
Modified cellular automaton model for modeling of microstructure and microsegregation in solidification of ternary alloys
ZHU Ming-fang(������)1, CAO Wei-sheng2, CHEN Shuang-lin3, XIE Fan-you2,
HONG Chun-pyo4, CHANG Y. Austin2
1. School of Materials Science and Engineering, Southeast University, Nanjing 210096, China;
2. Department of Materials Science and Engineering, University of Wisconsin-Madison,
1509 University Avenue, Madison, WI 53706, USA;
3. CompuTherm LLC, 437 S. Yellowstone Dr., Suite 217, Madison, WI 53719, USA;
4. Center for Computer-Aided Materials Processing (CAMP), Department of Metallurgical Engineering,
Yonsei University, Shinchon-dong 134, Seodaemun-ku, Seoul, 120-749 Korea
Received 20 April 2006; accepted 30 June 2006
Abstract: A modified cellular automaton (MCA) model has been extended to the ternary alloy system by coupling thermodynamic and phase equilibrium calculation engine PanEngine. In the present model the dendrite growth is driven by the difference between the local equilibrium liquidus temperature and local actual temperature, incorporating the effect of curvature. The local equilibrium liquidus temperature is calculated with PanEngine according to the local liquid concentrations of two solutes, which are determined by numerically solving the species transport equation in the domain. Model validation was carried out through the comparison of the simulated values to the prediction of the Scheil model for solute profiles in the primary dendrites. The simulated data with zero solid diffusivity and limited liquid diffusivity were increasingly close to the Scheil profiles as the solidification rate decreased. The simulated microstructure and microsegregation in an Al-Cu-Mg ternary alloy were compared with those obtained experimentally.
Key words: solidification; modeling; microstructure; microsegregation; Al-Cu-Mg ternary alloy
1 Introduction
Solidification microstructure and microsegregation of castings are very important since they are closely associated with the properties of final products. It is thus crucial to understand the mechanisms of microstructure formation and solute redistribution during alloy solidification. In recent years, with the development of powerful computers and advanced numerical techniques, numerical modeling has made significant progress and it is playing an increasing important role in studies on the microstructural evolution and microsegregation during solidification. The models based on the cellular automaton (CA) technique can reproduce most of the microstructure features observed experimentally with an acceptable computational efficiency[1-7].
Two of the key parameters needed in the microstructural modeling of alloys are the solute partition coefficient and the liquidus slope. For the simple binary model alloy systems, they are usually considered to be suitably taken as a constant. However, for multicomponent systems, which are the cases of most commercial alloys, the common assumption of the constant partition coefficient and liquidus slope is unlikely to be sufficient since the actual values may vary significantly with temperature and compositions.
In this paper, a two-dimensional (2-D) modified cellular automaton (MCA) model was coupled with a thermodynamic and phase equilibrium calculation engine PanEngine for simulating the primary dendrite formation and microsegregation during solidification of ternary alloy system. The model validation was performed by comparing the prediction of the Scheil model for solute concentration profile in the primary dendrites as a function of solid fraction with simulated values for an Al-3.9%Cu-0.9%Mg (mass fraction) alloy. The simulation results compared with those obtained xperimentally were presented.
2 Model description and numerical algorithm
The present work was performed based on a modified cellular automaton (MCA) approach for the simulations of dendrite growth, which was coupled with PanEngine to obtain phase equilibrium information of the ternary alloy systems. The 2-D calculation domain is divided into uniform square arrangement of cells. Each cell is characterized by several variables, such as temperature, concentrations of two solutes, crystallographic orientation and solid fraction, and marked as the state of liquid (fs=0), solid (fs=1) or interface (0��fs��1). For the sake of simplicity, the thermal field is assumed as uniform in a microscopic scale and the dendrite growth is governed by the diffusion of two solutes and interface curvature. The governing equation for solute diffusion within the entire domain is given by
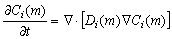
(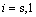 and
and 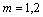 ) (1)
) (1)
where Ci(m) is the concentration and Ci(m) is the diffusion coefficient of solute element (m) in phase (i). Two solutes are considered to diffuse independently each other and cross diffusion is neglected. Eqn.(1) is solved using an explicit finite difference scheme. The zero-flux boundary condition is applied for the cells located at the four walls of the calculation domain.
The growth of the dendrite is driven by the local undercooling. The local undercooling at time tn, ��T(tn), is considered to be the difference between the local equilibrium liquidus temperature and local actual temperature, incorporating the effect of curvature and it is given by
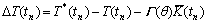 (2)
(2)
where T*(tn) is the local equilibrium liquidus temperature which is calculated using PanEngine according to the local interface liquid compositions of two solutes 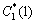 and
and 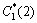 . T(tn) is the local actual temperature. ��(��) is the Gibbs-Thomson coefficient. The calculation of interface curvature
. T(tn) is the local actual temperature. ��(��) is the Gibbs-Thomson coefficient. The calculation of interface curvature 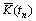 can be found elsewhere[2].
can be found elsewhere[2].
