AZ31镁合金在TMPAC-AlCl3离子液体中电镀铝
来源期刊:中国有色金属学报(英文版)2011年第9期
论文作者:刘奎仁 柳泉 韩庆 涂赣峰
文章页码:2104 - 2110
关键词:镁合金;电镀;离子液体;铝;腐蚀
Key words:magnesium alloy; electrodeposition; ionic liquids; aluminum; corrosion
摘 要:采用TMPAC-AlCl3离子液体,在浸锌后的镁合金AZ31表面实施恒流电镀铝,采用电化学技术评估镀铝层的耐蚀性,并采用SEM/EDX等技术表征镀层。结果表明:在浸锌后的镁合金表面可获得表面光洁的银白色镀铝层。在不同的电流密度下,呈现出两种形貌不同的镀铝层;在较佳电流密度12.3 mA/cm2时,镀铝层表面较致密;电镀时间越长,镀层越厚,镀铝层耐蚀性越好。
Abstract: Aluminum was electrodeposited with constant current on AZ31 magnesium alloy pretreated under optimized conditions from trimethyl-phenyl-ammonium chloride and anhydrous aluminum chloride (TMPAC-AlCl3) quaternary ammonium room temperature ionic liquids with benzene as a co-solvent. The corrosion resistance of the as-deposited Al layers was evaluated in 3.5% NaCl solution by the electrochemical technologies. The Al depositions were characterized by scanning electron microscopy equipped with energy dispersion X-ray. The results show that the microstructures of the Al depositions have spherical equiaxed grains obtained at a high current density, and bulk grains at a low current density. The Al deposition obtained at 12.3 mA/cm2 has a smooth and compact surface. The electrochemical measurements indicate that the thicker Al deposition can more effectively protect the Mg substrate. The Al deposition with bulk grains hardly protects the AZ31 Mg substrate from corrosion owing to its porosity.

LIU Kui-ren, LIU Quan, HAN Qing, TU Gan-feng
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 30 October 2010; accepted 27 May 2011
Abstract: Aluminum was electrodeposited with constant current on AZ31 magnesium alloy pretreated under optimized conditions from trimethyl-phenyl-ammonium chloride and anhydrous aluminum chloride (TMPAC-AlCl3) quaternary ammonium room temperature ionic liquids with benzene as a co-solvent. The corrosion resistance of the as-deposited Al layers was evaluated in 3.5% NaCl solution by the electrochemical technologies. The Al depositions were characterized by scanning electron microscopy equipped with energy dispersion X-ray. The results show that the microstructures of the Al depositions have spherical equiaxed grains obtained at a high current density, and bulk grains at a low current density. The Al deposition obtained at 12.3 mA/cm2 has a smooth and compact surface. The electrochemical measurements indicate that the thicker Al deposition can more effectively protect the Mg substrate. The Al deposition with bulk grains hardly protects the AZ31 Mg substrate from corrosion owing to its porosity.
Key words: magnesium alloy; electrodeposition; ionic liquids; aluminum; corrosion
1 Introduction
Magnesium and its alloys with many merits have been applied in plane, automobile and electric industries [1]. However, Mg alloys are generally known to have a poor corrosion resistance [2]. Aluminum as a light metal possesses many advantages, including good anti-corrosion property, high thermal and electrical conductivity, and excellent mechanical property. The electroplating of Al on the surface of magnesium alloy would not only prevent Mg substrate from corrosion, but also not markedly increase its mass. Meantime, both anodic oxidation technology and electrolytic coloring technology of Al alloys have been applied in the production industry. Those have the potential to be directly used for the Al layer [3]. There are some disadvantages to the common preparation methods of Al coatings or depositions including hot-dipping technology, thermal spraying and physical or chemical vapor deposition, except for plating. The plating has some advantages, such as cheap instruments, a smooth deposition with a low porosity, and room operation temperature. Al has a standard electrode potential of -1.66 V (vs SHE), by far lower than hydrogen. Furthermore, Al can only be electrodeposited from non-aqueous solution [4]. Among various Al-plating electrolytes, ionic liquids (ILs) have obtained attention because of their merits, such as adjustable Lewis acidity-basicity, a low vapor pressure, and a wide electrochemical window [5]. In 1951, the low- temperature melting salt with chloroaluminate ions was firstly applied to the electroplating of aluminum [6]. The most current AlCl3-based ILs used for the Al or its alloy plating included n-butyl pyridinium chloride (BPC- AlCl3) [7-8], 1-ethyl-3-methylimidazolium chloride ([EMIm]Cl-AlCl3) [9-11], and trimethyl-phenyl- ammonium (TMPAC-AlCl3) [12-13].
