Effect of introduction of exogenous strain Leptospirillum ferriphilum YSK on functional gene expression, structure, and function of indigenous consortium during pyrite bioleaching
来源期刊:中南大学学报(英文版)2020年第5期
论文作者:刘毅 申丽 王俊俊 刘宏伟 尹华群 刘学端 邱冠周
文章页码:1453 - 1465
Key words:Leptospirillum ferriphilum YSK; indigenous consortium; functional gene arrays (FGAs); pyrite bioleaching
Abstract: Leptospirillum ferriphilum YSK was added to a native consortium of bioleaching bacteria including Acidithiobacillus caldus, A. thiooxidans, A. ferrooxidans, Sulfobacillus thermosulfidooxidans, Acidiphilium spp., and Ferroplasma thermophilum cultured in modified 9K medium containing 0.5% (W/V) pyrite. The bioleaching efficiency markedly increased. Changes in community structure and gene expression were monitored with real-time PCR and functional gene arrays. Dynamic changes that varied in different populations in the consortium occurred after the addition of L. ferriphilum YSK, with growth of A. caldus S1, A. thiooxidans A01, Acidiphillum spp. DX1-1 promoted the growth of Ferroplasma L1, inhibited that of S. thermosulfidooxidans ST, and exerted little effect on that of A. ferrooxidans CMS. Genes encoding ADP heptose, phosphoheptose isomerase, glycosyltransferase, biotin carboxylase, and protoheme ferrolyase from L. ferriphilum, acetyl-CoA carboxylase from Acidiphillum spp., and doxD from A. caldus were up-regulated in 0–20 h. Genes encoding lipid A disaccharide synthase LpxB, glycosyl transferase, and ADP heptose synthase from A. ferrooxidans were up-regulated in 0–8 h and then down-regulated in 8–20 h. Genes encoding ferredoxin oxidoreductase from Ferroplasma sp. were up-regulated in 0–4 h, down-regulated in 4–16 h, and again up-regulated in 16–20 h. CbbS from A. ferrooxidans was down-regulated in 0–20 h.
Cite this article as: SHEN Li, WANG Jun-jun, LIU Hong-wei, YIN Hua-qun, LIU Xue-duan, QIU Guan-zhou, LIU Yi. Effect of introduction of exogenous strain Leptospirillum ferriphilum YSK on functional gene expression, structure, and function of indigenous consortium during pyrite bioleaching [J]. Journal of Central South University, 2020, 27(5): 1453-1465. DOI: https://doi.org/10.1007/s11771-020-4381-3.

J. Cent. South Univ. (2020) 27: 1453-1465
DOI: https://doi.org/10.1007/s11771-020-4381-3
SHEN Li(申丽)1, 2, WANG Jun-jun(王俊俊)1, 2, LIU Hong-wei(刘宏伟)1, 2, YIN Hua-qun(尹华群)1, 2,
LIU Xue-duan(刘学端)1, 2, QIU Guan-zhou(邱冠周)1, 2, LIU Yi(刘毅)1, 3
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Biohydrometallurgy of Ministry of Education, Central South University,Changsha 410083, China;
3. Key Laboratory of Agro-ecological Processes in Subtropical Regions and Taoyuan Station of Agro-ecology Research, Institute of Subtropical Agriculture, Chinese Academy of Sciences, Changsha 410125, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: Leptospirillum ferriphilum YSK was added to a native consortium of bioleaching bacteria including Acidithiobacillus caldus, A. thiooxidans, A. ferrooxidans, Sulfobacillus thermosulfidooxidans, Acidiphilium spp., and Ferroplasma thermophilum cultured in modified 9K medium containing 0.5% (W/V) pyrite. The bioleaching efficiency markedly increased. Changes in community structure and gene expression were monitored with real-time PCR and functional gene arrays. Dynamic changes that varied in different populations in the consortium occurred after the addition of L. ferriphilum YSK, with growth of A. caldus S1, A. thiooxidans A01, Acidiphillum spp. DX1-1 promoted the growth of Ferroplasma L1, inhibited that of S. thermosulfidooxidans ST, and exerted little effect on that of A. ferrooxidans CMS. Genes encoding ADP heptose, phosphoheptose isomerase, glycosyltransferase, biotin carboxylase, and protoheme ferrolyase from L. ferriphilum, acetyl-CoA carboxylase from Acidiphillum spp., and doxD from A. caldus were up-regulated in 0–20 h. Genes encoding lipid A disaccharide synthase LpxB, glycosyl transferase, and ADP heptose synthase from A. ferrooxidans were up-regulated in 0–8 h and then down-regulated in 8–20 h. Genes encoding ferredoxin oxidoreductase from Ferroplasma sp. were up-regulated in 0–4 h, down-regulated in 4–16 h, and again up-regulated in 16–20 h. CbbS from A. ferrooxidans was down-regulated in 0–20 h.
Key words: Leptospirillum ferriphilum YSK; indigenous consortium; functional gene arrays (FGAs); pyrite bioleaching
Cite this article as: SHEN Li, WANG Jun-jun, LIU Hong-wei, YIN Hua-qun, LIU Xue-duan, QIU Guan-zhou, LIU Yi. Effect of introduction of exogenous strain Leptospirillum ferriphilum YSK on functional gene expression, structure, and function of indigenous consortium during pyrite bioleaching [J]. Journal of Central South University, 2020, 27(5): 1453-1465. DOI: https://doi.org/10.1007/s11771-020-4381-3.
1 Introduction
Bioleaching [1-4] is widely used in the field of mineral processing. Studies with single strains have been done to improve the leaching efficiency of target metals, and have focused on single strains. Bioengineering technology has been used for domestication, mutagenesis, cell fusion and genetic engineered breeding of isolated strains. In addition, it also can select strains with high-efficiency leaching behavior that is temperature resistant and adapts to a wide range of acidity [1]. However, some researchers suggested the problems in the practical application of a single bacterial strain for leaching. The metabolic process of a single strain is relatively monotonic and therefore cannot effectively use various substances for energy. Secondly, a single strain can adapt relatively poorly to changes in environmental factors. In addition, the characteristics of some strains are unstable and easily lost, making their industrial use difficult.
