 8.
The particle size distributions of the Fe3O4 products obtained at different PEG-200 amounts are shown in Fig. 9. The size distributions of the Fe3O4 particles were self-similar, and the median particle sizes of Fe3O4 particles obtained at 0%, 2%, 4%, and 16% PEG-200 were 2.35, 2.35, 2.48 and 2.42 μm, respectively. It can be inferred that the addition of the PEG dispersant during the milling process could not change the particle size of the Fe3O4 product. This is because the shape of the powders is determined by the grinding mechanisms operating in the mill, but the grinding mechanisms cannot be altered by the presence of dispersants [35].
The XRD patterns of the Fe3O4 products obtained at different PEG-200 amounts are given in Fig. 10. All the XRD patterns had obvious diffraction peaks and matched well with the reflections of the standard Fe3O4. CaCO3 was only observed when the PEG-200 amount was 0%. This result indicates that it was not easy to obtain a pure-phase product without adding the PEG dispersant, which is consistent with the results shown in Fig. 7. Figure 11 gives the FT-IR spectra of the Fe3O4 products obtained at different PEG-200 amounts. All the products exhibited an absorption peak at around 577 cm-1, corresponding to the Fe―O bond in Fe3O4. Notably, the product obtained at 0% PEG-200 exhibited absorption peaks at 876 cm-1 and 1436 cm-1, which were attributed to the  group in CaCO3. These results are in agreement with the above XRD results.
group in CaCO3. These results are in agreement with the above XRD results.
The chemical composition of the Fe3O4 products obtained at different PEG-200 amounts was determined by titration and ICP-OES, and the results are presented in Table 3. It is shown that the product obtained by adding PEG contained higher concentrations of Fe, Ti, and Mn compared with the product obtained without adding PEG, whereas the product obtained by adding PEG contained less Ca, S and Mg, and the content of Ca was notably reduced from 2.01% to 0.81%. The results well accord with the previous results shown in Figs. 7 and 10. Therefore, it can be concluded that the effective magnetic separation of Fe3O4 can be realized by adding PEG dispersant during the wet milling of the mixed product, through a substantial reduction of the content of calcite in the Fe3O4 product.

Fig. 8 Schematic diagram of changes of potential in milling prior to magnetic separation process using PEG-200 as dispersant

Fig. 9 Size distributions of Fe3O4 products obtained at different PEG-200 amounts

Fig. 10 XRD patterns of Fe3O4 products obtained at different PEG-200 amounts

Fig. 11 FT-IR spectra of Fe3O4 products obtained at different PEG-200 amounts
3.3.2 Influence of relative molecular mass of PEG on magnetic separation of Fe3O4
To illustrate the influence of the relative molecular mass of the PEG dispersant on the magnetic separation of Fe3O4, a series of experiments were performed with the relative molecular mass of PEG ranging from 200 to 20000. Figure 12 shows the grade and recovery rate of iron in the Fe3O4 product obtained at different PEG relative molecular masses. It is shown that by increasing the relative molecular mass from 200 to 20000, the grade of iron was gradually reduced from 68.32% to 66.70% while the recovery rate of iron increased from 83.14% to 85.01%. These results indicate that the increase in the PEG relative molecular mass led to a reduction in the efficiency of the magnetic separation. REN et al [36] and YANG et al [37] reported that polymers with larger relative molecular masses can form a better conjunction with Fe3O4 particles, leading to well-enwrapped large particles, which results in the remarkable decrease of the magnetic property. Hence, the increase of the PEG relative molecular mass resulted in a poor magnetic separation probably because of a reduction in the magnetic property of the Fe3O4 particles.
