
Fabrication of Cr coating on AZ31 magnesium alloy by magnetron sputtering
WU Guo-song(吴国松)1, WANG Ai-ying(汪爱英)1, DING Ke-jian(丁克俭)2,
XU Cai-yun(徐采云)1, DAI Wei(代 伟)1, XU Ai-jiao(徐爱娇)1
1. Ningbo Institute of Materials Technology and Engineering, Chinese Academy of Sciences,
Ningbo 315201, China;
2. Institute of Biological Science and Technology, Beijing Jiao Tong University, Beijing 100044, China
Received 12 June 2008; accepted 5 September 2008
Abstract: Chromium-plating is considered an effective method to improve surface properties of metal materials. Magnetron sputtering was applied to prepare Cr coating on AZ31 magnesium alloy and the influence of bias voltage on properties of Cr coating was investigated. The obtained coatings present an (110) preferred texture and have a developed columnar structure. With increasing bias voltage, the surface structure of Cr coating becomes denser. All of the Cr-coated AZ31 have much higher surface microhardness than bare AZ31. The Cr coating deposited on AZ31 at bias voltage of -150 V has a higher corrosion potential than others. The result of the immersion test shows that it decreases the damaged area compared with bare AZ31. The failure of the coated AZ31 is mainly attributed to the existence of through-thickness defects in the coating.
Key words: magnesium alloy; Cr coating; magnetron sputtering; corrosion
1 Introduction
Magnesium alloy has been a technologically important material for its wide range of industrial applications in recent years. Unfortunately, its poor wear and corrosion resistance limit its extended utilization [1-4]. Today, surface treatment is used to increase the lifetime of magnesium alloy exposed to aggressive conditions in many engineering application[5]. Chromium coating is one of the most important protective coatings and has played a key role in surface treatment of metal materials. Usually, chromium coating is deposited on the substrate by chemical methods, e.g. electrochemical plating. However, these approaches make use of toxic reactants and produce toxic wastes and environmental pollutants[6-7]. Therefore, alternative technologies should be studied and developed.
Physical vapor deposition(PVD) is a clean environmental friendly technique, and does not produce wastes or appreciable atmospheric emissions[8]. Several PVD coating systems on magnesium alloy, such as Ti, Al, TiN, CrN and AlN, have already been well studied in the past years[9-15]. But, to the best of our knowledge, the preparation of Cr coating on magnesium alloy with PVD technique has not been reported. In this work, we attempted to deposit chromium coating on AZ31 magnesium alloy by a novel hybrid ion beam deposition system, which includes a linear ion source and a magnetron sputtering source. Furthermore, we deeply investigated the crystalline structure, morphology and anti-corrosion property of the obtain coating.
2 Experimental
As-extruded AZ31 plates were selected as substrates in this study. They were ground with emery paper up to 1500# and polished with Al2O3 paste (average size 1 μm). A hybrid ion beam deposition system, including a linear ion source and a magnetron sputtering source, was used to prepare Cr coating on AZ31 plate. The substrates were ultrasonically washed in pure alcohol for 5 min before they were sent into the vacuum chamber. When the base pressure of the chamber was below 2.66×10-3 Pa, the ion source with Ar gas was applied to clean the substrate for 30 min. The deposition was performed at room temperature with argon as sputtering gas. The specific parameters are shown as follows: base pressure of 2.66×10-3 Pa, Ar flux of 40 mL/s, DC sputtering current of 3 A and deposition time of 90 min. In this series of experiments, bias voltage was applied at pulse mode with frequency of 350 kHz and its value was chosen as 0, -50, -150 and -250 V for investigation, respectively.
After deposition, scanning electron microscopy (SEM) observation was performed to characterize the cross section and surface morphology of the obtained coatings. X-ray diffractometer with Cu Kα radiation was used to study the crystal structure of the obtained coatings. The surface mechanical property of the specimens was characterized by a KH-1000 microhardness meter. The load was selected as 245 mN and the load time was set at 10 s. For the electrochemical investigation, the experiments were controlled by PARSTAT 273A advanced electrochemical system, using the conventional three-electrode technique. The potential was referred to a saturated calomel electrode(SCE) and the counter electrode was a platinum sheet. Each sample was masked by paraffin wax with the surface area of 1.0×1.0 cm2 exposed in 3.5%(mass fraction) NaCl solution. The test was carried out at 1 mV/s at room temperature after samples were immersed in the above solution for 2 min. In addition, an immersion test, which used 3.5% NaCl solution as corrosive media, was also performed to evaluate the corrosion resistance of the
coated sample.
