Article ID: 1003-6326(2005)06-1429-04
Effect of ball milling and electrolyte on
properties of high-voltage LiNi<sub>0.5</sub>Mn<sub>1.5</sub>O4 spinel
FANG Hai-sheng(������)1, WANG Zhi-xing(��־��)1, YIN Zhou-lan(������)2,
LI Xin-hai(���º�)1, GUO Hua-jun(������)1, PENG Wen-jie(���Ľ�)1
(1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: Effect of ball milling and electrolyte on the properties of high-voltage LiNi0.5Mn1.5O4 was investigated. Ball milling has significant effect on the synthesis and property of LiNi0.5Mn1.5O4. The X-ray diffraction(XRD) patterns indicate that LiNi0.5Mn1.5O4 can��t be synthesized without ball milling even calcined at 900��. When synthesized with ball milling, LiNi0.5Mn1.5O4 almost exhibits only one plateau at around 4.7V. With the increase of ball milling time, the capacity of LiNi0.5Mn1.5O4 increases, but the cycling performance is not highly affected. The electrochemical property of LiNi0.5Mn1.5O4 highly depends on the electrolyte. The stable and high-voltage-resistant electrolyte is much beneficial to enhancement of electrochemical property of LiNi0.5Mn1.5O4, such as coulombic efficiency and cycling performance.
Key words: LiNi0.5Mn1.5O4; cathode; lithium ion batteries; ball milling; electrolyte CLC
number: TM912.9 Document code: A
1 INTRODUCTION
Recently, several research groups have reported transition-metal-substituted spinel materials (LiMxMn2-xO4, M: Cr, Co, Fe, Ni, Cu) with high-voltage plateaus above 4.5V[1-5]. Among these materials, LiNi0.5Mn1.5O4 is the most promising and attractive one because of its good cyclic property and relatively high capacity with a plateau at around 4.7V[3, 6].
Now, a variety of methods were used for preparation of LiNi0.5Mn1.5O4, such as solid-state reaction[4, 7, 8], sol-gel[4,6], co-precipitation[9], emulsion drying[10], composite carbonate process[11], molten salt[12], combustion[13] and ultrasonic spray pyrolysis method[14]. When the solid-state method is employed to synthesize transition metal oxides for the lithium-ion battery, ball milling as a useful technique is often introduced into the synthetic process in order to get homogeneous precursors[15, 16], which is very favorable for the process of solid-state reaction. The same technique is also an important aid in synthesis of LiNi0.5Mn1.5O4. In addition, LiNi0.5Mn1.5O4 is charged and discharged at high voltage, which requires the electrolyte is of good high voltage resistance. From the previous literatures[4, 7, 9, 10], severe electrolyte decomposition at high voltage and poor columbic efficiency for LiNi0.5Mn1.5O4 were often obtained, which indicated that the property of LiNi0.5Mn1.5O4 highly depends on the electrolyte.
So far, however, there is no literature about the influence of ball milling and electrolyte on the property of LiNi0.5Mn1.5O4 spinel. In the present paper, therefore, the effect of ball milling on the synthesis and property of LiNi0.5Mn1.5O4 was studied. The impact of electrolyte on the electro-chemical performance was also investigated.
2 EXPERIMENTAL
Appropriate amounts of Li2CO3, NiO and electrolytic MnO2 were initially ground in mortar and then were thoroughly mixed by ball milling for 0, 3 and 6h, respectively. Subsequently all mixed precursors were calcined at 900�� for 12h followed by an annealing treatment at 600�� for 24h in air. The powder X-ray diffraction (XRD, Rint-2000, Rigaku) measurement using CuK�� radiation was employed to identify the crystalline phase of the synthesized material.
The electrochemical characterizations were performed using CR2025 coin-type cell. For cathode fabrication, the prepared powders were mixed with 20% of carbon black and 10% of polyvinylidene fluoride in N-methyl pyrrolidinone until slurry was obtained. And then, the blended slurries were pasted onto an aluminum current collector, and the electrode was dried at 80�� for 1d in vacuum. The test cell consisted of the cathode and lithium foil anode separated by a porous polypropylene film, and 1mol/L LiPF6 in EC��EMC��DMC(1��1��1 in volume) or 1mol/L LiPF6 in PC��DMC(1��1 in volume) as electrolyte. The assembly of the cells was carried out in a dry Ar-filled glove box. The cells were charged and discharged over a voltage range of 3.5V to 4.9V versus Li/Li+ electrode at room temperature.