Table 3 Chemical compositions of Fe3O4 products obtained at different PEG-200 amounts (mass fraction, %)


Fig. 12 Effects of PEG relative molecular mass on grade and recovery rate of iron in Fe3O4 product
3.4 Magnetic properties
The magnetic properties of the Fe3O4 products obtained without and with the PEG dispersant were measured at room temperature, and the magnetic hysteresis (M-H) loops are presented in Fig. 13. It is shown that the Fe3O4 product obtained without adding PEG exhibited the highest magnetization saturation (Ms) of 79.963×10-3 A・m2/g, a coercivity value (Hc) of 5878.44 A/m, and a remanence value (Mr) of 8.8972×10-3 A・m2/g (Fig. 13(a)). While adding PEG during the preparation of Fe3O4, the Ms and Mr for the Fe3O4 product decreased to 58.796×10-3 and 7.7917×10-3 A・m2/g, respectively, but the Hc increased to 6527.60 A/m (Fig. 13(b)). These observations are consistent with the results in Figs. 5 and 6. It is well known that the decrease in the particle size can lead to an increasing surface-to-volume ratio, which in turn increases the surface spin disorder and consequently results in a reduction in the saturation magnetization [38]. The reduction in the overall magnetic phase concentration or poor crystallization may also affect the magnetic behavior of the as-synthesized powder by reducing the saturation magnetization and slightly increasing the coercive force [39,40]. However, when adding PEG during the wet milling of the mixed product, Ms for the Fe3O4 product increased to 83.656×10-3 A・m2/g, which is not far from the theoretical value of bare Fe3O4 (~90×10-3 A・m2/g) [41], and Hc decreased to 5729.28 A/m (Fig. 13(c)). This result indicates that the obtained Fe3O4 product possessed stronger magnetic properties after adding the PEG dispersant during the wet milling of the mixed product.

Fig. 13 Magnetization versus magnetic field curves of Fe3O4 products obtained without adding PEG (a), with adding PEG-6000 (4 g/L) during preparation of Fe3O4 (b) and with adding PEG-200 (4% of powder mass) during wet milling of mixed product (c)
4 Conclusions
1) The effect of PEG dispersant on the magnetic separation of Fe3O4 synthesized from a ferrous sulfate solution via co-precipitation with calcium hydroxide as the precipitant was investigated. The results indicated that the PEG dispersant had a significant influence on Fe3O4 recovery. When PEG was added during the preparation of Fe3O4 with PEG-6000 concentration increasing from 0 to 8 g/L, the grade of iron in the Fe3O4 product decreased from 65.58% to 57.79%. This result could be explained by the size reduction of the Fe3O4 particles, which was unfavorable for the magnetic separation of Fe3O4.
2) When PEG was added during the wet milling of the mixed product and the PEG-200 amount was increased from 0% to 4% of the powder mass, the grade of iron in the Fe3O4 product increased from 65.58% to 68.32%. The addition of PEG in the milling process made it possible to change the nature of the interparticle forces to be entirely repulsive, which caused an increase in the efficiency of the subsequent magnetic separation. While the relative molecular mass of PEG at an amount of 4% of the powder mass increased from 200 to 20000, the grade of iron was reduced from 68.32% to 66.70%. Polymers with larger molecular mass can give rise to improved conjunction to Fe3O4 particles and lead to a more remarkable decrease of the magnetic property, which results in a poor magnetic separation. In addition, the obtained Fe3O4 product possessed stronger magnetic properties after adding the PEG dispersant during the wet milling of the mixed product.
References
[1] HUANG Peng-hui, JIANG Bing, ZHANG Zhi-ye, WANG Xin-long, CHEN Xiao-dong, YANG Xiu-shan, YANG Lin. Recycling sulfur and iron resources in the waste ferrous sulfate [J]. Journal of Thermal Analysis and Calorimetry, 2015, 119(3): 2229-2237.
[2] HUANG Peng-hui, DENG Shao-gang, ZHANG Zhi-ye, WANG Xin-long, CHEN Xiao-dong, YANG Xiu-shan, YANG Lin. A sustainable process to utilize ferrous sulfate waste from titanium oxide industry by reductive decomposition reaction with pyrite [J]. Thermochimica Acta, 2015, 620: 18-27.
[3]  R, VACA F. Physicochemical characterization of raw materials and co-products from the titanium dioxide industry [J]. Journal of Hazardous Materials, 2009, 166(2-3): 1429-1440.
R, VACA F. Physicochemical characterization of raw materials and co-products from the titanium dioxide industry [J]. Journal of Hazardous Materials, 2009, 166(2-3): 1429-1440.
