化学腐蚀后的表面状态对硅片Fe沾污的影响
来源期刊:稀有金属2021年第1期
论文作者:赵江伟 宁永铎 周旗钢 钟耕杭 刘斌 肖清华
文章页码:62 - 69
关键词:硅片;Fe沾污;化学腐蚀;表面状态;
摘 要:利用不同的酸腐蚀和碱腐蚀条件对硅片进行了腐蚀,分析了腐蚀后的硅片表面状态对Fe沾污的影响。实验结果表明,不同酸腐蚀条件的硅片经过以HF酸结尾的改进型RCA(Radio Corporation of America)清洗法清洗后,表面疏水性增强,相对不易附着Fe离子,而当酸腐蚀硅片表面经过SC-1溶液处理后,表面亲水性增强,附着的Fe离子较多,且难以通过超纯水冲洗去除;随着酸腐蚀硅片表面粗糙度的增大,表面吸附的Fe离子也增多。不同碱腐蚀条件的硅片去除量越小,表面残留的研磨造成的机械损伤层厚度则越大,损伤层厚度较大时,表面吸附的Fe离子也越多,且难以通过超纯水冲洗去除;在90℃下腐蚀40 s的硅片,由于去除量约为4.2μm,研磨过程中造成的表面损伤层沾污没有完全去除,残留在损伤层中的Fe沾污经过改进型RCA清洗后也无法去除,沾污会在退火过程中扩散进入硅片体内。
网络首发时间: 2019-04-28 10:45
稀有金属 2021,45(01),62-69 DOI:10.13373/j.cnki.cjrm.xy19010031
赵江伟 宁永铎 周旗钢 钟耕杭 刘斌 肖清华
北京有色金属研究总院
有研半导体材料有限公司
利用不同的酸腐蚀和碱腐蚀条件对硅片进行了腐蚀,分析了腐蚀后的硅片表面状态对Fe沾污的影响。实验结果表明,不同酸腐蚀条件的硅片经过以HF酸结尾的改进型RCA(Radio Corporation of America)清洗法清洗后,表面疏水性增强,相对不易附着Fe离子,而当酸腐蚀硅片表面经过SC-1溶液处理后,表面亲水性增强,附着的Fe离子较多,且难以通过超纯水冲洗去除;随着酸腐蚀硅片表面粗糙度的增大,表面吸附的Fe离子也增多。不同碱腐蚀条件的硅片去除量越小,表面残留的研磨造成的机械损伤层厚度则越大,损伤层厚度较大时,表面吸附的Fe离子也越多,且难以通过超纯水冲洗去除;在90℃下腐蚀40 s的硅片,由于去除量约为4.2μm,研磨过程中造成的表面损伤层沾污没有完全去除,残留在损伤层中的Fe沾污经过改进型RCA清洗后也无法去除,沾污会在退火过程中扩散进入硅片体内。
中图分类号: TN304.12
作者简介:赵江伟(1993-),男,甘肃陇南人,硕士,研究方向:半导体硅材料,E-mail:huakuishehan@163.com;;*周旗钢,教授,电话:010-82087080,E-mail:zhouqigang@grinm.com;
收稿日期:2019-01-28
基金:国家重点研发计划项目(2017YFB0305603);北京市顺义区科技三项费项目(KS201824)资助;
Zhao Jiangwei Ning Yongduo Zhou Qigang Zhong Genghang Liu Bin Xiao Qinghua
General Research Institute for Nonferrous Metals
GRINM Semiconductor Materials Co.,Ltd.
