
Organic-magnesium complex conversion coating on
AZ91D magnesium alloy
CHEN Xiao-ming(陈晓明)1, 2, LI Guang-yu(李光玉)1, 2, LIAN Jian-she(连建设)1, 2
1. Key Laboratory of Automobile Materials, Ministry of Education, Jilin University, Changchun 130025, China;
2. College of Materials Science and Engineering, Jilin University, Changchun 130025, China
Received 23 September 2009; accepted 30 January 2010
Abstract: An organic-magnesium complex conversion (OMCC) coating on AZ91D magnesium alloy was obtained by treating in a solution containing organic compounds. SEM, FESEM and XPS were used to examine the surface morphology, thickness and structure of the conversion coatings. The results show that the continuous and uniform conversion coating is deposited on AZ91D alloy and the main component of the coatings is organic compound containing benzene ring, which forms a chemical bond with magnesium. The polarization measurement and salt spray test show that the corrosion resistance of the conversion coating is much higher than that of traditional chromate conversion coating.
Key words: chromium-free conversion coating; organic compound; corrosion; XPS; AZ91D magnesium alloy
1 Introduction
Magnesium alloys are promising materials for applications where weight reduction is essential, such as automotive, aerospace, electronics, computer and mobile phone because of their low density, good heat emitting, high specific mechanical strength and good resistance against electromagnetic wave. Because of their growing use in the transportation industry, the world production of magnesium grows steadily, reaching about 610 000 t in 2006. However, the poor corrosion resistance has limited their use. In recent years, researchers have focused on improving corrosion resistance of Mg alloys. Mg and its alloys are very reactive and corrode rapidly. Anions, such as Cl-, Br-, I- and 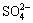 , in humid atmosphere or aqueous solutions as well as organic compounds like alcohols, ethers and phenols promote local and generalized corrosion[1]. The corrosion properties of magnesium can be improved by alloying with elements, such as manganese, aluminium, zinc, zirconium and rare earth. However, the surface treatments are still the effective ways to improve corrosion protection on alloys. Various surface treatments, such as chemical, electrochemical, laser cladding and thermal spraying on surfaces of magnesium and its alloys have been developed[2]. Among various surface treatments, the chemical conversion coating process is one of important corrosion protective methods of magnesium alloys for the advantages of low treatment cost and being easy to control the thickness precisely[3]. Such the coating can also improve the adhesion ability between the coating and the substrate which makes it easy for the next process. The most popular and traditional chemic conversion method is to treat magnesium alloys in chromate solutions like DOW 7, which is produced by Dow Chemical Company in Japan. But the use of chromate solutions is progressively restricted due to the waste water containing the high toxicity of hexavalent chromium compounds. Recently, some chromium-free conversion coatings for magnesium alloy have been developed in order to solve the pollution problem that can provide protection as effective as that of chromium conversion treatments, such as phosphate coatings phosphate/permanganate coatings[4-5], stannate-based coatings and cerium-based treatment coatings[6] and rare earth coatings[7-11].
, in humid atmosphere or aqueous solutions as well as organic compounds like alcohols, ethers and phenols promote local and generalized corrosion[1]. The corrosion properties of magnesium can be improved by alloying with elements, such as manganese, aluminium, zinc, zirconium and rare earth. However, the surface treatments are still the effective ways to improve corrosion protection on alloys. Various surface treatments, such as chemical, electrochemical, laser cladding and thermal spraying on surfaces of magnesium and its alloys have been developed[2]. Among various surface treatments, the chemical conversion coating process is one of important corrosion protective methods of magnesium alloys for the advantages of low treatment cost and being easy to control the thickness precisely[3]. Such the coating can also improve the adhesion ability between the coating and the substrate which makes it easy for the next process. The most popular and traditional chemic conversion method is to treat magnesium alloys in chromate solutions like DOW 7, which is produced by Dow Chemical Company in Japan. But the use of chromate solutions is progressively restricted due to the waste water containing the high toxicity of hexavalent chromium compounds. Recently, some chromium-free conversion coatings for magnesium alloy have been developed in order to solve the pollution problem that can provide protection as effective as that of chromium conversion treatments, such as phosphate coatings phosphate/permanganate coatings[4-5], stannate-based coatings and cerium-based treatment coatings[6] and rare earth coatings[7-11].