[4] SUN Min-min, ZHU Ai-mei, ZHANG Qiu-gen, LIU Qing-lin. A facile strategy to synthesize monodisperse superparamagnetic OA-modified Fe3O4 nanoparticles with PEG assistant [J]. Journal of Magnetism and Magnetic Materials, 2014, 369: 49-54.
[5] KARAPINAR N. Magnetic separation of ferrihydrite from wastewater by magnetic seeding and high-gradient magnetic separation [J]. International Journal of Mineral Processing, 2003, 71(1-4): 45-54.
[6] LI Yi-ran, WANG Jun, ZHAO Ying, LUAN Zhao-kun. Research on magnetic seeding flocculation for arsenic removal by superconducting magnetic separation [J]. Separation and Purification Technology, 2010, 73(2): 264-270.
[7] LAKSHMANAN R, RAJARAO G K. Effective water content reduction in sewage wastewater sludge using magnetic nanoparticles [J]. Bioresource Technology, 2014, 153: 333-339.
[8] XIA Hong, WANG juan, TIAN Ye, CHEN Qi-Dai, DU Xiao-bo, ZHANG Yong-lai, HE Yan, SUN Hong-bo. Ferrofluids for fabrication of remotely controllable micro-nanomachines by two-photon polymerization [J]. Advanced Materials, 2010, 22: 3204-3207.
[9] WANG juan, XIA Hong, XU Bin-bin, NIU Li-gang, WU Dong, CHEN Qi-dai, SUN Hong-bo. Remote manipulation of micronanomachines containing magnetic nanoparticles [J]. Optics Letters, 2009, 34(5): 581-583.
[10] WEI Xin-chao, VIADERO R C Jr. Synthesis of magnetite nanoparticles with ferric iron recovered from acid mine drainage: Implications for environmental engineering [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2007, 294: 280-286.
[11] NABIYOUNI G, JULAEE M, GHANBARI D, ALIABADI P C, SAFAIE N. Room temperature synthesis and magnetic property studies of Fe3O4 nanoparticles prepared by a simple precipitation method [J]. Journal of Industrial and Engineering Chemistry, 2015, 21: 599-603.
[12] ZHANG Dong-en, TONG Zhi-wei, LI Shan-zhong, ZHANG Xiao-bo, YING Ai-ling. Fabrication and characterization of hollow Fe3O4 nanospheres in a microemulsion [J]. Materials Letters, 2008, 62(24): 4053-4055.
[13] ALBORNOZ C, JACOBO S E. Preparation of a biocompatible magnetic film from an aqueous ferrofluid [J]. Journal of Magnetism and Magnetic Materials, 2006, 305(1): 12-15.
[14] HAW C Y, MOHAMED F, CHIA C H, RADIMAN S, ZAKARIA S, HUANG N M, LIM H N. Hydrothermal synthesis of magnetite nanoparticles as MRI contrast agents [J]. Ceramics International, 2010, 36(4): 1417-1422.
[15] VALENZUELA R, FUENTES M C, PARRA C, BAEZA J, DURAN N, SHARMA S K, KNOBEL M, FREER J. Influence of stirring velocity on the synthesis of magnetite nanoparticles (Fe3O4) by the co-precipitation method [J]. Journal of Alloys and Compounds, 2009, 488(1): 227-231.
[16] YU Wang, PENG Ying-lin, ZHENG Ya-jie. Effect of purification pretreatment on the recovery of magnetite from waste ferrous sulfate [J]. International Journal of Minerals, Metallurgy, and Materials, 2016, 23(8): 891-897.
[17] YU Wang, PENG Ying-lin, ZHENG Ya-jie. Recovery of iron from waste ferrous sulphate by co-precipitation and magnetic separation [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(1): 211-219.
[18] BISWAL A, DASH B, TRIPATHY B C, SUBBAIAH T, SHIN S M, SANJAY K, MISHRA B K. Influence of alternative alkali reagents on Fe removal during recovery of Mn as electrolytic manganese dioxide (EMD) from Mn sludge [J]. Hydrometallurgy, 2013, 140: 151-162.
