
ARTICLE
J. Cent. South Univ. (2019) 26: 383-392
DOI: https://doi.org/10.1007/s11771-019-4010-1

Alkalinity stabilization behavior of bauxite residue:Ca-driving regulation characteristics of gypsum
LI Xiao-fei(李晓飞)1, GUO Yin(郭颖)1, ZHU Feng(朱锋)1, HUANG Long-bin(黄隆斌)2,
William HARTLEY3, LI Yi-wei(李义伟)1, KONG Xiang-feng(孔祥峰)1, XUE Sheng-guo(薛生国)1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. The University of Queensland, SMI, Centre for Mined Land Rehabilitation, Brisbane, Qld 4072, Australia;
3. Crop and Environment Sciences Department, Harper Adams University, Newport, Shropshire,TF10 8NB, United Kingdom
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: Alkaline anions, include CO32–, HCO3–, Al(OH)4–, OH–, continuously released from bauxite residue (BR), will cause a potential disastrous impact on surrounding environment. The composition variation of alkaline anions, alkaline phase transformation pathway, and micro-morphological transition characteristics during the gypsum addition were investigated in an attempt to understand alkalinity stabilization behavior. Results demonstrated that alkaline anions stabilization degree in leachates can reach approximately 96.29%, whilst pH and alkalinity were reduced from 10.47 to 8.15, 47.39 mmol/L to 2 mmol/L, respectively. During the alkalinity stabilization, chemical regulation behavior plays significant role in driving the co-precipitation reaction among the critical alkaline anions (CO32–, HCO3–, Al(OH)4–, OH–), with calcium carbonate (CaCO3)) being the most prevalent among the transformed alkaline phases. In addition, XRD and SEM-EDX analyses of the solid phase revealed that physical immobilization behavior would also influence the stability of soluble alkali and chemical bonded alkali due to released Ca2+ from gypsum which aggregated the clay particles and stabilized them into coarse particles with a blocky structure. These findings will be beneficial for effectively regulating strong alkalinity of BR.
Key words: bauxite residue; gypsum; alkalinity stabilization; phase transformation; Ca-driven regulation
Cite this article as: LI Xiao-fei, GUO Yin, ZHU Feng, HUANG Long-bin, William HARTLEY, LI Yi-wei, KONG Xiang-feng, XUE Sheng-guo. Alkalinity stabilization behavior of bauxite residue: Ca-driving regulation characteristics of gypsum [J]. Journal of Central South University, 2019, 26(2): 383–392. DOI: https://doi.org/10.1007/s11771-019- 4010-1.
1 Introduction
Bauxite residue (BR) was identified as a hazardous alkaline solid waste, generated from the alumina extraction of bauxite ores using Bayer process [1–3]. On a global scale, annual BR production is estimated to be approximately 200 million tons, and the residue inventory reaches over 4.6 billion tons in 2018 [4–6]. The majority of BR is still stored on land and has the potential to cause serious environmental concerns due to its high pH (>13), alkalinity (>20000 mg/L as equivalent CaCO3) and sodicity (>50 g/kg) [7, 8]. For example, large amounts of residues stored in bauxite residue disposal area (BRDA) may cause dam collapse, whilst dissolution of soluble alkali from the residues will also create potential environmental risk [9–11]. In the past decade, dam collapse incidents occurred frequently, among which the most influential accident, e.g. approximately 1 million tons of BR suspension released from the Ajka BRDA, Hungary, in October 2010, leached large quantities of soluble alkaline substances, seriously destroyed the ecological environment and endangered the health of residents [2, 12]. Furthermore, the large amounts of soluble alkali formed on the surface of BRDAs by alkaline-salts migration may threaten the surrounding ecology [13, 14]. The underlying causes of these phenomena are the high solubility and mobility of soluble alkali (such as carbonate, bicarbonate, aluminate, hydroxide) in BR.
