铵盐柱浸风化壳淋积型稀土矿中的稀土和铝
来源期刊:中国有色金属学报(英文版)2016年第11期
论文作者:何正艳 张臻悦 余军霞 徐志高 徐源来 周芳 池汝安
文章页码:3024 - 3033
关键词:柱浸过程;风化壳淋积型稀土矿;稀土;铝;铵盐;传质
Key words:column leaching process; weathered crust elution-deposited rare earth ore; rare earth; aluminum; ammonium salt; mass transfer
摘 要:为了更好地了解风化壳淋积型稀土矿中稀土和铝的浸出过程,运用色谱塔板理论对稀土和铝的柱浸传质过程进行研究。结果表明,在一定范围内,提高铵根离子浓度能强化浸出传质过程。浸取剂pH在2~8的范围内对稀土的浸取传质效率几乎没有影响,而在强酸性条件下(pH<4)能提高铝的浸出传质效率。存在一最佳流速使浸取传质效率最高。稀土的最佳浸取条件如下:浸取剂pH 4~8、铵根离子浓度0.4 mol/L、流速0.5 mL/min。稀土和铝的浸出传质效率由低到高的顺序均为(NH4)2SO4
Abstract: In order to better understand the leaching process of rare earth (RE) and aluminum (Al) from the weathered crust elution- deposited RE ore, the mass transfer of RE and Al in column leaching was investigated using the chromatographic plate theory. The results show that a higher initial ammonium concentration in a certain range can enhance the mass transfer process. pH of leaching agent in the range of 2 to 8 almost has no effect on the mass transfer efficiency of RE, but plays a positive role in the mass transfer efficiency of Al under strong acidic condition (pH<4). There is an optimum flow rate that makes the highest mass transfer efficiency. The optimum leaching condition of RE is the leaching agent pH of 4-8, ammonium concentration of 0.4 mol/L and flow rate of 0.5 mL/min. The mass transfer efficiencies of RE and Al both follow the order: (NH4)2SO4
Trans. Nonferrous Met. Soc. China 26(2016) 3024-3033
Zheng-yan HE1, Zhen-yue ZHANG1, Jun-xia YU2, Zhi-gao XU2, Yuan-lai XU2, Fang ZHOU2, Ru-an CHI1,2
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory for Green Chemical Process of Ministry of Education, Wuhan Institute of Technology, Wuhan 430073, China
Received 30 October 2015; accepted 23 May 2016
Abstract: In order to better understand the leaching process of rare earth (RE) and aluminum (Al) from the weathered crust elution- deposited RE ore, the mass transfer of RE and Al in column leaching was investigated using the chromatographic plate theory. The results show that a higher initial ammonium concentration in a certain range can enhance the mass transfer process. pH of leaching agent in the range of 2 to 8 almost has no effect on the mass transfer efficiency of RE, but plays a positive role in the mass transfer efficiency of Al under strong acidic condition (pH<4). There is an optimum flow rate that makes the highest mass transfer efficiency. The optimum leaching condition of RE is the leaching agent pH of 4-8, ammonium concentration of 0.4 mol/L and flow rate of 0.5 mL/min. The mass transfer efficiencies of RE and Al both follow the order: (NH4)2SO4
Key words: column leaching process; weathered crust elution-deposited rare earth ore; rare earth; aluminum; ammonium salt; mass transfer
1 Introduction
Weathered crust elution-deposited rare earth (RE) ore, also named ion-adsorbed RE ore, is a unique mineral resource in China due to the rich middle and heavy RE [1]. For the tremendous commercial value, great attention is paid to the exploitation of this ore. It is widely distributed in southern China, such as Jiangxi, Guangdong, Fujian, Hunan, Yunnan, Guangxi and Zhejiang [2]. The main mineral compositions of the weathered crust elution-deposited RE ore are quartz, potash feldspar, plagioclase, kaolinite and white mica. In a warm and humid climate, original rocks containing RE are weathered and clay minerals are formed by biological, chemical and physical effects. During this weathering process, REs in the minerals are dissociated to be hydrated or hydroxyl-hydrated RE ions and further adsorbed on clay minerals with the migration of natural water [3]. As a result of the specific occurrence state, RE can be leached out from weathered crust elution- deposited RE ore with ammonium or sodium salt by ion-exchange method [4].
The leaching process of RE from weathered crust elution-deposited RE ore is a mass transfer process. The cations in the leaching agent diffuse from solution to the ore particles, and then the RE ions adsorbed on the ore particles are replaced and transferred into the solution [5]. TIAN et al [6-8] reported that the kinetics of RE leaching was controlled by inter-diffusion and the mass transfer efficiency would affect the leaching of RE. In the leaching process of RE, impurity ions, especially Al, accounting for 35%-75% of total amount of impurities in the leachate, are also exchanged into the leachate. The impurities in leachate would decrease the quality of RE product, increase the consumption of precipitator and even destroy the crystal forming of carbonate RE precipitation [9]. Currently, most researches focus on the leaching techniques, but limited literatures exist on the leaching mass transfer process of RE, let alone impurities [10,11]. Therefore, it is vitally necessary to investigate the mass transfer process of both RE and impurities (Al) to reduce the consumption of leaching agent, increase the leaching efficiency of RE, and reduce the content of impurities in the leachate [12].
Due to the strong exchangeability with RE, minor environmental pollution and low cost, ammonium salts are generally recognized as the optimal leaching agents at present. Ammonium sulfate ((NH4)2SO4), ammonium chloride (NH4Cl) and ammonium nitrate (NH4NO3) as the common ammonium salts are widely used in the actual RE recovery [13,14]. CHI and TIAN [1] discussed the leaching efficiencies of RE and impurity using the three different ammonium salts, which indicated that the leaching efficiency both followed the order of (NH4)2SO4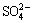 , Cl- and
, Cl- and 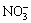 ) and search a novel leaching agent.