[19] LV Chao, XIA Hong, SHI Qing, WANG Gong, WANG Ying-shuai, CHEN Qi-dai, ZHANG Yong-lai, LIU Lian-qing, SUN Hong-bo. Sensitively humidity-driven actuator based on photopolymerizable PEG-DA films [J]. Advanced Materials Interfaces, 2017, 4(9): 1-8.
[20] YU Wang, PENG Ying-lin, ZHENG Ya-jie. Recovery of magnetite from FeSO4・7H2O waste slag by co-precipitation method with calcium hydroxide as precipitant [J]. Journal of Central South University, 2017, 24(1): 62-70.
[21] ISHIKAWA T, NAKAZAKI H, YASUKAWA A, KANDORI K, SETO M. Influences of Co2+, Cu2+ and Cr3+ ions on the formation of magnetite [J]. Corrosion Science, 1999, 41(8): 1665-1680.
[22] SHIRINOVA H, PALMA L D, SARASINI F,  J, RAMAZANOV M A, HAJIYEVA F, SANNINO D, POLICHETTI M, GALLUZZI A. Synthesis and characterization of magnetic nanocomposites for environmental remediation [J]. Chemical Engineering Transactions, 2016, 47: 103-108.
J, RAMAZANOV M A, HAJIYEVA F, SANNINO D, POLICHETTI M, GALLUZZI A. Synthesis and characterization of magnetic nanocomposites for environmental remediation [J]. Chemical Engineering Transactions, 2016, 47: 103-108.
[23] AMBASHTA R D,  M. Water purification using magnetic assistance: A review [J]. Journal of Hazardous Materials, 2010, 180(1-3): 38-49.
M. Water purification using magnetic assistance: A review [J]. Journal of Hazardous Materials, 2010, 180(1-3): 38-49.
[24] AROL A I, AYDOGAN A. Recovery enhancement of magnetite fines in magnetic separation [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2004, 232: 151-154.
[25] SHEN La-zhen, QIAO Yong-sheng, GUO Yong, TAN Jun-ru. Preparation and formation mechanism of nano-iron oxide black pigment from blast furnace flue dust [J]. Ceramics International, 2013, 39(1): 737-744.
[26] ZHANG Wen-xue, LU Bin, TANG Hui-hui, ZHAO Jing-xiang, CAI Qing-hai. Reclamation of acid pickling waste: A facile route for preparation of single-phase Fe3O4 nanoparticle [J]. Journal of Magnetism and Magnetic Materials, 2015, 381: 401-404.
[27] CHEN Ru-fen, SONG Shan-shan, WEI Yu. Study on the formation and phase transformation of 1D nanostructured Fe3O4 particles under an external magnetic field [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2012, 395: 137-144.
[28] GUNASEKARAN S, ANBALAGAN G. Spectroscopic study of phase transitions in natural calcite mineral [J]. Spectrochimica Acta: Part A, 2008, 69(4): 1246-1251.
[29] LI Tian-tian, SUI Feng-feng, LI Fu-chun, CAI Yuan-feng, JIN Zhang-dong. Effects of dry grinding on the structure and granularity of calcite and its polymorphic transformation into aragonite [J]. Powder Technology, 2014, 254: 338-343.
[30]  B, UCURUM M. An experimental study on the ultra-fine grinding of gypsum ore in a dry ball mill [J]. Powder Technology, 2016, 291: 186-192.
B, UCURUM M. An experimental study on the ultra-fine grinding of gypsum ore in a dry ball mill [J]. Powder Technology, 2016, 291: 186-192.
[31] BHAGAT R P, PATHAK P N. The effect of polymeric dispersant on magnetic separation of tungsten ore slimes [J]. International Journal of Mineral Processing, 1996, 47(3-4): 213-217.
[32] GREENWOOD R, ROWSON N, KINGMAN S, BROWN G. A new method for determining the optimum dispersant concentration in aqueous grinding [J]. Powder Technology, 2002, 123(2-3): 199-207.