Abstract:
With the development of very large-scale integrated circuits(VLSI),the integration of the circuit was getting higher andhigher,and the yield of the device was more and more sensitive to the metal impurities and defects in the silicon wafer.As a commonmetal impurity in the process of integrated circuits(IC)manufacturing,iron(Fe)had a large diffusion coefficient at high temperatureand easy to diffuse into the bulk of wafer through surface contamination.Through different etching processes,the effect of the surfacestate of the etched silicon wafer on Fe contamination was studied.The surface photovoltage(SPV)method was used to analyze the Feconcentration in silicon wafer.All experimental silicon wafers were first etched under different acid and alkali etching conditions.Thenall silicon wafers were cleaned by an improved Radio Corporation of America(RCA)cleaning method,mainly through two SC-1 solu-tions(5% NH4OH and 10% H2O2),one SC-2 solution(volume ratio of HCl∶H2O2∶H2O=1∶1∶20),one dilute HF solution(containing 1% HF)and several times quickly drain(QDR)cleaning.The first part of the experiment:the cleaned acid-etched silicon wafers werepided into five groups,and each group contains silicon wafers with four different acid etching conditions.The first group of silicon wa-fers were directly annealed;The second group of silicon wafers were contaminated by FeCl3 solution with a concentration of 5×10-9 mol・L-1 for 10 min and then annealed.The third group of silicon wafers were contaminated by FeCl3 solution and rinsed with ultrapure waterfor 10 s,and then annealed;The fourth and fifth group of silicon wafers were contaminated by the FeCl3 solution after respectively im-mersing half and whole of silicon wafers in the SC-1 solution for 2 min,and then annealed after rinsing with ultrapure water.The sec-ond part of the experiment:the cleaned alkali-etched silicon wafers were pided into three groups,and each group contains silicon wa-fers with four different alkali etching conditions.The first group of silicon wafers were directly annealed;The second group of siliconwafers were contaminated by FeCl3 solution with a concentration of 5×10-9 mol・L-1 for 10 min and then annealed.The third group of sili-con wafers were contaminated by FeCl3 solution and rinsed with ultrapure water for 10 s,and then annealed.The silicon wafers weredried in air before annealing at 650 ℃ for 30 min.The annealed silicon wafers were polished and measured for Fe by SPV method.Theresults of the Groups 1~3 in the first part of the experiment showed that the average concentration of bulk Fe of silicon wafers contami-nated with FeCl3 solution was higher than 9.1×1010 atom・cm-3 after annealing.The roughness of silicon wafers with a removal amount of 30μm was smaller than that of silicon wafers with a removal amount of 15 μm.And when the removal amount was the same,the roughness ofsilicon wafers etched by the new acid was smaller than that of silicon wafers etched by the exhausted acid.And silicon wafers with a small-er roughness had a lower degree of Fe contamination.The experimental results of the fourth and fifth group of silicon wafers showed thatthe surface hydrophilicity of acid-etching silicon wafers was enhanced by immersion in SC-1 solution.After silicon wafers were contami-nated by FeCl3 solution,the Fe ions adsorbing on the surface could not be cleaned by ultra-pure water.The average bulk Fe concentrationof silicon wafers was more than 1×1011 atom・cm-3 after annealing.And the contamination amount of the whole silicon wafer immersed inSC-1 solution was larger than that of half of the silicon wafer immersed in SC-1 solution.The results of Groups 1~3 in the second part ofthe experiments showed that the surface removal amount of silicon wafers is 4.2 μm after alkali-etching at 90 ℃ for 40 s.And as the sili-con wafers cleaned by improved RCA cleaning,the average concentration of bulk Fe was still high after annealing.When silicon waferswere contaminated with FeCl3 solution,the average concentration of bulk Fe of silicon wafers washed by ultrapure water was lower thanthat of silicon wafers without being washed by ultrapure water.But it was still higher than the initial level of silicon wafers.And the con-tamination degree of silicon wafers with larger amount of surface removal was lower.The surface hydrophobicity of silicon wafers with dif-ferent acid etching conditions enhanced after an improved RCA cleaning method ending with HF acid,and Fe ions were relatively diffi-cult to adhere to the surface,while the acid etching wafers were treated with SC-1 solution,the surface hydrophilicity of the wafers en-hanced and more Fe ions adhering to the surface could not be removed by rinsing with ultra-pure water.As the surface roughness of acidetching silicon wafers increased,the Fe ions adhering to the surface also increased.The smaller removal amount of the silicon wafers withdifferent alkali etching conditions,the greater residual thickness of the mechanical damage layer caused by the surface grinding and themore Fe ions adsorbed on the surface.The Fe ions could not be removed by rinsing with ultra-pure water.After etching the silicon wafersfor 40 s at 90 ℃,the contamination of surface damage layer caused by the grinding process was not completely removed due to the 4.2 μmremoval amount.The Fe contamination in the damage layer also could not be removed after an improved RCA cleaning,and would diffuseinto the bulk of silicon wafers during the annealing process.