) and search a novel leaching agent.
In this study, the mass transfer of RE and Al in the leaching process by the three ammonium salts was investigated in the fixed bed column. The effects of leaching agent pH, ammonium (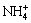 ) concentration and flow rate on the mass transfer efficiencies of RE and impurities (Al) were studied and results were evaluated by chromatographic plate theory. The effects of anions (
) concentration and flow rate on the mass transfer efficiencies of RE and impurities (Al) were studied and results were evaluated by chromatographic plate theory. The effects of anions (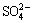 , Cl- and
, Cl- and 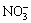 ) on the mass transfer of RE and Al were also discussed.
) on the mass transfer of RE and Al were also discussed.
2 Experimental
2.1 Materials
The experimental RE ore sample is original RE ores collected from Dingnan (DN) County in Jiangxi Province of southern China. The RE ore sample was mixed thoroughly and dried in an oven at 50 °C for 8 h.
All chemicals in this study were purchased from Sinopharm Chemical Reagent Co., Ltd., Shanghai, China, with analytical grade. The used deionized water was produced by an ultra-pure water system (Super series, Nison Instrument Co., Ltd., Shanghai, China).
2.2 Column leaching experiments
The experiments were processed at ambient temperature (25 °C) in a glass column with an inner diameter of 45 mm and a column length of 500 mm, which can be seen as a chromatographic column. 250 g of dried RE ore sample was packed uniformly in the glass column and the packed ore height was measured. A sand core filter plate was fixed at the bottom of the leaching column to retain soil particles and two layers of filter paper were put on the upper of the RE ore sample to resist preferential flow. The leaching agent was added from the top of the leaching column at a constant flow rate by a peristaltic pump. Leachate was collected from the bottom of the leaching column and the concentrations of RE and Al were analyzed by titration method.
2.3 Analytical methods
The total content of RE in the leachate was determined by EDTA titration using xylenol orange as indicator and hexamethylene tetramine as buffer. After that, excess EDTA solution was added into the studied liquid sample and placed in a water bath at 90 °C for 10 min. Then, the content of Al was analyzed by back titration using a known concentration of zinc solution. The error of the titration method was ±2%.
3 Results and discussion
3.1 Characterization of RE ore
The main chemical composition of the RE ore was analyzed by X-ray fluorescence (Axios advanced, Panalytical B.V.) and the results are listed in Table 1. It reveals that the RE ore contains 0.10% RE and 19.53% Al. The occurrence states of RE and Al in RE ore were determined with the sequential fractionating extraction method [15] and shown in Tables 2 and 3. For RE, it exists as aqueous soluble state, ion-exchangeable state, colloid sediment state and mineral state. The exchangeable RE grade is 0.090%, demonstrating that 90.00% of RE in this ore exists as ion-exchangeable state. The partitioning of the exchangeable RE in the leachate was analyzed by inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7700x, Agilent Technologies Inc.) and the results are shown in Table 4.
Table 1 Main chemical compositions of RE ores (mass fraction, %)
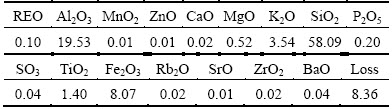
Table 2 Occurrence states of RE in rare earth ore
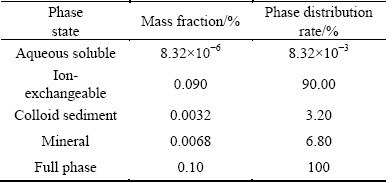
Table 3 Occurrence states of Al in rare earth ore
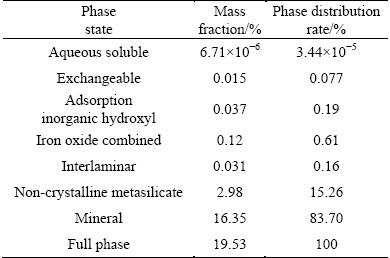
Table 4 Partitioning of exchangeable RE (mass fraction, %)
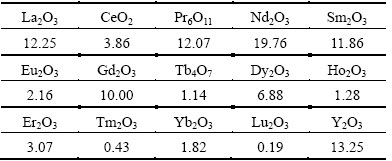
It indicates that 52.06% of REs are the middle and heavy REs with tremendous commercial value. For Al, the occurrence states include soluble state, exchangeable state, adsorption inorganic hydroxyl state, iron oxide combined state, interlaminar state, non-crystalline metasilicate state and mineral state. The exchangeable Al takes only 0.077% of the total amount, while the mineral Al takes 83.70%. The main occurrence state of Al in the RE ores is mineral state. For the high content of Al (19.53%) in the ores, the small part of the exchangeable Al (0.015%) still has a great effect on the downstream production, thus the mass transfer process of Al should be studied.
3.2 Leaching process of rare earth ore
In the weathered crust elution-deposited RE ore, RE in ion-exchangeable state, mainly adsorbed on clay mineral, could be exchanged by electrolyte solution through ion-exchange method [16,17]. The main clay minerals are aluminosilicate which can be described as [Al4(Si4O10)(OH)8]m. The leaching chemical reaction with ammonium salt can be expressed as [1]:
[Al4(Si4O10)(OH)9]m・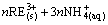
 [Al4(Si4O10)(OH)9]m・
[Al4(Si4O10)(OH)9]m・ (1)
(1)
where s and aq represent solid phase and aqueous phase, respectively.
