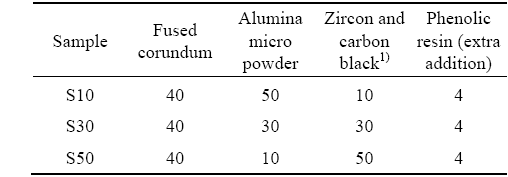
Preparation of β-Sialon/ZrN bonded corundum composites from zircon by nitridation reaction sintering process
来源期刊:中南大学学报(英文版)2009年第5期
论文作者:马北越 于景坤 刘涛 颜正国
文章页码:725 - 729
Key words:β-Sialon; ZrN; zircon; corundum; composites; nitridation reaction sintering process
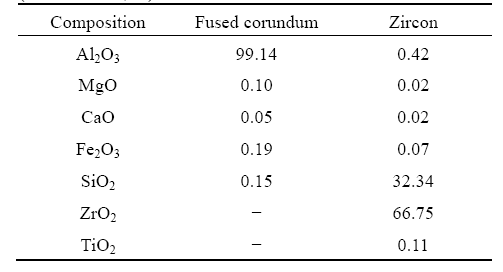
Abstract: β-Sialon/ZrN bonded corundum composites were synthesized using fused white corundum, alumina micro powder, zircon and carbon black by nitridation reaction sintering process. Phase composition and microstructure of the synthesized composites were investigated by X-ray powder diffraction and scanning electronic microscope, and the formation process of the composites was discussed. The results show that the composites with different compositions can be obtained by controlling the heating temperature and contents of zircon and carbon black. The proper temperature to synthesize the composites is 1 773 K.
基金信息:the National Natural Science Foundation of China and Baoshan Iron and Steel Co., Ltd
J. Cent. South Univ. Technol. (2009) 16: 0725-0729
DOI: 10.1007/s11771-009-0120-5
![]()
3.1 Thermodynamic analysis
During the synthesis process of the β-Sialon(z=3)/ ZrN bonded corundum composites, the following reactions are given as:
ZrSiO4(s)=ZrO2(s)+SiO2(s) (2)
3Al2O3(s)+6SiO2(s)+15C(s)+5N2(g)=
2Si3Al3O3N5(s)+15CO(g) (3)
2ZrO2(s)+4C(s)+N2(g)=2ZrN(s)+4CO(g) (4)
Al2O3(s)+3C(s)+N2(g)=2AlN(s)+3CO(g) (5)
3Al2O3(s)+6ZrSiO4(s)+15C(s)+5N2(g)=
6ZrO2(s)+2Si3Al3O3N5(s)+15CO(g) (6)
3Al2O3(s)+6ZrSiO4(s)+27C(s)+8N2(g)=
6ZrN(s)+2Si3Al3O3N5(s)+27CO(g) (7)
4Al2O3(s)+6ZrSiO4(s)+30C(s)+9N2(g)=
2AlN(s)+6ZrN(s)+2Si3Al3O3N5(s)+30CO(g) (8)
According to the correlated thermodynamic data [15], the standard Gibbs free energies for reactions (3)-(5), the expression formulas between partial pressure of CO gas (p(CO)) at p(N2)=![]() 0.1 MPa and temperature can be obtained respectively as follows.
0.1 MPa and temperature can be obtained respectively as follows.
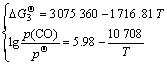 (9)
(9)
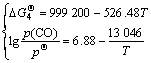 (10)
(10)
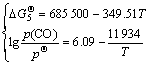 (11)
(11)
Fig.1 shows the equilibrium relationship of the condensed phases in Al2O3-ZrO2-SiO2-C-N2 system plotted by the thermodynamic data as shown in formulas (9)-(11), which indicates that when p(CO) in the reacting furnace remains constant, with the increase of the heating temperature, C in sample will react with SiO2 and N2 to form Si3Al3O3N5 (reaction (3)), and the stability domain of the condensed phases changes from Al2O3(s)+ZrO2(s)+SiO2(s)+C(s) to Al2O3(s)+ZrO2(s)+ Si3Al3O3N5(s)+C(s) and AlN(s)+ZrN(s)+Si3Al3O3N5(s)+ C(s). For example, when p(CO) is 0.1 MPa, the initial temperatures to form Si3Al3O3N5, ZrN and AlN are about 1 791, 1 898 and 1 961 K, respectively. In addition, when the temperature remains constant, with the decrease of the partial pressure of CO gas, the stability domain will change from Al2O3(s)+ZrO2(s)+SiO2(s)+C(s) to Al2O3(s)+ ZrO2(s)+Si3Al3O3N5(s)+C(s) and AlN(s)+ZrN(s)+Si3Al3- O3N5(s)+C(s).