The organic compound solution is usually used as the corrosion resistant inhibitor for the paint or pigment systems. Only a few works are reported on the application of the organic compound conversion coatings on magnesium alloys[12] which show that the organic compound conversion coating can significantly improve the corrosion resistance of metal. The organic compound with polyphenol structure having hydroxyl (OH) groups can react with metal ions to form organic-magnesium complex and improve the corrosion resistance of magnesium alloy.
In this study, the organic-magnesium complex conversion (abbreviated as OMCC coating) was deposited on AZ91D magnesium alloy through solution containing organic compounds. The morphology, structure and corrosion resistance of the conversion coating were investigated by scanning electron microscopy (SEM), field emission scanning electron microscopy (FESEM), X-ray photo-emission spectroscopy (XPS), electrochemical polarization test in 3% NaCl (mass fraction) and salt spray test. The corrosion resistance of the OMCC coating was discussed in comparison with the results of the phosphate coating and Dow7 coating.
2 Experimental
2.1 Preparation of conversion coating on Mg alloy
The die-cast AZ91D magnesium alloy was cut into square shape specimens with cross-section area of 50 mm×50 mm and thickness of 3 mm by a wire electro-discharging machine. The chemical compositions of the alloy are shown in Table 1. All the specimens were polished with SiC sandpaper up to 2 000 grit to obtain uniform surfaces. Then, the substrate was cleaned in 10% sodium hydroxide solution (60-70 ?C) for 15 min to remove soils or greases of the surface and rinsed thoroughly in running water and deionized water to remove all the alkalis. The cleaned specimens were immersed in the conversion solution at room temperature for 5 min. The ingredients of the solution are shown in Table 2. The main ingredients are Na3PO4, Na2B7O4 and organic compound. The pH of the bath was adjusted by adding HCl to about 4.0. A visible coating on the specimens is formed after the treatment, and then the samples were dried. The traditional chromate conversion coating obtained in a solution called Dow 7 and the phosphate coating on AZ91D magnesium alloy[13] were prepared for comparison.
Table 1 Chemical compositions of magnesium alloy AZ91D (mass fraction, %)

Table 2 Ingredients of OMCC conversion bath (g/L)

2.2 Examination
SEM, FESEM and XPS were used to examine the morphology, thickness and structure of the conversion coating. The surface morphologies of the conversion coating were observed by using scanning electron microscopy (SEM, JSM-5310, Japan electronics). X-ray photoemission spectroscopy (XPS) was used to analyze the component of the conversion coating. XPS spectra of the coatings were acquired with an ESCALABMk II (Vacuum Generators) spectrometer using unmono- chromatized Al Kα X-ray (240 W).
Cycles of XPS measurements were done in a high vacuum chamber with a base pressure of 1.33×10-6 Pa.
2.3 Polarization measurement and salt spray test
Polarization measurement and salt spray test were carried out to evaluate the corrosion resistance of the OMCC coating. The polarization measurements of the conversion coatings were performed on an Electrochemical Analyzer (VersaSTAT 3, Princeton Applied Research) by Linear Sweep Voltammeter technique at room temperature in 3% NaCl aqueous solution using a classic three-electrode cell. A saturated calomel electrode (SCE) was used as the reference electrode and the platinum plate was used as the auxiliary electrode. All potentials quoted were on the SCE. The coated specimens were masked with epoxy resin (EP651) so that only 1 cm2 was exposed to the electrolyte. Before the potentiodynamic sweep experiments, the specimens were immersed into electrolyte for about 30 min to stabilize the open-circuit potential (OCP, φ0). The scanning rate was 50 mV/min for all measurements. The salt spray tests were conducted in a SF850 salt spay cabin (Atlas Electric Devices) and ASTM B117-03 standard was adopted for the salt spray test[14].
