
Preparation and characterization of silica microcapsules containing butyl-stearate via sol-gel method
MIAO Chun-yan(缪春燕)1, 2, YAO You-wei(姚有为)1, TANG Guo-yi(唐国翌)1,WENG Duan(翁 端)2
1. Graduated School at Shenzhen, Tsinghua University, Shenzhen 518055, China;
2. Department of Materials Science and Engineering, Tsinghua University, Beijing 100086, China
Received 15 July 2007; accepted 10 September 2007
Abstract: For thermal energy storage application in energy-saving building materials, silica microcapsules containing phase change material were prepared using sol-gel method in O/W emulsion system. In the system droplets in microns are formed by emulsifying an organic phase consisting of butyl-stearate as core material. The silica shell was formed via hydrolysis and condensation from tetraethyl silicate with acetate as catalyst. The SEM photographs show the particles possess spherical morphology and core-shell structure. The as-prepared silica microcapsules mainly consist of microsphere in the diameter of 3-7 μm and the median diameter of these microcapsules equals to 5.2 μm. The differential scanning calorimetry (DSC) curves indicate that the latent heat and the melting point of microcapsules are 86 J/g and 22.6 ℃, respectively. The results of DSC and TG further testify the microcapsules with core-shell structure.
Key words: silica microcapsules; sol-gel; butyl-stearate; phase change materials
1 Introduction
Increasing energy cost and associated environmental problems have intensified efforts towards the energy storage and sustainable energy technologies. Over the past decade, the integration of phase change materials (PCMs) into building fabrics have been investigated as a potential technology for minimizing energy consumptions in buildings because PCMs allow large amounts of heat to be stored during their melting process and to be released during their solidifying process[1-6]. Butyl-stearate (BS) as a kind of PCMs with moderate energy-storing density, proper melting point and economic price has been studied in buildings.
A laboratory scale energy-storing gypsum wallboard was produced by the direct incorporation of 21%-22%(mass fraction) commercial grades BS at the mixing stage of conventional gypsum board production. Compared with gypsum wallboard alone, the energy-storing capability of this PCM wallboard has a tenfold increase in capacity for the storage and release of heat[7]. ZHANG et al[8-10] produced the PCMs clay which contained BS as PCM and expanded perlite as matrix via penetrating method. In recent years, microencapsulation of PCM has been studied and applied in thermal energy fields due to its advantages, such as protection of the core materials, increasing the heat transfer area, and permitting the core material to withstand frequent changes in volume when the PCMs change their state from solid to liquid or vice versa. Many microencapsulation methods have been developed for paraffin[11-13], such as interfacial polymerization, polymerization in situ, and sol-gel, but micro- encapsulation of BS has not been reported. Microcapsules with silica as shell materials possess hydrophilic surface and anti-oxidization property compared with organic polymer microcapsules, and silica microcapsules with paraffin as PCM have been prepared via sol-gel method from O/W emulsion[14-15].
In this study, spherical microcapsules with silica as shell materials and BS as core materials are successfully prepared from an O/W emulsion in the presence of polyvinyl alcohol (PVA) as stabilizer, and sorbitan monooleate (Span80) and polyoxyethylene(20) sorbitan monooleate (Tween80) as emulsifiers.
2 Experimental
The particle size and surface morphology of silica microcapsules were examined using a scanning electron microscope (S-4700). The particle size distribution was measured adapting particle size analyzer (Rise-2008). Thermogravimetry (TG) analysis was carried out on TA-2. Differential scanning calorimetry (DSC) curves were evaluated using DSC (Mettler Toledo, DSC823e) between the scales of 0-50 ℃ at a heating or cooling rate of 5 ℃/min and under nitrogen atmosphere.
A typical microencapsulation procedure was carried out as follows: 1) 1.0 g of PVA was dissolved in 49.0 mL of distilled water; 2) an organic solution of 8 mL of BS and 1.5 g of mixed emulsifiers (45.0% Span 80 and 55.0% Tween 80) was prepared, then the organic solution was heated to 80 ℃; 3) maintaining the temperature of reaction system between 85 ℃ and 90 ℃, 10 g of PVA aqueous solution was added to the organic solution, and the mixture was emulsified mechanically at stirring rate of 300 r/min for 10 min, then the remains of PVA aqueous was added to the mixture and the mixture was emulsified at 600 r/min to form an O/W emulsion; 4) while stirring, 1.0 g of sodium chloride solution (2.5 mol/L) was added into the emulsion; 5) after stirring for 30 min, 8 mL of tetraethyl silicate (TEOS) and 0.2 g of acetate acid solution (10.0%) were slowly added into the emulsion system to start the hydrolysis and condensation of TEOS; 6) after the addition, the reaction mixture was cooled to 55.0 ℃ for 3 h. The resultant microcapsules were centrifuged, washed with distilled water and dried at 55.0 ℃ in oven for 24 h.