Keyword:
silicon wafer; Fe contamination; chemical etching; surface state;
Received: 2019-01-28
随着超大规模集成电路的发展,电路的集成度越来越高,线宽不断减小,器件的成品率对硅片中存在的金属杂质和缺陷也越来越敏感
硅片制造要经过单晶生长、切片、倒角、研磨、腐蚀、抛光等一系列工艺过程,经过研磨的硅片会在表面形成机械损伤层,一般通过碱腐蚀或者酸腐蚀来去除这些损伤层
主要通过不同的腐蚀工艺,研究了腐蚀后的硅片表面状态对Fe沾污的影响。从而为硅片生产过程中Fe沾污的预防以及沾污后的处理措施提供参考,提高硅片的品质。
1 实验
1.1 材料及方法
实验主要包括两个部分:酸腐蚀后的硅片表面状态对Fe沾污的影响,以及碱腐蚀后的硅片表面状态对Fe沾污的影响。
实验选取轻掺B<100>晶向的研磨硅片,研磨使用Speed Fam 20B研磨机,研磨压力为45 MPa,转速为50 r・min-1,采用FUJIMI PWA-12磨砂,磨砂中值粒径为(7.8±0.6)μm,硅片的平均体Fe浓度均值为1×109atom・cm-3的水平。硅片的酸腐蚀采用HNO3-HF-CH3COOH混酸溶液,碱腐蚀采用KOH溶液。所有实验硅片首先经过不同的酸腐蚀条件和碱酸腐蚀条件进行腐蚀,腐蚀条件如表1和表2所示。表1中的酸液分为新酸和乏酸,新酸为刚配置的酸液,乏酸指新配的酸液腐蚀500片硅片之后的酸液。腐蚀完成之后,为了排除腐蚀过程中的Fe沾污对实验结果的影响,将所有的硅片均经过改进型RCA清洗工艺,主要为经过2次SC-1溶液(5%NH4OH与10%H2O2)、1次SC-2溶液(体积比为HCl∶H2O2∶H2O=1∶1∶20)、1次稀HF溶液(含1%HF)并配合多次QDR清洗。第一部分实验为:将清洗后的酸腐蚀硅片分为五组,每组均包含四种不同的酸腐蚀条件的硅片,第一组硅片直接退火;第二组硅片在浓度为5×10-9mol・L-1的Fe Cl3溶液中浸泡沾污10 min后退火;第三组硅片经过Fe Cl3溶液沾污后用超纯水冲洗10 s,再退火;第四组和第五组分别将硅片的一半和全部浸入SC-1溶液中2 min后,在Fe Cl3溶液中沾污,然后经过超纯水冲洗后退火。第二部分实验:将清洗后的碱腐蚀硅片分为三组,每组均包含四种不同的碱腐蚀条件的硅片,第一组硅片直接退火;第二组硅片在浓度为5×10-9mol・L-1的Fe Cl3溶液中浸泡沾污10 min后退火;第三组硅片经过Fe Cl3溶液沾污后用超纯水冲洗10 s,再退火。硅片退火前均在空气中晾干,退火温度为650℃,退火时间30 min。退火后的硅片经过抛光后测体Fe。
表1 硅片的酸腐蚀条件 下载原图
Table 1 Acid etching conditions of silicon wafers

表2 硅片的碱腐蚀条件 下载原图
Table 2 Alkali etching conditions of silicon wafers

1.2 实验分析方法
实验采用表面光电压(SPV)法测量硅片体内的Fe浓度,利用Semilab公司开发的WT-2000,能够对硅抛光片进行非接触性的全面扫描,测量硅片Fe杂质含量。因为SPV技术主要是测量硅片体内的Fe离子沾污浓度,因此硅片表面的Fe沾污需要通过退火将表面Fe离子驱入到体内,才能准确测量硅片的Fe污染,本实验主要利用硅片生产过程中经常利用的消除氧施主的退火。采用上海中晨数字技术设备有限公司生产的接触角测量仪(JC2000C1)对硅片的表面液滴接触角进行测量分析。
