Trans. Nonferrous Met. Soc. China 26(2016) 551-556
Electrochemical behavior of galena and jamesonite flotation in high alkaline pulp
Wei SUN, Chen SUN, Run-qing LIU, Xue-feng CAO, Hong-biao TAO
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 28 March 2015; accepted 13 October 2015
Abstract: In order to effectively separate galena and jamesonite and improve the recovery during the mixing flotation, the interaction mechanisms between the minerals and the collector of diethyl dithiocarbamate (DDTC) were investigated. Single mineral flotation test was organized to research the effect of pulp pH value on the flotation behavior of galena and jamesonite. Electrochemistry property of the interaction of these two minerals with DDTC was investigated by cyclic voltammetry and Tafel tests. Flotation test shows that the recovery of jamesonite in high alkaline pulp is strongly depressed by lime (Ca(OH)2). The cyclic voltammetry and Tafel tests results show that the interaction between galena and DDTC is an electrochemical process. High pH value has little influence on the interaction between galena and DDTC, while it has great effect on jamesonite due to self-oxidation and specific adsorption of OH- and CaOH+ on jamesonite surface. Non-electroactive hydroxyl compound and low-electroconductive calcium compounds cover the surface of jamesonite, which impedes electron transfer and DDTC adsorption, thus leads to very low floatability of jamesonite.
Key words: galena; jamesonite; flotation; electrochemical behavior; cyclic voltammetry; Tafel test
1 Introduction
Galena and jamesonite are respectively important sources of antimony and lead in China. In the traditional process, the mixed concentrate of jamesonite and galena is sent directly to the smelter for separation, which is with high energy consumption, heavy pollution, and not in conformity with the requirement of economical and environment-friendly society. Galena associated with jamesonite has poor floatability [1], because the floatability of jamesonite is similar to that of galena [2,3]. To get a better understanding of collector-mineral interaction mechanism, flotation property and electrochemical behavior of these minerals, flotation test and flotation electrochemistry are essential. Meanwhile, the tests results will provide foundations for flotation separation and recovery increase. But few electrochemical investigations have been devoted to the separation of galena and jamesonite by flotation.
Researchers studied the mechanism of flotation reagents on galena, pyrite and other sulfide minerals by applying various electrochemical techniques. CHEN et al [4] calculated the electronic structures of PbS with vacancy defects, and concluded that the movement states of electrons are changed due to vacancy defects, which influenced the flotation behaviors of galena. LAN [5] studied the effects of impurity on electrochemical oxidation, collecting and depression of galena by cyclic voltammetry. From the cyclic voltammetry curves, the depression effect of calcium hydroxide on doping galena is stronger than that of sodium hydroxide. ZHANG et al [6] studied the influences of lime and sodium hydroxide on the flotation behaviors of pyrite. In alkaline conditions, nonconductive material and adsorption of calcium membrane appear on pyrite��s surface, which is why lime has stronger depression effect on the pyrite flotation than sodium hydroxide. QIN et al [7] concluded that pulp potential was an important parameter for sulfide ore flotation process. The regulation of pulp potential can change redox properties of the system, thus the flotation characteristics of the entire system can be improved. WANG et al [8] concluded that in strong alkaline media, lime was not only regulator of pH, but also could stabilize special potential very well, and this special potential was exactly flotation potential of galena. GU et al [9] concluded that when pH>12.5 and ��h<0.17 V, S0 is excessive due to HPbO2- dissolution and oxidation on the surface of galena, which promotes the floatability of galena. The interfacial electrochemical behaviors of the interaction of marmatite were studied by Tafel tests and impedance measurements in Ref. [10]. With the rising of pH, the corrosive current rises, and hydrophobic sulfur on mineral surface decreases rapidly and hydroxylation becomes obvious. HUANG and SUN [11] indicated that the adsorption of 2-aminothiophenol onto