3 RESULTS AND DISCUSSION
3.1 Effect of ball milling on synthesis and electrochemical performance of LiNi0.5Mn1.5O4
Fig.1 shows the XRD patterns of LiNi0.5-Mn1.5O4 powders prepared under the different conditions. It��s evident that all fundamental peaks can be indexed to the cubic spinel structure. However, clear impurity (NiO) peaks denoted by asterisk are observed for LiNi0.5Mn1.5O4 powders prepared without ball milling, which indicates that the amount of Ni in the spinel phase is smaller than that in LiNi0.5Mn1.5O4, and Mn3+ reduced from Mn4+ exists in the spinel phase. With the increase of ball milling time, the impurity peaks are greatly suppressed and almost not observed for 6h, which suggests that the prepared powders with ball milling assistance are almost pure LiNi0.5Mn1.5O4 phase. Comparing the patterns, it can be concluded that the impurity in the sample without ball milling would be mainly ascribed to unreacted NiO. The above results could be explained below: although the precursors, in the case of no ball milling process, are ground in mortar, they can��t be thoroughly mixed, so the contact area of the precursors particles is limited, which makes the process of the solid-state reaction more difficult. Therefore, the solid-state reaction of the precursors without ball milling assistance is hard to proceed completely even at high temperature, and some unreacted NiO still exists in the prepared powders. When ball milling processing was employed into the synthesis route, fine and homogeneous precursors particles can be obtained, and their contact area and reactivity can be greatly enhanced, which highly facilitated the completeness of solid-state reaction. Thus, the prepared powders are almost pure LiNi0.5Mn1.5O4 phase. Here, we should also point out that faint impurity peaks were still observed in the pattern of LiNi0.5Mn1.5O4 ball-milled for 3h and 6h. However, these peaks may not be completely ascribed to unreacted NiO, especially for 6h, since they also can be attributed to LixNi1-xO caused by oxygen loss of LiNi0.5-Mn1.5O4 at high temperature[4]. Of course, it��s in all possibility that the impurity in the sample without ball milling also contain LixNi1-xO. In addition, sharp and intensive peaks of samples with ball milling process indicate well crystallization of LiNi0.5Mn1.5O4 powders. Hence, the results demonstrate that ball milling is a very effective and necessary process.
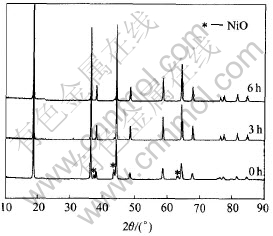
Fig.1 XRD patterns of LiNi0.5Mn1.5O4 processed with different ball milling times
1mol/L LiPF6 in EC��EMC��DMC (1��1��1 in volume) was used as electrolyte to study the effect of ball milling on the electrochemical performance of LiNi0.5Mn1.5O4. The charge��discharge curves of LiNi0.5Mn1.5O4 cycled between 3.5V and 4.9V at the rate of 1/7C are shown in Fig.2. It��s clear that the ball milling processing has significant effect on the charge��discharge profile and capacity of LiNi0.5Mn1.5O4 powders. The charge��discharge profiles of samples with different ball milling time are completely different. In the case of no ball milling process, the sample shows a large plateau at around 4.1V based on Mn3+/Mn4+ redox couple in addition to the 4.7V plateau based on Ni2+/Ni4+ redox couple[4], and delivers much smaller capacity. The existence of Mn3+ was reduced from Mn4+ caused by Ni deficiency in the spinel phase, and the existence of inactive impurity NiO diminished the amount of active material in the powders and resulted in low capacity. This result is in good agreement with the XRD pattern. On the other hand, a large 4.7V plateau with a faint 4.1V plateau is observed for the sample ball-milled for 3h, and the sample ball-milled for 6h almost exhibits only one plateau at around 4.7V. As for capacity, it increases quickly with the introduction of ball milling process, as clearly seen in Fig.2. From the XRD results, in-active impurity amount in the samples drastically decreases when the ball milling process is employed, and hence, the amount of active material in the powders increases. This is a possible explanation why the capacity is higher for the ball-milled materials. Fig.3 shows the electrochemical cycling performance of LiNi0.5Mn1.5O4 processed with different ball milling time, at the rate of 1/3C. With the increase of ball milling time, the capacity of Li-Ni0.5Mn1.5O4 is slightly enhanced which is also due to the decrease of inactive impurity amount in the prolonged ball-milled sample and reaches up to 133mA��h��g-1 in the case of 6h, but the cycling performances are similar.