It indicates that the leaching process is a reversible and typical noncatalytic heterogeneous reaction in liquid-solid systems [18]. The leaching process can be seen as a chromatographic column elution process, in which the RE ore is a stationary phase and the leaching agent is a mobile phase [19]. When the leaching agent flows through the packed bed, an ion-exchange reaction takes place between the RE and 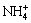 . The mass transfer efficiency in the leaching process can be evaluated by chromatographic plate theory [20]. For the same packed ore length (L), the high mass transfer efficiency can be obtained at a lower height equivalent to a theoretical plate (HETP) or a large plate number (n) [21]. According to the leaching curves obtained from column leaching experiments, HETP and n can be calculated by the following equations [22]:
. The mass transfer efficiency in the leaching process can be evaluated by chromatographic plate theory [20]. For the same packed ore length (L), the high mass transfer efficiency can be obtained at a lower height equivalent to a theoretical plate (HETP) or a large plate number (n) [21]. According to the leaching curves obtained from column leaching experiments, HETP and n can be calculated by the following equations [22]:
HETP=L/n (2)
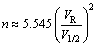 (3)
(3)
where n is the true plate number; VR (mL) is the retention volume, which represents the volume of the leachate collected from the bottom of the leaching column; V1/2 (mL) is the peak width at half height; HETP (mm) is the height equivalent to a theoretical plate; L (mm) is the packed ore length.
3.3 Effects of pH on leaching process of RE and Al
pH may affect the exchangeability and diffusivity of the leaching agent [19]. Figure 1 shows the effects of pH on leaching curves of RE and Al from the RE ores by (NH4)2SO4, NH4Cl and NH4NO3. For the three ammonium salts, the leaching concentration of RE and Al increase rapidly to the maxima at first, and then decrease sharply till to zero. The leaching curves are all similar to the chromatographic elution curves, which indicates that the mass transfer process of RE and Al can be evaluated by chromatographic plate theory. The leaching HETPs of RE and Al at different pH were calculated and shown in Fig. 2. It is observed that pH almost has no effects on the values of HETP for RE leaching, while HETP for Al leaching decreases obviously with pH decreasing from 4 to 2. These are consistent with the phenomena shown in Fig. 1. For Al leaching, the highest peak concentration and the broadest elution profile are obtained at pH 2. The mass transfer efficiency of Al can be enhanced in strong acidic condition. This can be explained by the occurrence states and contents of RE and Al in the RE ore listed in Tables 2 and 3. Under a strong acidic condition (pH<4), exchangeable state can be exchanged by ammonium salt, and colloid sediment state RE and adsorption inorganic hydroxyl state Al can be dissolved [23]. The content of adsorption inorganic hydroxyl state Al (0.037%) in RE ores is more than double contents of exchangeable state Al (0.015%). Nevertheless, the content of colloid sediment state RE (0.0032%) is much lower than that of exchangeable state RE (0.090%). Therefore, pH has greater effect on the leaching of Al than on the leaching of RE. According to the results above, the optimal pH range is from 4 to 8. Too high pH would lead to the precipitate of RE and Al, whereas too low pH would increase the leaching of the impurities, especially Al. The initial pH of the three ammonium salts is 5.5-6.0 in the experimental conditions, thus it could be used directly without pH adjustment.
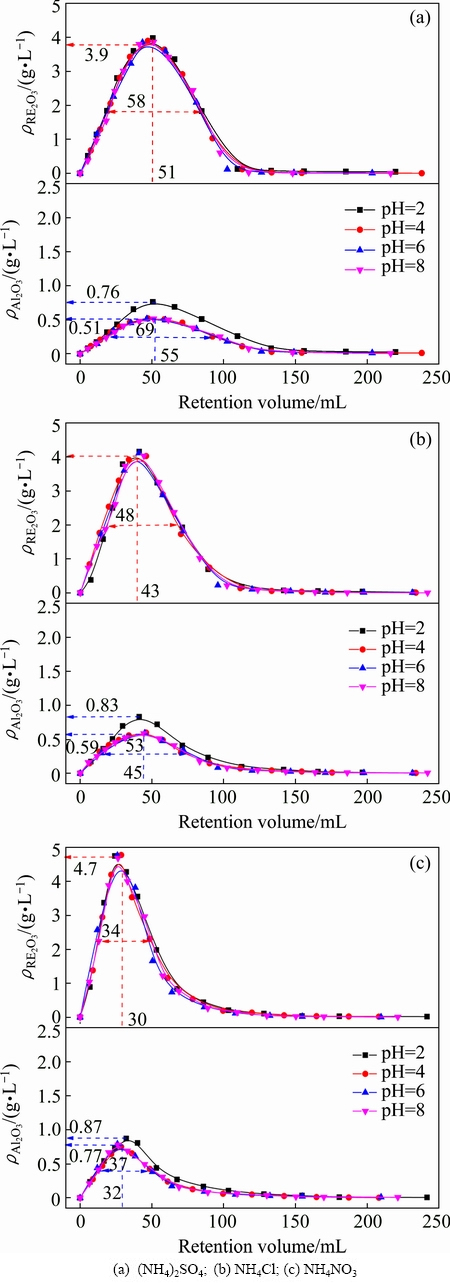
Fig. 1 Leaching curves of RE and Al by ammonium salts at different pH values (250 g RE ore, 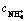 =0.2 mol/L, flow rate of leaching agent u=0.5 mL/min, t=25 °C)
=0.2 mol/L, flow rate of leaching agent u=0.5 mL/min, t=25 °C)
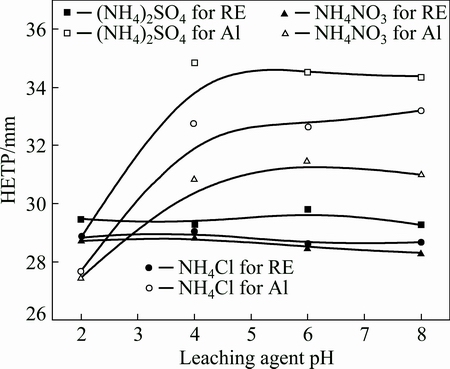
Fig. 2 HETP curves of leaching RE and Al by three ammonium salts at different pH (250 g RE ore, 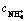 = 0.2 mol/L, u=0.5 mL/min, t=25 °C)
= 0.2 mol/L, u=0.5 mL/min, t=25 °C)
The effects of different ammonium salts on the leaching process of RE and Al can also be seen from Fig. 1. The peak concentrations of RE and Al with NH4NO3 are the highest and those with (NH4)2SO4 are the lowest. Peak shape with NH4NO3 is the narrowest and that with (NH4)2SO4 is the broadest. These results agree with those obtained in Fig. 2. HETP decreases following the order of (NH4)2SO4>NH4Cl>NH4NO3. It reveals that the mass transfer efficiencies of RE and Al both follow the order of (NH4)2SO4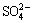