3 Results and discussion
3.1 Morphology of microcapsules
The SEM photographs of silica microcapsules are shown in Fig.1. From Fig.1 (a), it is clear that the as-prepared silica microcapsules mainly consist of microsphere in diameter of 3-7 μm. The SEM photograph of the fractured microcapsules in Fig.1(b) illustrates the core/shell structure of microcapsules. According to the particle size distribution of silica microcapsules containing BS (shown in Fig.2), the median diameter of silica microcapsules is 5.2 μm and the diameter of 95% silica microcapsules is not more than 13 μm.
3.2 TG analysis
The thermal gravimetry (TG) curves of BS and microcapsules are shown in Fig.3. The temperature of initial mass loss (5%) of BS is 170 ℃ and the mass loss ends at 235 ℃, while the initial mass loss of micro-

Fig.1 SEM micrographs of silica microcapsules containing BS
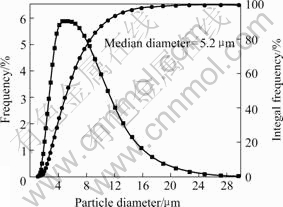
Fig.2 Particle size distribution of silica microcapsules containing BS
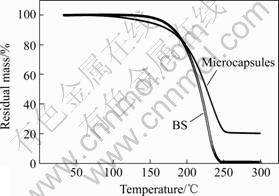
Fig.3 TG curves of BS and microcapsules containing BS
capsules is different from that of the BS. In the temperature range of 100-170 ℃, the obvious mass loss (about 9%) of microcapsules appears, which is due to the water absorbed by silica gel, and the maximum mass loss of microcapsules is 78% at 250 ℃. This result, together with the SEM observation, verifies the formation of microcapsules with core-shell structure and the average content of core materials is about 69%(mass fraction).
3.3 DSC curves of microcapsules
Fig.4 shows the typical melting and solidifying curves of DSC for BS alone and silica microcapsules containing BS. As shown in Fig.4, BS and silica microcapsules exhibit similar thermal properties; the ΔHf of BS and silica microcapsules are determined to be 132 J/g and 86 J/g, and the melting points of BS and microcapsules are 22.6 and 23.0 ℃, respectively.
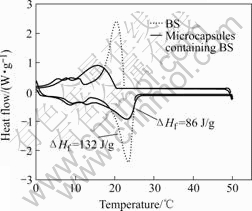
Fig.4 DSC curves of BS and microcapsules containing BS
The average content of BS in a silica microcapsule can be estimated by dividing the ΔHf of microcapsules by the ΔHf of BS alone, assuming that the energy of core materials does not change before and after microencapsulation. Accordingly, the average content of BS in microcapsules is 65%, which is basically in accordance with TG analysis.
It remains to identify the micro-shell formation mechanism. In this study, nonionic surfactant is enriched at the oil-water interface and contributes to the stabilization of this emulsion; sodium chloride is added into the emulsion and the Na+ ions interact with the oxygen atom of the ethylene oxide group of the nonionic surfactant to form the complex Span80-Na+ and Tween80-Na+[16], which increases the volume of terminal hydrophilic group of the surfactants, brings the hydrophilic group of surfactants a spot of positive charge and contributes to the oil in water emulsion more stable. With acetic acid as a catalyst, on the one hand, microcapsules possess the lowest porosity among catalyst (HCl, H2SO4, HNO3, HF, NH3, HAc )[17], on the other hand, in acid condition, atomic group such as ―OH, ―OSi≡ can instabilize the positive charge around Si nucleus or increase the steric hindrance, which reduces the hydrolysis rate[18]. Under these conditions, the hydrolysis rate is smaller than the condensation rate. The oligomer of TEOS exists in the system primarily with the form of Si(OR)2(OH)2 or Si(OR)3OH that are both lipophilic and hydrophilic, and they are apt to gather around the oil droplets in emulsion. Thus, silica micro-shell formation is expected.
4 Conclusions
1) Silica microcapsules encapsulating BS as PCM are prepared via a combination of O/W emulsion technique with a sol-gel method.