3 Results and discussion
3.1 Morphology and composition analysis of coating
Fig.1 shows the optical morphology of the OMCC coating on AZ91D magnesium alloy. From Fig.1, it is clear that the surface of AZ91D magnesium alloy is covered with uniform grey-white coating. The coating is so thin that some mechanically polished scratches are still observed.
Fig.2(a) shows the morphology of OMCC coating on AZ91D magnesium alloy. It is seen from Fig.2(a) that the magnesium alloy substrate is entirely covered by the coating and some irregular cracks form on the surface.

Fig.1 Optical photo of OMCC coating on AZ91D magnesium
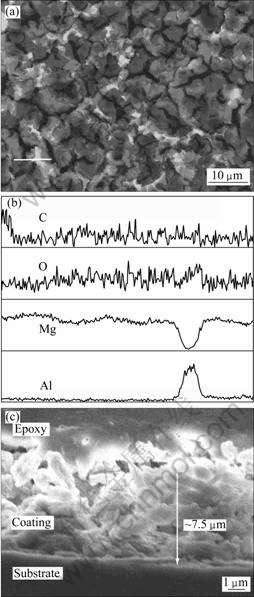
Fig.2 Morphology of OMCC coating on AZ91D alloy(a), corresponding EDS analysis of coating on surface substrate (C, O, Mg, Al) (b) and FESEM image of cross-section of OMCC on AZ91D alloy (c)
But the cracks do not penetrate to substrate. The line scanning of element analysis along the line is marked in Fig.2(a) and the corresponding EDS analysis is shown in Fig.2(b). The surface element analysis shows that there are Mg, Al, O and C elements on the surface of the coating. The surface morphology of the coating can be closely related to the microstructure of the substrate[15]. The AZ91D magnesium alloy exhibits two-phase structure consisting of Mg-rich α phases and Al-rich β phases. The comparison of Fig.2(a) with (b) shows clearly a close relationship between the coating morphology and the substrate microstructure. The Al-rich β phases correspond to the whitish network and Mg-rich α phases correspond to the black part (shown in Fig.2(a)). Fig.2(c) shows FESEM image of cross-section of OMCC coating on AZ91D magnesium alloy. The thickness of the coating is about 7.5 μm. The images confirm that the conversion coating is dense and has indeed adhered to the substrate.
Fig.3(a) shows the XPS spectrum of the OMCC coating. Except for peaks of Mg and Al element, there are also evident peaks of C and O elements. Figs.3(b)-(d) show the XPS spectra of Mg 2p, C 1s and O 1s peaks. As shown in Fig.3(b), the Mg 2p peaks at 49.6 eV and 50.3 eV are observed, which are attributed to the bonds of Mg―Mg and Mg―O, respectively. As shown in Fig.3(c), the C 1s core levels reveal three peaks structures with binding energies at 284.4 eV, 285.7 eV and 288.4 eV, respectively. The peak at 284.40 eV is caused by pollution during XPS measurement. The peak at 285.84 eV is attributed to C=O bond in the group ―C6H4―C(O)― in the organic polymer[16], which should be the reaction product of organic compound. The peak at 288.7 eV is attributed to C=O bond in the C(O)―O group. The detail of O 1s core levels is shown in Fig.3(d), which reveals four-peak structures with binding energies of 530.9, 531.6, 532.3 and 533.1 eV, respectively. The feature of 530.9 eV is attributed to Mg―O bond and the O 1s peak at 531.6 eV is assigned to C=O in ―(O)C― groups in organic polymer. The peak at 532.3 eV is attributed to O―H bond in the group C(O)―OH and the peak at 533.2 is attributed to C=O bond in the group ―C(O)―O―[17]. Based on the results of the XPS and the element analysis, the OMCC coating mainly consists of organic compound containing benzene ring, which forms a chemical bond with magnesium.