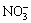 , and the mass transfer efficiency of the leaching agent was consistent with its complexing ability. It can also be observed from Fig. 1 that the peak concentration of RE (3.9-4.7 g/L) is far higher than that of Al (0.51-0.87 g/L). Due to the broader half peak width and lower peak concentration, the mass transfer efficiency of Al is lower compared with that of RE. It is also demonstrated by the higher HETP of Al shown in Fig. 2. The retention volume at the peak concentration of RE is only 2-4 mL less than that of Al. This reveals that the leaching of RE and Al is mostly simultaneous. It is hard for the separation of RE and Al in the leaching process under this experiment condition. However, the resolution of RE and Al may be improved by increasing the packed column length or changing the mobile phase [19]. In the studied three ammonium salts, NH4NO3 obtains the highest mass transfer efficiency of RE, while (NH4)2SO4 obtains the lowest content of impurities in the leachate.
, and the mass transfer efficiency of the leaching agent was consistent with its complexing ability. It can also be observed from Fig. 1 that the peak concentration of RE (3.9-4.7 g/L) is far higher than that of Al (0.51-0.87 g/L). Due to the broader half peak width and lower peak concentration, the mass transfer efficiency of Al is lower compared with that of RE. It is also demonstrated by the higher HETP of Al shown in Fig. 2. The retention volume at the peak concentration of RE is only 2-4 mL less than that of Al. This reveals that the leaching of RE and Al is mostly simultaneous. It is hard for the separation of RE and Al in the leaching process under this experiment condition. However, the resolution of RE and Al may be improved by increasing the packed column length or changing the mobile phase [19]. In the studied three ammonium salts, NH4NO3 obtains the highest mass transfer efficiency of RE, while (NH4)2SO4 obtains the lowest content of impurities in the leachate.
3.4 Effects of ammonium concentration on leaching process of RE and Al
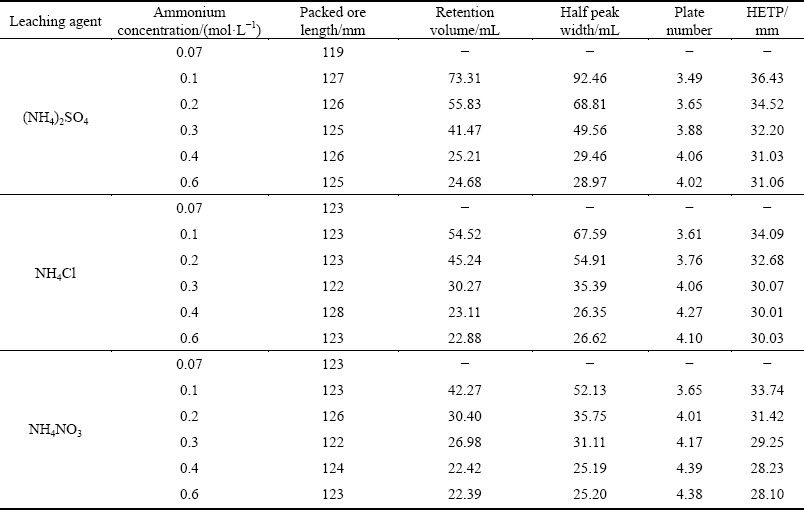
Concentration is a key factor that affects the exchangeability and diffusivity of the leaching agent to the mineral particles [25]. Figure 3 depicts the effects of ammonium concentration on the leaching process of RE and Al from the RE ores. It is found that the peak concentrations of RE and Al increase sharply when the ammonium concentration of the three ammonium salts increases from 0.07 to 0.4 mol/L, and almost no increase is observed with the continuous concentration increase. Similar results are present in the elution profile of RE and Al. Elution profile becomes narrower and narrower till the concentration of ammonium increases to 0.4 mol/L and then the half peak width keeps at a constant value. According to the data in Fig. 3, the HETP and plate number of RE and Al under different ammonium salts are calculated and listed in Tables 5 and 6. For the three ammonium salts, the plate number increases while HETP decreases with increasing ammonium concentration till 0.4 mol/L, and after that no obvious change is observed with the continuous increase in the ammonium concentration. The results indicate that the leaching mass transfer efficiency can be improved by increasing the ammonium concentration to a certain high concentration range. Higher initial concentration can provide an important driving force to overcome the mass transfer resistances of ammonium diffusing from solution to ore particles. However, the positive effect of ammonium concentration on the mass transfer efficiency can be negligible when the initial ammonium concentration is high enough. At this moment, the leaching kinetics of RE and Al is controlled by chemical reaction of ion exchange instead of solid-film diffusion [26]. Therefore, the positive effect of high ammonium concentration on the leaching process of RE and Al is unobvious after 0.4 mol/L. Blindly improving the concentration of leaching agent will not only increase the production cost, but also aggravate the environmental burden. It is better to choose a suitable concentration of leaching agent on the basis of the characteristics of a specific RE ore in the actual exploitation of weathered crust elution-deposited RE ore.