3 Results and discussion
Fig.1 shows the XRD patterns of these chromium coatings deposited on AZ31 substrate at different bias voltages. Compared with the XRD curve of AZ31, two peaks of Cr coating are clearly observed, which is corresponded to the planes (110) and (211), respectively. According to the comparison of peak intensity, it is indicated that Cr(110), densely packed plane, is the preferred growth orientation in all the Cr coatings.
Fig.2 shows the cross section morphologies of the obtained coatings on AZ31 substrate. From the SEM observation, it can be seen that all the coatings cover the substrate homogeneously. Furthermore, it can be determined that the average thickness of the coating is about 2.5 ?m. The insert shows the morphology at a high magnification and it reveals that all the Cr coatings have a developed columnar structure.
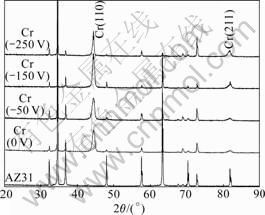
Fig.1 XRD patterns of Cr coatings deposited on AZ31 substrate at different bias voltages

Fig.2 Cross sectional SEM images of Cr coatings deposited on AZ31 at different bias voltages: (a) 0 V; (b) -50 V; (c) -150 V; (d) -250 V
Fig.3 shows the surface morphology of Cr coatings deposited on AZ31 at different bias voltages. The left of these panels shows the surface structure at a lower magnification and the right shows the one at a higher magnification. The surface of Cr coating becomes smoother and the microstructure of Cr coating becomes denser with the increase of bias voltage, which is due to the effect of ions impact. Therefore, it is indicated that increasing bias voltage is beneficial to the densification of Cr coating.

Fig.3 Surface SEM images of Cr coatings deposited on AZ31 at different bias voltages: (a), (a′) 0 V; (b), (b′) -50 V; (c), (c′) -150 V; (d), (d′) -250 V
Fig.4 shows the surface microhardness of those samples prepared in this study. The surface microhardness of AZ31 is greatly improved due to the formation of Cr coating. As shown in this figure, there is no distinct difference on the microhardness between these coated samples deposited at different bias voltages. Because the coatings prepared in this study are thin, the obtained values are greatly influenced by the microhardness of AZ31 substrate.
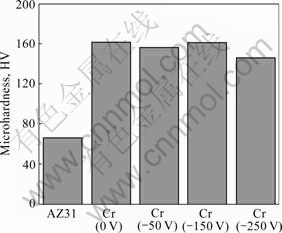
Fig.4 Microhardness of bare AZ31 and Cr-coated samples
Fig.5 shows the polarization curves of all samples in this study. From this image, it can be seen that the coated AZ31 prepared at bias voltage of -150 V has a higher corrosion potential than other samples and its corrosion potential is shifted positively about 200 mV compared with that of the uncoated sample. Although the coating deposited at bias voltage of -150 V is much more compact than those deposited at bias voltage of 0 V and -50 V, it can be speculated that there must be some through-thickness defects still existing in this coating, according to the theory of the mixed potential. From the SEM images, the coating deposited at bias voltage of -250 V is more compact and smoother than that deposited at bias voltage of -150 V. But its corrosion potential is lower. Until now, the true reason is unknown. It is probably due to the poorer adhesion between coating and substrate and the higher expanding rate of corrosion pit. Fig.6 shows the surface appearance of the Cr-coated AZ31 and bare AZ31 after 1 h of immersion in 3.5% NaCl solution. Obviously, the Cr-coated sample has much less corrosion pits than bare AZ31. It means that this Cr coating protects the substrate effectively during this period of investigation. With increasing the immersion time, the domain of corrosion pits is gradually enlarged and the amount is also increased due to the progress of galvanic corrosion. Finally, it induces the failure of the Cr-coated AZ31.
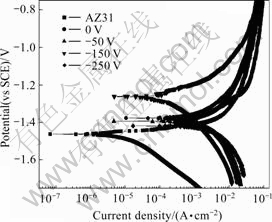
Fig.5 Polarization curves of Cr coatings deposited on AZ31 at different bias voltages

Fig.6 Surface appearance of samples after 1 h of immersion
4 Conclusions
1) Chromium coating has been successfully deposited on AZ31 magnesium alloy by a hybrid ion beam deposition system that includes a linear ion source and a magnetron sputtering source.