To overcome these hurdles, metabolically diverse acidophilic microorganisms have been explored in the construction of various co-culture systems for mixed microbial community leaching. Leaching with mixed microbial community can be more efficient than that with a single strain of bacteria. LIAO et al [5] studied the that amount of recycled metals was equal if the tailings were leached by mixed cultures of Leptospirillum ferriphilum and Acidithiobacillus thiooxidans at three different ratios or by pure culture of L. ferriphilum, which was better than the pure culture of Acidithiobacillus ferrooxidans. FALCO et al [6] studied the comparison of covellite bioleaching using a pure culture of Acidithiobacillus ferrooxidans or A. thiooxidans and a mixed culture of Leptospirillum ferrooxidans and A. thiooxidans. BATTAGLIA-BRUNET et al [7] carried out cobalt-bearing pyrite bioleaching with mixed strains of Thiobacillus ferrooxidans, T. thiooxidans, and Microspirilla ferrooxidans. The leaching effect of the mixed bacteria was significantly higher than that of monocultures. DOPSON et al [8] described that the sole use of single A. caldus was ineffective in oxidatively decomposing sulfide minerals, while A. caldus mixed with iron-oxidizing bacteria leached sulfide ore with markedly improved efficiency. OKIBE et al [9] used the pure and mixed moderate thermophilic acidophilic microorganisms to leach pyrite in bioreactors operating at a constant pH and optimized the microbial composition in the mixed leaching system using fluorescence in-situ hybridization. These studies reported that the leaching capacity of the mixed bacteria was much higher than that of either monocultur.
The foregoing supports the view that mixed microbial leaching is better than the use of a single strain in bio-metallurgy. Sulfide leaching efficiency can be effectively improved by cooperative bioleaching [10], in which sulfur- and iron-oxidizing microorganisms are mixed and inoculated in the leaching system. A leaching microbial community is typically composed of many types of microorganisms, such as A. caldus, L. ferriphilum, and Acidiphillum spp., which use elemental sulfur, ferrous iron, and organic materials, respectively [11, 12]. The relationships among these microorganisms are complex, and involve competition, mutualism, and synergy [13], with the leaching microbial community often stabilizing and performing a certain ecological function.
In the bio-metallurgy process, the structure of the leaching microbial community is closely linked to the leaching efficiency of the metallic sulfide minerals [14, 15]. Differences in the population composition of leaching microbial communities lead to distinction in leaching efficiency [6]. However, the structure of a leaching microorganism community can change during the leaching process, with changes of various environmental factors including temperature, pH, and heavy metal ions influencing the leaching efficiency at different stages of leaching [16].
Exogenous species have been introduced to the indigenous community and the impact of the introduced species on population diversity and ecological function of the indigenous community analyzed, to study the relationship between community structure and function [17, 18]. This approach has been widely used in many fields including animals and plants [19, 20]. However, little is known in the field of biological metallurgy. In previous studies, the gene expression of microorganisms has been analyzed in the process of bioleaching. However, most of them are performed without the introduction of foreign strains, and there are few studies on gene expression and structure in the whole leaching process after the introduction of foreign strains into the indigenous leaching microbial community.
This work investigated the influence on the functional gene expression, and the structure and function of the indigenous leaching microbial community was studied after the introduction of L. ferriphilum at different growth stages in the indigenous leaching microbial community. The findings provide a theoretical foundation for the introduction of exogenous strains to indigenous leaching communities in industrial applications.
2 Materials and methods
2.1 Microorganisms
A mixed culture derived from acid mine drainage samples from several copper mines in China was adapted in our previous research [21], and was used as the indigenous consortium in this study. L. ferriphilum YSK strain isolated by our laboratory was also adapted during the low-grade copper sulfide bioleaching and was used as the exogenous strain. Details are provided in Table 1.
2.2 Bioleaching experiments
Pyrite bioleaching experiments were carried out in 250-mL shake flasks containing 100 mL of 0K basal salts medium (without ferrous sulfate supplement) and 0.5% (W/V) pulp density at 40 °C, 170 r/min, and an initial pH of 1.5. Consortium A was inoculated only with the indigenous community, as a control group in 33 bottles. Consortium B was simultaneously inoculated with L. ferriphilum YSK and indigenous community (the final bacterial concentration was approximately 107 cells/mL) in 18 bottles. Consortium C was inoculated with L. ferriphilum YSK during the period of rapid growth of indigenous microbial community (at 66 h) in 12 bottles. Consortium D was inoculated with L. ferriphilum YSK when growth of the indigenous microbial community had stabilized after 138 h in 21 bottles. The bioleaching experiments were carried out in triplicate. The experiment performed for 240 h.
At 24, 72, 96, 144, 168 and 216 h, genomic DNA was extracted from consortiums A and B. Genomic DNA from consortium C was extracted at 96, 144, 168, and 216 h. Genomic DNA from consortium D was extracted at 168 and 216 h. Total RNA was extracted from consortiums A and D at 146, 150, 154, and 158 h. In addition, before the introduction of L. ferriphilum YSK, the RNA from pure culture was extracted as a control. Deionized water was added to flasks to compensate for evaporation loss.
2.3 Analytical techniques
Planktonic bacteria were counted using a Thomas chamber with an optical microscope. Total dissolved iron concentrations in solution were measured by atomic absorption spectrophotometry. Ferrous iron concentration was determined by titration with potassium dichromate (K2Cr2O7). The pH of the leaching systems was measured with a pH meter. Planktonic and biofilm-associated cells were collected in Ref. [22]. Samples from flasks with consortium A and consortium D were collected, filtered, dried in air, and examined by scanning electron microscopy (SEM) at 10 h after the introduction of L. ferriphilum YSK.
2.4 DNA extraction and purification
Planktonic and sessile cells were collected as previously described in Ref. [22]. DNA from planktonic, sessile, and total cells was extracted using a TIANamp genomic DNA purification kit (Tiangen Biotech, Co., Ltd., Beijing, China). The crude DNA was purified by the mini-column purification method and quantified by ethidium bromide-ultraviolet detection on an agarose gel. The concentration of the purified genomic DNA was measured by spectrophotometry using a ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA).
Table 1 Acidophiles used in pyrite leaching experiments

2.5 16S rRNA gene PCR, fractionation, cloning, restriction digestion, sequencing, and analysis
The 16S rRNA gene PCR, fractionation, cloning, restriction digestion, sequencing, and analysis were done as previously described in Ref. [23].
2.6 PCR and real-time PCR
The primers used in this study are listed in Table 2 in Ref. [21]. They have been previously described in Ref. [24] and were synthesized by Shanghai Sangon Company (Shanghai, China). The conventional PCR program, purification of PCR products, and the calculation of DNA copy numbers have been described in Ref. [24]. The PCR products were diluted serially from 103 to 1010 copies/μL by real-time PCR to construct standard curves. The real-time PCR was carried out with iCycler iQ Real-time PCR detection system (Bio-Rad Laboratories Inc., Hercules, CA, USA). The detailed steps for quantitative analysis were previously described in Ref. [24].