2 结果与讨论
2.1 酸腐蚀表面状态对Fe沾污的影响
图1(a~c)为酸腐蚀实验第一组到第三组硅片的实验结果,从图1(a)可以看到,经过改进型RCA清洗的酸腐蚀硅片退火后,体内的平均Fe浓度很低,和硅片的初始Fe浓度水平一致,均值在1×109atom・cm-3的水平。从图1(b)可知,经过Fe Cl3溶液沾污的酸腐蚀硅片,退火后硅片体内的平均Fe浓度较高,均大于9.1×1010atom・cm-3,腐蚀去除量为30μm的硅片比去除量为15μm的硅片沾污量低,且当腐蚀去除量相同时,新酸腐蚀的硅片Fe沾污程度小于乏酸腐蚀的硅片。从图1(c)可以看到,硅片在Fe Cl3溶液中沾污后经过超纯水冲洗,硅片的平均Fe浓度和初始Fe浓度水平一致,均值在1×109atom・cm-3的水平。说明经过不同酸腐蚀条件的硅片在Fe Cl3溶液中沾污后,表面沾污的Fe浓度较高,且沾污程度也有差异,而经过超纯水冲洗后,能够冲洗掉附着在硅片表面的Fe离子。这主要是因为酸腐蚀硅片经过了以稀HF酸清洗结尾的改进型RCA清洗,呈现疏水性,表面以Si-H键为主,氢终端表面非常稳定,可以避免氧化层的产生,不容易和Fe离子形成强吸附,因此表面沾污的Fe离子能够很容易地被超纯水冲洗带走,而沾污后不经过超纯水冲洗的硅片,表面的Fe最终会在退火过程中扩散进入硅片中。
图1 酸腐蚀硅片沾污Fe Cl3溶液前后的平均体Fe浓度对比
Fig.1 Comparison of average concentration of bulk Fe before and after acid etching wafers were contaminated with Fe Cl3solution
(a)Uncontaminated;(b)Contaminated;(c)Rinsed with ultrapure water after contamination
硅片经过改进型RCA清洗后,表面疏水性增强,而浸入SC-1溶液后,亲水性又会增强。为了进一步探究酸腐蚀后的硅片表面亲水性对Fe沾污的影响,将酸腐蚀后的硅片一半或者全部浸入SC-1溶液中进行亲水处理,利用表面接触角测量仪对硅片处理前后的表面接触角测量得到如图2所示的结果。能够知道经过SC-1溶液的处理,硅片表面的接触角减小,硅片的亲水性增强。
经过SC-1溶液处理后的硅片再经过Fe Cl3溶液的沾污和超纯水的冲洗,退火测得硅片体内的平均Fe浓度如图3所示。由图3可以看到,酸腐蚀硅片浸入SC-1溶液使表面亲水性增强后,经过Fe Cl3溶液沾污,表面吸附的Fe离子用超纯水冲洗不掉,退火后进入硅片体内的平均Fe浓度超过了1×1011atom・cm-3,且经过不同酸腐蚀条件的硅片平均体Fe浓度大小关系和图1(b)一致,全部浸入SC-1溶液的硅片比一半浸入SC-1溶液的硅片Fe沾污程度严重。为了更好地观察硅片的Fe沾污情况,图4给出了上述实验硅片的SPV体Fe分布扫描图。
图2 清洗后的酸腐蚀硅片浸入SC-1溶液前后的表面接触角
Fig.2 Surface contact angle of cleaned acid-etched wafer be-fore and after immersion in SC-1 solution