The electrodeposition of Al from anhydrous electrolytes has been studied widely on various kinds of metal substrates [5]. It is hard for Al to be electroplated on magnesium alloys owing to its high chemical reactivity. CHANG et al [3, 11] plated aluminum on the surface of Mg alloys using [EMIm]Cl-AlCl3 ILs. Sequentially, ZHANG et al [14] achieved the amorphous Al-Mn deposition on AZ31B from inorganic molten salts AlCl3-NaCl-KCl-MnCl2. These works exhibit an important significance to exploit a novel surface treatment technology used for Mg alloys. Nevertheless, the study on the Al plating of Mg alloy substrate still continues on the early stage. The more information urgently needed will make its knowledge system more various and rich. TMPAC-AlCl3 has a low melting point of -75 °C as x(AlCl3)=66.7%, a good electrochemical and thermal stability [4], and a relatively cheap price [12]. But its electric conductivity is low [15]. The addition of benzene can improve the problem [16]. The electrodeposition of Al from TMPAC-AlCl3 had been achieved on the W [12-13, 17] and Al [13] substrates. A thick silvery-colored Al layer was electrodeposited on copper alloy surface from TMPAC-AlCl3+benzene mixed solution, which can prevent substrate from corrosion. However, to our knowledge, no report has been published on the electroplating of Al on Mg alloy substrate from TMPAC-AlCl3 ionic liquids.
In the present study, the influences of activation time and zinc-immersion time on the corrosion resistance of activation films and zinc coatings were studied using potentiodynamic polarization curves in order to optimize procedure parameters. The electrodeposition of Al was carried out on the surface of AZ31 magnesium alloys from TMPAC-AlCl3 ionic liquids. The effects of current density and plating time on the microstructure and thickness of the as-deposited Al coatings were studied. The anti-corrosion property of Al depositions was characterized in 3.5% NaCl solution by the electrochemical technologies, including potentiodynamic polarization curves, electrochemical impedance spectroscopy and the variation of open circuit potential of tested samples with different periods of immersion.
2 Experimental
The trimethyl-phenyl-ammonium (TMPAC) of technical grade quality (>98%) was dried under vacuum for 24 h at 100 °C. All other reagents were of analytical grade. The acidic ionic liquids with 1:2 of molar ratio of TMPAC to AlCl3 was prepared by the slow addition of AlCl3 to TMPAC at room temperature under a continuous magnetic bar stirring [12].
The cathodes (40 mm?15 mm?3 mm) were made of AZ31 magnesium alloys with a composition of Al 3%, Zn 1%, Mn 0.4% and Mg balance (mass fraction). The AZ31 samples were pretreated using 600# grit SiC sand paper polishing, alkaline cleaning, chemical pickling, HF acid surface activation and zinc-immersion. And then they were fully cleaned with ethanol and toluene. Table 1 shows the detail of the pretreatment procedure of the magnesium alloy. The entire previous steps including alkaline cleaning, chemical pickling and surface activation served the zinc-immersion procedure. The compact zinc-immersion film achieved provided an ideal intermediate layer for the subsequent Al-electroplating process. The pure Al sheets with a mass fraction of 99.9% as anodes (20 mm?20 mm?1 mm) were cleaned with acetone, fully rinsed with water, and then pickled in 1 mol/L NaOH solution at 80 °C. Eventually, both the cathode and anode were immediately rinsed with ethanol and toluene, respectively, before being stored in a flask of toluene. In the ionic liquids with a benzene content of 50% (volume fraction), the Al plating was performed at 40-42 °C under an agitating speed of 200 r/min.
Table 1 Formulations and operation conditions of magnesium alloy pretreatment procedure
The morphology and components of the deposition cross-section or surface were examined with a scanning electron microscope SSX-550 equipped with energy dispersion X-ray spectrometer (SEM-EDX, SHIMAZDU, Japan). The corrosion-resistance of Al depositions was investigated using electrochemical impedance spectroscopy (EIS), potentiodynamic polarization measurement (IE) and sequentially sampling the open circuit potential (OCP) with the variable of immersion time in a NaCl solution. All the measurements were performed in an ordinary three-electrode cell filled with a 3.5% NaCl solution (mass fraction) at room temperature with an electrochemical workstation IM6e (ZAHNER, Germany). The working electrode was the Al deposition with a exposed surface area of 1 cm2. The remainder was sealed with ethoxyline resin. The counter electrode and reference electrode were a platinum sheet and a saturated calomel electrode (SCE), respectively. The EIS measurements were carried out at open circuit potential. The detected frequency was in a range of 10 000-0.01 Hz, and the voltage amplitude was 5 mV.