jamesonite was physical adsorption. ZHANG et al [12] found that by using calcium oxide as regulator, jamesonite had low floatability at pH>12. They also indicated that pulp potential and pH value had a significant influence on flotation behavior of jamesonite [13]. With sodium diethyl dithiocarbamate (DDTC) as a collector, lead diethyl dithiocarbamate is generated on jamesonite��s surface as major adsorption product, shown by FTIR spectroscopy analysis. Also, ZHANG et al [14] studied electrochemical flotation behavior of jamesonite with ethyl xanthate, DDTC and ammonium dibuyldithio- phosphate (ADDP) as collectors, respectively. LI et al [15] investigated flotation performance of jamesonite in DDTC solution using cyclic voltammetry method, and concluded that PbD2 and SbOD formed on jamesonite by chemical adsorption. YU et al [16] found that when electrode potential is 178-472 mV, DDTC, PbD2 and S0 will be adsorbed on jamesonite; when electrode potential is higher than 472 mV, they will be oxidized into Pb2+, SO32- and SO42-. Under these conditions, the hydrophilic hydroxyl precipitate occurs on jamesonite surface, so that its collector-less floatability is poor [17]. YU et al [18,19] also used cyclic voltammetry method to investigate interaction mechanism of jamesonite with diethyl dithiocarbamate and diethyl xanthate as collectors, and found that at around the corrosive potential, collector-metal ions passivated on the electrode surface, therefore, this process drastically depressed corrosive reactions on mineral surface.
In this work, the electrochemical mechanisms of galena and jamesonite with DDTC as a collector under high alkaline pulp were investigated by using pH modifier, cyclic voltammetry and Tafel tests.
2 Experimental
2.1 Materials
The galena and jamesonite used in this work were obtained respectively from Fankou Mine, Guangdong Province, China, and Dachang No. 100 ore body, Guangxi, China. Mineral lumps were crushed and mineral grains of several millimeters in diameter were handpicked and were ground in a ceramic ball mill. Then, fractions with sizes of 0.074-0.038 mm were obtained by screening as flotation samples.
2.2 Electrodes
Four kinds of electrode were used in this work. A graphite piece served as the counter electrode, while an Ag/AgCl electrode served as the reference electrode. The galena electrode was prepared from pure crystal, which was picked out from this mineral sample. It was gently polished on the abrasive papers of different types step by step into smooth face, and then washed with acetone and put in specialized electrode sleeve. Another working electrode was a kind of carbon paste electrode consisted of 82.5% (mass fraction) of particles (less than 0.147 mm) of jamesonite, 10% (mass fraction) of graphite powder and 7.5% (mass fraction) of paraffin. The graphite power was spectrum grade. In order to renew the working surface, the working electrode was gently polished with 600 grit SiC papers and then rinsed with distilled water during every measurement. Experimental device was the potentiostat/galvanostat model 283A from EG & PAR Company.
2.3 Flotation tests
Flotation test was conducted in a 40 mL XFG type hitch groove flotation machine at a rotating speed of 1800 r/min. In flotation process, 2.0 g mineral sample was added into a TCX-SOW type ultrasonic cleaner in order to remove the surface oxide, and then the sample was transferred into the flotation cell for further processing. The water used in the process was distilled water. The sequence of reagent addition was pH regulator, collector and frother. The conditioning time for collector and frother was 2 min. The flotation time was 4 min. The flotation recovery (R) was calculated from R=m1/(m1+m2)��100%, where m1 and m2 were masses of the floated and un-floated fractions, respectively.
3 Results and discussion
3.1 Effect of pulp pH value
The effect of regulator on the flotation recovery of galena at pH 10 and 12 is given in Fig. 1. It shows that the flotation recovery decreased slightly with lime (Ca(OH2)) as regulator, compared with that with NaOH as regulator. But overall, high pH value has little influence on the floatability of galena.
Figure 2 shows that flotation recovery of jamesonite falls more sharply in high alkali pulp than that of galena under the same condition. At pH value of 12, the recovery of jamesonite is only 39%, suggesting that lime has a strong depressive effect on jamesonite.