2) Micron size (3-7 μm) spherical silica capsules containing BS can be prepared from weak acidic solution by using nonionic surfactant as the emulsifiers, PVA as stabilizer and TEOS as silica resource.
3) The microcapsule has a relatively higher energy-storing density of 86 J/g and proper melting point of 22.6 ℃.
References
[1] KHUDHAIR A M, FARID M M. A review on energy conservation in building applications with thermal storage by latent heat using phase change materials[J]. Energy Conversion and Management, 2004, 45: 263-275.
[2] DARKWA K. Evaluation of regenerative phase change drywalls: low-energy buildings application[J]. Int J Energy Res, 1999, 23: 1205-1212.
[3] AHMET K. Energy storage applications in greenhouse by means of phase change materials (PCMs): a review[J]. Renewable Energy, 1998, 13(1): 89-103.
[4] HALAWA E, BRUNO F, SAMAN W. Numerical analysis of a PCM thermal storage system with varying wall temperature[J]. Energy Conversion and Management, 2005, 46(15/16): 2592-2604.
[5] ZALBA B, MARIN J M, CABEZA L F, MEHLING H. Review on the thermal energy storage with phase change: materials, heat transfer analysis and applications[J]. Appl Therm Eng, 2003, 23(3): 251-283.
[6] SCHOSSIG P, HENNING, GSCHWANDER S, HAUSSMANN T. Micro-encapsulated phase-change materials integrated into construction materials[J]. Solar Energy Material and Solar Cells, 2005, 89: 297-306.
[7] FELDMAN D, BANU D. Obtaining an energy storing building material by direct incorporation of an organic phase change material in gypsum wallboard[J]. Solar Energy Mater, 1991, 22: 231-242.
[8] ZHANG Dong, ZHOU Jian-min, WU Ke-ru, LI Zong-jin. Granulated phase change composite for energy storage[J]. Acta Material Composite Sinica, 2004, 21(5): 103-107. (in Chinese).
[9] ZHANG Dong, ZHOU Jian-min, WU Ke-ru, LI Zong-jin. Study on fabrication method and energy-storing behavior of phase-changing energy-storing concrete[J]. Journal of Building Materials, 2003, 6(4): 374-377.(in Chinese).
[10] ZHOU Jian-min, ZHANG Dong, WU Ke-ru. Experiment study and analysis on obtaining and energy storing composite material by direct incorporating organic phase change materials into porous granule[J]. Energy Conservation Technology, 2003, 21(6): 5-7.
[11] ZOU G L, LAN X Z, TAN Z C, SUN L X. Microencapsulation of n-hexadecane as phase change material in polyurea[J]. Acta Phys-Chim Sin, 2004, 20: 90-93.
[12] HAWLADER M N A, UDDIN M S, KHIN M M. Microencaopsulated PCM thermal-energy storage system[J]. Applied Energy, 2003, 74: 195-202.
[13] ZHANG X X, FAN Y F, TAO X M, YICK K L. Fabrication and properties of microcapsules and nanocapsules containing n-octadecane[J]. Materials Chemistry and Physics, 2004, 88: 300-307.
[14] WANG L Y, TSAI P S, YANG Y M. Preparation of silica microspheres encapsulating phase-change material by sol-gel method in O/W emulsion[J]. Journal of Microencapsulation, 2006, 23(1): 3-14.
[15] MIAO C Y, LU G, YAO Y W, TANG G Y, WENG D. Preparation of silica microcapsules containing octadecane as temperature adjusting powder[J]. Chemistry Letters, 2007, 36(4): 494-495.
[16] MATSUBARA H, OHTA A, KAMEDA M, VILLENEUVET, IKEDA N, ARATONO M. Interaction between ionic and nonionic surfactants in the adsorbed film and micelle: hydrochloric acid, sodium chloride, and tetraethylene glycol monooctyl ether[J]. Langmuir, 1999, 15: 5496-5499.
[17] POPE E J A, MACKENZIE J D. Sol-gel processing of silica Ⅱ. The role of the catalyst[J]. J Non-Cryst Solids 1986, 87: 185-198.
[18] LIN J. The effect of catalysts on TEOS hydrolysis-condensation mechanism[J]. Journal of Inorgic Materials, 1997, 12(3): 363-369.(in Chinese)
(Edited by YUAN Sai-qian)
Foundation item: Project(50572045) supported by the National Natural Science Foundation of China; project supported by Innovation Fund from the PetroChina Company Limited
Corresponding author: TANG Guo-yi; Tel: +86-755-26036752; E-mail: Tanggy@mail.tsinghua.edu.cn