3 Results and discussion
3.1 Surface pretreatment of Mg alloy substrate
The magnesium and its alloys, wherever possible, are extremely easy to be oxidized under the existence of oxygen. It forms a porous oxidate film on the surface. There is a replacement reaction to happen between the plating electrolyte and the Mg alloy substrate. The replacement products are harmful to the Al-plating process. It is difficult to be realized that Al would be directly electroplated on the AZ31 Mg alloy surface. A compact intermediate layer deposited on the Mg alloy surface will be beneficial to the electroplating of Al. The surface activation and zinc-immersion belong to the traditional surface treatments used for Mg alloys. If the treatment was used as a fraction of the entire Al-electroplating pretreatment process, it would have practice significance. Even though the similar pretreatments have been used as the fraction [14, 18], their processing technology or formula is more or less different from the present work. This study systematically confirms the effects of activation time and zinc-immersion time on the corrosion resistance of activation and zinc-immersion films.
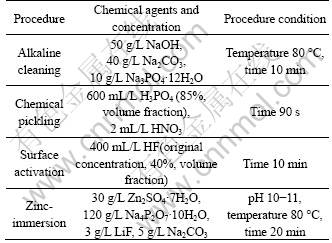
The samples were obtained for different reaction time (t) in the HF activation solution and zinc-immersion solution, respectively. Their corrosion-resistance properties were tested by polarization curves. A parallel team includes three samples obtained under the same condition. The previous step of zinc-immersion procedure chose the activation time of 10 min. The polarization resistance (Rp) of those samples was calculated according to their polarization curves. Accordingly, the mean value and standard error of the parallel team were calculated. Figure 1 shows the relationship curves between the reaction time and the polarization resistance of activation films and zinc-immersion depositions. The Rp of the activation films or zinc depositions rapidly increases with the prolonged time. After 10 min, Rp has a slow increase rate. Considering the standard error of the mean Rp, it is of no significant difference for the resistance distribution of the samples obtained for above 10 min (see Fig. 1). As time increased from 2 min to 10 min, the chemical reaction reacted fully in the whether the HF activation or zinc-immersion process. Correspondingly, the surface of the films or depositions became a bit more compact. As time was beyond 10 min, the relatively compact films or depositions were fully laid on the substrate surface. It follows that the mean value of the Rp calculated has no significant increase considering the standard error. In short, it is sufficient for the next step to prepare the compact activation films and the zinc depositions beyond 10 min. In the present work, the activation time and zinc-immersion time were defined as 10 min and 20 min, respectively.
3.2 Electrodeposition of Al
The silvery-colored satin Al layer was constant- current electrodeposited in the TMPAC-AlCl3 ionic liquids mixed with 50% benzene (volume fraction). Some depositions were prepared at 5.8, 12.3 and 19.2 mA/cm2 for 60 min, respectively, and others at 12.3 mA/cm2 for 30 and 120 min, respectively. The SEM images and EDX analysis are shown in Fig. 2. The Al deposition has a bulk grain structure prepared at a low current density (5.8 mA/cm2) from Fig. 2(a). Nevertheless, they have a spherical grain structure at a relatively high current density (12.3 mA/cm2, 19.2 mA/cm2), as shown in Figs. 2(b) and (c). In Fig. 2(a), there are many fine pores distributed on the surface of the Al deposition. This results in the less compact surface. The Al deposition with spherical-shape equiaxed grains is more compact than that with bulk grains. As current density rises, the Al grains obtained grow. It is quite different for the microstructures, respectively at a high and low current. The nucleation rate of a new grain increases more rapidly than its growing rate as current density rises. At a relatively high current, the nucleation rate controls the growth of the Al deposition. This promotes the formation of the equiaxed crystal grains with spherical shape [19]. Thanks to no pore and crack, the deposition (Fig. 2(b)) is smoother and more compact than those shown in Figs. 2(a) and (c). Accordingly, the current density is defined as about 12.3 mA/cm2 to prepare a compact aluminum layer. The grains grow fully and their size increases with rising of plating time (see Figs. 2(b), (d) and (e)). The EDX analysis of this case (Fig. 2(b)) shows that the as-deposited layer is made of Al (Fig. 2(f)). The other depositions are extremely similar to that in Fig. 2(f).