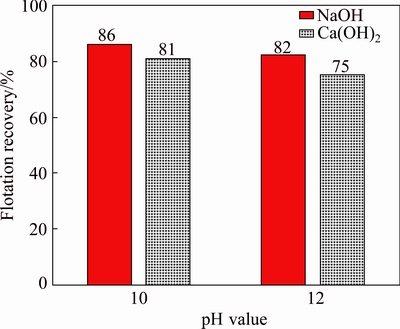
Fig. 1 Effect of pH value on flotation recovery of galena with NaOH and Ca(OH)2 as regulators at 25 ��C and DDTC concentration of 1��10-4 mol/L

Fig. 2 Effect of pH value on recovery of jamesonite with NaOH and Ca(OH)2 as regulators at 25 ��C and DDTC concentration of 1��10-4 mol/L
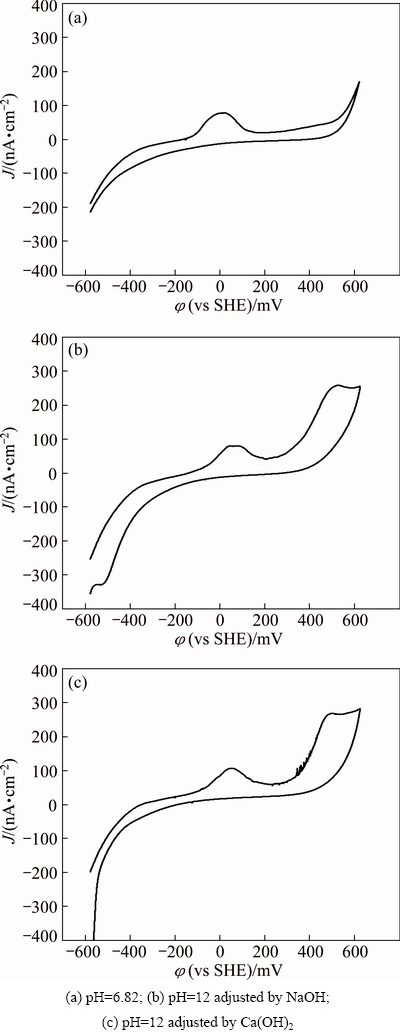
Fig. 3 Cyclic voltammogram curves of galena with DDTC as collector at scan rate of 20 mV/s, 25 ��C, KNO3 concentration of 0.1 mol/L and DDTC concentration of 1��10-4 mol/L under different pH values

Fig. 4 Cyclic voltammogram curves of jamesonite with DDTC as collector at scan rate of 20 mV/s, 25 ��C, KNO3 concentration of 0.1 mol/L and DDTC concentration of 1��10-4 mol/L under different pH conditions
3.2 Cyclic voltammogram
The cyclic voltammogram curves of galena under natural pH condition, pH 12 adjusted by NaOH and by lime respectively are presented in Fig. 3. It can be seen from Fig. 3 that anodic peak emerges at a potential of about 0. These phenomena may correspond to the following reaction:
PbS+2D-=PbD2+S+2e (1)
Galena itself can be oxidized to Pb2+ and S at pH 6.82. As the generation of PbD2 in the system occurs simultaneously with oxidation of galena, there may have another reaction:
Pb2++2D-=PbD2 (2)
The anodic oxidation peaks of galena and PbD2 almost overlap in high alkali pump with low scanning potential. The oxidation product of galena is HPbO2- and S, so, the reaction of generation of PbD2 is
HPbO2-+2D-+3H+=PbD2+2H2O (3)
As positive scanning processes, anodic peaks of PbD2 and galena are separated, PbD2 can be produced according to the following reaction as oxidative product of galena is S2O32-.