3.2 Phase composition and microstructure
Fig.2 shows the XRD patterns of the samples heated at 1 673-1 823 K for 6 h, with the mass fraction of zircon and carbon black of 10% (sample S10), 30% (sample S30)
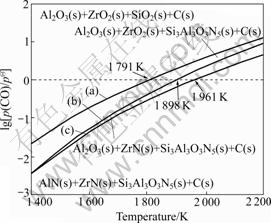
Fig.1 Equilibrium relationship of condensed phases in Al2O3- ZrO2-SiO2-C-N2 system: (a) 3Al2O3(s)+6SiO2(s)+15C(s)+ 5N2(g)=2Si3Al3O3N5(s)+15CO(g); (b) 2ZrO2(s)+4C(s)+ N2(g)=2ZrN(s)+4CO(g); (c) Al2O3(s)+3C(s)+N2(g)= 2AlN(s)+3CO(g)
and 50% (sample S50), respectively.![]() It indicates that the crystalline phases of the samples involve O′-Sialon (Si2-xAlxO1+xN2-x, x=0.17), β-Sialon(z=3), ZrO2, ZrN, Al2O3 and Al4Si3C6. When the heating temperature remains constant, with the increase of the contents of zircon and carbon black, the diffraction peak intensities
It indicates that the crystalline phases of the samples involve O′-Sialon (Si2-xAlxO1+xN2-x, x=0.17), β-Sialon(z=3), ZrO2, ZrN, Al2O3 and Al4Si3C6. When the heating temperature remains constant, with the increase of the contents of zircon and carbon black, the diffraction peak intensities
of β-Sialon and ZrN strengthen gradually. However, the intensities of O′-Sialon and ZrO2 weaken. Meanwhile, the Al2O3 phase nearly remains constant. It can also be seen that Al4Si3C6 forms at 1 823 K. The reason for this is that SiO2 and Al2O3 in the sample react with C to form SiC and Al4C3, respectively.
Moreover, in the temperature range of 1 673-1 823 K, for sample S10, the crystalline phases are composed of β-Sialon, ZrO2, ZrN and Al2O3, but there is no O′-Sialon. It can be deduced that SiO2 in the sample can be nitridized completely to form Si3N4, and Si3N4 reacts with Al2O3 to form β-Sialon. For sample S30 heated at 1 673-1 723 K, the crystalline phases involve O′-Sialon. Meanwhile, O′-Sialon and β-Sialon appear in the XRD pattern at 1 723 K. Compared with the diffraction peak intensity of O′-Sialon at 1 673 K, the intensity of O′-Sialon at 1 723 K is lower. This reveals that O′-Sialon can be converted into β-Sialon with increasing the temperature. When the temperature reaches 1 773-1 823 K, O′-Sialon vanishes completely, and the crystalline phases involve β-Sialon, ZrO2, ZrN and Al2O3. For sample S50 heated at 1 673-1 723 K, the diffraction peak intensity of O′-Sialon is higher than that of sample S30. In the temperature range of 1 773-1 823 K, there is no O′-Sialon in the sample, which indicates that O′-Sialon can be converted into β-Sialon at high
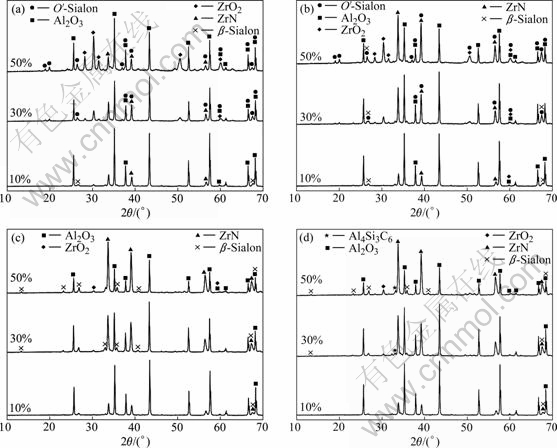
Fig.2 XRD patterns of samples heated at different temperatures for 6 h: (a) T=1 673 K; (b) T=1 723 K; (c) T=1 773 K; (d) T=1 823 K
temperature. Hence, increasing temperature favors the formation of Si3N4 and the preparation of β-Sialon/ZrN bonded corundum composites.
It was reported that the β-Sialon powder was synthesized from clay by carbothermal reduction and nitridation reaction, and then the β-Sialon bonded corundum composites were prepared by pressureless sintering process [10]. The composites sintered at 1 873 K had still two distinguishable stable crystalline phases involving corundum and β-Sialon, and there were no any other phases like O′-Sialon and X-Sialon. In this work, O′-Sialon can be observed in the samples synthesized at temperatures below 1 773 K. However, it vanishes completely at temperatures above 1 773 K. Therefore, during the preparation of β-Sialon/ZrN bonded corundum composites, O′-Sialon is an intermediate phase.