2.7 Microarray design, construction, hybridization, and data analysis
The design and construction of the microarray has been previously described in Ref. [23] by DNA labeling, microarray hybridization, and data analysis [25, 26]. Three biological replicates were used per treatment (each treatment was labeled three times to eliminate possible dye-labeling bias).
3 Results and discussion
3.1 Comparison of pyrite leaching among consortia
During the leaching process, the total iron (Fe2+ and Fe3+) after oxidative decomposition comprises two metallic sulfide ores. One is in the form of ions in leaching solution and the other forms a solid precipitate with other elements (mainly to form jarosite) [27]. To confirm the production and amount of jarosite during the leaching of pyrite, thus ensuring the reliability of the calculation methods and results of leaching efficiency, pyrite residuals of consortiums A and D for X-ray diffraction analysis were collected after 10 d of pyrite leaching (Figure 1). The content of jarosite was only 2.4% in consortium D with a higher total iron concentration in solution, while no jarosite was shown in consortium A. A small amount of jarosite formed during pyrite leaching has been reported, especially when pH is low (pH<1.7) [9]. Therefore, the pyrite leaching efficiency can be calculated with the total iron concentration of the solution by determining the content of total iron in the solution divided by the content of total iron in the ore.
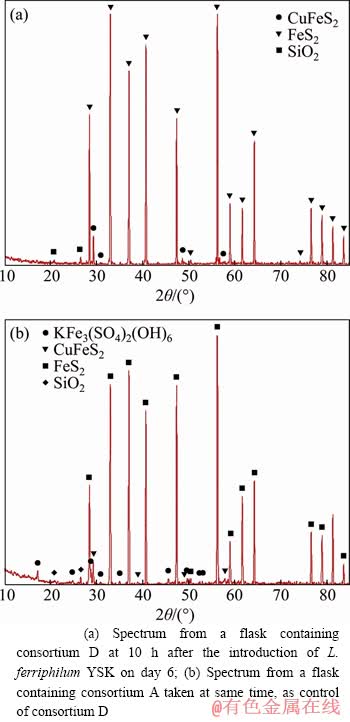
Figure 1 X-ray diffraction spectra of two different pyrite residuals: (a) Spectrum from a flask containing consortium D at 10 h after the introduction of L. ferriphilum YSK on day 6; (b) Spectrum from a flask containing consortium A taken at same time, as control of consortium D
Figure 2 shows the comparison of pyrite leaching after the introduction of L.ferriphilum YSK at different periods to the indigenous leaching community. The Fe leaching efficiency during pyrite bioleaching of different consortia is described in Figure 2(a). The leaching efficiency of pyrite reached 96.4% after the introduction of L. ferriphilum YSK at the stable period (Consortium D). In the fast growth period (Consortium C), the leaching efficiency was 91.1%. When L. ferriphilum YSK was inoculated simultaneously with the indigenous community (Consortium B), the extraction rate was relatively poor (86.1%). The leaching rate of pyrite in Consortium A was only 77.1%. The results indicated that the introduction of L. ferriphilum YSK can enhance the pyrite leaching efficiency of the indigenous microbial community, especially when introduced when the community is stable. Compared with consortium A, the leaching rate was improved by approximately 19%. The rapid oxidation of pyrite produced a large amount of Fe2+ after the growth period, while the iron-oxidizing bacteria L. ferriphilum YSK grew faster due to the increase in energy. According to the indirect mechanism [28], a large amount of Fe2+ was oxidized into Fe3+, providing oxidant for minerals, thereby accelerating the ore dissolution.
Figure 2(b) depicts the effect on Fe2+ concentration after the introduction of L. ferriphilum YSK at different periods in leaching solution. The ferrous iron concentration was relatively decreased in the leaching solution after the introduction of L. ferriphilum YSK, compared to Consortium A. This was likely due to the fact that L. ferriphilum YSK used Fe2+ as the sole energy source [29]. The introduction of L. ferriphilum YSK accelerated the use of Fe2+, which led to the decrease in ferrous iron.
The pH changes after the introduction of L. ferriphilum YSK at different periods in the leaching solution are presented shown in Figure 2(c). The pH of the leaching system increased at the pre-leaching period (1-5 d), followed by a decrease at the later leaching stage (5-10 d) after the initial introduction of L. ferriphilum YSK (Consortium B), compared to the control group (Consortium A). In the leaching systems where L. ferriphilum YSK was introduced when growth was rapid and stable (Consortium C and D, respectively), the pH of the leaching solution was relatively increased. The chemical formulae related to acid production and acid consumption in the leaching of pyrite are [28]:
4Fe2++O2+4H+→4Fe3++2H2O
2S0+2H2O+3O2→2SO42-+4H+
Consortium B featuring the initial introduction of L. ferriphilum YSK was influenced by the iron-oxidizing bacterium with a strong oxidizing ability to Fe2+. Accordingly, acid consumption was higher than acid consumption in the early leaching stage (1-5 d), resulting in the increase in pH. In the later period (5-10 d), the rapid oxidation of pyrite provided more energy for sulfur-oxidizing bacteria, resulting in more acid production than acid consumption, and thus a relative decrease in pH. For Consortiums C and D, the introduction of L. ferriphilum YSK significantly affected the oxidation of Fe2+. According to the literature in Ref. [28], at the middle or later period of pyrite leaching, the competitive adsorption of Fe2+/Fe3+ ion leads to the formation of a passivation film on the ore surface. The introduction of L. ferriphilum YSK promoted the elimination of this film due to the oxidation of Fe2+, which led to the pH increase.
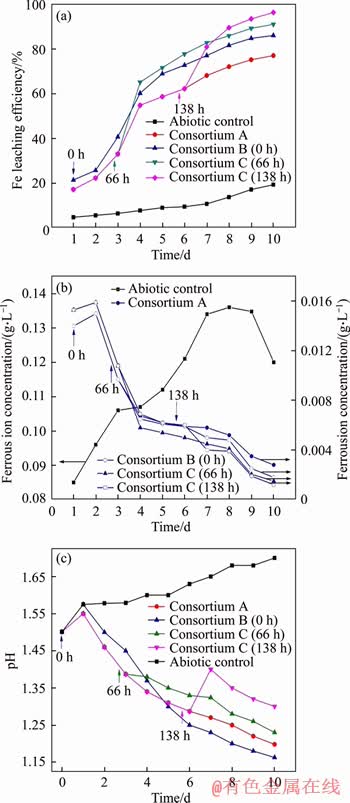
Figure 2 Total iron extraction rate (a), ferrous iron concentrations (b), and pH values (c) during pyrite bioleaching by pure (Consortium A) and mixed cultures (Consortiums B, C, and D) (Each point in the figure is a mean value ± standard deviation (n=3) of samples)
Figure 3 shows a representative SEM image of pyrite residuals following the introduction of L. ferriphilum YSK during the rapid growth period (Consortium C). Compared to the control group (Figures 3(b) and (d)), the number of bacteria on the surface of pyrite residuals increased significantly on day 3 and 6 after the introduction of L. ferriphilum YSK, indicating that the introduced bacteria were mainly adsorbed on the ore surface.