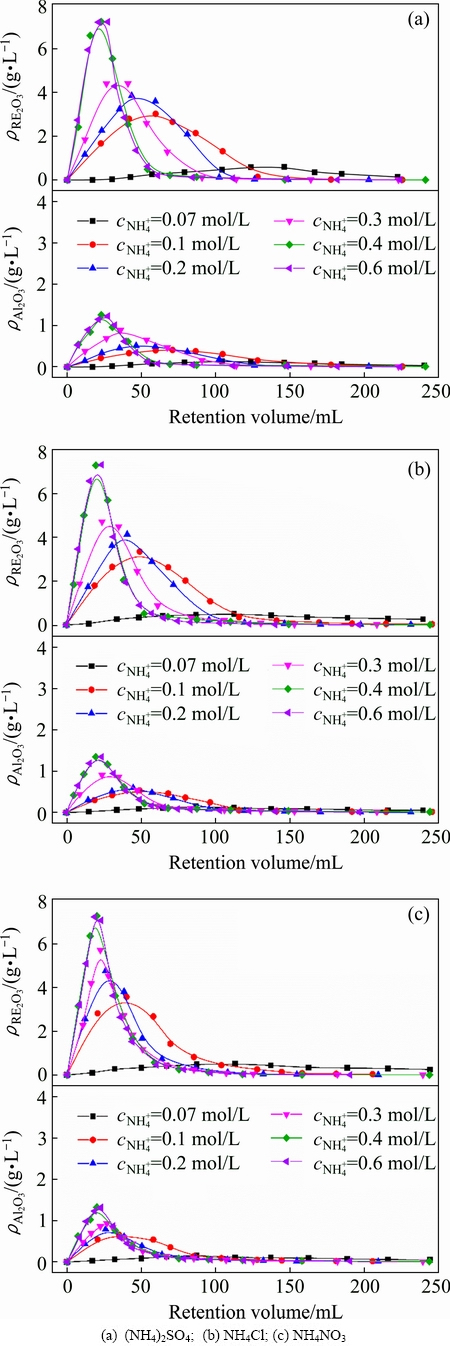
Fig. 3 Leaching curves of RE and Al by ammonium salts under various ammonium concentration (250 g RE ore, u=0.5 mL/min, t=25 °C, initial pH 5.5-6.0)
Compared with Al, the half peak width of RE is narrower and the peak concentration of RE is much higher in the studied range of ammonium concentration. These all can be better expressed by HETP. The HETP of Al is higher than that of RE, namely the mass transfer efficiency of Al is lower. Furthermore, it can also be found that the HETPs of RE and Al increase following the order of NH4NO3
Table 5 Calculated HETP of RE under various ammonium concentrations
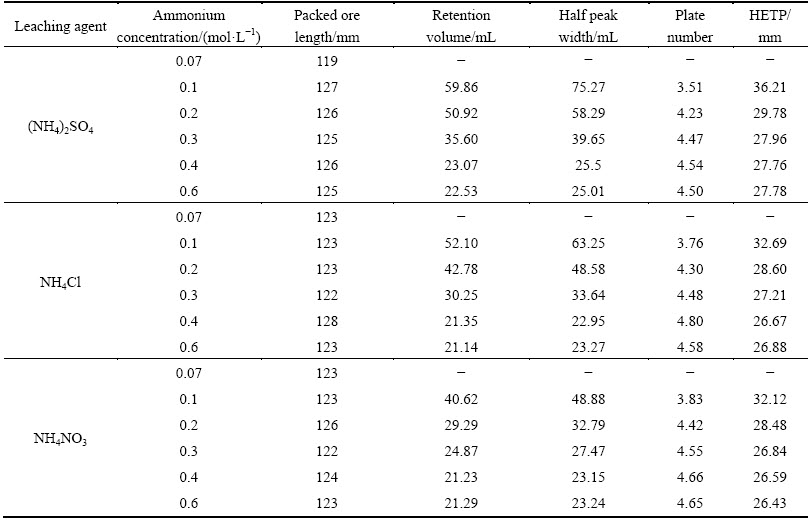
Table 6 Calculated HETP of Al under various ammonium concentrations
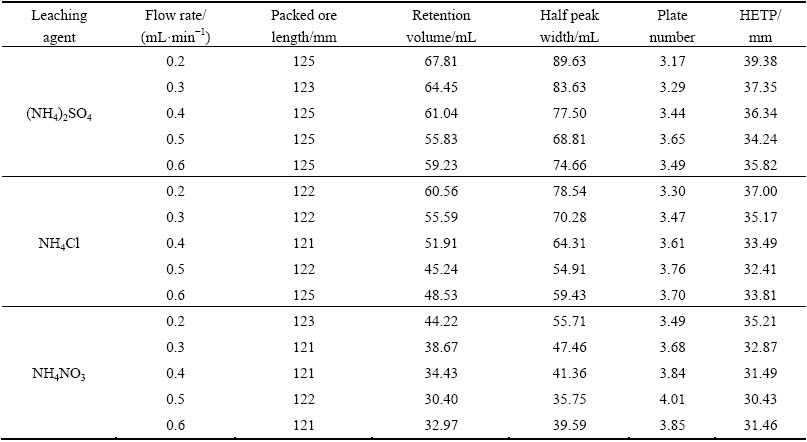
3.5 Effects of flow rate on leaching process of RE and Al
The flow rate of leaching agent is one of the important factors that influence the mass transfer efficiency [5]. Figure 4 shows the effects of flow rate of leaching agent on leaching process of RE and Al by different ammonium salts. With the flow rate increasing to 0.5 mL/min, the peak concentrations of RE and Al increase and the retention volumes at the peak concentrations reduce. The leached-out RE can be taken away timely by the flow of solution to prevent the reabsorption. After the flow rate exceeding 0.5 mL/min, the peak concentration decreases and the retention volume increases. The flow rate of solution among the ore particles is too fast and the contact between leaching agent and ore particles is insufficient. The equilibrium of the ion exchange reaction has not been established [6]. The optimal mass transfer efficiencies of RE and Al in the leaching process are obtained at the flow rate of 0.5 mL/min. Plates number and HETP obtained at different flow rates are shown in Tables 7 and 8. For RE and Al, plate numbers firstly increase and then decrease with the increase of the flow rate, and maximum is obtained at flow rate of 0.5 mL/min. While HETP has the opposite trend, the minimum is obtained at the flow rate of 0.5 mL/min. According to van Deemter equation (Eq. (4)), there is an optimal flow rate of leaching agent (uopt) at which HETP reaches the smallest value [27].
