An investigation on electrochemical performances of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x = 0-0.2) alloy electrodes for Ni/MH battery application
来源期刊:中南大学学报(英文版)2014年第6期
论文作者:张羊换 陈莉翠 赵宸 杨泰 徐超 赵栋梁
文章页码:2125 - 2135
Key words:A2B7-type electrode alloy; adding Si; annealing treatment; structure; electrochemical performances
Abstract: The La–Mg–Ni-based A2B7-type La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) electrode alloys were prepared by casting and annealing. The influences of the additional silicon and the annealing treatment on the structure and electrochemical performances of the alloys were investigated systemically. Both of the analyses of XRD and SEM reveal that the as-cast and annealed alloys are of a multiphase structure, involving two main phases (La, Mg)2Ni7 and LaNi5 as well as one minor phase LaNi3. The addition of Si and annealing treatment bring on an evident change in the phase abundances and cell parameters of (La, Mg)2Ni7 and LaNi5 phase for the alloy without altering its phase structure. The phase abundances decrease from 74.3% (x=0) to 57.8% (x=0.2) for the (La, Mg)2Ni7 phase, and those of LaNi5 phase increase from 20.2% (x=0) to 37.3% (x=0.2). As for the electrochemical measurements, adding Si and performing annealing treatment have engendered obvious impacts. The cycle stability of the alloys is improved dramatically, being enhanced from 80.3% to 93.7% for the as-annealed (950 °C) alloys with Si content increasing from 0 to 0.2. However, the discharge capacity is reduced by adding Si, from 399.4 to 345.3 mA·h/g as the Si content increases from 0 to 0.2. Furthermore, such addition makes the electrochemical kinetic properties of the alloy electrodes first increase and then decrease. Also, it is found that the overall electrochemical properties of the alloys first augment and then fall with the annealing temperature rising.
J. Cent. South Univ. (2014) 21: 2125-2135
DOI: 10.1007/s11771-014-2162-6
ZHANG Yang-huan(张羊换)1, 2, CHEN Li-cui(陈莉翠)1, 2, ZHAO Chen(赵宸)1, 2,YANG Tai(杨泰)1, 2, XU Chao(徐超)1, 2, ZHAO Dong-liang(赵栋梁)2
1. Key Laboratory of Integrated Exploitation of Baiyun Obo Multi-Metal Resources,
Inner Mongolia University of Science and Technology, Baotou 014010, China;
2. Department of Functional Material Research, Central Iron and Steel Research Institute, Beijing 100081, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2014
Central South University Press and Springer-Verlag Berlin Heidelberg 2014
Abstract: The La–Mg–Ni-based A2B7-type La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) electrode alloys were prepared by casting and annealing. The influences of the additional silicon and the annealing treatment on the structure and electrochemical performances of the alloys were investigated systemically. Both of the analyses of XRD and SEM reveal that the as-cast and annealed alloys are of a multiphase structure, involving two main phases (La, Mg)2Ni7 and LaNi5 as well as one minor phase LaNi3. The addition of Si and annealing treatment bring on an evident change in the phase abundances and cell parameters of (La, Mg)2Ni7 and LaNi5 phase for the alloy without altering its phase structure. The phase abundances decrease from 74.3% (x=0) to 57.8% (x=0.2) for the (La, Mg)2Ni7 phase, and those of LaNi5 phase increase from 20.2% (x=0) to 37.3% (x=0.2). As for the electrochemical measurements, adding Si and performing annealing treatment have engendered obvious impacts. The cycle stability of the alloys is improved dramatically, being enhanced from 80.3% to 93.7% for the as-annealed (950 °C) alloys with Si content increasing from 0 to 0.2. However, the discharge capacity is reduced by adding Si, from 399.4 to 345.3 mA·h/g as the Si content increases from 0 to 0.2. Furthermore, such addition makes the electrochemical kinetic properties of the alloy electrodes first increase and then decrease. Also, it is found that the overall electrochemical properties of the alloys first augment and then fall with the annealing temperature rising.
Key words: A2B7-type electrode alloy; adding Si; annealing treatment; structure; electrochemical performances
1 Introduction
The Ni/MH batteries, by virtue of their major advantages, such as high-energy density, excellent power density and long cycle life, have been deemed to be a leading technology as the battery power source for electric vehicles (EVS) [1], which open up a broaden field for the development and application of the Ni/MH battery. However, further improvements of the battery are necessary for superior performances, especially discharge capacity and electrochemical hydrogen storage kinetics. A series of metal hydride electrode materials, including the rare-earth-based AB5-type alloys [2], the AB2-type Laves phase alloys, the V-based solid solution alloys and the Mg-based alloys [3-5], are selectable as potential electrode materials. In particular, the rare earth-based AB5-type alloys have been industrialized in large scale in China and Japan. And yet, no one among the above-mentioned hydrogen storage materials can meet the transport application perfectly because there is a certain limit on the alloy properties, such as the low discharge capacity of the AB5-type electrode alloy, the poor activation capability of the AB2-type Laves phase as well as V-based solid solution electrode alloys and the poor cycle stability of the Mg-based electrode alloy. Thus, the demand for new electrode materials with superior performances has become increasingly imperative. In such circumstance, La–Mg–Ni-system A2B7-type alloys have been considered to be the most promising candidates as the negative electrode materials of Ni/MH rechargeable battery due to their higher discharge capacities (380-410 mA·h/g) and lower production costs since KADIR et al [6] and KOHNO et al [7] have reported their research results. In order to commercialize this product as early as possible, researchers have dedicated enormous efforts and made a dramatic progress, for example, LIU et al [8-9] have published detailed reviews recently. However, the Chinese researchers in this area are deeply frustrated by the fact that the production of this new type alloys being the negative electrode in Ni/MH battery has not been put into application in China on account of their rather poor electrochemical cycle stability. So, how to enhance the cycle stability of the alloy without reducing its discharge capacity is still a serious challenge to these researchers.