Fig.3 shows the SEM photographs and EDS pattern of sample S30 heated at 1 773 K for 6 h.
It can be found that the surface of the sample is smooth. EDS pattern (Fig.3(c)) shows that zone 1 belongs to Zr-Si-Al-O-N material. From XRD pattern, the sample obtained at 1 773 K involves β-Sialon, ZrN and Al2O3 (corundum particle, zone 2 in Fig.3(b)). So it can be concluded that the particle as shown in zone 1 is the composite body of β-Sialon and ZrN. In Ref.[16] the β-Sialon bonded corundum composites were prepared from bauxite by reaction sintering process under nitrogen atmosphere at 1 773-1 823 K for 5 h, and the SEM photographs show that the β-Sialon matrix and the corundum particles are bonded to a compact body, and β-Sialon crystalline grain mostly exists as columnar and interlaced. In this work, compact microstructure can be obtained (Fig.3(a)), β-Sialon and ZrN form a composite body which is granular (Fig.3(b)).
3.3 Analysis of formation process
The overall reaction equation of synthesizing β-Sialon(z=3)/ZrN bonded corundum composites using zircon, alumina micro powder, carbon black and fused corundum can be expressed by
3Al2O3(s)+6ZrSiO4(s)+27C(s)+8N2(g)=6ZrN(s)+2Si3Al3O3N5(s)+27CO(g) (12)
The formation process of β-Sialon (z=3) is given by reactions (13)-(16). At high temperature, ZrSiO4 can be decomposed to form ZrO2 and SiO2 (reaction (13)), and SiO2 reacts with C and N2 to form Si3N4 (reactions (14)-(16)), and finally the solid solution (β-Sialon) can be obtained.
ZrSiO4(s)=ZrO2(s)+SiO2(s) (13)
SiO2(s)+C(s)=SiO(g)+CO(g) (14)
3SiO(g)+3C(s)+2N2(g)=Si3N4(s)+3CO(g) (15)

Fig.3 SEM photographs of sample S30 heated at 1 773 K for 6 h and EDS pattern: (a) Secondary electron image; (b) Back- scattered electron image; (c) EDS pattern of zone 1 in Fig.3(b)
3SiO2(s)+6C(s)+2N2(g)=Si3N4(s)+6CO(g) (16)
The samples heated at 1 673-1 723 K (Figs.2(a) and (b)) include O′-Sialon that vanishes completely at 1 773-1 823 K (Figs.2(c) and (d)). It can be deduced that the following reactions favor the formation and vanishment of O′-Sialon (x=0.17) as well as the conversion of it into β-Sialon.
2SiO2(s)+3C(s)+N2(g)=Si2N2O(s)+3CO(g) (17)
1.83Si2N2O(s)+0.17Al2O3(s)=2Si1.83Al0.17O1.17N1.83(s)
(18)
Si1.83Al0.17O1.17N1.83(s)+1.33Al2O3(s)+3.33C(s)+
1.25N2(g)=0.50Si3Al3O3N5(s)+0.33SiO(g)+
3.33CO(g) (19)
SiO2 in sample reacts with C and N2 to form Si2N2O (reaction (17)), and it reacts with Al2O3 to form O′-Sialon (x=0.17) (reaction (18)). As shown in reaction (19), with the increase of temperature, O′-Sialon reacts with Al2O3 and C to form β-Sialon (z=3).
In the temperature range of 1 773-1 823 K, ZrO2 is converted into ZrN gradually.
2ZrO2(s)+4C(s)+N2(g)=2ZrN(s)+4CO(g) (20)
In this experiment, the formation process of β-Sialon(z=3)/ZrN bonded corundum composites can be summarized as follows.
(1) When the temperature is below 1 773 K, SiO2, one of the decomposition product from zircon, reacts with C to form Si2N2O under nitrogen atmosphere, and Si2N2O reacts with Al2O3 to form O′-Sialon. During the formation process of O′-Sialon, the other decomposition product (ZrO2) is nitridized to form ZrN.
(2) When the temperature reaches 1 773 K, the formation of β-Sialon includes two aspects. On one hand, SiO2 in the sample reacts with C and N2 to form Si3N4, β-Sialon can be obtained directly from Al2O3 and Si3N4, namely, β-Sialon is a solid solution of Al2O3 in the crystal lattice of Si3N4. On the other hand, the formed O′-Sialon as step (1) reacts with Al2O3, C and N2 to form β-Sialon. During the formation process of O′-Sialon and β-Sialon, ZrO2 is nitridized to form ZrN.