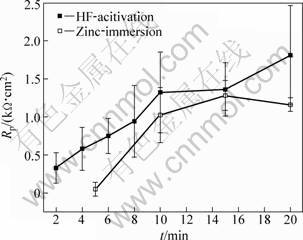
Fig. 1 Relationship between polarization impedance of AZ31 magnesium alloy and time of HF acid-activation or zinc- immersion
The depositions were prepared at 12.3 mA/cm2 for 30, 60 and 120 min on the AZ31 Mg alloy surface, respectively. The SEM images of their cross-section are shown in Fig. 3. A white-colored zinc immersion coating with a thickness of 1 μm exists between the Al deposition and Mg alloy substrate. The thickness of the Al depositions obtained increases with the prolonged plating time. As time reaches 120 min, the Al layer has a thickness of 13.3 μm (Fig. 3(c)). Its low magnification image is shown in Fig. 3(d). The Al deposition is uniformly laid on the substrate surface. A close bonding e among Al, zinc and the substrate is beneficial to protect the AZ31 Mg alloy substrate.

Fig. 2 SEM images and EDX spectrum of as-deposited Al depositions on surface of AZ31 magnesium alloy from direct plating: (a) 5.8 mA/cm2, 60 min; (b) 12.3 mA/cm2, 60 min; (c) 19.2 mA/cm2, 60 min; (d) 12.3 mA/cm2, 30 min; (e) 12.3 mA/cm2, 120 min; (f) EDX analysis of (d)
3.3 Corrosion electrochemical measurements
The sample 1 (5.8 mA/cm2, 60 min) and sample 2 (12.3 mA/cm2, 60 min) were measured in a 3.5% NaCl aqueous solution by potentiodynamic polarization curves (Fig. 4). Sample 1 with bulk grains has no passive behavior. It hardly protects the AZ31 Mg substrate from corrosion. The anodic passivation behavior of sample 2 is found. The passive current is 6.8?10-2 mA/cm2 as the same order of magnitude as the Al deposition obtained in Ref. [11]. Beyond this point positive to -1.1 V, the pitting corrosion broke the thin passive oxide layer in the active corrosion region. Sample 2 with spherical equiaxed grains has a better anti-corrosion property than sample 1 with bulk grains.
The samples 1, 2 and 3 (12.3 mA/cm2, 120 min) were measured in a 3.5% NaCl aqueous solution by electrochemical impedance spectroscopy (Fig. 5). The AZ31 Mg alloy has an electrochemical reaction resistance (Rct) of about 500 Ω?cm2 according to the diameter of one single capacitive loop in the AZ31 impedance plot (see Fig. 5(a), showing that it is easy to be corroded in the NaCl solution. Nevertheless, the electrodeposition of Al can significantly improve the corrosion-resistance of the substrate. The Rct values of samples 2 and 3 are at least above 1 100 Ω?cm2 and 2 100 Ω?cm2. The Rct of sample 1 reaches only 50 Ω?cm2, by far less than the substrate, samples 2 and 3. Many pores exist in the sample 1 deposition with bulk grains, as seen from Fig. 2(a). The NaCl solution permeates the Al deposition through the pores up to the Mg substrate. It follows that the Al deposition does not stop AZ31 from corrosion, but promotes its destruction. A rapid corrosion between a big cathode (Al deposition) and a small anode (AZ31 substrate) works instead of the protection of cathodic Al coating. Obviously, a cathodic coating will not prevent rusting if there are pinholes or cracks in it [20]. This explains the Rct of sample 1 considerably less than other samples, especially AZ31. The samples above were immersed in the NaCl solution for 60 min. The experimental results showed the Mg substrate was easy to be corroded owing to the hydrogen evolution reaction during the immersion process. There were some deep etching pits on AZ31 surface. However, the surface of samples 2 and 3 had no obvious change. The porosity of sample 1 invalidated the protection of the cathodic Al deposition. Furthermore, sample 1 had such a poor corrosion resistance that its silvery-colored Al deposition lost metallic luster and was destroyed instead of the black corrosion products. The immersion experimental results are in accordance with the EIS analysis results. To confirm the conclusion above, the Al deposition with bulk grains was prepared at 5.8 mA/cm2 for 180 min, which is thicker than sample 1. It was tested using the EIS and immersion experimental. The results showed that it still had a poor anti-corrosion similar to sample 1. The possible reason is that the surface of zinc-immersion films is quite coarse in microscopic view, resulting from the corrosion reaction of the chemical pickling. It is too hard for the bulk grains formed to occupy the everywhere of zinc layer surface. Meantime, the bulk grains are difficult to grow fully at a low current, even if with a prolonged plating time. So many pinholes among bulk grains are probable to connect with each other through the entire Al deposition, resulting in the poor corrosion.