It was convinced that the element substitution or addition is an effective method for improving the overall properties of the hydrogen storage alloys. In the case of La–Mg–Ni series hydrogen-storage alloys, the partial replacement of Ni with Co, Fe, Mn, Al and Cu [10-12] as well as La with Ce, Pr and Nd [13-15] were studied systematically. Moreover, it was also confirmed that the capacity deterioration of the La–Mg–Ni system alloy electrodes is mainly attributed to the pulverization of the alloy particles and the oxidation/corrosion of the elements Mg and La [16]. Therefore, it can be expected that adding Si can make the alloy resist corrosion and oxidation more effectively, for a dense silicon oxide film would form on the surface of the alloy electrode. To confirm this, the effects of the addition of Si and annealing treatment were systematically investigated on the structures and electrochemical hydrogen storage performances of the La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) electrode alloys, and some major experiment results were provided.
2 Experimental
The raw materials La, Ni, Co, Mg and Si have a purity of 99.8% at least. The chemical compositions of the electrode alloys were La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2). For convenience, the alloys were denoted with Si content as Si0, Si0.05, Si0.1, Si0.15 and Si0.2, respectively. The alloy ingots were prepared by using a vacuum induction furnace in a helium atmosphere under the pressure of 0.04 MPa in order to prevent element Mg from volatilizing during the melting. A part of the alloys were annealed at 900 °C, 950 °C, 1000 °C and 1050 °C for 8 h in vacuum, respectively.
The phase structure and composition of the as-cast and annealed alloys were characterized by XRD (D/max/2400). The diffraction, with the experimental parameters of 160 mA, 40 kV and at a scan rate of 10 (°)/min respectively, were performed with Cu Kα1 radiation filtered by graphite. The morphologies of the as-cast and annealed alloys were examined by SEM (QUANTA 400).
The round electrode pellets with a 15 mm diameter were prepared by cold pressing the mixture of alloy powder and carbonyl nickel powder with the mass ratio of 1:4 under the pressure of 35 MPa. After being dried for 4 h, the electrode pellets were immersed in a 6 mol/L KOH solution for 24 h in order to wet itself fully before the electrochemical measurement.
We have the electrochemical measurements be performed at 30 °C by using a tri-electrode open cell, consisting of a working electrode (the metal hydride electrode), a sintered Ni(OH)2/NiOOH counter electrode and a Hg/HgO reference electrode, which were immersed in the electrolyte of 6 mol/L KOH, and define the voltage between the negative electrode and the reference one as the discharge voltage. In every cycle, the alloy electrode was first charged with a constant current density. After resting for 15 min, it was discharged with the same current density to cut-off voltage of -0.500 V.
The electrochemical impedance spectra (EIS) and the Tafel polarization curves of the alloys were measured by electrochemical workstation (PARSTAT 2273). The fresh electrodes were fully charged and then rested for 2 h up to the open circuit potential being stable. For the EIS measurements, the frequency ranged from 10 kHz to 5 mHz at 50% depth of discharge (DOD), the amplitude of signal potentiostatic or galvanostatic measurements was 5 mV, and the number of points per decade of frequencies was 60. For the Tafel polarization curves, the potential range was from -1.2 to +1.0 V (0 Hg/HgO) with a scan rate of 5 mV/s. For the potentiostatic discharge, the test electrodes in the fully charged state were discharged at 500 mV potential steps for 5000 s on electrochemical workstation PARSTAT 2273, using the electrochemistry corrosion software CorrWare.
3 Results and discussion
3.1 Structural characteristics
The XRD patterns of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys are depicted in Fig. 1. The identification by means of ICDD reveals that all the as-cast and annealed alloys have a multiphase structure, containing two major phases (La, Mg)2Ni7 and LaNi5 as well as a residual phase LaNi3. We have noticed that adding a small amount of Si and the annealing treatment can hardly change the phase component but they incur the phase abundances change evidently. The lattice parameters listed in Tables 1 and 2 are together with the abundances of the (La, Mg)2Ni7 and LaNi5 major phases in the alloys, which were calculated by Jade 6.0 software based on the XRD data. It is found that the addition of Si makes the lattice constants and cell volumes of the two major phases enlarged visibly, which justifies the successful alloying of Si with (La, Mg)2Ni7 and LaNi5. Noticeably, adding Si makes the lattice constants of the LaNi5 phase increased much larger than those of the (La, Mg)2Ni7 phase, which means that Si prefers to form solid solution with the LaNi5 phase. Besides, it is evident that the addition of Si leads to an obvious increase in the LaNi5 phase and a decrease in the (La, Mg)2Ni7 phase. Furthermore, it can be seen from Table 2 that the annealing treatment also induces a notable increase in the lattice constants and cell volume of the major phases in the alloys. The abundance of the (La, Mg)2Ni7 phase first increases and then decreases with the rising of annealing temperature, while the LaNi5 phase shows a completely opposite tendency, first decreasing then increasing.