(3) β-Sialon/ZrN matrix formed as steps (1) and (2) can be distributed regularly around corundum particles. It is stable in thermodynamics. Therefore, β-Sialon/ZrN bonded corundum composites can be prepared.
(1) By using zircon, carbon black, alumina micro powder and fused corundum as the starting materials, the composites with different compositions can be obtained by controlling the heating temperature and the contents of zircon and carbon black.
(2) The proper temperature to synthesize the β-Sialon/ZrN bonded corundum composites is 1 773 K.
(3) The formation process of the β-Sialon(z=3)/ZrN bonded corundum composites includes the decomposition of zircon, the formation of Si2N2O and Si3N4, the formation of O′-Sialon and β-Sialon and the nitridation process of ZrO2.
[1] ZHU Qiang, YU Jing-kun. Synthesis of SiC-Al2O3 refractory composite powder [J]. J Chin Ceram Soc, 2008, 36(1): 40-43.
[2] BERNARDOW E, COLOMBO P, HAMPSHIRE S. Sialon-based ceramics from filled preceramic polymers [J]. J Am Ceram Soc, 2006, 89(12): 3839-3842.
[3] FALAMAKI C, FBADZADEH T. Kinetic investigation of the carbothermal reduction of an Iranian clay [J]. Ceram Int, 2002, 28(8): 887-892.
[4] YU J K, UENO S. Synthesis of β-Sialon whiskers from pyrophyllite [J]. J Ceram Soc Jpn, 1997, 105(9): 821-823.
[5] KURAMA S, SCHULZ I, HERRMANN M. Wear behaviour of α- and α/β-Sialon ceramics stabilized with Nd2O3 and Y2O3 [J]. J Eur Ceram Soc, 2009, 29(1): 155-162.
[6] HOTTA M, TATAMI J, KOMEYA K, ZHANG C, MEGURO T, TERNER M R, CHENG Y B. Formation process of calcium-α Sialon hollow balls composed of nanosized particles by carbothermal reduction-nitridation [J]. J Am Ceram Soc, 2008, 91(3): 860-864.
[7] MACKENZIE K J D, BARNEVELD D V. Carbothermal synthesis of β-Sialon from mechanochemically activated precursors [J]. J Eur Ceram Soc, 2006, 26(1/2): 209-215.
[8] ZHANG Hai-jun, HAN Bo, LIU Zhan-jie. Preparation and oxidation of bauxite-based β-Sialon-bonded SiC composite [J]. Mater Res Bull, 2006, 41(9): 1681-1689.
[9] WANG Xiao-yi, PENG Da-yan, WEN Hong-jie. Current situations of research and application of Sialon bonded corundum composite refractories [J]. Refractories, 2005, 39(3): 199-202. (in Chinese)
[10] XU Li-hua, LIAN Fang, ZHANG Han, BI Yu-bao, CHENG Ke, QIAN Yang-bao. Optimal design and preparation of beta-Sialon multiphase materials from natural clay [J]. Mater Design, 2006, 27(7): 595-600.
[11] HAN Bo, ZHANG Hai-jun, LIU Zhan-jie. Study on the cryolite corrosion resistance of bauxite based β-Sialon bonded corundum/SiC composites [J]. Bull Chin Ceram Soc, 2007, 26(4): 40-43. (in Chinese)
[12] LI Ya-wei, LI Nan, WANg Bin-yao, LIU Jing, CHENG Fang-yu. β-Sialon/corundum composite refractories by nitridation sintering process[J]. J Inorg Mater, 2000, 15(4): 612-618. (in Chinese).
[13] ALBANO M P, SCIAN A N. Mullite/Sialon/alumina composition by infiltration processing [J]. J Am Ceram Soc, 1997, 80(1): 117-124.
[14] MA Bei-yue, YU Jing-kun. Synthesis of ZrO2-SiC composite powder and effect of its addition on properties of Al2O3-C refractories [J]. Trans Nonferrous Met Soc China, 2007, 17(5): 996-1000.
[15] LIANG Ying-jiao, CHE Yin-chang. Handbook of thermodynamic data in inorganic [M]. Shenyang: Northeastern University Press, 1993: 449-479. (in Chinese)
[16] ZHANG Hai-jun, LIU Zhan-jie, ZHONG Xiang-chong. Properties of β-Sialon bonded corundum composites [J]. J Chin Ceram Soc, 2005, 33(11): 1308-1313. (in Chinese)
(Edited by YANG You-ping)