3.2 Analysis of population dynamics
3.2.1 Community dynamics analysis in Consortium A
Community dynamics in Consortium A (Figure 4(a)) indicated that A. caldus S1 was the dominant strain during the entire bioleaching process. Prior research reports [6, 20] have documented that in the leaching of pyrite, the concentration of sulfur-oxidizing microorganisms often exceeds that of iron-oxidizing organisms. This is mainly because the complete oxidation of sulfur releases more energy than that of ferrous iron [9, 30], thus meeting the growth requirements of the number of sulfur-oxidizing bacteria. Ferroplasma L1 and S. thermosulfidooxidans ST were the second and third dominant species in the indigenous community, respectively. Without the competition of L. ferriphilum YSK, Fe2+ was exclusively utilized by Ferroplasma L1. In addition, the use of organic matter guaranteed its dominant position in the community [31]. S. thermosulfidooxidans ST was the third dominant group in Consortium A, which was very different from a previous study [21] that reported fewer S. thermosulfidooxidans ST in the presence of L. ferriphilum YSK in the indigenous community. However, without the involvement of L. ferriphilum YSK in the competition for energy, the growth of S. thermosulfidooxidans ST was better. The growth of At. thiooxidans A01, A. ferrooxidans CMS, and Acidiphillum spp. DX1-1 were relatively poor in Consortium A. Among them, the pattern of A. thiooxidans A01 was slightly lower from a previous study [21]. The reason was that the initial inoculum of A. thiooxidans A01 used previously was higher in Ref. [21]. The dynamic changes in A. ferrooxidans CMS and Acidiphillum spp DX1-1 were previously reported as not being significantly different.

Figure 3 SEM images of four different pyrite residuals:
3.2.2 Community dynamics analysis in Consortium B
Compared to Consortium A, the introduction of L. ferriphilum YSK had a great effect on the growth of various populations in the indigenous community (Figure 4(b)). Among them, A. caldus S1 remained the dominant population. However, the bacterial concentration increased slightly in every time period, compared with Consortium A. For example, in Consortium B, the highest concentration of A. caldus S1 was on day 7 (7.63×107 cells/mL) during the entire leaching period, while the highest concentration in Consortium A 5.71×107 cells/mL on day 7. Since L. ferriphilum YSK displays a relatively strong oxidative decomposition of pyrite, its introduction can accelerate the decomposition of pyrite to release more elemental sulfur, which provided more energy for the growth of sulfur-oxidizing bacteria, such as A. caldus S1. This was also the reason why the bacterial concentration of A. thiooxidans A01, a sulfur-oxidizing bacterium, was higher in Consortium B than in Consortium A. However, the introduction of L. ferriphilum YSK significantly inhibited the growth of Ferroplasma L1, and especially S. thermosulfidooxidans ST. In Consortium A, without the competition of L. ferriphilum YSK, although S. thermosulfidooxidans ST did not grow under optimum environmental conditions, it still had a higher bacterial concentration since it could obtain sufficient energy for growth. However, in Consortium B, due to competitive disadvantages, energy was limited and growth was significantly inhibited. The optimum growth conditions of Ferroplasma L1 were similar to the conditions in this experiment, and this microorganism can use organic matter, which could compensate for the detriment of competing for ferrous iron. Therefore, Ferroplasma L1 still formed a high proportion of the community in Consortium B. Although other disadvantaged populations were slightly changed in Consortium B (for example, the increase in Acidiphillum spp. DX1-1), the changes were not significant.
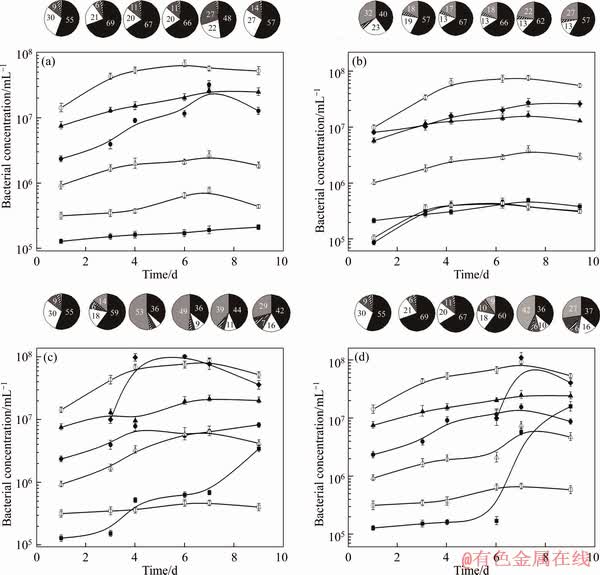
Figure 4 Microbial community dynamics analysis of A. caldus S1 (○, black part in pie charts), F. thermophilum L1(▲, white part in pie charts), L. ferriphilum YSK ( , light gray part in pie charts), S. thermosulfidooxidans ST(●), A. thiooxidans A01(△), A. ferrooxidans DMS(□), and Acidiphilium spp (■) in Consortiums A and B (at 24, 72, 96, 144, 168, and 216 h), Consortium C (at 96, 144, 168, and 216 h), and Consortium D (at 168 and 216 h) during pyrite bioleaching, as determined by RT-PCR. The cell counts are calculated based on the equivalents of gene copy numbers. Each point in the figure is the mean ± standard deviation (n=3). The pie-charts show the relative numbers of the bacteria in consortiums A (a), B (b), C (c) and D (d) during pyrite bioleaching, as determined by RT-PCR (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article)
, light gray part in pie charts), S. thermosulfidooxidans ST(●), A. thiooxidans A01(△), A. ferrooxidans DMS(□), and Acidiphilium spp (■) in Consortiums A and B (at 24, 72, 96, 144, 168, and 216 h), Consortium C (at 96, 144, 168, and 216 h), and Consortium D (at 168 and 216 h) during pyrite bioleaching, as determined by RT-PCR. The cell counts are calculated based on the equivalents of gene copy numbers. Each point in the figure is the mean ± standard deviation (n=3). The pie-charts show the relative numbers of the bacteria in consortiums A (a), B (b), C (c) and D (d) during pyrite bioleaching, as determined by RT-PCR (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article)
3.2.3 Community dynamics analysis in Consortium C
Community dynamics in consortium C are shown in Figure 4(c). L. ferriphilum YSK quickly adapted to the growth environment, and grew rapidly on day 3. On day 4, it was the dominant population with bacterial concentration of 9.83×107 cells/mL. The optimal growth conditions of L. ferriphilum YSK were similar to the experimental conditions, which lead to its rapid adaptation to the growth environment. However, according to Ref. [32], there is a relatively high concentration of Fe2+ in the early stages of the metal sulfide minerals bioleaching. Therefore, the introduction of L. ferriphilum YSK during the rapid growth period, in which Fe2+ is relatively abundant, provides the energy source for rapid growth. Due to their growth inhibition, the concentration of Ferroplasma L1 and S. thermosulfidooxidans ST decreased, compared to Consortium A. For specific sulfur-oxidizing bacteria, such as A. caldus S1 and A. thiooxidans A01, the bacterial concentration was increased, compared to Consortium B, but the overall trend of change was very similar to that of Consortium B. Among the low-abundance populations, the concentration of Acidiphillum spp. DX1-1 significantly increased. This may be related to the increased metabolic activity of the community in response to disturbances from exotic species.