3.2 Corrosion characteristics of coating
Fig.4 shows the electrochemical polarization curves of the AZ91D magnesium alloy substrate (curve 1), the phosphate coating (curve 2), the Dow7 coating (curve 3) and the OMCC coating (curve 4) on the AZ91D magnesium alloy in 3% NaCl aqueous solution at room temperature. The cathode reaction on the polarization
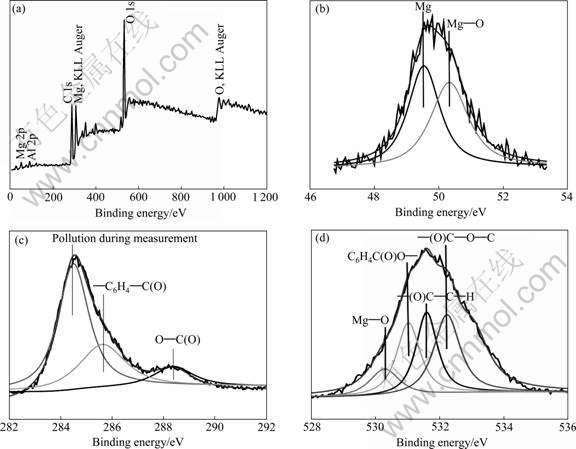
Fig.3 XPS spectra of OMCC coating(a), Mg 2p(b), C 1s(c) and O 1s(d)

Fig.4 Electrochemical polarization curves in 3% NaCl aqueous solution
curves corresponds to the evolution of the hydrogen. The anodic polarization curve part shows the most important features related to the corrosion resistance. The polarization current increases with increasing the applied anodic potential. The corrosion potential φcorr of the AZ91D magnesium alloy substrate, the phosphate coating, the Dow7 coating and the OMCC coating on the AZ91D magnesium alloy are -1.43, -1.10, -0.99, -0.47 V, respectively. So, the corrosion potential φcorr of the OMCC coating is the most positive, which signifies that corrosion is the most difficult to happen on the OMCC coating in the same environment. The corrosion current density Jcorr of the AZ91D magnesium alloy substrate, the phosphate coating, the Dow7 coating and the OMCC coating on the AZ91D magnesium alloy are 927, 6.84, 39.3, 3.1 μA/cm2, respectively. The corrosion current density Jcorr of the OMCC coating is the lowest, which signifies that the anodic passivation coating formed by corrosion can last for longer time before it is erodedly broken. The polarization curve analysis indicates that the OMCC coating exhibits an improved corrosion resistance in comparison with the phosphate coating and Dow7 coating and can effectively protect the magnesium alloy substrates from corrosion.
In order to further prove the corrosion resistance of the OMCC coating, the salt spray test were conducted. Figs.5(a) and (b) show the surface morphologies of the OMCC coating and the Dow7 coating after 3 d salt spray test, respectively. It is seen that the surface of the Dow7 coating is rusted by almost 40% after 3 d salt spray test, but the surface of the OMCC coating is rusted with only several small spots. This implies that the passage of corrosion is interdicted, even though corrosion happens, and it does not rapidly spread. Therefore, the corrosion resistance of the OMCC coating is higher than that of the Dow7 coating. This result is in coincidence with that of the above electrochemical polarization behaviors.

Fig.5 Photos of corrosion surfaces after 3 d of salt spray: (a) OMCC coating; (b) Dow7 coating
4 Conclusions
1) A chemical conversion coating on AZ91D magnesium alloy substrate is obtained. The main component of the coating is organic-magnesium compound containing benzene ring.
2) In the polarization test, the conversion coating shows more positive potential and obviously lower corrosion current density in comparison with those of the traditional chromate conversion coating (Dow7 coating) and the popular phosphate coating. The salt spray tests also show the improved corrosion resistance of the OMCC conversion coating.
3) The present OMCC conversion coating can be a preferential substituting conversion coating to the chromate conversion coating on magnesium alloy surface. The present organic chromium-free conversion can also be utilized as the pretreatment layer to be favorable to further organic painting on magnesium alloy.