Soluble alkali regulation of BR is therefore critical [15, 16]. Methodologies for removing or stabilizing soluble alkali to promote safety management of BRDAs have been proposed, such as multi-stages washing, seawater precipitation, carbonation conversion, waste acid interaction, microbially-driven and gypsum amelioration [17–23]. However, few practices are successful due to the potential secondary contamination and technical costs that restrict the large-scale application of these methods. Furthermore, sodium, the common dealkalization indicator, plays an important role in the above researches. However, it maybe unfit to appropriately describe the transformation process of alkaline anions in BR due to its complex existing forms [15]; moreover, composition variation of alkaline anions and its alkalinity regulation reactions cannot be systematically analyzed with the changes of Na content. Although gypsum amelioration appears to be promising for large-scale remediation of BR among these methods, which significantly reduces pH and Al content, improves physical properties, and has been used to amend alkaline residue for grassland restoration [12, 19, 24]. Unfortunately, the composition distribution of alkaline anions may also be the main factor that restricts effective amelioration [8]. LI et al [16] demonstrated that CO32– was the dominant anions and accounts for 75% of total alkalinity by the leaching experiments. However, limited information on the quantified analysis of main alkaline anions (CO32–, HCO3–, Al(OH)4–, OH–) variation, originated from soluble alkali and chemical bonded alkali, following gypsum stabilization exists. Furthermore, less attention has been paid to the transformation behaviors of these alkaline anions and their physical immobilization processes during gypsum stabilization in solid–liquid phases.
The objectives of this paper were to 1) investigate the composition distribution and concentration variation of alkaline anions during gypsum stabilization, 2) assess alkaline anions transformation pathway and the detailed stabilization reactions based on the optimized gypsum stabilization condition, and 3) evaluate the effect of micro-morphological transition during gypsum-stabilization in BR on component variation of alkaline anions and the alkaline stability of chemical bonded alkali and soluble alkali. These findings will supplement the research deficiencies in alkalinity regulation mechanisms of BR by gypsum, and is beneficial to the research in ecological remediation for BRDAs.
2 Materials and methods
2.1 Sample collection and treatment
Bauxite residue was collected from Guangxi Branch Alumina Corporation of China Limited in 2015 (Lat 23°18'28.68'' N, Long 107°31'8.15'' E). The residues were sampled with an auger to a depth of 20 cm and stored in polyethylene bags before being returned to the laboratory. Samples were placed in plastic trays, and subsequently air-dried at room temperature for one week, then thoroughly homogenized and dried at 70 °C for 60 h. Finally, the sample was disaggregated using a mortar and pestle, and sieved (100 mesh) for use in the present study.
2.2 Gypsum stabilization experiments
The gypsum stabilization experiment was conducted based on the optimal conditions of alkaline anions leaching, ratio of Liquid to solid 2 mL/g, 23 h, 30 °C [16], to investigate the effect of gypsum addition, stabilization time, stirring strength on the alkalinity regulation of BR. Gypsum addition was 0 wt%, 0.4 wt%, 0.8 wt%, 1.2 wt%, 1.6 wt%, 2.0 wt%, 2.4 wt% on a dry BR basis; Time was controlled to 0.5, 1, 2, 4, 8, 12, 16, 24,48 h; Stirring strength was controlled at 60, 80, 100, 120, 240 r/min.
BR (20 g) and gypsum (CaSO4·H2O) powder were put in a beaker, then mixed with deionized water for 15 s by hand-stirring vigorously. And the beaker was subsequently sealed with the membrane to prevent air flow and water evaporation. The gypsum stabilization processes were conducted in a constant temperature water bath. Supernatants were separated through vacuum suction filters and then stored in volumetric flasks prior to analysis. Meanwhile, the residual solids were subsequently oven dried at 70 °C for 60 h and stored in hermetically sealed plastic bags for the solid phase analysis.
2.3 Analytical methods
2.3.1 Leachate analysis
The pH was immediately determined in all experiments through a PHS-3C and the concentration of OH– was obtained from pH data. CO32– and HCO3– were analyzed by the titration methods using 0.005 mol/L H2SO4 [14]. Firstly, a 5 mL leachate was diluted with 10 mL deionized water; 0.05 mL phenolphthalein was added to the supernatant, and then titrated to pH 8.3 (T1). Secondly, the leachate was titrated to pH 4.1 (T2) using 0.1 mL bromophenol blue (the value reached to the titration endpoint of bromophenol blue). Concentrations of CO32– and HCO3– were calculated respectively according to T1 and T2. In addition, contents of Al and Ca were determined by inductively coupled plasma auto emission spectrometry and the concentration of Al(OH)4– was then obtained from Al content [8, 25].