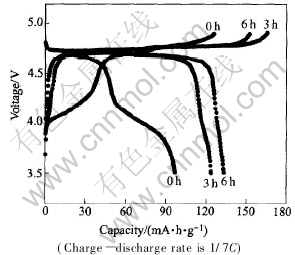
Fig.2 Charge��discharge curves of LiNi0.5Mn1.5O4 processed with different ball milling time
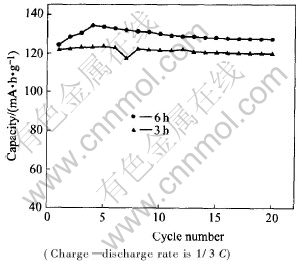
Fig.3 Electrochemical cycling performance of LiNi0.5Mn1.5O4 processed with different ball-milling time
3.2 Effect of electrolyte on electrochemical performance of LiNi0.5Mn1.5O4
As for high voltage cathode materials, electrolyte is an essential factor for the electrochemical performance of LiNi0.5Mn1.5O4. Here, two kinds of electrolyte were selected to show the effect of electrolyte on the electrochemical performance of LiNi0.5Mn1.5O4: one is 1mol/L LiPF6 in EC��EMC��DMC (1��1��1 in volume), the other is 1mol/L LiPF6 in PC��DMC (1��1 in volume). Fig.4 shows the coulombic efficiency of LiNi0.5Mn1.5O4 ball-milled for 6h in both electrolyte at the rate of 1/3C. In general, poor columbic efficiency is observed for both kinds of electrolyte, which is mainly due to the electrolyte decomposition taken place by irreversible reaction with active materials at high voltage[4]. When 1mol/L LiPF6 in EC��EMC��DMC (1��1��1 in volume) is selected as electrolyte, the coulombic efficiency is around 85% and almost keeps invariable upon cycling which indicates the electrolyte is relatively stable. On the other hand, in the case of 1mol/L LiPF6 in PC��DMC (1��1 in volume) as electrolyte, the coulombic efficiency is much poor and quickly decreases from the second cycle, which indicates that the electrolyte is very unstable at high voltage and very easy to react continuously with the active materials during the cyclic process.
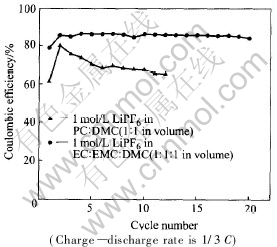
Fig.4 Comparison of coulombic efficiency of electrolytic dependence of LiNi0.5Mn1.5O4 ball-milled for 6h
Fig.5 shows comparison of cycling performance of electrolytic dependence of LiNi0.5Mn1.5O4 ball-milled for 6h, at the rate of 1/3C. When 1mol/L LiPF6 in EC��EMC��DMC (1��1��1 in volume) is selected as electrolyte, it exhibits high capacity as well as good cycling performance, whereas the capacity is reduced and faded quickly from the second cycle upon cycling, in the case of 1mol/L LiPF6 in PC��DMC (1��1 in volume) as electrolyte, as clearly seen in Fig.5. Comparing Fig.4 with Fig.5, it��s obvious that poor columbic efficiency is corresponding to poor cycling performance. The instability of electrolyte and continuous side reaction between electrolyte and active materials result in the amount decrease and structural degradation of the LiNi0.5Mn1.5O4 spinel. These effects lead to a quick capacity fade on cycling in the case of 1mol/L LiPF6 in PC��DMC (1��1 in volume) as electrolyte .
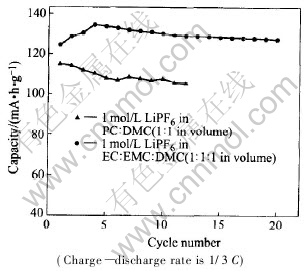
Fig.5 Comparison of cycling performance of electrolytic dependence of LiNi0.5Mn1.5O4 ball-milled for 6h
Therefore, the electrochemical performance depends strongly on the electrolyte, and the development of suitable electrolyte is a key factor for the effective use of LiNi0.5Mn1.5O4 spinel. In order to solve the problem on electrolyte decomposition at high voltage, we think that three approaches must be taken: 1) searching for suitable electrolyte that is highly resistant against oxidation at high voltage, 2) optimizing the LiNi0.5Mn1.5O4 powders, 3) modifying the LiNi0.5Mn1.5O4/electrolyte interface.
4 CONCLUSIONS
Ball milling has great effect on the synthesis and property of LiNi0.5Mn1.5O4. X-ray diffraction(XRD) patterns indicate that LiNi0.5Mn1.5O4 can��t be synthesized without ball milling even calcined at 900��. When synthesized with ball milling, LiNi0.5Mn1.5O4 almost exhibits only one plateau at 4.7V. With the increase of ball milling time, the capacity of LiNi0.5Mn1.5O4 increases, but the cycling performance is not highly affected. The electrochemical property of LiNi0.5Mn1.5O4 highly depends on the electrolyte. The stable and high-voltage-resistant electrolyte is very beneficial to enhancement of electrochemical performance of LiNi0.5Mn1.5O4.