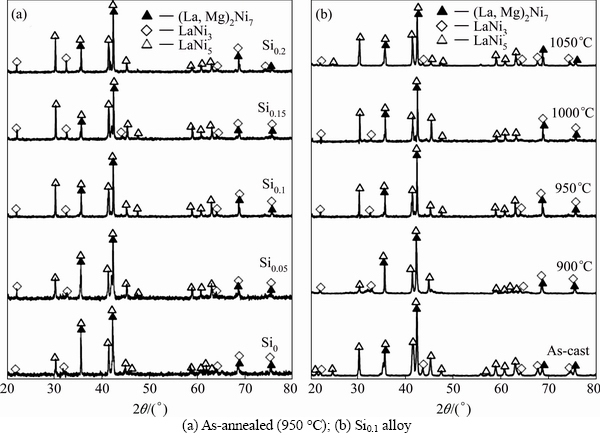
Fig. 1 XRD profiles of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys:
Table 1 Lattice parameters, abundances of (La, Mg)2Ni7 and LaNi5 major phases of as-annealed (950 °C) alloys
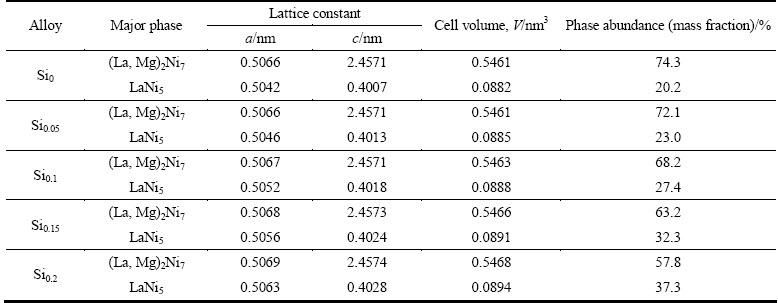
Table 2 Lattice parameters, abundances of (La, Mg)2Ni7 and LaNi5 major phases of Si0.1 alloy annealed at different temperatures
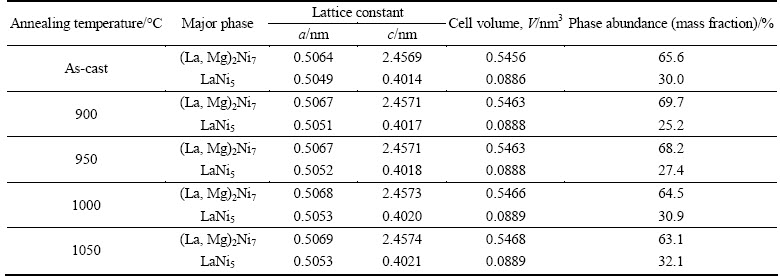
The SEM images of the as-cast and annealed Si0.1 alloy are shown in Fig. 2. Evidently, the morphologies of the as-cast and annealed Si0.1 alloys display a dendrite structure. The annealing treatment brings about an obvious homogeneity of the composition segregation in the alloys. Apparently, when the annealing temperature rises to 1050 °C, the grains become very coarse. The analysis of SEM equipped with an energy dispersive spectrometry (EDS) detects that all the experimental alloys have multiphase structure, containing (La, Mg)2Ni7 (denoted as A), LaNi5 (denoted as B) as well as LaNi3 (denoted as C) phases. Moreover, the EDS patterns exhibit that the Si concentration in the LaNi5 phase is visibly higher than that in (La, Mg)2Ni7 one, which is very well consistent with the results observed by XRD.
3.2 Electrochemical hydrogen storage performances
3.2.1 Cycle stability
The cycle stability of the electrode alloy, a decisive factor for the life of the Ni/MH battery, is symbolized by capacity retaining rate (Sn), which is defined as Sn=Cn/Cmax×100%, where Cmax is the maximum discharge capacity and Cn is the discharge capacity at the n-th charge-discharge cycle with a current density of 300 mA/g, respectively. Figure 3 shows the variations of the Sn values of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys depending on the charge-discharge cycle number. The slopes of the curves reflect the degradation rate of the discharge capacity during the charge-discharge cycles. The smaller the slope of the curve is, the better the cycle stability of the alloy will be. With respect to the degradation rate of the discharge capacity, it markedly declines with the rising of Si content, implying that the addition of Si exerts a positive impact on the cycle stability. Also, it is found from Fig. 3(b) that the degradation rate of the discharge capacity of the Si0.1 alloy first falls and then rises with the annealing temperature increasing. In order to quantitatively describe the effects of the Si content and the annealing temperature on the cycle stability of the alloy, we take the capacity retaining rate (S100) at 100th cycle as a criterion for mutual comparison and establish the relationships between the S100 value and Si content as well as the S100 value and annealing temperature, as depicted in Fig. 3. Clearly, we find that the S100 values grow with the Si content rising, being enhanced from 80.3% to 93.7% for the as-annealed (950 °C) alloys with Si content increasing from 0 to 0.2. A maximum S100 value also appears for the Si0.1 alloy, augmenting from 70.2% to 89.2% when the temperature rises to 1000 °C, and then it unexpectedly falls to 83.2% when 1050 °C.
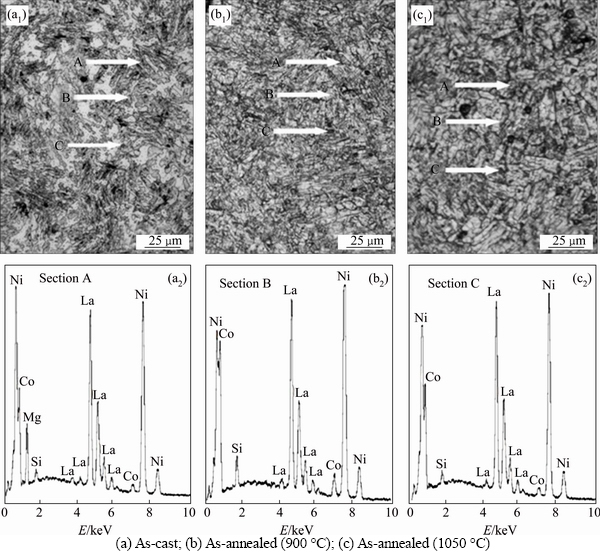
Fig. 2 SEM images of as-cast and annealed Si0.1 alloy:
Some interpretations can be provided as reasons for the cycle stability changed by adding Si and annealing treatment. The invalidation of an electrode is characterized by the decay of discharge capacity and the drop of discharge voltage [17]. It is universally believed that the degradation of the discharge capacity of the A2B7-type alloy electrode results from the forming and ever-thickening of Mg(OH)2 and La(OH)3 surface layers which hinder the hydrogen atoms from diffusing in or out, in alkaline solutions [18]. Moreover, the hydrogen storage material suffers a volume change during the charge-discharge process which aggravates the cracking and pulverizing of the alloy, and then makes the surface of the material apt to be oxidized. Generally, the positive contribution generated by adding Si to the cycle stability of the alloy is ascribed to the following aspects. Firstly, the addition of Si facilitates the forming of a compact silicon oxide layer on the surface of the alloy electrode which effectively protects it from being corroded [2, 19]. Secondly, the enlarged cell volume caused by adding Si decreases the ratios of expansion/contraction in the process of hydrogen absorption/desorption, enhancing the anti-pulverization capability. Thirdly, the increased LaNi5 phase induced by adding Si exerts a beneficial effect on the cycle stability for the cycle stability of the LaNi5 phase is greatly superior to that of the (La, Mg)2Ni7 phase. The result shown in Fig. 3 (b) indicates that the annealing treatment generates beneficial and harmful influences on the cycle stability of the alloy. The positive contribution is ascribed to the enlarged cell volumes and the more homogeneous compositional distribution created by the annealing treatment which can retard the pulverization of the alloy. And the negative impact is attributed to the coarsening of the grains as shown in Fig. 2. As considered by SAKAI et al [20], a passivation layer formed on the grain boundaries could play an effectively protective role until the alloy was pulverized below the grain size. Thus, it can be believed that the alloy with the smaller grain size has longer cycle life.