2.3.2 Residue analysis
The gypsum stabilization experimented residual solids were thoroughly homogenized, and subsequently dried at 70 °C for 60 h, then sieved to retain its particle size <0.38 μm. The residues were then analyzed by X-ray powder diffraction (XRD) on a Bruker D8 discover 2500 with Cu Kα1 tube using a Sol-X detector. The X-ray diffraction patterns were performed from 10° to 80° at a step size of 0.04° of 2θ and a scan rate of 1°/min. XRD data analysis used the Materials Data Jade 6.0 software with PANalytical analysis package to identify and quantify phases.
The morphological structure and surface element distribution of BR were analyzed by scanning electron microscopy (SEM). Dried residue powders were deposited on a Cu support plate with Au support film (conductive coating, deposited by low vacuum sputter coating) and examined by a GSED field emission gun.
3 Results and discussion
Soluble alkaline anions were determined following water leaching as presented in Table 1 [16]. CO32– was the critical alkali in BR, dissolving easily, and significantly influencing total alkalinity. Meanwhile, CO32–, HCO3– and Al(OH)4– would also promote pH rise in the solution phase by the following hydrolysis reactions (Eqs. (1)–(3)).
Table 1 Composition of alkaline anions in raw BRa (mmol/kg)

CO32–+H2O→HCO3–+OH– (1)
HCO3–+H2O→H2CO3+OH– (2)
Al(OH)4–+H2O→Al(OH)3+OH– (3)
Therefore, information regarding the composition distribution and concentration of alkaline anions during gypsum stabilization is essential.
An XRD pattern of raw BR is presented in Figure 1, indicating that minerals of chemical bonded alkali mainly consist of calcite (CaCO3), cancrinite ([Na6Al6Si6O24]·2[CaCO3]) and hydrogarnet (Ca3Al2(SiO4)x(OH)12-4x). Some alkaline anions in the leachate originated from the dissolution of these insoluble chemical bonded alkalis as follows [2, 26]:
Calcite,
CaCO3→Ca2++CO32– (4)

Figure 1 XRD pattern collected from raw bauxite residue
Cancrinite,
[Na6Al6Si6O24]·2[CaCO3]+26H2O→6Na++2Ca2++6H4SiO4+6Al(OH)3+2HCO3–+8OH– (5)
Hydrogarnet,
Ca3Al2(SiO4)x(OH)12–4x→3Ca2++(6–4x)OH–+xH4SiO4+2Al(OH)3 (6)
The solubility of the chemical bonded alkali may be limited by the continuous supply of Ca2+ with the dissolution of gypsum [27, 28]. Additionally, changes in the physical characteristics of BR may be another reason for the restrictive dissolution of chemical bonded alkali [29].
3.1 Effect of gypsum addition
Based on soluble alkali leaching optimization conditions, ratio of liquid to solid 2 mL/g, 30 °C, 23 h, alkaline anion composition variation, alkalinity, pH, calcium release behavior in the leachate were investigated under different gypsum additions (0, 0.4 wt%, 0.8 wt%, 1.2 wt%, 1.6 wt%, 2.0 wt%, 2.4 wt%). As shown in Figure 2, pH significantly was reduced from 10.47 to 8.11 when gypsum was added at 1.2%, whilst leachate alkalinity dramatically decreased from 54.54 mmol/L to 2.03 mmol/L. Dissolution of gypsum will affect alkaline anions composition distribution due to a series of stabilization reactions (Figure 3). The alkaline anions concentration, CO32–, HCO3–, Al(OH)4– and OH–, were reduced from 38.1, 14.2, 1.94, 0.30 mmol/L to 0, 2.02, 0.0072,0.0015 mmol/L, respectively, which will be beneficial to the regulation of alkalinity and pH in BR. Solubility of gypsum was also closely linked to the leaching and diffusion of BR alkaline anions. Calcium concentration gradually increased with gypsum concentration from 0% to 1.6%, but then leveled out (Figure 4). This indicated that Ca2+ may be involved in the stabilization reactions of alkaline anions, promoting alkaline phase transformation.
1.2% gypsum addition could effectively regulate the alkaline anions, especially carbonate, which were mainly originated from soluble alkali. Meanwhile, gypsum would also suppress the dissolution of chemical bonded alkali by releasing of Ca2+.
3.2 Effect of stabilized time
Alkalinity and pH of leachate, composition variation of alkaline anions, and the calcium release behavior related to stabilization time are presented in Figures 5–7 (gypsum addition 1.2%, ratio of liquid to solid 2 mL/g, 30 °C, 23 h).