REFERENCES
[1]Sigala C, Guyomard D, Verbaere A, et al. Positive electrode materials with high operating voltage for lith-ium batteries: LiCryMn2-yO4 (0��y��1) [J]. Solid State Ionics, 1995, 81(3-4): 167-170.
[2]Kawai H, Nagata M, Tukamoto H, et al. A novel cathode Li2CoMn3O8 for lithium ion batteries operating over 5 volts [J]. J Mater Chem, 1998, 8(4): 837-839.
[3]Ohzuku T, Takeda S, Iwanaga M. Solid-state redox potentials for Li[Me1/2Mn3/2]O4 (Me:3d-transition metal) having spinel-framework structures: a series of 5 volt materials for advanced lithium-ion batteries [J]. J Power Sources, 1999, 81-82: 90-94.
[4]Zhong Q, Bonakdarpour A, Zhang M, et al. Synthesis and electrochemistry of LiNixMn2-xO4 [J]. J Electrochem Soc, 1997, 144(1): 205-213.
[5]Ein-Eli Y, Howard W F Jr. LiCu��xCu��yMn��, ��[2-(x+y)]-O4: 5V cathode materials [J]. J Electrochem Soc, 1997, 144(8): L205-L207.
[6]Wu X, Kim S B. Improvement of electrochemical properties of LiNi0.5Mn1.5O4 spinel [J]. J Power Sources, 2002, 109(1): 53-57.
[7]Idemoto Y, Narai H, Koura N. Crystal structure and cathode performance dependence on oxygen content of LiMn1.5Ni0.5O4 as a cathode material for secondary lithium batteries [J]. J Power Sources, 2003, 119-121: 125-129.
[8]Ohzuku T, Ariyoshi K, Yamamoto S. Synthesis and characterization of Li[Ni1/2Mn3/2]O4 by two-step solid state reaction [J]. J Ceram Soc Jpn, 2002, 110(5): 501-505.
[9]Alcantara R, Jaraba M, Lavela P, et al. Optimizing preparation conditions for 5V electrode performance, and structural changes in Li1-xNi0.5Mn1.5O4 spinel [J]. Electrochim Acta, 2002, 47(11): 1829-1835.
[10]Myung S T, Komaba S, Kumagai N, et al. Nano-crystalline LiNi0.5Mn1.5O4 synthesized by emulsion drying method [J]. Electrochim Acta, 2002, 47(15): 2543-2549.
[11]Lee Y S, Sun Y K, Ota S, et al. Preparation and characterization of nano-crystalline LiNi0.5Mn1.5O4 for 5V cathode material by composite carbonate process [J]. Electrochem Commun, 2002, 4(12): 989-994.
[12]Kim J H, Myung S T, Sun Y K. Molten salt synthesis of LiNi0.5Mn1.5O4 spinel for 5V class cathode material of Li-ion secondary battery [J]. Electrochim Acta, 2004, 49(2): 219-227.
[13]Lazarraga M G, Pascual L, Gadjov H, et al. Nano-size LiNiyMn2-yO4 (0��y��0.5) spinels synthesized by a sucrose-aided combustion method: characterization and electrochemical performance [J]. J Mater Chem, 2004, 14(10): 1640-1647.
[14]Park S H, Oh S W, Myung S T, et al. Effect of synthesis condition on LiNi0.5Mn1.5O4 cathode material for prepared by ultrasonic spray pyroylsis method [J]. Solid State Ionics, 2005, 176(5-6): 481-486.
[15]Kosova N V, Uvarov N F, Devyatkina E T, et al. Mechanochemical synthesis of LiMn2O4 cathode material for lithium batteries [J]. Solid State Ionics, 2000, 135(1-4): 107-114.
[16]Jeong W T, Joo J H, Lee K S. Improvement of electrode performances of spinel LiMn2O4 prepared by mechanical alloying and subsequent firing [J]. J Power Sources, 2003, 119-121: 690-694.
(Edited by LI Xiang-qun)
Foundation item: Project supported by the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry; Project supported by the Postdoctoral Science Foundation of Central South University, China
Received date: 2005-03-24; Accepted date: 2005-07-07
Correspondence: WANG Zhi-xing, Associate Professor, PhD; Tel: +86-731-8836633; E-mail: zxwang@mail.csu.edu.cn