(a)Before immersion in SC-1 solution;(b)After immersion inSC-1 solution
由图4可以看到,颜色较亮的部分沾污比较严重,硅片初始的体Fe浓度分布比较均匀,而硅片一半浸入SC-1溶液后,浸入SC-1溶液的部分Fe沾污比较严重,硅片全部浸入SC-1溶液后,中心大块部分的Fe沾污都比较严重。这说明硅片浸入SC-1溶液使表面亲水性增加后,对Fe离子的沾污更为严重。经过分析可以知道,当硅片浸入SC-1溶液后,硅片表面的Si-OH键增多,表面亲水性增强,亲水性表面相对疏水性表面能量更高
图3 浸泡SC-1溶液的酸腐蚀硅片沾污Fe Cl3溶液后的平均体铁浓度
Fig.3 Average concentration of bulk Fe after acid etching wafers immersed in SC-1 solution were contaminated with Fe Cl3solution
(a)Half of wafer was immersed in SC-1;(b)Whole wafer was immersed in SC-1
图4 酸腐蚀硅片的体Fe浓度SPV扫描图
Fig.4 SPV scanning maps of bulk Fe concentration of acid etching wafers
(a)Uncontaminated;(b)Contaminated after half of wafer was immersed in SC-1;(c)Contaminated after whole wafer was immersed in SC-1
为了分析经过不同酸腐蚀条件的硅片Fe沾污程度的差异,利用表面粗糙度测量仪对酸腐蚀硅片的表面粗糙度(Ra)值进行测量,测量结果如图5所示。由图可以看到硅片在不同的酸腐蚀条件下,表面的粗糙度(Ra)也不一样。去除量相同时,新酸腐蚀的硅片要比乏酸腐蚀的硅片的粗糙度低,且在同一种酸液中腐蚀去除量大的硅片表面粗糙度值低于去除量小的硅片。不同酸腐蚀条件下的硅片表面粗糙度和硅片的Fe沾污程度表现出了相关性,说明表面粗糙度大的硅片,比表面积也较大,增加了硅片对Fe离子的吸附面积,因此酸腐蚀后表面粗糙度大的硅片Fe沾污程度也较大。
2.2 碱腐蚀表面状态对Fe沾污的影响
图6(a~c)为碱腐蚀实验第一组到第三组硅片的实验结果。由图6(a)可知硅片在90℃下碱腐蚀40 s时,经过改进型RCA清洗后退火,体内的平均Fe浓度依然较高,而经过其他碱腐蚀条件的硅片,体内的平均Fe浓度和硅片的初始Fe浓度水平一致,均值在1×109atom・cm-3的水平。由图6(b)可以看到清洗过的碱腐蚀硅片在Fe Cl3溶液中沾污,退火后硅片的体Fe沾污程度较高,且不同碱腐蚀条件的硅片沾污程度不同。图6(c)的硅片在Fe Cl3溶液中沾污后,经过了超纯水冲洗,相比图6(b)硅片体内的平均Fe浓度降低了,但仍然高于硅片的初始水平。
图5 不同酸腐蚀条件下的硅片表面粗糙度(Ra)值
Fig.5 Surface roughness (Ra) of wafers with different acid etching conditions
图6 碱腐蚀硅片沾污Fe Cl3溶液前后的平均体铁浓度对比