Figure 2 Effect of gypsum on pH in leachate of bauxite residue

Figure 3 Effect of gypsum on alkaline anions in leachate of bauxite residue

Figure 4 Change of Ca2+ in leachate of bauxite residue
pH rapidly was reduced to 8.54 at 0 (excluding initial stirring time), during which pH reached 8.15 after 4 h (Figure 5), indicating that OH– had been transformed and stabilized [7]. Additionally, most CO32– and Al(OH)4– in solution was stabilized by the release of Ca2+, with little HCO3– and OH– left in the leachate (Figure 6). Therefore, CO32– was involved in the stabilization reaction, whilst HCO3–, OH– and Al(OH)4– were also driven to co- precipitate due to the supply of Ca2+, and the physical structure of BR was apparently improved due to Ca2+ addition [28]. Furthermore, dissolution of gypsum was reduced after 4 h due to alkaline anions being stabilized. Aging or 4 h, alkaline anions were in a dynamic state of equilibrium and the concentration of Ca2+ reached 7.28 mmol/L at this stage and remained almost constant thereafter (Figure 7).
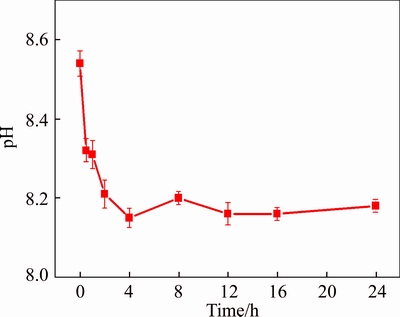
Figure 5 Effect of stabilization processes on pH in leachate of bauxite residue

Figure 6 Effect of stabilization processes on alkaline anions in solution of bauxite residue

Figure 7 Effect of stabilization processes on concentration of Ca2+ in solution of bauxite residue
3.3 Effect of stirring strength
Effects of stirring intensity on composition distribution of alkaline anions, alkalinity, pH, and the calcium release behavior are presented in Table 2 and Figure 8 (gypsum addition 1.2%, ratio of liquid to solid 2 mL/g, 30 °C, 4 h).
Table 2 Effect of stirring intensity on alkalinity in leachate of BR


Figure 8 Effect of stirring intensity on concentration of Ca2+ in solution of bauxite residue
Compared with the different stirring intensity, pH changed only slightly. Composition distributions of alkaline anions in leachate were unaffected by increasing stirring intensity. Furthermore, Ca2+ concentration in the leachate remained unchanged. These results indicated that the increase in stirring intensity made little difference on the degree of stabilization of alkaline anions and the dissolution equilibrium of chemical alkali, whilst solid–liquid system possesses high stability. For systematically understanding the alkalinity stabilization behavior, related investigations had been conducted from different angles, such as chemical reactions pathway, physical structure and element distribution, under the optimized gypsum stabilization conditions (gypsum addition 1.2%, ratio of liquid to solid 2 mL/g, 30 °C, 4 h). Of course, considering the effect of alkalinity regulation and cost, stirring intensity was set as 0.
3.4 Behavior of chemical regulation
Changes to the main ions following alkaline anion stabilization are presented in Table 3. Chemical reaction mechanisms include two parts:1) the soluble alkaline anions (CO32–, HCO3–, Al(OH)4–, OH–) were precipitated by Ca2+, being generated from the dissolution of gypsum; the alkalinity of BR was effectively stabilized and the stabilization rate of alkaline anions (generated from the 1st leaching of soluble alkali) reached 100%. 2) The alkaline anions originated from dissolution of chemical alkali (CaCO3, [Na6Al6Si6O24]·2[CaCO3], Ca3Al2(SiO4)x(OH)12–4x) were suppressed by the supply of Ca2+, and the concentrations of CO32–, HCO3–, Al(OH)4–, OH– were reduced to 0, 2.025, 0.0072, 0.0015 mmol/L, respectively, whilst the stabilization ratio of chemical bonded alkali reached 71.55% and the pH reduced from 9.57 to 8.15.
Table 3 Distribution and content of main ions during process of soluble alkali stabilization

According to the above gypsum-stabilized experimental data, CO32– maybe thoroughly reacted with Ca2+ to generate calcium carbonate (CaCO3), whilst the HCO3–, Al(OH)4–, OH– would be stabilized and the reaction species products may be determined by the solubility of chemical bonded alkali, quantitative relation of alkaline anions and Ca2+. The following precipitation reactions may exist in BR [8, 13].