3.2.4 Community dynamics analysis in Consortium D
Community dynamics in Consortium D are depicted in Figure 4(d). The population trends were similar to those of Consortium C. The causes of these changes were basically the same as the other consortia. The difference was mainly reflected in the bacteria concentration. Compared with other communities, there were two phenomena in Consortium D. Firstly, L. ferriphilum YSK rapidly become a dominant species. At the later stage of pyrite leaching, a Fe2+/Fe3+ film forms on the ore surface due to competitive adsorption [28]. The microorganisms were initially adsorbed on the ore surface in the early leaching stage [33]. Therefore, Fe2+ accumulated on the ore surface provided energy for the growth of L. ferriphilum YSK. With the removal of the Fe2+/Fe3+ film, the available ore surface area was relatively increased, which was beneficial to further oxidative decomposition of pyrite. Secondly, Acidiphillum spp. DX1-1 was a disadvantaged population in Consortium D, and its concentration dramatically increased in the later leaching stage. This can be explained in two ways. On one hand, the introduction of exotic bacteria promoted the metabolic activity of the community. On the other hand, due to the lack of energy required for growth at the later stage of leaching and the influence of environmental factors, each microorganism was about to enter a period of decline. Death and autolysis of the bacteria gradually increased, which provided more energy for the growth of Acidiphillum spp. DX1-1, thereby promoting its growth.
3.3 Functional gene expression analysis
The introduction of L. ferriphilum YSK during the different growth stages of indigenous leaching microbial community revealed that the best introduction period was the stable growth. Therefore, the stable growth period (Consortium D) was selected to analyze the changes of community functional gene expression levels after the introduction of indigenous community. Sixty-four functional genes involved in carbon, nitrogen, iron, and sulfur metabolism, metal resistance, extracellular polymers, and electron transport were selected for analysis at 138–158 h after the introduction of L. ferriphilum YSK (Figure 5).
The leaching microorganisms were mainly adsorbed on the ore surface during the initial stage of leaching. The number of adsorbed microorganisms reached a certain amount after which the microorganisms began to increase and gradually reached the maximum value in the solution [33]. When L. ferriphilum YSK was introduced, it was primarily adsorbed on the ore surface, then the large number of extracellular polymeric substances (EPS) would be produced. It has been reported that the production of EPS plays a very important role in bioleaching [33, 34]. Therefore, the analysis of the expression of functional genes encoding EPS was important in elucidating the adsorption behavior of microorganisms. Figure 5 shows that the genes encoding ADP heptose, phosphoheptose isomerase, and glycosyltransferase from L. ferriphilum were up-regulated from 0 to 20 h after the introduction of the bacteria compared to the control (expression of L. ferriphilum related genes before introduction). The reason may be that L. ferriphilum introduced was mainly adsorbed on the ore surface and required the production of relatively more EPS to remain viable. For genes encoding lipid A disaccharide synthase LpxB, glycosyl transferase and ADP-heptose synthase from A. ferrooxidans, the expression levels were up-regulated within 0–8 h and down-regulated within 8–20 h after introduction. This may reflect that the initial introduction of L. ferriphilum YSK promoted the adsorption or growth of A. ferrooxidans on the ore surface, while A. ferrooxidans was inhibited with the constant adsorption and growth of L. ferriphilum YSK on the ore surface.
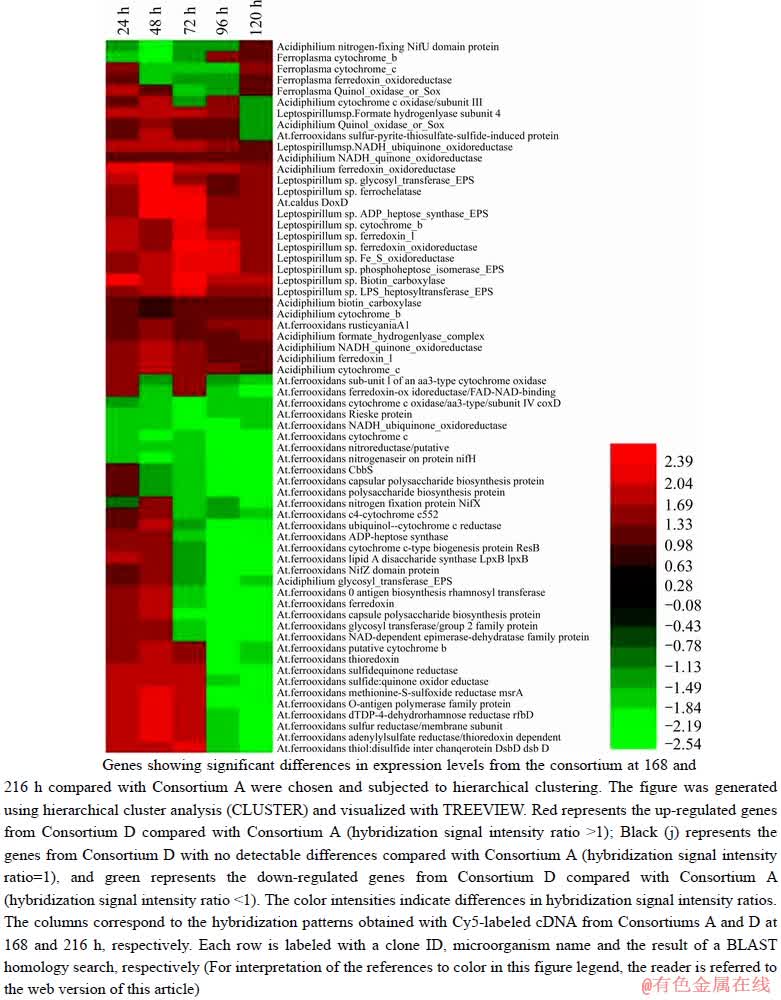
Figure 5 Hierarchical cluster analysis of select genes based on ratios obtained from hybridization signal intensity of genes from Consortium D.