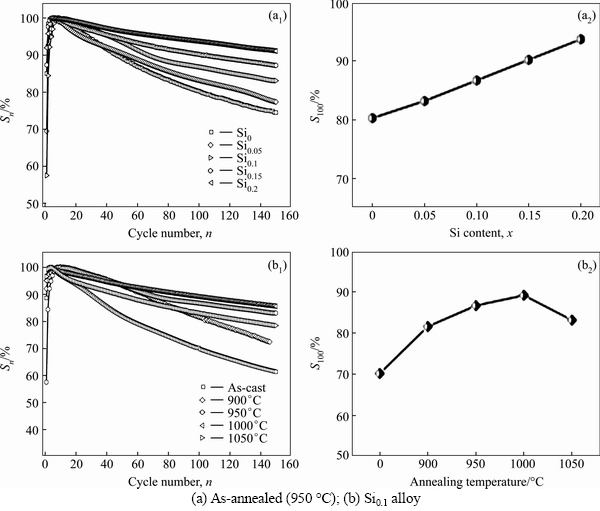
Fig. 3 Evolution of Sn values of La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys with cycle number:
3.2.2 Activation capability and discharge capacity
The activation capability of an alloy electrode is evaluated by the number of charging-discharging cycles, which is required for attaining the greatest discharge capacity through the cycle at a constant current density of 60 mA/g. The fewer the number of charging-discharging cycle is, the better the activation property will be. The variations of the discharge capacities of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys with the cycle number are illustrated in Fig. 4, indicating that all the alloys can attain their maximum discharge capacities at the first charging-discharging cycle. Whatever the addition of Si or the annealing treatment does not affect the activation capability of the alloy. It is convinced that the activation capability of an alloy electrode is associated with the change of the internal energy of the hydride system before and after hydrogen absorption. The larger the added internal energy which involves the surface energy originated from oxidative film forming on the electrode alloy surface, the larger the stain energy produced by hydrogen atom entering the interstitial of the tetrahedron or octahedron of the alloy lattice, the poorer the activation performance of the alloy [21]. The superior activation performances of the as-cast and annealed alloys are mainly ascribed to their multiphase structures because the phase boundary can decrease the lattice distortion and the strain energy formed in the process of hydrogen absorption and the phase boundary can also provide good diffusion tunnels for hydrogen atoms, improving the activation performance of the alloy. Additionally, the enlarged cell volume by adding Si decreases the expansion/contraction ratio of the alloy in the process of the hydrogen absorption/desorption, reducing the strain energy and the diffusion activation energy.
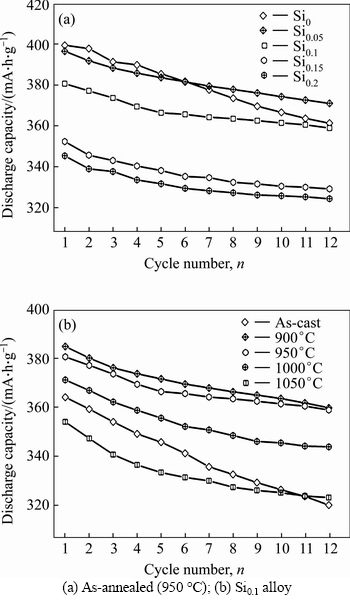
Fig. 4 Evolution of discharge capacity of La0.8Mg0.2Ni3.3- Co0.2Six (x=0-0.2) alloys with cycle number:
Figure 5 depicts the evolutions of the discharge capacity of the as-cast and annealed La0.8Mg0.2Ni3.3- Co0.2Six (x=0-0.2) alloys with the Si content and the annealing temperature. Obviously, the addition of Si leads to a visible decline in the discharge capacity which reduces from 399.4 to 345.3 mA·h/g as the Si content increases from 0 to 0.2. Also, it is found that the Si0.1 alloy yields a maximum discharge capacity with the annealing temperature rising. When the annealing temperature rises from 0 (as-cast was defined as the annealing temperature of 0 °C) to 1050 °C, the discharge capacity of the Si0.1 alloy grows from 364.1 (0 °C) to 384.8 mA·h/g (900 °C) and then declines to 342.5 mA·h/g (1050 °C). Thus, it is considered that two reasons are basically responsible for the decreased discharge capacity resulting from adding Si. On one hand, the reduction of the (La, Mg)2Ni7 phase caused by adding Si is detrimental to the discharge capacity since the (La, Mg)2Ni7 phase possesses much higher electrochemical capacity than that of the LaNi5 phase. On the other hand, the addition of Si is universally convinced to be harmful to the discharge capacity of the LaNi5 phase [19, 22-23]. The reason why Si0.1 alloy can have the maximum discharge capacity is related to the structural changes by annealing treatment. When the annealing temperature is 900 °C, the abundance of (La, Mg)2Ni7 phase increases and the cell volume becomes enlarged, which can facilitate to improve the discharge capacity. However, as the annealing temperature continue to rise, the abundance of (La, Mg)2Ni7 phase actually dropped, making the Si0.1 alloy have a maximum discharge capacity.