Ca2++CO32– CaCO3 (7)
CaCO3 (7)
3Ca2++4OH–+2Al(OH)4– Ca3Al2(OH)12 (8)
Ca3Al2(OH)12 (8)
4Ca2++4OH–+2Al(OH)4–+CO32– Ca3Al2(OH)12·CaCO3·5H2O (9)
Ca3Al2(OH)12·CaCO3·5H2O (9)
6Ca2++4OH–+2Al(OH)4–+3SO42– Ca3Al2(OH)12·3CaSO4·26H2O (10)
Ca3Al2(OH)12·3CaSO4·26H2O (10)
The solubility product constant (Ksp) of chemical bonded alkali were in the following order: CaCO3>Ca3Al2(OH)12>Ca3Al2(OH)12·CaCO3·5H2O>Ca3Al2(OH)12·3CaSO4·26H2O. Therefore, most of the critical anion CO32– was converted to CaCO3. Other alkaline anions including HCO3–, Al(OH)4–, and OH– were also effectively stabilized as insoluble CaCO3 with the dissolution of gypsum and Ca2+. However, XRD analyses revealed that TCA (Ca3Al2(OH)12), hydrocalumite (Ca3Al2(OH)12·CaCO3·5H2O) and ettringite (Ca3Al2(OH)12·3CaSO4·26H2O) were not present (Figure 9) and therefore consumption of Ca2+ may cause co-precipitation with HCO3– and Al(OH)4–, HCO3– and OH–, resulting in the generation of simpler insoluble alkali, and the prominent reactions may include Eqs. (4), (11) and (12).
Ca2++HCO3–+Al(OH)4– CaCO3+Al(OH)3+H2O(11)
CaCO3+Al(OH)3+H2O(11)
Ca2++HCO3–+OH– CaCO3+H2O (12)
CaCO3+H2O (12)

Figure 9 XRD analyses of BR with different treatments: BR: Raw bauxite residue; G0, G0.8, G1.2: Addictive of gypsum of 0, 0.8, 1.2 respectively under optimal condition ratio of liquid to solid 2 mL/g, 30 °C, 4 h
Consequently, gypsum stabilization affects both alkalinity speciation and alkalinity solubility, and the CaCO3 should be the dominant product of gypsum stabilization. Moreover, aluminium hydroxide (Al(OH)3) and tricalcium aluminate (TCA) may also exist in the residue [24]; However, the corresponding peaks were not detected by the XRD analysis, which maybe attributed to the existing forms, such as amorphous state and low abundance.
3.5 Behavior of physical immobilization
Although alkaline anions were partially stabilized by the precipitation reactions, some anions may still exist in the complex physical structure of BR, which would not easily dissolve and diffuse in the solution through the 1st stage leaching [16, 30]. Furthermore, gypsum addition has potentially profound effects on bauxites morphological structure (Figure 10). SEM-EDX results in Figure 10 demonstrate changes in microstructure and surface element content of BR directly related to the addition of gypsum. According to SEM images, the size fraction significantly increased with the gypsum addition,whilst the number of pores obviously decreased between microaggregates, and particle distribution on the surface converted to uniform and microaggregates structure gradually ranged from loose to dense [24]. The micro-morphological transition indicated that the released calcium and its interaction on the surface of BR particles may contribute to the sequestration of alkali, such as the alkalinity transformation products (CaCO3, Al(OH)3), soluble alkali and raw alkaline mineral, and combined with the XRD results in Figure 9, which suggesting that the Ca-associated solids, such as calcium carbonate and gypsum, successfully adhered to the surface of BR, and promoted the fine particle and fragmented structure to convert to coarse particles and a blocky structure, then retarded the dissolution and diffusion of soluble alkali [23].