L. ferriphilum YSK is an iron-oxidizing bacterium. When introduced into the indigenous community, the functional genes involved in iron metabolism promoted the use of Fe2+ to a certain extent and affected the ultimate leaching effect of the indigenous community. The expression of the functional genes encoding protoheme ferrolyase from L. ferriphilum was up-regulated within 0-20 h after introduction, perhaps due to the rapid growth after introduction L. Ferriphilum YSK. The genes encoding ferredoxin oxidoreductase from Ferroplasma sp. were up-regulated in 0-4 h, then were down-regulated in 4-16 h, and gradually up-regulated in 16-20 h. The reason for this complex change in the trend may be that the introduction of L. ferriphilum YSK promoted the oxidative decomposition of pyrite, leading to the production of more Fe2+. Therefore, Ferroplasma L1 could obtain relatively more energy for growth in the short term, which would impose a competitive disadvantage for energy with the rapid increase in L. ferriphilum YSK, leading to inhibited growth. When the competition between the two microorganisms reached a dynamic equilibrium, Ferroplasma L1 gradually adapted and the use of Fe2+ gradually increased.
For functional genes involved in sulfur metabolism, doxD from A. caldus S1 were up-regulated within 0–20 h after introduction. The reason may be that the introduction of L. ferriphilum YSK accelerated the oxidative decomposition of pyrite, generating a large amount of sulfur energy for sulfur-oxidizing bacteria, which led to the up-regulation of the expression of functional genes.
Generally speaking, the autotrophic leaching microorganisms mainly use carbon dioxide as the carbon source in the leaching system [34]. However, the content of dissolved carbon dioxide will be very low when pH is low. Therefore, the competition for carbon sources was intense among various autotrophic microorganisms in the community. Figure 5 illustrates certain differences in the expression of functional genes related to carbon metabolism from various populations. For example, the genes encoding biotin carboxylase from L. ferriphilum and the genes encoding acetyl-CoA carboxylase from Acidiphillum spp. were gradually up-regulated within 0-20 h after introduction, but expression level of CbbS from A. ferrooxidans was gradually up-regulated within 0-20 h after introduction. L. ferriphilum was favorably positioned to compete for carbon sources, and competitiveness was relatively insufficient for disadvantaged populations in the community. Heterotrophic Acidiphillum spp. was able to use organic matter as an energy source, and thus the disturbance of the indigenous community intensified after the introduction of an exotic strain and metabolites gradually increased, which may be one of the reasons for the up-regulation of gene expression level related to carbon metabolism in 0–20 h after introduction.
4 Conclusions
The introduction of L. ferriphilum YSK at different growth stages can improve the pyrite leaching of the indigenous microbial community to varying degrees. Introduction at the stable growth period was the best, and the leaching efficiency of pyrite was increased from 77.1% to 96.4%. After the introduction of L. ferriphilum YSK, the dynamic changes of each population were different in the indigenous microbial community. Growth of sulfur-oxidizing microorganisms, such as A. caldus S1 and A. thiooxidans A01, was synergistically promoted by L. ferriphilum YSK. For iron- oxidizing microorganisms and L. ferriphilum YSK, such as Ferroplasma L1 and S. thermosulfidooxidans ST, because the introduction of similar metabolic microorganisms brought about energy competition, growth was inhibited at a certain period. For heterotrophic microorganisms, such as Acidiphillum spp.DX1-1, the introduction of L. ferriphilum YSK could promote growth, mainly because of the symbiotic relationship between them. Facultative autotrophic bacteria, such as A. ferrooxidans CMS, were at disadvantaged position in different growth periods, and the introduction of L. ferriphilum YSK had little influence.
Hierarchical cluster analysis was used to study the changes in functional gene expression level at 138 h. The genes encoding ADP heptose, phosphoheptose isomerase, glycosyltransferase, biotin carboxylase and protoheme ferrolyase from L. ferriphilum, and the genes encoding acetyl-CoA carboxylase from Acidiphillum spp. and doxD from A. caldus were up-regulated in 0-20 h. The genes encoding lipid A disaccharide synthase LpxB, glycosyl transferase and ADP heptose synthase from A. ferrooxidans were up-regulated in 0-8 h and down-regulated in 8-20 h. The genes encoding ferredoxin oxidoreductase from Ferroplasma sp. were up-regulated in 0-4 h, down-regulated in 4- 16 h, and finally up-regulated in 16-20 h. CbbS from A. ferrooxidans was down-regulated at 0–20 h. This showed that after the introduction of L. ferriphilum YSK, the increase of the leaching efficiency of pyrite was related to the genes encoding enzyme involved in carbon metabolism, iron metabolism, sulfur metabolism, extracellular polymer and electron transport, which were up/down-regulation in the leaching process. These findings reveal the possibility of successful introduction of indigenous leaching bacteria, and clarify the optimal type and timing of introduction. The data will have important practical significance and potentially broad applications.
References
[1] NGUYEN V K, MU H L, PARK H J, LEE J U. Bioleaching of arsenic and heavy metals from mine tailings by pure and mixed cultures of Acidithiobacillus spp [J]. Journal of Industrial & Engineering Chemistry, 2015, 21(1): 451-458. DOI: 10.1016/j.jiec.2014.03.004.
[2] WANG Jun, LIAO Rui, LAO Lang, ZHAO Hong-bo, ZHAI Rui, QIN Wen-qing, QIU Guan-zhou. A comprehensive utilization of silver-bearing solid wastes in chalcopyrite bioleaching [J]. Hydrometallurgy, 2017, 169: 152-157. DOI: 10.1016/j.hydromet.2017.01.006.
[3] FENG Shuo-shuai, YANG Hai-lin, WANG Wu. Insights into the enhancement mechanism coupled with adapted adsorption behavior from mineralogical aspects in bioleaching of copper-bearing sulfide ore by Acidithiobacillus sp [J]. RSC Adv, 2015, 5: 98057-98066. DOI: 10. 1039/c5ra15934b.