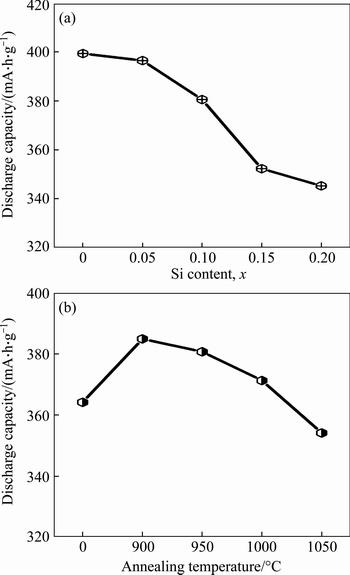
Fig. 5 Evolutions of discharge capacity of as-cast and annealed alloys with Si content (a) and annealing temperature (b)
3.2.3 Electrochemical hydrogen storage kinetics
The electrochemical hydrogen storage kinetics of an alloy electrode is evaluated by means of high rate discharge ability (HRD) which is defined as follows: RHRD=Ci,max/C60,max×100%, where Ci,max and C60,max are the maximum discharge capacities of the alloy electrode charged-discharged at the current densities of i and 60 mA/g, respectively. The variations of HRD values of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x= 0-0.2) alloys with the discharge current density are shown in Fig. 6. It is found that both the addition of Si and the annealing treatment engender an evident impact on the RHRD of the alloy. In order to describe these influences more intuitively, we have established a relationship between the RHRD and Si content as well as another relationship between the RHRD and annealing temperature for a fixed current density of 300 mA/g respectively, as presented in Fig. 6. Apparently, the as-annealed (950 °C) alloys attain a maximum HRD value of 94.6% when Si content x = 0.05, and the Si0.1 alloy has a maximum HRD value of 95.2% when annealed at 900 °C.
It has come to light that the electrochemical hydrogen storage kinetics of the alloy electrode is determined by the charge-transfer rate on the surface of an alloy electrode and the hydrogen diffusion capability in the alloy bulk [24]. Thereby, it is extremely necessary to investigate the hydrogen diffusion coefficient and the charge-transfer rate. As interpreted and modeled by KURIYAMA et al [25], the charge-transfer ability on the surface of an alloy electrode can be qualitatively evaluated by its electrochemical impedance spectrum (EIS), which of the as-cast and annealed La0.8Mg0.2Ni3.3- Co0.2Six (x=0-0.2) alloys are demonstrated in Fig. 7. Clearly, each EIS spectrum contains two distorted capacitive loops at high and middle frequencies as well as an almost straight line at low frequency. Among of them, as semicircle in the high frequency region corresponds to the contact resistance between the alloy powder and the conductive material being small, the semicircle in the middle frequency region equates to the charge-transfer resistance on the alloy surface and the line in low frequency related to the atomic hydrogen diffusion in the alloy become larger. Hence, it seems to be self-evident that the larger the radius of the semicircle in the middle frequency region is, the higher the charge-transfer resistance of the alloy electrode will be. It is found that the radii of the large semicircles of the as-cast and annealed alloys in the middle frequency first shrink and then expand with the rising of Si content and the annealing temperature.
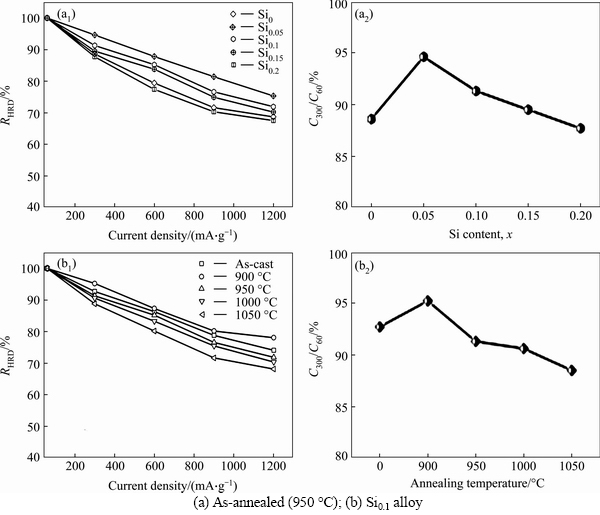
Fig. 6 Evolution of HRD values of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys with discharge current density:
The diffusion coefficients of hydrogen atoms can be obtained by measuring the semilogarithmic curves of anodic current versus working duration of an alloy, which of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys are presented in Fig. 8. Based on the model built by WHITE et al [26], the diffusion coefficient of the hydrogen atoms in the bulk of the composite could be calculated through the slope of the linear region of the corresponding plots by the following formulae:
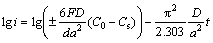 (1)
(1)
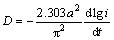 (2)
(2)
where i is the diffusion current density (A·g–1), D is the hydrogen diffusion coefficient (cm2·s–1), F is the Faraday constant, C0 is the initial hydrogen concentration in the bulk of the alloy (mol·cm–3), Cs is the hydrogen concentration on the surface of the alloy particles (mol·cm–3), a is the alloy particle radius (cm), d is the density of the hydrogen storage alloy (g·cm–3), and t is the discharge time (s), respectively. In Eq. (2), dlgi/dt is the slope of the linear region in Fig. 8. Thus, D values can be easily obtained, and the variation of D values of the as-cast and annealed alloys depending on the Si content and the annealing temperature are also provided in Fig. 8. Clearly, the D values of the alloys first augment and then decline with the rising of the Si content and the annealing temperature, which conforms well to the results in Fig. 6.