Figure 10 Morphological structure (a1, b1, c1) and surface element analysis (a2, b2, c2) of BR with gypsum stabilization:
Additionally, dissolution of chemical bonded alkalis in the raw BR (calcite, cancrinite, hydrogarnet) also may be suppressed with the changes to its physical structure. For instance, the microaggregates in BR will be transformed into the macroaggregates by gypsum addition, and this process can further promote the soil formation of BR [24]. In addition, the alkaline phase (soluble alkali and chemical bonded alkali), existing in BR, could be sufficiently stabilized by the Ca-driving. Therefore, physical regulation behavior and chemical regulation behavior are the critical mechanisms, and the related gypsum stabilization processes are presented in Figure 11. Moreover, the stabilization product, aluminum hydroxide (Al(OH)3), may be covered with the Ca-bearing solid according to the analysis of the SEM-EDX and XRD, which will lead to the decreasing of Al content on the surface of BR after stabilization reactions, then avoid the secondary environmental issue from the releasing of excessive aluminum.
4 Conclusions
1) The stabilization ratio of alkaline anions reached approximately 96.29% at the 1st leaching stag. Alkalinity of leachates reached 2 mmol/L whilst pH was reduced from 10.47 to 8.15 with a gypsum addition 1.2%, ratio of liquid to solid 2 mL/g, at 30 °C, 4 h.

Figure 11 Alkalinity stabilization mechanisms of gypsum in bauxite residue
2) Chemical regulation behavior of gypsum was the main pathway for significantly reducing the critical anions (CO32–). Meanwhile, the Ca-driven would promote the transformation of alkaline anions (HCO3–, Al(OH)4–, OH–); Co-precipitation reaction was established among the HCO3– and Al(OH)4–, HCO3– and OH– and, the dominant alkalinity speciation was CaCO3. The dissolution of chemical bonded alkali (calcite, cancrinite, hydrogarnet ) was suppressed by the supply of Ca2+.
3) Physical regulation behavior of gypsum would also prevent the dissolution and diffusion of soluble alkali and chemical bonded alkali due to the released Ca2+ from gypsum, which aggregated the clay particles and stabilized them into coarse particles with a blocky structure. The stability of alkaline phase had been further improved by the physical sequestration.
Acknowledgements
The authors sincerely acknowledge the anonymous reviewers for their insights and comments to further improve the quality of the manuscript.
References
[1] POWER G, GRAFE M, KLAUBER C. Bauxite residue issues: I. Current management, disposal and storage practices [J]. Hydrometallurgy, 2011, 108: 33–45. DOI: 10.1016/ j.hydromet.2011.02.006.
[2] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science and Pollution Research, 2016, 23: 12822–12834. DOI: 10.1007/s11356-016-6478-7.
[3] CAO Jing-ya, WU Qian-hong, LI Huan, OUYANG Cheng-xin, KONG Hua, XI Xiao-shuang. Metallogenic mechanism of Pingguo bauxite deposit, western Guangxi, China: Constraints from REE geochemistry and multi-fractal characteristics of major elements in bauxite ore [J]. Journal of Central South University, 2017, 24: 1627–1636. DOI: 10.1007/s11771-017-3568-8.
[4] PAN Xiao-lin, YU Hai-yan, TU Gan-feng. Reduction of alkalinity in bauxite residue during Bayer digestion in high-ferrite diasporic bauxite [J]. Hydrometallurgy, 2015, 151: 98–106. DOI: 10.1016/j.hydromet.2014.11.015.
[5] XUE Sheng-guo, ZHU Feng, KONG Xiang-feng, WU Chuan, HUANG Ling, HUANG Nan, HARTLEY W. A review of the characterization and revegetation of bauxite residues (Red mud) [J]. Environmental Science and Pollution Research, 2016, 23: 1120–1132. DOI: 10.1007/s11356- 015-4558-8.
[6] XU Guang, DING Xu-han, KURUPPU M, ZHOU Wei, BISWAS W. Research and application of non-traditional chemical stabilizers on bauxite residue (red sand) dust control, a review [J]. Science of the Total Environment, 2018, 616–617: 1552–1565. DOI: 10.1016/j.scitotenv.2017.10.158.
[7] JOHNSTON M, CLARK M W, MCMAHON P, WARD N. Alkalinity conversion of bauxite refinery residues by neutralization [J]. Journal of Hazardous Materials, 2010, 182: 710–715. DOI: 10.1016/j.jhazmat.2010.06.091.
[8] KIRWAN L J, HARTSHORN A, MCMONAGLE J B, FLEMING L, FUNNELL D. Chemistry of bauxite residue neutralisation and aspects to implementation [J]. International Journal of Mineral Processing, 2013, 119: 40–50. DOI: 10.1016/j.minpro.2013.01.001.