Fig. 3 Cross-section SEM images of as-deposited Al deposition on AZ31 Mg alloy obtained at 12.3 mA/cm2 for different plating time: (a) 30 min; (b) 60 min; (c), (d) 120 min
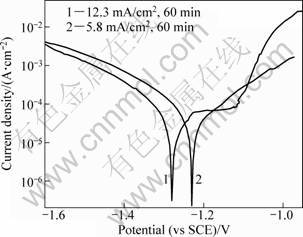
Fig. 4 Polarization curves of as-deposited Al coating on AZ31 magnesium alloy obtained at 5.8 mA/cm2 and 12.3 mA/cm2 for 60 min
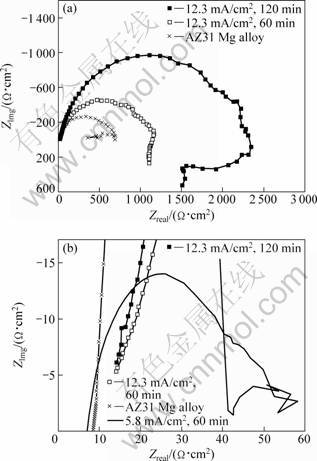
Fig. 5 Electrochemical impedance spectra: (a) As-deposited Al coatings prepared at high current and AZ31 Mg alloy; (b) Al coatings prepared at low current
Figure 6 shows the time variation curve of open circuit potential (OCP) of samples 2 and 3 immersed in a NaCl solution. The samples 2 and 3 have a relatively steady OCP with the prolonged immersion time. However, the OCP of sample 3 less fluctuant than sample 2 shows that the former with a thicker Al layer has a better anti-corrosion property than the latter. The results of OCP vs time are in accordance with the EIS analysis results. As for a direct current plating, the thicker Al deposition can more effectively protect the Mg substrate. It is considered that an intermediate layer with less metallic activity than Zn and Al would be prepared on the Mg alloy surface. It probably makes OCP of the Al-deposited Mg alloy shift toward a positive orientation to more effectively protect Mg substrate. Besides, the Al deposition with a microstructure of fine grains could be electroplated on Mg alloys using the pulse current plating. The methods maybe improve more significantly the anti-corrosion property of Mg alloys than the present work.
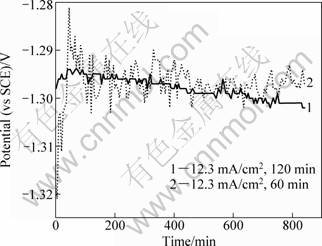
Fig. 6 Relationship between open circuit potential (OCP) and time in 3.5% NaCl solution
4 Conclusions
1) The silvery-colored satin Al deposition was electroplated using the constant current method from TMPAC-AlCl3 ionic liquids with 50% benzene. The microstructures show spherical equiaxed grains obtained at the high current and bulk grains at the low current, respectively.
2) The Al deposition with spherical equiaxed grains has a compact surface obtained at 12.3 mA/cm2. The electrochemical measurements indicate that the thicker Al deposition can more effectively protect the Mg substrate. The Al deposition with bulk grains hardly protects the AZ31 Mg substrate from corrosion owing to its porosity, even if it is thicker.
References
[1] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys―A critical review [J]. J Alloys Compd, 2002, 336: 88-113.
[2] LIU L J, SCHLESINGER M. Corrosion of magnesium and its alloys [J]. Corros Sci, 2009, 51: 1733-1737.
[3] CHANG J K, CHEN S Y, TSAI W T, DENG M J, SUN I W. Electrodeposition of aluminum on magnesium alloy in aluminum chloride (AlCl3)-1-ethyl-3-methylimidazolium chloride (EMIC) ionic liquid and its corrosion behavior [J]. Electrochem Commun, 2007, 9: 1602-1606.
[4] ZHAO Y, VANDERNOOT T J. Review: Electrodeposition of aluminium from nonaqueous organic electrolytic systems and room temperature molten salts [J]. Electrochim Acta, 1997, 42: 3-13.
[5] SIMKA W, PUSZCZYK D, NAWRAT G. Electrodeposition of metals from non-aqueous solutions [J]. Electrochim Acta, 2009, 54: 5307-5319.