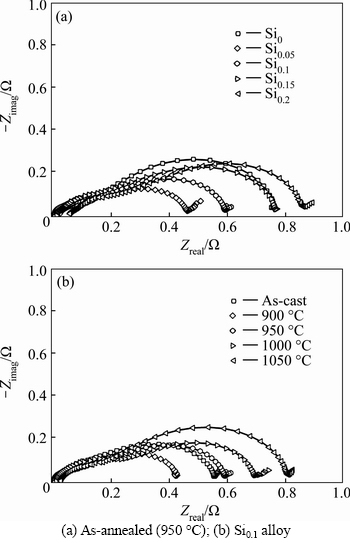
Fig. 7 Electrochemical impedance spectra (EIS) of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six(x=0-0.2) alloys:
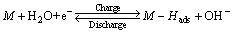
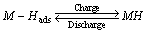
Limiting current density (IL), another important electrochemical kinetic characteristic of an alloy electrode, can be obtained by measuring the Tafel polarization curve, which of the as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six(x=0-0.2) alloys are demonstrated in Fig. 9. It is found that, whatever the case is, a clear point of inflection appears in each anodic polarization curve, implying the existent of critical value which is termed as limiting current density (IL). It reflects that an oxidation reaction took place on the surface of the alloy electrode, and the generated oxidation product hinders hydrogen atoms further penetrating [27]. Thereby, the limiting current density (IL) can be viewed as a critical current density of passivation, which is mainly determined by the solid state diffusion of hydrogen in metal-hydride electrode [24]. In order to illustrate the influences of the Si content and the annealing temperature on the limiting current density (IL), we exhibit the variations of the IL values of the alloys depending on the Si content and the annealing temperature, as presented in Fig. 9. It is clear that the IL values of the alloys first mount up and then go down with the growing of the Si content and the annealing temperature. To sum up, based on the above-mentioned results, we can conclude that the high rate discharge ability (HRD) of the experimental alloys is dominated by the charge-transfer rate and the hydrogen diffusion capability jointly. Undoubtedly, a clear and full understanding of the mechanism of electrochemical hydriding/dehydriding is the premiss to know the influences of adding Si and the annealing treatment on the electrochemical hydrogen storage kinetics. The electrochemical hydriding/dehydriding reaction taking place at the hydrogen storage electrode in an alkaline solution during charging and discharging process can be summarized as
 (3)
(3)
 (4)
(4)
where M is the hydrogen storage alloy, M-Hads refers to the hydrogen on the surface of the metal hydride and MH refers to the hydrogen in its bulk. Metal hydrides compose of hydrogen atoms and metal atoms which constitute a host lattice. Metal and hydrogen usually form two different kinds of hydrides: one is α-phase in which only some hydrogen is absorbed, and another is β-phase where hydride is fully formed. We can see from the above two equations that when the alloy electrode is charging in KOH solution, hydrogen atoms which are obtained by electrolyzing H2O diffusing from the interface between alloy and electrolyte into bulk alloy, and then store themselves in the metallic lattice in the form of hydride. In the process of discharging, the hydrogen stored in the bulk alloy diffuses toward the alloy surface where it is reacted. The formation of metal hydrides consists of several mechanistic steps and the hydrogen absorption rate depends on the slowest one. As interpreted by KLEPERIS [28], the charge-transfer step is controlled by both crystallographic and electronic structures. The compositional variation on the alloy surface results in an evident impact on the valence electron configurations, which dominate the charge-transfer reaction, namely the hydrogen dissociation reaction [29]. The hydrogen atoms attached to the grain surface of the alloy electrode will have two possible directions at last, becoming hydrogen molecule or diffusing into the bulk alloy and being formed metal hydrides. That is to say, the diffusion rate of hydrogen atom within the surface layer of alloy is a vital factor determining the utilization of charging current, which is just the ratio of the diffusion current to the imposed current. In terms of hydrogen diffusion, it is approved that the diffusion coefficient of hydrogen atoms in the metallic lattices is dominated by the interaction between the metal and hydrogen as well as the structure of the alloy [30]. As elaborated by CUI and LUO [31], an increase in the lattice constants and cell volume facilitates to decrease the diffusion activation energy of hydrogen atoms, thus enhancing hydrogen diffusion. Also, the diffusion ability of hydrogen atoms is very sensitive to grain size [32] because grain boundaries can provide many sites with low diffusing activation energy, accelerating the diffusion rate of hydrogen atom in the alloy bulk. The measurements of the HRD, EIS, limiting current density (IL) and hydrogen diffusion coefficient (D) all exhibit that the electrochemical kinetics of the alloys first augment and then fall with the rising of the Si content and the annealing temperature, implying that both the addition of Si and the annealing treatment engender beneficial and harmful impacts on the electrode hydrogen storage kinetics. Here, we will first explain the effects brought about by adding Si. The positive contribution behaves in two ways, the enlarged cell volume incurred by adding Si reduces diffusion activation energy of hydrogen atoms and the increased LaNi5 phase by adding Si improves the electrocatalytic activity of the alloy electrodes dramatically. The adverse action results from the compact silicon oxide layer created by adding Si, which not only severely weakens the charge-transfer rate on the alloy surface but also hinders the hydrogen diffusion from inner of the bulk to the surface, consequently impairing the electrochemical kinetic property. As for the effect contributed by annealing treatment, it also has both the positive and the negative sides, improving the alloy’s electrochemical kinetic due to the increased cell volume and coarsening the alloy grains at the same time.