2PbS+3H2O+4D-=2PbD2+S2O32-+6H++8e (4)
The cyclic voltammogram curves of jamesonite under the same condition are presented in Fig. 4. It is shown in Fig. 4 that an anodic current peak emerges at potential of about 0 when DDTC is added. PbS occurs on jamesonite surface under neutral and highly alkaline conditions. Here are the corresponding reactions:
Pb4FeSb6S14+8D-+18H2O=6SbO3-+4PbD2+Fe3++14S0+36H++41e (5)
Pb4FeSb6S14+8D-+21H2O=6SbO3-+4PbD2+Fe(OH)3+14S0+39H++41e (6)
After collector was added, the current densities of oxidation peak and cathodic reduction peak declined, indicating that the inactivation occurred. And Fe3+ and Fe(OH)3 were produced according to Eqs. (5) and (6), respectively. Compared with the natural pH, high alkaline conditions have a sharper decline, which might result from the fact that the hydrophilic ferric hydroxide covered the surface of jamesonite electrode [3,12,20], thus hindering the collector to react with mineral particles.
3.3 Tafel curves
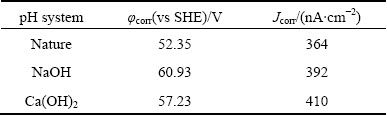
The Tafel curves and galvanic corrosion parameters of galena in electrolyte solution are presented in Fig. 5 and Table 1, with DDTC as collector and under natural pH condition, pH 12 fixed by NaOH and pH 12 fixed by lime, respectively. As can be seen from the figure, when DDTC is added, corrosion current of galena declined drastically at the same degree under different conditions. This is due to the high alkaline system and the system of high alkaline calcium has little effect on the interaction between DDTC and galena. DDTC generated sodium diethyl dithiocarbamate which is hydrophobic, covering the surface of galena and inhibit its oxidation.
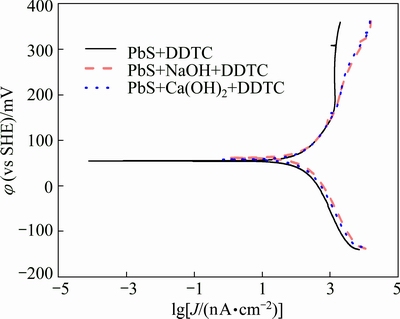
Fig. 5 Tafel curves of galena with DDTC as collector at scan rate of 20 mV/s, 25 ��C, KNO3 concentration of 0.1 mol/L and DDTC concentration of 1��10-4 mol/L under different pH systems
Table 1 Electrochemical corrosion parameters of galena under different pH systems with DDTC as collector
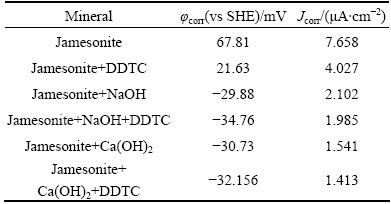
The Tafel curves and galvanic corrosion parameters of jamesonite in electrolyte solution are presented in Fig. 6 and Table 2, with DDTC as collector and under natural pH condition, pH 12 adjusted by NaOH and pH 12 adjusted by Ca(OH)2, respectively. Figure 6 shows that under natural pH condition, corrosion potential of the system reduces from 67.81 to 21.63 mV. The corrosion current density decreases from 7.658 to 4.027 ��A/cm2, which means that DDTC is absorbed on the surface of jamesonite. But changes of corrosion potential and corrosion current are less than those of galena, indicating that DDTC has stronger collecting capacity to galena than to jamesonite under the same condition.
At high pH value, with addition of the collector, there is no obvious change in the corrosion potential and corrosion current density, while they decline more slightly in high alkaline system than that in high alkaline calcium system. This may result from the adsorption and coverage of hydroxyl compounds and calcium compounds [3,12,16]. The interaction between DDTC and jamesonite is very weak at high pH value. This can be the foundation of separating galena and jamesonite with DDTC under high alkaline calcium system.