[6] HURLEY F H, WIER T P Jr. The electrodeposition of aluminum from nonaqueous solutions at room temperature [J]. J Electrochem Soc, 1951, 98: 207-212.
[7] ALI M R, NISHIKATA A, TSURU T. Electrodeposition of Co-Al alloys of different composition from the AlC13-BPC-CoC12 room temperature molten salt [J]. Electrochim Acta, 1997, 42: 1819-1828.
[8] ALI M R, NISHIKATA A, TSURU T. Electrodeposition of Al-Ni intermetallic compounds from aluminum chloride-N-(n-butyl) pyridinium chloride room temperature molten salt [J]. J Electroanal Chem, 2001, 513: 111-118.
[9] JIANG T, CHOLLIER BRYM M J, DUBE G, LASIA A, BRISARD G M. Electrodeposition of aluminium from ionic liquids: Part I―Electrodeposition and surface morphology of aluminium from aluminium chloride (AlCl3)-1-ethyl-3-methylimidazolium chloride ([EMIm]Cl) ionic liquid [J]. Surf Coat Technol, 2006, 201: 1-9.
[10] LIU Q X, ZEIN E L, ABEDIN S, ENDRES F. Electroplating of mild steel by aluminium in a first generation ionic liquid: A green alternative to commercial Al-plating in organic solvents [J]. Surf Coat Technol, 2006, 201: 1352-1356.
[11] CHANG J K, CHEN S Y, TSAI W T, DENG M J, SUN I W. Improved corrosion resistance of magnesium alloy with a surface aluminum coating electrodeposited in ionic liquid [J]. J Electrochem Soc, 2008, 155: C112-C116.
[12] ZHAO Y, VANDERNOOT T J. Electrodeposition of aluminium from room temperature AlCl3-TMPAC molten salts [J]. Electrochim Acta, 1997, 42: 1639-1643.
[13] JIANG T, CHOLLIER BRYM M J, DUBE G, LASIA A, BRISARD G M. Electrodeposition of aluminium from ionic liquids: Part Ⅱ―Studies on the electrodeposition of aluminum from aluminum chloride (AlCl3)-trimethylphenylammonium chloride (TMPAC) ionic liquids [J]. Surf Coat Technol, 2006, 201: 10-18.
[14] ZHANG J F, YAN C W, WANG F H. Electrodeposition of Al-Mn alloy on AZ31B magnesium alloy in molten salts [J]. Appl Surf Sci, 2009, 255: 4926-4932.
[15] PAPAGEORGIOU N, EMMENEGGER F P. The effect of cosolvents and additives on the electrochemical properties of [(Me)3PhN][Al2Cl7] melts [J]. Electrochim Acta, 1993, 38: 245-252.
[16] MOY R, EMMENEGGER F P. Co-cosolvents for chloroaluminate electrolytes [J]. Electrochim Acta, 1992, 37: 1061-1068.
[17] MOFFAT T P. Electrodeposition of Al-Cr metallic glass [J]. J Electrochem Soc, 1994, 141: L115-117.
[18] QIAN Hong-mei, LI Yan, LING Guo-ping. Influence of acid pickling on electrodeposition of aluminum on magnesium alloy in room temperature molten salts [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(5): 854-860. (in Chinese)
[19] PAUNOVIC M, SCHLESINGER M. Fundamentals of electrochemical deposition [M]. 2nd ed. New York: John Wiley & Sons Inc, 2006: 276.
[20] EVANS U R. An introduction of metallic corrosion [M]. 2nd. London: Edward Arnold, LTD, 1963: 170.
刘奎仁,柳 泉,韩 庆,涂赣峰
东北大学 材料与冶金学院,沈阳 110004
摘 要:采用TMPAC-AlCl3离子液体,在浸锌后的镁合金AZ31表面实施恒流电镀铝,采用电化学技术评估镀铝层的耐蚀性,并采用SEM/EDX等技术表征镀层。结果表明:在浸锌后的镁合金表面可获得表面光洁的银白色镀铝层。在不同的电流密度下,呈现出两种形貌不同的镀铝层;在较佳电流密度12.3 mA/cm2时,镀铝层表面较致密;电镀时间越长,镀层越厚,镀铝层耐蚀性越好。
关键词:镁合金;电镀;离子液体;铝;腐蚀
(Edited by LI Xiang-qun)
Corresponding author: LIU Kui-ren; Tel/Fax: +86-24-83686997; E-mail: liukr@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(11)60980-1