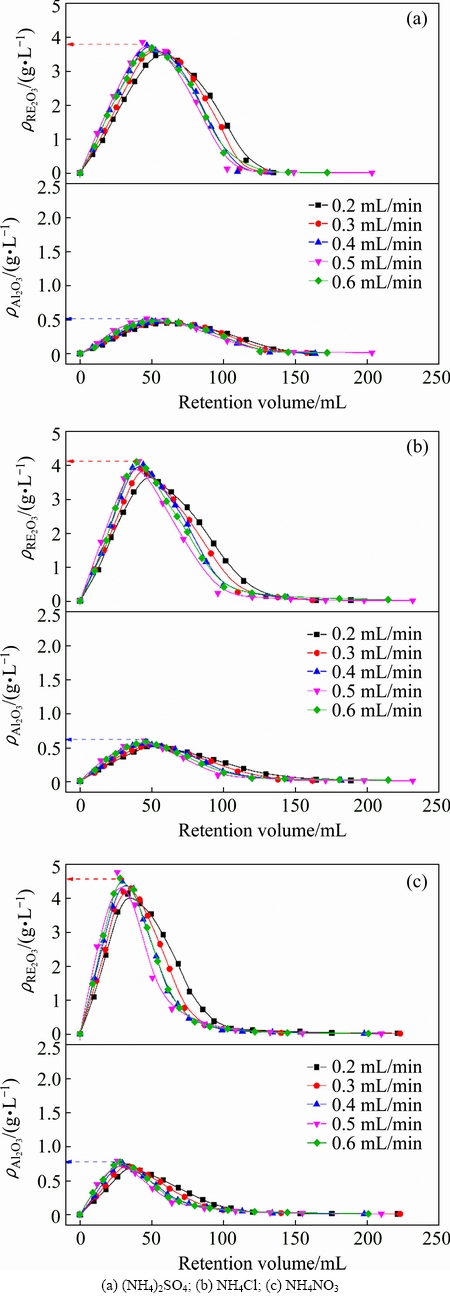
Fig. 4 Leaching curves of RE and Al by ammonium salts under various flow rates (250 g RE ore, 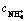 =0.2 mol/L, t=25 °C, initial pH 5.5-6.0)
=0.2 mol/L, t=25 °C, initial pH 5.5-6.0)
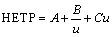 (4)
(4)
where A is the multiple paths diffusion item or eddy diffusion; B is the longitudinal diffusion coefficient; C is the mass transfer impedance coefficient; u is the flow rate of leaching agent. When u<uopt, HETP increases with decreasing the flow rate. B/u plays a leading role in HETP, and Cu can be negligible. The longitudinal diffusion (B/u) mainly decides the mass transfer efficiencies of RE and Al leaching. When u>uopt, HETP decreases with decreasing the flow rate. Cu plays a leading role in HETP, and B/u can be negligible. The mass transfer impedance (Cu) mainly decides the mass transfer efficiency [20]. Figure 5 shows the HETP curves of leaching RE and Al with different ammonium salts under various flow rates of leaching agent. It can be seen obviously that there is an optimum flow rate for a lowest HETP. This is consistent with the van Deemter equation. When the flow rate is less than 0.5 mL/min, the mass transfer efficiencies of RE and Al leaching are controlled by the longitudinal diffusion velocity. HETP decreases with increasing the flow rate. When the flow rate is larger than 0.5 mL/min, the mass transfer efficiency depends on the diffusion rate of the solute between the leaching agent and the ore particle. HETP increases with increasing the flow rate.
The retention volume of RE at the peak concentration is just a little less than that of Al, suggesting the impossibility for the separation of RE and Al in the leaching process under the experiment condition. Among the three ammonium salts, NH4NO3 leads to the narrowest half peak width and the highest peak concentration, while (NH4)2SO4 leads to the broadest half peak width and the lowest peak concentration. Mass transfer efficiencies of RE and Al follow the order of NH4NO3>NH4Cl>(NH4)2SO4. It is in accordance with the results obtained in the discussion of the leaching agent pH and the ammonium concentration.