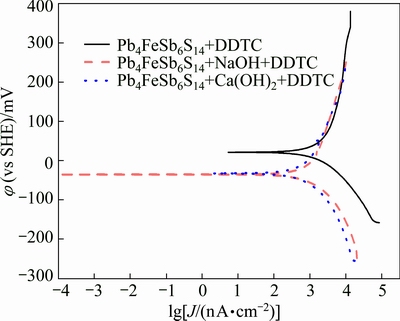
Fig. 6 Tafel curves of jamesonite with DDTC as collector at scan rate of 20 mV/s, 25 ��C, KNO3 concentration of 0.1 mol/L and DDTC concentration of 1��10-4 mol/L under different pH systems
Table 2 Electrochemical corrosion parameters of jamesonite under different pH systems with DDTC as collector
4 Conclusions
1) Pulp pH has a significant influence on the flotation of galena and jamesonite. In high alkaline condition adjusted by lime, better separation between galena and jamesonite is obtained than that in high alkaline condition adjusted by NaOH.
2) Through potential scan of cyclic voltammograms and analysis of Tafel tests, it can be concluded that electrochemical reactions play an important role in the interaction of DDTC with galena and jamesonite. High pH value has little effect on electrochemical property on galena surface and the interaction between galena and DDTC. As to jamesonite, due to self-oxidation and specific adsorption of OH- and CaOH+, non- electroactive hydroxyl compound and low-electrical- conductive calcium compounds are generated on the jamesonite surface, hindering the transfer of the electrons on the surface of the electrode. Thus, the adsorption of DDTC on jamesonite surface is difficult, leading to the drastic decline of floatability of jamesonite.
References
[1] SUN Wei, HAN Hai-sheng, TAO Hong-biao, LIU Run-qing. Study on the flotation technology and adsorption mechanism [J]. International Journal of Mining Science and Technology, 2015, 25: 53-57.
[2] CHEN Jian-hua, LI Yu-qiong, LONG Qiu-rong, WEI Zong-wu, CHEN Ye. Improving the selective flotation of jamesonite using tannin extract [J]. International Journal of Mineral Processing, 2011, 100: 54-56.
[3] ZHAO Cui-hua, CHEN Jian-hua, LI Yu-qiong, HE Qian, WU Bo-zeng. Electronic structure and flotation behavior of complex mineral jamesonite [J]. The Chinese Journal of Nonferrous Metals, 2015, 25(3): 590-596. (in Chinese)
[4] CHEN Jian-hua, WANG Lei, CHEN Ye, LI Yu-qiong, GUO Jin. Density functional theory of effects of vacancy defects on electronic structure and flotation of galena [J]. The Chinese Journal of Nonferrous Metals, 2010, 20(9): 1815-1821. (in Chinese)
[5] LAN Li-hong. The effect of lattice defect of galena on the surface property, molecular absorption of flotation reagents and electrochemical behavior [D]. Nanning: Guangxi University, 2012. (in Chinese)
[6] ZHANG Ying, QIN Wu-lin, SUN Wei, HE Guo-yong. Electrochemical behaviors of pyrite flotation using lime and sodium hydroxide as depressantors [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(3): 675-679. (in Chinese)
[7] QIN Wen-qing, YAO Guo-cheng, GU Guo-hua, QIU Guan-zhou, WANG Dian-zuo. Electrochemistry of sulfide minerals and its floatability [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(10): 2669-2677. (in Chinese)
[8] WANG Dian-zuo, GU Guo-hua, LIU Ru-yi. Potential adjustment flotation of galena�Clime-diethyldithiocarbamate system [J]. The Chinese Journal of Nonferrous Metals, 1998, 8(2): 322-326. (in Chinese)
[9] GU Guo-hua, HU Yue-hua, QIU Guan-zhou, WANG Hui, WANG Dian-zuo. Electrochemistry of galena in hight alkaline flotation [J]. Mining and Metallurgical Engineering, 2002, 22(1): 52-55. (in Chinese)
[10] YU Run-lan, QIU Guan-zhou, HU Yue-hua, QIN Wen-qing. The corrosive electrochemical study of marmatite [J]. Journal of Chinese Society for Corrosion and Protection, 2004, 24(4): 226-229. (in Chinese)
[11] HUANG Hong-jun, SUN Wei. Effect of 2-aminothiophenol on the separation of jamesonite and marmatite [J]. Mining Science and Technology, 2010, 20: 425-427. (in Chinese)
[12] ZHANG Qin, HU Yue-hua, GU Guo-hua, XU Jing. Selective flotation separation of jamesonite from pyrrhotite by lime [J]. Mining and Metallurgical Engineering, 2004, 24(2): 30-32. (in Chinese)
[13] ZHANG Qin, HU Yue-hua, XU Jing, CHEN Tie-jun. FTIR spectroscopic study of electrochemical flotation of jamesonite- diethyldithiocarbamate system [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(3): 493-496.