References
[1] ARDELEAN H, FRATEUR I, MARCUS P. Corrosion protection of magnesium alloys by cerium, zirconium and niobium-based conversion coatings [J]. Corrosion Science, 2008, 50: 1907-1918.
[2] CHEN Xiao-ming, LI Guang-yu, LIAN Jian-she. JIANG Qing. An organic chromium-free conversion coating on AZ91D magnesium alloy [J]. Applied Surface Science, 2008, 255(5): 2322-2328.
[3] GRAY J E. LUAN B. Protective coatings on magnesium and its alloys―A critical review[J]. J Alloys Compd, 2002, 336(1/2): 88-113.
[4] KOUISNI L, AZZI M, DALARD F, MAXIMOVITCH S. Phosphate coatings on magnesium alloy AM60 (part 1): Study of the formation and the growth of zinc phosphate films [J]. Surface Coating and Technology, 2004, 185(1): 58-67.
[5] LI Guang-yu, LIAN Jian-she, JIANG Zhong-hao. A black phosphate coating for C1008 steel [J]. Surface Coating and Technology, 2004, 176(2): 215-221.
[6] YANG K, GER M, HWU W, SUNG Y, LIU Y. Study of vanadium-based chemical conversion coating on the corrosion resistance of magnesium alloy [J]. Materials Chemistry and Physics, 2007, 101(2/3): 480-485.
[7] HEMINGWAY R, LAKES P. Plant polyphenols [M]. New York: Plenum Press, 1992.
[8] MANSFELD F, WANG Y, SHIH H. The Ce-Mo process for the development of a stainless aluminum [J]. Electrochimica Acta, 1992, 37(12): 2277-2282.
[9] HUGHES A, TAYLOR R, HINTON B, WILSON L. XPS and SEM characterization of hydrated cerium oxide conversion coatings [J]. Surface and Interface Analysis, 1995, 23(7/8): 540-550.
[10] DABALA M, ARMELAO L, BUCHBERGER A, CALLIARI I. Cerium-based conversion layers on aluminum alloys [J]. Applied Surface Science, 2001, 172(3/4): 312-322.
[11] YU Xing-wen, CAO Chun-an, YAO Zhi-ming, ZHOU De-rui, YIN Zhong-da. Study of double layer rare earth metal conversion coating on aluminum alloy LY12 [J]. Corrosion Science, 2001, 43(7): 1283-1294.
[12] KULICK L, SAAD K I. Protective coatings metals and other surfaces. U.S 3975214[P]. 1976.
[13] LI Guang-yu, LIAN Jian-she, NIU Li-yuan, JIANG Zhong-hao. Growth of zinc phosphate coatings on AZ91D magnesium alloy [J]. Surface Coating and Technology, 2006, 201(3/4): 1814-1820.
[14] ASTMB117―03. Practice for operating salt spray, 2004, ASTM International. http//www.nnnastm.org.
[15] LUNDER O, WALMSLEY J, MACK P, NISANCIOGLU K. Formation and characterisation of a chromate conversion coating on AA6060 aluminium [J]. Corrosion Science, 2005, 47(7): 1604-1624.
[16] BEAMSON G, BRIGGS D. High resolution XPS of organic[M]. Polymers: The Scienta ESCA300 Database. 1992.
[17] CLARK D T, FEAST W J, TWEEDALE P J, THOMAS H R. ESCA applied to polymers. XXVI. Investigation of a series of aliphatic, aromatic, and fluorine-containing polycarbonates [J]. Journal of Polymer Science Polymer Chemistry Edition, 1980, 18(6): 1651-1664.
(Edited by LI Yan-hong)
Foundation item: Project(50871046) supported by the National Natural Science Foundation of China; Project(2007KZ09) supported by the 2007 Science and Technology Support Plan of Changchun City, China
Corresponding author: LI Guang-yu; Tel: +86-431-85095875; Fax: +86-431-85095876; E-mail: guangyu@jlu.edu.cn