2) All Cr coatings prepared in this study present an (110) preferred texture and has a developed columnar structure. The surface structure of chromium coating turns more compact with increasing bias voltage.
3) The surface microhardness of the Cr coated AZ31 is distinctly improved compared with that of bare AZ31. The Cr-coated sample prepared at bias voltage of -150 V has a higher corrosion potential than other samples in this study. The immersion test shows that this Cr coating protects the substrate effectively during the period of investigation. The failure of the coated AZ31 is mainly attributed to the existence of through-thickness defects in the coating.
References
[1] SONG G, ATRENS A, WU X, ZHANG B. Corrosion behavior of AZ21, AZ501, and AZ91 in sodium chloride [J]. Corrosion Science, 1998, 40(10): 1769-1791.
[2] SONG G, STJOHN D. The effect of zirconium grain refinement on the corrosion behaviour of magnesium-rare earth alloy MEZ [J]. Journal of Light Metals, 2002, 2(1): 1-16.
[3] SONG G, BOWLES A, STJOHN D. Corrosion resistance of aged die cast magnesium alloy AZ91D [J]. Mater Sci Eng A, 2004, 366(1): 74-86.
[4] FRACASSI F, DAGOSTINO R, PALUMBO F, ANGELINI E. Application of plasma deposited organosilicon thin films for the corrosion protection of metals [J]. Surface & Coatings Technology, 2003, 174/175(9): 107-111.
[5] GRAY J E, LUAN B. Protective coating on magnesium and its alloys―A critical review [J]. Journal of Alloys and Compounds, 2002, 336(1/2): 88-113.
[6] UMEHARA H, TAKAYA M, TERAUCHI S. Chrome-free surface treatments for magnesium alloy [J]. Surface and Coatings Technology, 2003, 169/170(6): 666-669.
[7] ZENG Zhi-xiang, WANG Li-ping, LIANG Ai-min, ZHANG Jun-yan. Tribological and electrochemical behavior of thick Cr-C alloy coatings electrodeposited in trivalent chromium bath as an alternative to conventional Cr coatings [J]. Electrochimica Acta, 2006, 52(3): 1366-1373.
[8] NAVINSEK B, PANJAN P, MILOSEV I. PVD coatings as an environmentally clean alternative to electroplating and electroless processes [J]. Surface & Coatings Technology, 1999, 116/119(9): 476-487.
[9] ZHANG Er-lin, XU Li-ping, YANG Ke. Formation by ion plating of Ti-coating on pure Mg for biomedical applications [J]. Scripta Materialia, 2005, 53(5): 523-527.
[10] WU S K, YEN S C, CHOU T S. A study of r.f.-sputtered Al and Ni thin films on AZ91D magnesium alloy [J]. Surface & Coatings Technology, 2006, 200(8): 2769-2774.
[11] HOCHE H, SCHEERER H, PROBST D. Development of a plasma surface treatment for magnesium alloys to ensure sufficient wear and corrosion resistance [J]. Surface & Coatings Technology, 2003, 174/175(9): 1018-1023.
[12] HOCHE H, ROSENKRANZ C, DELP A. Investigation of the macroscopic and microscopic electrochemical corrosion behaviour of PVD-coated magnesium die cast alloy AZ91 [J]. Surface & Coatings Technology, 2005, 193(1/3): 178-184.
[13] HOCHE H, BLAWERT C, BROSZEIT E. Galvanic corrosion properties of differently PVD-treated magnesium die cast alloy AZ91 [J]. Surface & Coatings Technology, 2005, 193(1/3): 223-229.
[14] ALTUN H, SINICI H. Corrosion behaviour of magnesium alloys coated with TiN by cathodic arc deposition in NaCl and Na2SO4 solutions [J]. Materials Characterization, 2008, 59(3): 266-270.
[15] ALTUN H, SEN S. The effect of DC magnetron sputtering AlN coatings on the corrosion behaviour of magnesium alloys [J]. Surface & Coatings Technology, 2005, 197(7): 193-200.
(Edited by HE Xue-feng)
Foundation item: Project(200701A6305013) supported by the Natural Science Foundation of Ningbo City, China
Corresponding author: WU Guo-song; Tel: +86-574-86685171; E-mail: wuguosong@nimte.ac.cn