[4] PENG Tang-jian, CHEN Lei, WANG Jing-shu, MIAO Jie, SHEN Li, YU Run-lan, GU Guo-hua, QIU Guan-zhou, ZENG Wei-min. Dissolution and passivation of chalcopyrite during bioleaching by Acidithiobacillus ferrivorans at low temperature [J]. Minerals, 2019, 9: 332. DOI: 10.3390/min9060332.
[5] LIAO Xiao-jian, SUN Shui-yu, ZHOU Si-yu, YE Mao-you, LIANG Jia-lin, HUANG Jin-jia, GUAN Zhi-jie, LI Shou- peng. A new strategy on biomining of low grade base-metal sulfide tailings [J]. Bioresource Technology, 2019, 294: 122187. DOI: 10.1016/j.biortech.2019.122187.
[6] FALCO L, POGLIANI C, CURUTCHET G, DONATI E A. A comparison of bioleaching of covellite using pure cultures of Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans or a mixed culture of Leptospirillum ferrooxidans and Acidithiobacillus thiooxidans [J]. Hydrometallurgy, 2003, 71(1): 31-36. DOI: 10.1016/S0304-386X(03)00170-1.
[7] BATTAGLIA-BRUNET F, D′HUGUES P, CABRAL T, CEZAC P, GARCIA J L, MORIN D. The mutual effect of mixed Thiobacillus and Leptospirillum populations on pyrite bioleaching [J]. Minerals Engineering, 1998, 11(2): 195-205. DOI: 10.1016/S0892-6875(97)00151-9.
[8] DOPSON M, LINDSTROEM E B. Potential role of Thiobacillus caldus in arsenopyrite bioleaching [J]. Applied & Environmental Microbiology, 1999, 65(1): 36-40. DOI: 10.1002/abio.370190413.
[9] OKIBE N, JOHNSON D B. Biooxidation of pyrite by defined mixed cultures of moderately thermophilic acidophiles in pH controlled bioreactors: Significance of microbial interactions [J]. Biotechnology and Bioengineering, 2004, 87(5): 574-583. DOI: 10.1002/bit.20138.
[10] LI Shu-zhen, ZHONG Hui, HU Yue-hua, ZHAO Jian-cun, HE Zhi-guo, GU Guo-hua. Bioleaching of a low-grade nickel-copper sulfide by mixture of four thermophiles [J]. Bioresource Technology, 2014, 153: 300-306. DOI: j.biortech.2013.12.018.
[11] JOHNSON D B. Biodiversity and ecology of acidophilic microorganisms [J]. FEMS Microbiology Ecology, 1998, 27(4): 307-317. DOI: 10.1016/s0168- 6496(98)00079-8.
[12] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review part A: Progress in bioleaching: fundamentals and mechanisms of bacterial metal sulfide oxidation [J]. Applied Microbiology and Biotechnology, 2003, 63(3): 239-248. DOI: 10.1007/s00253-003-1448-7.
[13] JOHNSON D B. Biodiversity and interactions of acidophiles: Key to understanding and optimizing microbial processing of ores and concentrates [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1367-1373. DOI: CNKI: SUN:ZYSY.0.2008-06-013.
[14] MA Li-yuan, WANG Xing-jie, TAO Jie-meng, FENG Xue, ZOU Kai, XIAO Yun-hua, LIANG Yi-li, YIN Hua-qun, LIU Xue-duan. Bioleaching of the mixed oxide-sulfide copper ore by artificial indigenous and exogenous microbial community [J]. Hydrometallurgy, 2017, 169: 41-46. DOI: 10.1016/ j.hydromet.2016.12.007.
[15] HE Zhi-guo, YIN Zhen, WANG Xin, ZHONG Hui, SUN Wei. Microbial community changes during the process of pyrite bioleaching [J]. Hydrometallurgy, 2012, 125-126(8): 81-89. DOI: 10.1016/j.hydromet.2012.05.010.
[16] WANG Jun, GAN Xiao-wen, ZHAO Hong-bo, HU Ming-hao, LI Kai-yun, QIN Wen-qin, QIU Guan-zhou. Dissolution and passivation mechanisms of chalcopyrite during bioleaching: DFT calculation, XPS and electrochemistry analysis [J]. Minerals Engineering, 2016, 98: 264-278. DOI: 10.1016/j.mineng.2016.09.008.
[17] BRYAN C G, JOULIAN C, SPOLAORE P, EI ACHBOUNI, CHALLAN-BELVAL S, MORIN D, D’HUGUES P. The efficiency of indigenous and designed consortia in bioleaching stirred tank reactors [J]. Minerals Engineering, 2011, 24(11): 1149-1156. DOI: 10.1016/ j.mineng.2011.03.014.
[18] GIAVENO A, LAVALLE L, CHIACCHIARINI P, DONATI E. Bioleaching of zinc from low-grade complex sulfide ores in an airlift by isolated Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2007, 89(1, 2): 117-126. DOI: 10.1016/ j.hydromet.2007.07.002.
[19] WALLENTINUS I, NYBERG C D. Introduced marine organisms as habitat modifiers [J]. Marine Pollution Bulletin, 2007, 55(7-9): 323-332. DOI: 10.1016/ j.marpolbul.2006.11.010.
[20] MOSEMAN S M, ZHANG Rui, QIAN Peng-yuan, LEVIN L A. Diversity and functional responses of nitrogen-fixing microbes to three wetland invasions [J]. Biological Invasions, 2009, 11(2): 225-239. DOI: 10.1007/s10530-008-9227-0.
[21] LIU Yi, YIN Hua-qun, ZENG Wei-min, LIANG Yi-li, LIU Yao, NGOM B, QIU Guan-zhou, SHEN Li, FU Xian, LIU Xue-duan. The effect of the introduction of exogenous strain Acidithiobacillus thiooxidans A01 on functional gene expression, structure and function of indigenous consortium during pyrite bioleaching [J]. Bioresource Technology, 2011, 102(17): 8092-8098. DOI: 10.1016/j.biortech.2011.06.012.
[22] SAND W, GEHRKE T. Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacial processes involving iron (III) ions and acidophilic bacteria [J]. Research in Microbiology, 2006, 157(1): 49-56. DOI: 10.1016/j.resmic.2005.07.012.
[23] YIN Hua-qun, GAO Lin-hui, QIU Guan-zhou, WANG Dian-zuo, KELLOGG L, ZHOU Ji-zhong, DAI Zhi-min, LIU Xue-duan. Development and evaluation of 50-mer oligonucleotide arrays for detecting microbial populations in acid mine drainages and bioleaching systems [J]. Journal of Microbiological Methods, 2007, 70: 165-178. DOI: 10.1016/j.mimet.2007.04.011.