Table 7 Calculated HETP of RE under various flow rates of leaching agent

Table 8 Calculated HETP of Al under various flow rates of leaching agent
4 Conclusions
1) In the studied pH range of 2 to 8, only the mass transfer efficiency of Al can be improved with pH decreasing from 4 to 2. The strong acidic condition (pH<4) will cause the leachate with high content of impurities due to the dissolution of a large amount of the adsorbed inorganic hydroxyl Al. Ammonium concentration plays a positive role in the mass transfer efficiency by reducing the resistances of diffusion. An excessive concentration cannot enhance the mass transfer process, but increase the cost. The relationship between the flow rate and HETP follows the van Deemter equation. There is an optimum flow rate for a lowest HETP. This indicates that the best leaching condition for all the studied ammonium salts is pH 4-8 of the leaching agent, 0.4 mol/L of ammonium concentration 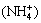 and 0.5 mL/min of flow rate.
and 0.5 mL/min of flow rate.
2) The comparison on the three ammonium salts indicates the HETPs for RE and Al both follow the order of (NH4)2SO4>NH4Cl>NH4NO3. NH4NO3 is the optimal leaching agent for the recovery of RE, while (NH4)2SO4 is the best one for the lowest content of impurities in the leachate.
3) The retention volume of RE at the peak concentration is just a little less than that of Al, suggesting the impossibility for the separation of RE and Al in the leaching process under the experiment condition. The resolution between RE and Al may be improved by a higher packed ore length or/and different leaching agents.
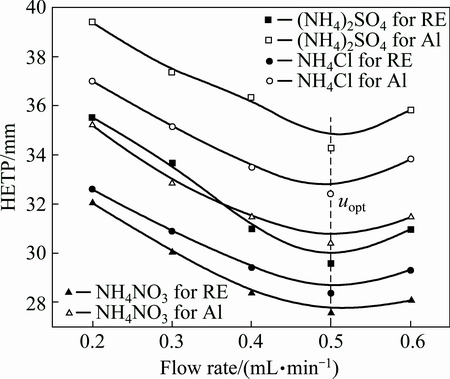
Fig. 5 HETP curves of leaching RE and Al by ammonium salts under various flow rates (250 g RE ore, 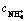 =0.2 mol/L, t=25 °C, initial pH 5.5-6.0)
=0.2 mol/L, t=25 °C, initial pH 5.5-6.0)
References
[1] Chi Ru-an, Tian Jun. Weathered crust elution-deposited rare earth ores [M]. New York: Nova Science Publishers, 2008.
[2] ZHANG Zhen-yue, HE Zheng-yan, XU Zhi-gao, YU Jun-xia, ZHANG Yue-fei, CHI Ru-an. Rare earth partitioning characteristics of China rare earth ore [J]. Chinese Rare Earths, 2016, 37(1): 121-127. (in Chinese)
[3] Nesbitt H W. Mobility and fractionation of rare earth elements during weathering of a granodiorite [J]. Nature, 1979, 279: 206-210.
[4] Chi Ru-an, Tian Jun, Li Zhong-jun, Peng Cui, Wu Yuan-xin, Li Shi-rong, Wang Cun-wen, Zhou Zhi-ang. Existing state and partitioning of rare earth on weathered ores [J]. Journal of Rare Earths, 2005, 23(6): 756-759.
[5] Tian Jun, Yin Jing-qun, Chen Kai-hong, Rao Guo-hua, Jiang Min-tao, Chi Ru-an. Optimization of mass transfer in column elution of rare earths from low grade weathered crust elution-deposited rare earth ore [J]. Hydrometallurgy, 2010, 103: 211-214.
[6] Tian Jun, Yin Jing-qun, Tang Xue-kun, Chen Ji, Luo Xian-ping, Rao Guo-hua. Enhanced leaching process of a low-grade weathered crust elution-deposited rare earth ore with carboxymethyl sesbania gum [J]. Hydrometallurgy, 2013, 139: 124-131.
[7] Tian Jun, Chi Ru-an, Yin Jing-qun. Leaching process of rare earths from weathered crust elution-deposited rare earth ore [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(5): 892-896.
[8] Tian Jun, Yin Jing-qun, Chi Ru-an, Rao Guo-hua, Jiang Min-tao, Ouyang Ke-xian. Kinetics on leaching rare earth from the weathered crust elution-deposited rare earth ores with ammonium sulfate solution [J]. Hydrometallurgy, 2010, 101: 166-170.
[9] HE Zheng-yan, ZHANG Zhen-yue, YU Jun-xia, CHI Ru-an. Behaviors of rare earth, aluminum and ammonium in leaching process of weathered crust elution-deposited rare earth ore [J]. Chinese Rare Earths, 2015, 36(6): 18-24. (in Chinese)
[10] Li Chun. The generalization and application of new technology on lixiviating mineral at the original place for ionic rare earths [J]. Nonferrous Metals Science and Engineering, 2011, 2(1): 63-67. (in Chinese)
[11] Xiao Zhi-zheng, Tang Xun-zhong, Wang Xin-ming, Li Jian-xiong. Study on in-situ leaching stope of deeply soleplate ion-absorbed rare earth [J]. Industrial Minerals & Processing, 2003(12): 9-11. (in Chinese)
[12] Qiu Ting-sheng, Fang Xi-hui, Wu Hong-qiang, Zeng Qing-hua, Zhu Dong-mei. Leaching behaviors of iron and aluminum elements of ion-absorbed-rare-earth ore with a new impurity depressant [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(9): 2986-2990.