[9] MAYES W M, JARVIS A P, BURKE I T, WALTON M, FEIGL V, KLEBERCZ O, GRUIZ K. Dispersal and attenuation of trace contaminants downstream of the ajka bauxite residue (red mud) depository failure, Hungary [J]. Environmental Science & Technology, 2011, 45: 5147–5155. DOI: 10.1021/es200850y.
[10] LIU Wan-Chao, CHEN Xiang-qing, LI Wang-xing, YU Yan-fen, YAN Kun. Environmental assessment, management and utilization of red mud in China [J]. Journal of Cleaner Production, 2014, 84: 606–610. DOI: 10.1016/ j.jclepro.2014.06.080.
[11] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573: 155–163. DOI: 10.1016/j.scitotenv.2016.08.108.
[12] BURKE I T, PEACOCK C L, LOCKWOOD C L, STEWART D I, MORTIMER R J G, WARD M B, RENFORTH P, GRUIZ K, MAYES W M. Behavior of aluminum, arsenic, and vanadium during the neutralization of red mud leachate by HCl, gypsum, or seawater [J]. Environmental Science & Technology, 2013, 47: 6527–6535. DOI: 10.1021/es4010834.
[13] GRAFE M, POWER G, KLAUBER C. Bauxite residue issues: III. Alkalinity and associated chemistry [J]. Hydrometallurgy, 2011, 108: 60–79. DOI: 10.1016/ j.hydromet.2011.02.004.
[14] KONG Xiang-feng, JIANG Xing-xing, XUE Sheng-guo, HUANG Ling, HARTLEY W, WU Chuan, LI Xiao-fei. Migration and distribution of salinity in bauxite residue during water leaching [J]. Transactions of Nonferrous Metals Society of China, 2017, 28(3): 534–541. DOI: 10.1016/ S1003-6326(18)64686-2.
[15] HAYNES R J. Bauxite processing residue: A critical review of its formation, properties, storage, and revegetation [J]. Critical Review Environmental Science and Technology, 2011, 41: 271–315. DOI: 10.1080/ 10643380902800000.
[16] LI Xiao-fei, YE Yu-zhen, XUE Sheng-guo, WU Chuan, KONG Xiang-feng, HARTLEY W, LI Yi-wei. Leaching optimization and dissolution behavior of alkaline anions in bauxite residue [J]. Transactions of Nonferrous Metals Society of China, 2018, 28: 1248–1255. DOI: 10.1016/ S1003-6326(18)64763-6.
[17] MENZIES N W, FULTON I M, MORRELL W J. Seawater neutralization of alkaline bauxite residue and implications for revegetation [J]. Journal of Environmental Quality, 2004, 33: 1877–1884. DOI: 10.2134/jeq2004.1877.
[18] XUE Sheng-guo, WU Yu-jun, LI Yi-wei, KONG Xiang-feng, ZHU Feng, WILLIAM Hartley, LI Xiao-fei, YE Yu-zhen. Industrial wastes applications for alkalinity regulation in bauxite residue: A comprehensive review [J]. Journal of Central South University, 2019, 26(2): 268–288.
[19] COURTNEY R, KIRWAN L. Gypsum amendment of alkaline bauxite residue-plant available aluminium and implications for grassland restoration [J]. Ecological. Engineering, 2012, 42: 279–282. DOI: 10.1016/j.ecoleng. 2012.02.025.
[20] SANTINI T C, FEY M V. Spontaneous vegetation encroachment upon bauxite residue (red mud) as an indicator and facilitator of in situ remediation processes [J]. Environmental Science & Technology, 2013, 47: 12089–12096. DOI: 10.1021/es402924g.
[21] KINNARINEN T, LUBIENIECKI B, HOLLIDAY L, HELSTO J J, HAKKINEN A. Recovery of sodium from bauxite residue by pressure filtration and cake washing [J]. International Journal of Mineral Processing, 2015, 141: 20–26. DOI: 10.1016/j.minpro.2015.06.006.
[22] HAN Y S, JI S, LEE P K, OH C. Bauxite residue neutralization with simultaneous mineral carbonation using atmospheric CO2 [J]. Journal of Hazardous Materials, 2016, 326: 87–93. DOI: 10.1016/j.jhazmat.2016.12.020.