[33] ZHANG Xue, HU Hui-ren. Synthesis and application of a polyacrylate dispersant on the preparation of ultrafine ground calcium carbonate in a laboratory stirred media mill [J]. Powder Technology, 2014, 266: 218-227.
[34] LEE H H, YAMAOKA S, MURAYAMA N, SHIBATA J. Dispersion of Fe3O4 suspensions using sodium dodecylbenzene sulphonate as dispersant [J]. Materials Letters, 2007, 61(18): 3974-3977.
[35] KAPUR P C, HEALY T W, SCALES P J, BOGER D V, WILSON D. Role of dispersants in kinetics and energetics of stirred ball mill grinding [J]. International Journal of Mineral Processing, 1996, 47(1-2): 141-152.
[36] REN Jie, HONG Hai-yan, REN Tian-bin, TENG Xin-rong. Preparation and characterization of magnetic PLA-PEG composite particles [J]. Materials Letters, 2005, 59(21): 2655-2658.
[37] YANG Jing-hai, ZOU Ping, YANG Li-li, CAO Jian, SUN Yun-fei, HAN Dong-lai, YANG Shuo, WANG Zhe, CHEN Gang, WANG Bing-ji, KONG Xiang-wang. A comprehensive study on the synthesis and paramagnetic properties of PEG-coated Fe3O4 nanoparticles [J]. Applied Surface Science, 2014, 303: 425-432.
[38] SHEN La-zhen, QIAO Yong-sheng, GUO Yong, MENG Shuang-ming, YANG Guo-chen, WU Mei-xia, ZHAO Jian-guo. Facile co-precipitation synthesis of shape-controlled magnetite nanoparticles [J]. Ceramics International, 2014, 40(1, Part B): 1519-1524.
[39] FAIYAS A P A , VINOD E M, JOSEPH J, GANESAN R, PANDEY R K. Dependence of pH and surfactant effect in the synthesis of magnetite (Fe3O4) nanoparticles and its properties [J]. Journal of Magnetism and Magnetic Materials, 2010, 322(4): 400-404.
[40] KUMAR R, SAKTHIVEL R, BEHURA R, MISHRA B K, DAS D. Synthesis of magnetite nanoparticles from mineral waste [J]. Journal of Alloys and Compounds, 2015, 645: 398-404.
[41] LIN Chia-chang, HO Jui-min, WU Min-shan. Continuous preparation of Fe3O4 nanoparticles using a rotating packed bed: Dependence of size and magnetic property on temperature [J]. Powder Technology, 2015, 274: 441-445.
彭映林1,余 旺2,郑雅杰2
1. 湖南城市学院 材料与化学工程学院,益阳 413000;
2. 中南大学 冶金与环境学院,长沙 410083
摘 要:以CaOH为沉淀剂通过共沉淀从绿矾溶液中回收Fe3O4,研究分散剂PEG对Fe3O4磁分离的影响。结果表明,分散剂PEG对Fe3O4的回收有显著影响。在Fe3O4制备过程中添加PEG不利于Fe3O4的回收。当PEG-6000浓度从0增加到8 g/L时,Fe3O4产品的铁品位从65.58%降至57.79%,中值粒径从2.35 μm 降至1.35 μm。然而,在混合产物的湿磨过程中添加PEG可促进随后的Fe3O4回收。当PEG-200用量从粉末质量的0%增加到4%时,Fe3O4产物中铁的品位从65.58% 升高到68.32%。当PEG-200用量为4%时,改变PEG相对分子质量从200增加到20000,Fe3O4产物中铁品位从68.32%降至66.70%。
关键词:绿矾;Fe3O4;PEG;共沉淀;磁分离;湿磨
(Edited by Bing YANG)
Foundation item: Project (2016JJ6017) supported by Hunan Provincial Natural Science Foundation of, China; Project (15C0245) supported by the Research Foundation of Education Bureau of Hunan Province, China; Project (2013A090100013) supported by the Special Foundation of Major Science & Technology Program of Guangdong Province, China
Corresponding author: Ya-jie ZHENG; Tel: +86-731-88836285; E-mail: zyj@csu.edu.cn
DOI: 10.1016/S1003-6326(18)64785-5