[13] Chi Ru-an, Wang Dian-zuo. Rare earth mineral processing [M]. Beijing: Science Press, 2014. (in Chinese)
[14] Xiao Yan-fei, Liu Xiang-sheng, Feng Zong-yu, Huang Xiao-wei, Huang Li, Chen Ying-ying, Wu Wen-yuan. Role of minerals properties on leaching process of weathered crust elution-deposited rare earth ore [J]. Journal of Rare Earths, 2015, 33(5): 545-552.
[15] Yang Xiu-li, Zhang Jun-wei. Recovery of rare earth from ion-adsorption rare earth ores with a compound lixiviant [J]. Separation and Purification Technology, 2015, 142: 203-208.
[16] Moldoveanu G A, Papangelakis V G. Recovery of rare earth elements adsorbed on clay minerals: II. Leaching with ammonium sulfate [J]. Hydrometallurgy, 2013, 131-132: 158-166.
[17] Moldoveanu G A, Papangelakis V G. Recovery of rare earth elements adsorbed on clay minerals: I. Desorption mechanism [J]. Hydrometallurgy, 2012, 117-118: 71-78.
[18] Xiao Yan-fei, CHEN Ying-ying, FENG Zong-yu, HUANG Xiao-wei, HUANG Li, LONG Zhi-qi, CUI Da-li. Leaching characteristics of ion-adsorption type rare earths ore with magnesium sulfate [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(11): 3784-3790.
[19] He Zheng-yan, Zhang Zhen-yue, Yu Jun-xia, Xu Zhi-gao, Chi Ru-an. Process optimization of rare earth and aluminum leaching from weathered crust elution-deposited rare earth ore with compound ammonium salts [J]. Journal of Rare Earth, 2016, 34(4): 413-419.
[20] Tian Jun, Tang Xue-kun, Yin Jing-qun, Chen Ji, Luo Xiao-ping, Rao Guo-hua. Enhanced leachability of a lean weathered crust elution-deposited rare-earth ore: Effects of sesbaniagum filter-aid reagent [J]. Metallurgical and Materials Transactions B, 2013, 44: 1070-1077.
[21] Shimpi R P, Patel H G. A two variable refined plate theory for orthotropic plate analysis [J]. International Journal of Solids and Structures, 2006, 43(22-23): 6783-6799.
[22] Kim S E, Thai H T, Lee J. Buckling analysis of plates using the two variable refined plate theory [J]. Thin-Walled Structures, 2009, 47: 455-462.
[23] Xiao Yan-fei, Feng Zong-yu, Huang Xiao-wei, Huang Li, Chen Ying-ying, Wang Liang-shi, Long Zhi-qi. Recovery of rare earths from weathered crust elution deposited rare earth ore without ammonia-nitrogen pollution: I. Leaching with magnesium sulfate [J]. Hydrometallurgy, 2015, 153: 58-65.
[24] He Zheng-yan, Zhang Zhen-yue, Yu Jun-xia, Zhou Fang, Xu Yuan-lai, Xu Zhi-gao, Chen Zhe, Chi Ru-an. Kinetics of column leaching of rare earth and aluminum from weathered crust elution-deposited rare earth ore with compound ammonium salt [J]. Hydrometallurgy, 2016, 163: 33-39.
[25] Tian Jun, Tang Xue-kun, Yin Jing-qun, Luo Xiao-ping, Rao Guo-hua, Jiang Min-tao. Process optimization on leaching of a lean weathered crust elution-deposited rare earth ores [J]. International Journal of Mineral Processing, 2013, 119: 83-88.
[26] Xiao Yan-fei, Feng Zong-yu, Hu Gu-hua, Huang Li, Huang Xiao-wei, Chen Ying-ying, Li Ming-lai. Leaching and mass transfer characteristics of elements from ion-adsorption type rare earth ore [J]. Rare Metal, 2015, 34(5): 357-365.
[27] van Deemter J J, Zuiderweg F J, Klinkenberg A. Longitudinal diffusion and resistance to mass transfer as causes of nonideality in chromatography [J]. Chemical Engineering Science, 1956, 5(6): 271-289.
何正艳1,张臻悦1,余军霞2,徐志高2,徐源来2,周 芳2,池汝安1, 2
1. 中南大学 资源加工与生物工程学院,长沙 410083;
2. 武汉工程大学 绿色化工过程教育部重点实验室,武汉 430073
摘 要:为了更好地了解风化壳淋积型稀土矿中稀土和铝的浸出过程,运用色谱塔板理论对稀土和铝的柱浸传质过程进行研究。结果表明,在一定范围内,提高铵根离子浓度能强化浸出传质过程。浸取剂pH在2~8的范围内对稀土的浸取传质效率几乎没有影响,而在强酸性条件下(pH<4)能提高铝的浸出传质效率。存在一最佳流速使浸取传质效率最高。稀土的最佳浸取条件如下:浸取剂pH 4~8、铵根离子浓度0.4 mol/L、流速0.5 mL/min。稀土和铝的浸出传质效率由低到高的顺序均为(NH4)2SO4
关键词:柱浸过程;风化壳淋积型稀土矿;稀土;铝;铵盐;传质
(Edited by Wei-ping CHEN)
Foundation item: Projects (51274152, 41472071) supported by the National Natural Science Foundation of China; Project (T201506) supported by the Program for Excellent Young Scientific and Technological Innovation Team of Hubei Provincial Department of Education, China
Corresponding author: Ru-an CHI; Tel: +86-27-87905258; E-mail: rac@wit.edu.cn
DOI: 10.1016/S1003-6326(16)64433-3