[14] ZHANG Qin, HU Yue-hua, XU Jing, HU Ding-guo. Study on electrochemical behavior of jamesonite-collector systems [J]. Metal Mine, 2006, 7: 33-36. (in Chinese)
[15] LI Wei-zhong, QIN Wen-qing, QIU Guan-zhou, DONG Qing-hai. Electrochemical behaviors and floatation performance of jamesonite in diethyldithiocarbamate solution [J]. Mining and Metallurgical Engineering, 2006, 26(2): 42-44. (in Chinese)
[16] YU Run-lan, HU Yue-hua, QIU Guan-zhou, QIN Wen-qing. An electrochemical study of DDTC adsorption on jamesonite [J]. Electrochemistry, 2004, 10(2): 145-152.
[17] QIU Guan-zhou, YU Run-lan, HU Yue-hua, QIN Wen-qing. Corrosive electrochemistry of jamesonite [J]. Transactions of Nonferrous Metals Society of China, 2004, 14(6): 1169-1173.
[18] YU Run-lan, HU Yue-hua, QIU Guan-zhou, QIN Wen-qing. Interaction mechanism of jamesonite with flotation collectors by cyclic voltammetry [J]. Journal of Central South University: Science and Technology, 2004, 35(2): 201-205. (in Chinese)
[19] YU Run-lan, HU Yue-hua, QIU Guan-zhou, QIN Wen-qing. Interface electrochemistry of interaction of collector with jamesonite [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(1): 127-131. (in Chinese)
[20] YU Run-lan, HU Yue-hua, QIU Guan-zhou. QIN Wen-qing. Electrochemistry of jamesonite in system of diethyl dithiocarbamate and saturated Ca(OH)2 [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(10): 1763-1769. (in Chinese).
��Ǧ���������Ǧ���ڸ���ϵ�еĸ�ѡ�绯ѧ��Ϊ
�� ΰ���� ���������壬��ѧ�棬�պ��
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ��Ϊ��Ч���뷽Ǧ��ʹ�����Ǧ��������ڻ�ϸ�ѡ�еĻ����ʣ��о������������ռ�(DDTC)�����û�����ͨ�������︡ѡʵ���о���pHֵ�����߸�ѡ��Ϊ��Ӱ�졣ͨ��ѭ�������������ƶ����߲����о��������������õĵ绯ѧ���ʡ���ѡʵ�����������ڸ���ϵ�д�����Ǧ��ʯ��ǿ�����ơ�ѭ���������ߺ����ƶ����߱����������뷽Ǧ��ʹ�����Ǧ��������ǵ绯ѧ���̡�����ϵ�Է�Ǧ������������Ӱ�첻���ǶԴ�����Ǧ��Ӱ��ܴ����������������Լ�OH-��CaOH+�����ӵ��������������·ǵ���Ե��ǻ�������͵͵����Եĸ�ϵ�����︽��������棬�谭�����ڵ缫����Ĵ��ݣ��Ӷ�ʹ������Ǧ��Ŀɸ��Խ��͡�
�ؼ��ʣ���Ǧ������Ǧ��ѡ���绯ѧ��Ϊ��ѭ�������������ƶ�����
(Edited by Wei-ping CHEN)
Foundation item: Projects (51104179, 51374247) supported by the National Natural Science Foundation of China
Corresponding author: Run-qing LIU; Tel: +86-13875851194; E-mail: liurunqing@126.com
DOI: 10.1016/S1003-6326(16)64114-6