[24] LI Qi-hou, TIAN Ye, FU Xian, YIN Hua-qun, ZHOU Zhi-jun, LIANG Yi-ting, QIU Guan-zhou, LIU Jie, LIU Hong-wei, LIANG Yi-li, SHEN Li, CONG Jing, LIU Xue-duan. The community dynamics of major bioleaching microorganisms during chalcopyrite leaching under the effect of organics [J]. Current Microbiology, 2011, 63(2): 164-172.
[25] LIANG Yu-ting, van NOSYRAND J D, WANG Jian, ZHANG Xu, ZHOU Ji-zhong, LI Guang-he. Microarray-based functional gene analysis of soil microbial communities during ozonation and biodegradation of crude oil [J]. Chemosphere, 2009, 75(2): 193-199. DOI: 10.1016/ j.chemosphere.2008.12.007.
[26] EISEN M B, SPELLMAN P T, BROWN P O, BOTSTEIN A D. Correction: Cluster analysis and display of genome-wide expression patterns [J]. Proceedings of the National Academy of Sciences of the United States of America, 1999, 96(19): 14863-14868. DOI: 10.2307/48859.
[27] STOTT M B, WALITING H R, FRANZMANN P D, SUTTON D. The role of iron hydroxy precipitates in the passivation of chalcopyrite during bioleaching [J]. Minerals Engineering, 2000, 13(10): 1117-1127. DOI: 10.1016/ S0892-6875(00)00095-9.
[28] RODRIGUEZ Y, BALLESTER A, BLAZQUEZ M L, GONZA L'EZ F, MUNOZ J A. New information on chalcopyrite bioleaching mechanism at low and high temperature [J]. Hydrometallurgy, 2003, 71(1): 37-46. DOI: 10.1016/S0304-386X(03) 00173-7.
[29] SMITH SARSH L, JOHNSON D B. Growth of Leptospirillum ferriphilum in sulfur medium in co-culture with Acidithiobacillus caldus [J]. Extremophiles, 2018, 22(2): 1-7. DOI: 10.1007/s00792-018-1001-3.
[30] PENG Tang-jian, ZHOU Dan, LIU Ya-nan, YU Run-lan, QIU Guan-zhou, ZENG Wei-ming. Effects of pH value on the expression of key iron/sulfur oxidation genes during bioleaching of chalcopyrite on thermophilic condition [J]. Annals of Microbiology, 2019, 69: 627-635. DOI: 10.1007/s13213-019-01453-y.
[31] FRANZMANN P D, HADDAD C M, HAWKES R B, ROBERTSON W J, PLUMB J J. Effects of temperature on the rates of iron and sulfur oxidation by selected bioleaching bacteria and archaea: Application of the Ratkowsky equation [J]. Minerals Engineering, 2005, 18(13, 14): 1304-1314. DOI: 10.1016/j. mineng. 2005. 0 4.006.
[32] ZENG Wei-min, TAN S, CHEN Miao, QIU Guan-zhou. Detection and analysis of attached microorganisms on the mineral surface during bioleaching of pure chalcopyrite with moderate thermophiles [J]. Hydrometallurgy, 2011, 106(1, 2): 46-50. DOI: 10.1016/j.hydromet.2010.11.014.
[33] ZENG Wei-min, QIU Guan-zhou, ZHU Hong-bo, PENG Juan-hua, CHEN Miao, TAN Su-ne, CHAO Wei-liang, LIU Xue-duan, ZHANG Yan-sheng. Community structure and dynamics of the free and attached microorganisms during moderately thermophilic bioleaching of chalcopyrite concentrate [J]. Bioresource Technology, 2010, 101(18): 7068-7075. DOI: 10.1016/j.biortech.2010.04.003.
[34] KINZLER K, GEHRKE T, TELEGDI J, SAND W. Bioleaching a result of interfacial processes caused by extracellular polymeric substances (EPS) [J]. Hydrometallurgy, 2003, 71(1, 2): 83-88. DOI: 10.1016/ s0304-386x(03) 00176-2.
(Edited by YANG Hua)
中文导读
黄铁矿生物浸出过程中在不同生长时期引入Leptospirillum ferriphilum YSK对土著浸矿微生物群落结构和功能的影响
摘要:在含有0.5%(W/V)黄铁矿的改良9K培养基中,添加Leptospirillum ferriophilum YSK到天然生物浸出菌群中,包括Acidithiobacillus caldus,A.thiooxans,Sulfobacillus thermosfidooxans,Acidiphilium spp.和Ferrodium thermophilum,浸出率明显提高,并用实时荧光定量PCR和功能基因芯片监测群落结构和基因表达的变化。加入L. ferriophilum YSK后群落内不同种群间的动态变化不同,促进A. caldus S1,A. thiooxans A0,Acidiphillum spp. DX1-1的生长,抑制了Ferroplasma L1和S. Thermosulfidooxidans ST的生长,但对A. ferrooxidans CMS的影响不大。功能基因芯片分析发现:来自L. ferriphilum 的编码ADP heptose, phosphoheptose isomerase, glycosyltransferase, biotin carboxylase和protoheme ferrolyase的基因,以及来自Acidiphillum spp编码的 acetyl-CoA carboxylase 和来自A. caldus 的doxD基因在引种后0~20 h内表达水平上调;来自A. ferrooxidans编码的lipid A disaccharide synthase LpxB, glycosyl transferase和 ADP heptose synthase的基因在引种后0~8 h表达水平上调,在8~20 h下调;来自Ferroplasma sp.编码的ferredoxin oxidoreductase的基因在引种后0~4 h表达水平上调,而在4~16 h表达水平下调,在16~20 h表达上调;来自A. ferrooxidans 的CbbS 基因在引种0~20 h表达水平下调。
关键词:Leptospirillum ferriphilum YSK;土著群落;功能基因芯片(FGAs);黄铁矿生物浸出
Foundation item: Projects(51604308, 41771300, 41301274) supported by the National Natural Science Foundation of China; Project(2017QNCXTD_GTD) supported by the Youth Innovation Team Project of Institute of Subtropical Agriculture, Chinese Academy of Sciences; Project(2017YFD0202000) supported by the National Key Research and Development Program of China; Project(2020GDASYL-20200402001) supported by the special Project of Science and Technology Development, China
Received date: 2019-06-28; Accepted date: 2020-04-07
Corresponding author: LIU Yi, PhD, Associate Researcher; Tel: +86-731-84619716; E-mail: liuyi@isa.ac.cn; ORCID: 0000-0002-5688- 1324