[23] XUE Sheng-guo, LI Meng, JIANG J, MILLAR G J, LI Chu-xuan, KONG Xiang-feng. Phosphogypsum stabilization of bauxite residue: conversion of its alkaline characteristics [J]. Journal of Environmental Sciences, 2019, 77: 1–10. DOI: 10.1016/j.jes.2018. 05.016.
[24] ZHU Feng, HOU Jing-tao, XUE Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [H]. Land Degradation & Development, 2017, 28(7): 2109–2120. DOI: 10.1002/ldr.2737.
[25] HUANG Yan-fang, CHAI Wen-cui, HAN Gui-hong, WANG Wen-juan, YANG Shu-zhen, LIU Jiong-tian. A perspective of stepwise utilisation of Bayer red mud: Step two—Extracting and recovering Ti from Ti-enriched tailing with acid leaching and precipitate flotation [J]. Journal of Hazardous Materials, 2016, 307: 318–327. DOI: 10.1016/ j.jhazmat.2016.01.010.
[26] WHITTINGTON B I, FLETCHER B L, TALBOT C. The effect of reaction conditions on the composition of desilication product (DSP) formed under simulated Bayer conditions [J]. Hydrometallurgy, 1998, 49: 1–22. DOI: 10.1016/S0304-386X(98)00021-8.
[27] WONG J W C, HO G E. Use of waste gypsum in the revegetation on red mud deposits: A greenhouse study [J]. Waste Mangement & Research, 1993, 11: 249–256. DOI: 10.1006/wmre.1993.1024.
[28] MAYES W M, AND P L Y, AUMO N J. Buffering of alkaline steel slag leachate across a natural wetland [J]. Environmental Science & Technology, 2006, 40: 1237–1243. DOI: 10.1021/es051304u.
[29] XUE Sheng-guo, YE Yu-zhen, ZHU Feng, WANG Qiong-li, JIANG Jun, HARTLEY W. Changes in distribution and microstructure of bauxite residue aggregates following amendments addition [J]. Journal of Environmental Sciences, 2019, 78: 276–286. DOI: 10.1016/j.jes.2018.10.010.
[30] LI Yi-wei, JIANG Jun, XUE Sheng-guo, MILLAR G, KONG, Xiang-feng, LI Xiao-fei, LI Meng, LI Chu-xuan. Effect of ammonium chloride on leaching behavior of alkaline anion and sodium ion in bauxite residue [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(10): 2125–2134. DOI: 10.1016/S1003-6326(18)64857-5.
(Edited by YANG Hua)
中文导读
赤泥碱性调控行为:石膏的钙驱动稳定化特性
摘要:赤泥是一种强碱性固体废弃物,其碱性阴离子(CO32–, HCO3–, Al(OH)4–, OH–)的释放极易引发重大环境问题。本文研究了施用后石膏赤泥碱性阴离子组分的变化、碱性物相转化路径以及固相微观形态转变特征,结果表明:石膏作用下的赤泥液相体系碱性阴离子稳定率高达96.29%,pH和液相碱性分别由10.47降至8.15,47.39 mmol/L降至2 mmol/L;在赤泥碱性稳定化过程中,化学调控行为表现在驱动赤泥典型碱性离子(CO32–, HCO3–, Al(OH)4–, OH–)的沉淀及离子间的共沉淀,碳酸钙(CaCO3)是主要的碱性转化产物。借助X射线衍射分析、扫描电镜和能谱等方法分析发现石膏钙离子溶出可促使赤泥表面细小颗粒溶解转化为稳定碱性物相,同时赤泥微观形态向致密的大颗粒块状结构转变,含钙物相附着于致密块状团聚体表面形成的物理封存方式提高了碱性转化产物及原生赤泥碱性组分的稳定性。研究结果将为应用石膏类工业废弃物开展赤泥碱性调控及堆场生态修复提供理论基础。
关键词:赤泥;石膏;碱性稳定;物相转化;钙驱动调控
Foundation item: Project(41877511) supported by the National Natural Science Foundation of China; Project(201509048) supported by the Environmental Protection’s Special Scientific Research for the Chinese Public Welfare Industry, China
Received date: 2018-10-10; Accepted date: 2018-12-10
Corresponding author: XUE Sheng-guo, PhD, Professor; Tel: +86-13787148441; E-mail: sgxue70@hotmail.com; sgxue@csu.edu.cn; ORCID: 0000-0002-4163-9383