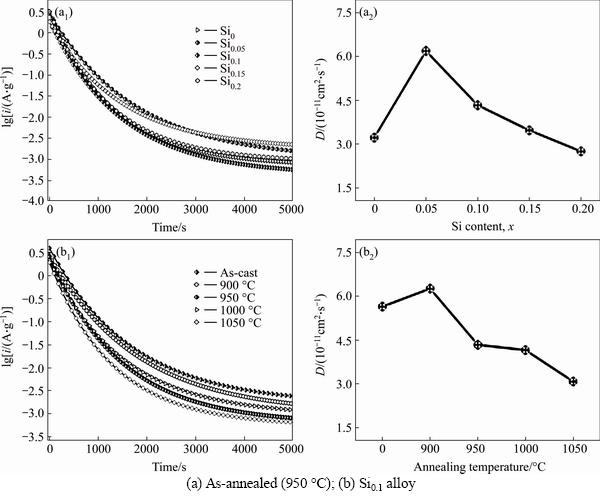
Fig. 8 Semilogarithmic curves of anodic current vs time responses of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x = 0-0.2) alloys:
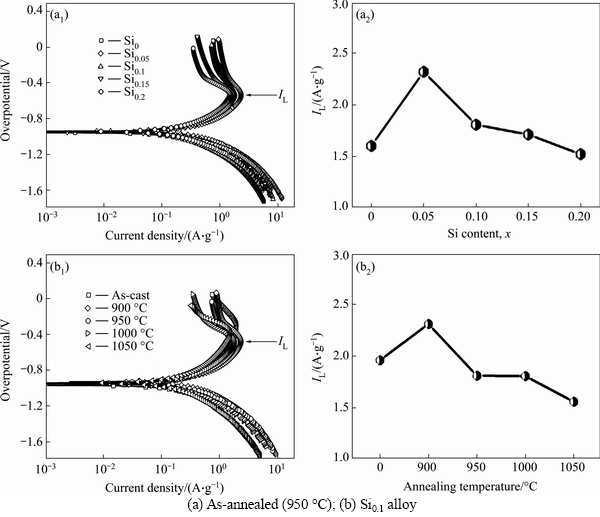
Fig. 9 Tafel polarization curves of as-cast and annealed La0.8Mg0.2Ni3.3Co0.2Six (x=0-0.2) alloys:
4 Conclusions
1) The addition of Si gives rise to an increase in the LaNi5 phase and a decrease in the (La, Mg)2Ni7 phase without changing the major phase structures of the alloys and also incurs a visible increment in the lattice constants and cell volume of the alloys. Such element addition reduces the discharge capacities of the as-cast and annealed alloys, but it enhances their cycle stabilities considerably. The HRD values of the as-cast and annealed alloys first increase then decrease with the rising of Si content, to which a rather similar trend is obtained by measuring the hydrogen diffusion coefficient (D), Tafel polarization curves and electrochemical impedance spectra (EIS).
2) The annealing treatment makes the lattice constants and cell volumes of the alloys visibly enlarged and have the alloy grains coarsened obviously at the same time. Furthermore, the amount of the (La, Ni)2Ni7 phase first grows and then declines with the annealing temperature increasing, but it is a completely opposite change for the LaNi5 phase. The electrochemical performances of the alloys, including cycle stability, discharge capacity and high rate discharge ability (HRD), first increase and then decrease with the rising of the annealing temperature.
References
[1] SAKINTUNA B, LAMARI-DARKRIM F, HIRSCHER M. Metal hydride materials for solid hydrogen storage: A review [J]. International Journal of Hydrogen Energy, 2007, 32(9): 1121-1140.
[2] WILLEMS J J G, BUSCHOW K H J. From permanent magnets to rechargeable hydride electrodes [J]. Journal of the Less-Common Metals, 1987, 129(15): 13-30.
[3] OVSHINSKY S R, FETCENKO M A, ROSS J. A nickel metal hydride battery for electric vehicles [J]. Science, 1993, 260(5105): 176-181.
[4] TSUKAHARA M, KAMIYA T, TAKAHASHI K, KAWABATA A, SAKURAI S, SHI J, TAKESHITA H T, KURIYAMA N, SAKAI T. Hydrogen storage and electrode properties of V-based solid solution type alloys prepared by a thermic process [J]. Journal of The Electrochemical Society, 2000, 147(8): 2941-2944.
[5] SUN D L, ENOKI H, GINGL F, AKIBA E. New approach for synthesizing Mg-based alloys [J]. Journal of Alloys and Compounds, 1999, 285(1/2): 279-283.
[6] KADIR K, SAKAI T, UEHARA I. Synthesis and structure determination of a new series of hydrogen storage alloys; RMg2Ni9 (R=La, Ce, Pr, Nd, Sm and Gd) built from MgNi2 laves-type layers alternating with AB5 layers [J]. Journal of Alloys and Compounds, 1997, 257(1/2): 115-121.
[7] KOHNO T, YOSHIDA H, KAWASHIMA F, INABA T, SAKAI I, YAMAMOTO M, KANDA M. Hydrogen storage properties of new ternary system alloys: La2MgNi9, La5Mg2Ni23, La3MgNi14 [J]. Journal of Alloys and Compounds, 2000, 311(2): L5-L7.
[8] LIU Y F, CAO Y H, HUANG L, GAO M X, PAN H G. Rare earth–Mg–Ni-based hydrogen storage alloys as negative electrode materials for Ni/MH batteries [J]. Journal of Alloys and Compounds, 2011, 509(3): 675-686.
[9] LIU Y F, PAN H G, GAO M X, WANG Q D. Advanced hydrogen storage alloys for Ni/MH rechargeable batteries [J]. Journal of Materials Chemistry, 2011, 21(13): 4743-4755.
[10] ZHANG X B, SUN D Z, YIN W Y, CHAI Y J, ZHAO M S. Effect of Mn content on the structure and electrochemical characteristics of La0.7Mg0.3Ni2.975-xCo0.525Mnx(x=0-0.4) hydrogen storage alloys [J]. Electrochimica Acta, 2005, 50(14): 2911-2918.
[11] LIU Y F, PAN H G, GAO M X, LI R, LEI Y Q. Effect of Co content on the structural and electrochemical properties of the La0.7Mg0.3Ni3.4-xMn0.1Cox hydride alloys: II. Electrochemical properties [J]. Journal of Alloys and Compounds, 2004, 376(1/2): 304-313.
[12] ZHANG Y H, LI B W, REN H P, CAI Y, DONG X P, WANG X L. Cycle stabilities of the La0.7Mg0.3Ni2.55-xCo0.45Mx (M=Fe, Mn, Al; x=0, 0.1) electrode alloys prepared by casting and rapid quenching [J]. Journal of Alloys and Compounds, 2008, 458(1/2): 340-345.