Fig.6 Comparison of average concentration of bulk Fe before and after alkali etching wafers were contaminated with Fe Cl3solution
(a)Uncontaminated;(b)Contaminated;(c)Rinsed with ultrapure water after contamination
对碱腐蚀后的硅片表面粗糙度Ra测量得到图7所示的结果。由图7可以看到不同碱腐蚀后的硅片表面粗糙度差别较小,且沾污程度较小的硅片粗糙度较大,又因为沾污后的硅片经过超纯水冲洗后仍然存在沾污,由此可知表面粗糙度对不同碱腐蚀条件下的硅片Fe沾污程度的差异性未见显著影响。
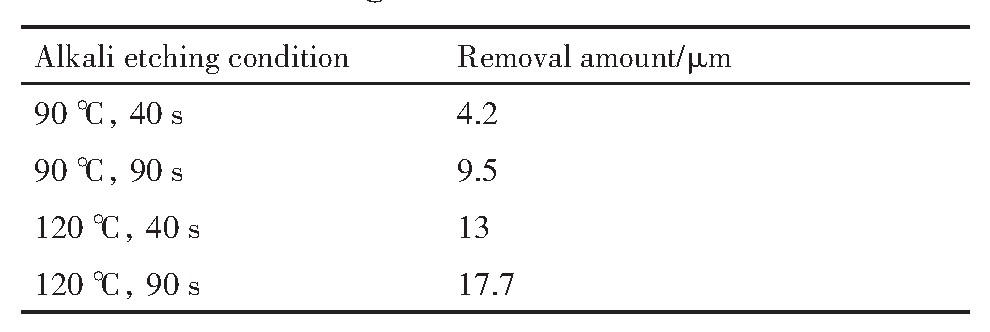
为了进一步分析图6的实验结果,对碱腐蚀前后的硅片厚度进行了测量,从而得到了硅片的腐蚀去除量,如表3所示。
由表3可以看到硅片在90℃下腐蚀40,90 s以及在120℃下腐蚀40,90 s时,硅片的去除量依次增大。因为硅片在腐蚀之前经过了研磨,研磨过程中会在硅片表面形成一定厚度的机械损伤层,机械损伤层由破碎层、裂纹层和位错层等构成,一般认为正常研磨压力下机械损伤层厚度约等于磨砂粒径
图7 不同碱腐蚀条件下的硅片表面粗糙度(Ra)值
Fig.7 Surface roughness(Ra)of wafers with different alkali etching conditions
表3 不同碱腐蚀条件下的硅片去除量 下载原图
Table3 Removal amount of silicon wafers with different alkali etching conditions
为了验证硅片表面损伤对Fe离子的吸附作用,在硅片表面轻微划出了特定形状“A”的损伤,然后根据碱腐蚀硅片的处理方法在Fe Cl3溶液中沾污,用超纯水冲洗后退火并观察硅片的SPV体Fe分布扫描图,如图8所示。由图可以看到,在硅片有损伤的区域,硅片的体Fe浓度较高,体Fe分布也呈现出了特定的形状“A”,由此可以证实,硅片表面损伤会影响Fe离子在表面的吸附,损伤区域的Fe离子吸附作用较强。
图8 硅片表面损伤后沾污的体Fe浓度SPV扫描图
Fig.8 SPV scanning maps of bulk Fe concentration of contami-nated wafer after surface damage
3 结论
1.不同酸腐蚀条件的硅片在以HF酸清洗结尾的改进型RCA清洗后,表面疏水性增强,相对不易附着Fe离子,表面沾污的Fe离子较易通过超纯水冲洗去除,而当酸腐蚀硅片经过SC-1溶液处理后,表面亲水性增强,表面附着的Fe离子难以通过超纯水冲洗去除;在新酸和乏酸中去除30μm以及在新酸和乏酸中去除15μm的硅片,表面粗糙度依次增大,附着的Fe离子也增多。
2.不同碱腐蚀条件的硅片去除量越小,表面残留的研磨造成的机械损伤层厚度则越大,表面吸附的Fe离子也越多,且难以通过超纯水冲洗去除;在90℃下腐蚀40 s的硅片,由于去除量只有4.2μm,研磨过程中造成的表面损伤层沾污没有完全去除,残留在损伤层中的Fe沾污经过改进型RCA清洗后无法清洗干净,沾污会在退火过程中扩散进入硅片体内。
参考文献