[13] ZHANG Y H, HOU Z H, LI B W, REN H P, CAI Y, ZHAO D L. Electrochemical hydrogen storage characteristics of as-cast and annealed La0.8-xNdxMg0.2Ni3.15Co0.2Al0.1Si0.05 (x=0-0.4) alloys [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(5): 1403-1412.
[14] ZHANG Y H, HOU Z H, YANG T, ZHANG G F, LI X, ZHAO D L. Structure and electrochemical hydrogen storage characteristics of La0.8-xPrxMg0.2Ni3.15Co0.2Al0.1Si0.05(x=0-0.4) electrode alloys [J]. Journal of Central South University of Technology, 2013, 20(5): 1142-1150.
[15] SHEN X Q, CHEN Y G, TAO M D, WU C L, DENG G, KANG Z Z. The structure and 233 K electrochemical properties of La0.8-xNdxMg0.2Ni3.1Co0.25Al0.15 (x = 0.0–0.4) hydrogen storage alloys [J]. International Journal of Hydrogen Energy, 2009, 34(6): 2661-2669.
[16] LIU Y F, PAN H G, YUE Y J, WU X F, CHEN N, LEI Y Q. Cycling durability and degradation behavior of La–Mg–Ni–Co-type metal hydride electrodes [J]. Journal of Alloys and Compounds, 2005, 395(1/2): 291-299.
[17] ZHANG Y H, LI B W, REN H P, CAI Y, DONG X P, Wang X L. Investigation on structures and electrochemical performances of the as-cast and -quenched La0.7Mg0.3Co0.45Ni2.55-xFex (x=0-0.4) electrode alloys [J]. International Journal of Hydrogen Energy, 2007, 32(18): 4627-4634.
[18] DORNHEIM M, DOPPIU S, BARKHORDARIAN G, BOESENBERG U, KLASSEN T, GUTFLEISCH O, BORMANN R. Hydrogen storage in magnesium-based hydrides and hydride composites [J]. Scripta Materialia, 2007, 56(10): 841-846.
[19] WILLEMS J J G. Metal hydride electrodes stability of LaNi5-related compounds [J]. Philips Journal of Research, 1984, 39(1): 1-94.
[20] SAKAI T, HAZAMA T, MIYAMURA H, KURIYAMA N, KATO A, ISHIKAWA H. Rare-earth-based alloy electrodes for a nickel-metal hydride battery [J]. Journal of the Less-Common Metals, 1991, 172-174(3): 1175-1184.
[21] SIMICIC M V, ZDUJIC M, DIMITRIJEVIC R, NIKOLIC- BUJANOVIC L, POPOVIC N H. Hydrogen absorption and electrochemical properties of Mg2Ni-type alloys synthesized by mechanical alloying [J]. Journal of Power Sources, 2006, 158(1): 730-734.
[22] MELI F, ZUETTEL A, SCHLAPBACH L. Surface and bulk properties of LaNi5-xSix alloys from the viewpoint of battery applications [J]. Journal of Alloys and Compounds, 1992, 190(1): 17-24.
[23] SAKAI T, OGURO K, MIYAMURA H, KURIYAMA N, KATO A, ISHIKAWA H, IWAKURA C. Some factors affecting the cycle lives of LaNi5-based alloy electrodes of hydrogen batteries [J]. Journal of the Less-Common Metals, 1990, 161(2): 193-202.
[24] RATNAKUMAR B V, WITHAM C, BOWMAN R C, HIGHTOWER A, FULTZ B. Electrochemical studies on LaNi5-xSnx metal hydride alloys [J]. Journal of The Electrochemical Society, 1996, 143(8): 2578-2584.
[25] KURIYAMA N, SAKAI T, MIYAMURA H, UEHARA I, ISHIKAWA H, IWASAKI T. Electrochemical impedance and deterioration behavior of metal hydride electrodes [J]. Journal of Alloys and Compounds, 1993, 202(1/2): 183-197.
[26] ZHENG G, POPOV B N, WHITE R E. Electrochemical determination of the diffusion coefficient of hydrogen through a LaNi4.25Al0.75 electrode in alkaline aqueous solution [J]. Journal of The Electrochemical Society, 1995, 142(8): 2695-2698.
[27] ZHAO X Y, DING Y, MA L Q, WANG L Y, YANG M, SHEN X D. Electrochemical properties of MmNi3.8Co0.75Mn0.4Al0.2 hydrogen storage alloy modified with nanocrystalline nickel [J]. International Journal of Hydrogen Energy, 2008, 33(22): 6727-6733.
[28] KLEPERIS J, WOJCIK G, CZERWINSKI A, SKOWRONSKI J, KOPCZYK M, BELTOWSKA-BRZEZINSKA M. Electrochemical behavior of metal hydrides [J]. Journal of Solid State Electrochemistry, 2001, 5(4): 229-249.
[29] NOBUHARA K, KASAI H, DINO W A, NAKANISHI H. H2dissociative adsorption on Mg, Ti, Ni, Pd and La surfaces [J]. Surface Science, 2004, 566-568(Part 2): 703-707.
[30] FENG F, NORTHWOOD D O. Hydrogen diffusion in the anode of Ni/MH secondary batteries [J]. Journal of Power Sources, 2004, 136(2): 346-350.
[31] CUI N, LUO J L. Electrochemical study of hydrogen diffusion behavior in Mg2Ni-type hydrogen storage alloy electrodes [J]. International Journal of Hydrogen Energy, 1999, 24(1): 37-42.
[32] XIE D H, LI P, ZENG C X, SUN J W, QU X H. Effect of substitution of Nd for Mg on the hydrogen storage properties of Mg2Ni alloy [J]. Journal of Alloys and Compounds, 2009, 478(1/2): 96-102.
(Edited by DENG Lü-xiang)
Foundation item: Projects(51371094, 51161015) supported by the National Natural Science Foundations of China; Project(2011ZD10) supported by Natural Science Foundation of Inner Mongolia, China
Received date: 2013-01-17; Accepted date: 2013-08-20
Corresponding author: ZHANG Yang-huan, Professor, PhD; Tel: +86-10-62183115; E-mail: zhangyh59@sina.com