The growth velocity Vg and local undercooling is related by the classical sharp interface model[8]
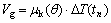 (3)
(3)
where ��k(��) is the interface kinetic coefficient.
It is well known that dendrites always grow in specific crystallographic orientations. Therefore, it is necessary to consider anisotropy in either the surface energy or interfacial attachment kinetics (or both) in the models of dendritic growth[9]. The present model accounts for the anisotropy in both surface energy and interfacial kinetics. For a fcc-lattice crystal of Al-rich Al-Cu-Mg alloys used in the present simulations, it exhibits a fourfold anisotropies of the surface energy and kinetics at the solid/liquid (SL) interface. The Gibbs-Thomson coefficient G(��) and the interface kinetics coefficient ��k(��) are thus given by
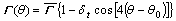 (4)
(4)
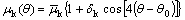 (5)
(5)
where  ,
,  ,
, 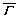 , and dt are the average interface kinetic coefficient, the degree of the kinetic anisotropy, the average Gibbs-Thomson coefficient, and the degree of the surface energy anisotropy, respectively. �� is the angle between the normal of the SL interface and the horizontal direction, and ��0 is the preferred growth orientation of the crystal. The angle �� can be calculated according to the gradient of solid fraction at the SL interface using the following equation.
, and dt are the average interface kinetic coefficient, the degree of the kinetic anisotropy, the average Gibbs-Thomson coefficient, and the degree of the surface energy anisotropy, respectively. �� is the angle between the normal of the SL interface and the horizontal direction, and ��0 is the preferred growth orientation of the crystal. The angle �� can be calculated according to the gradient of solid fraction at the SL interface using the following equation.
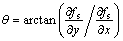 (6)
(6)
where 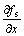 and
and 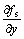 indicate the gradient of solid
indicate the gradient of solid
fraction at the SL interface along x axis (horizontal direction) and y axis (vertical direction) respectively. The growth velocities of interface cells were calculated with Eqns.(2)-(6). The change in the rate of the solid fraction in an interface cell can thus be evaluated from the crystal growth velocity Vg as follows:
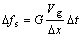 (7)
(7)
where ��x is the cell spacing. ��t is the time step. G is a geometrical factor related to the state of neighbor cells. The details of the growth algorithm can be found elsewhere[7].
As dendrite grows, the growing cells reject solutes at the SL interface. The rejected amount of solute element (m) is evaluated by
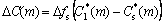 (8)
(8)
where 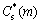 is the interface solid composition of solute element (m) which is calculated from PanEngine according to the interface liquid composition
is the interface solid composition of solute element (m) which is calculated from PanEngine according to the interface liquid composition  of solute (m). The rejected
of solute (m). The rejected  is added to the remaining liquid in the same cell and its surrounding neighbor cells. Thus, the overall solutes in the domain can be kept constant. The physical parameters used in the present work are obtained from Refs.[7,10]. The average interface kinetic coefficient
is added to the remaining liquid in the same cell and its surrounding neighbor cells. Thus, the overall solutes in the domain can be kept constant. The physical parameters used in the present work are obtained from Refs.[7,10]. The average interface kinetic coefficient  is taken as 3.0��10-4 m/(s��K).
is taken as 3.0��10-4 m/(s��K).
3 Results and discussion
To validate the present model, the simulated solute profiles of an Al-3.9%Cu-0.9%Mg alloy were compared with the predictions of the Scheil model. It is known that for Al-rich ternary alloys, the solidification sequence is primary a phase?binary eutectic?ternary eutectic. However, in the present work, we only consider primary a dendrites solidified from liquid. According to the Scheil calculation, the Al-3.9%Cu-0.9%Mg alloy solidifies from 919 K and �� phase comes out at 792 K. Thus, the simulations started from the liquidus temperature of 919 K and stopped at 792 K. The calculation domain consists of 200��200 meshes with mesh size ��x=2 ��m. Four solid seeds with different preferred growth orientations were randomly assigned in the domain. The temperature in the entire domain was assumed to be homogeneous and cooled down with a constant cooling rate. Fig.1 shows the simulated dendrite morphology and composition maps of an Al-3.9%Cu-0.9%Mg alloy with a cooling rate of 10 K/s for two solutes. It can be seen that the center region of the dendrites exhibits lower composition than the outside shell for both Cu and Mg solutes. The variation of solid compositions with the increase of solid fraction was then recorded for the comparison with the predictions of the Scheil model. It is known that the Scheil model is derived based on the assumptions of no diffusion in the solid and complete mixing in the liquid. To analyze the effect of diffusion in liquid on the microsegregation, zero solid diffusivity (Ds(m)=0) and finite liquid diffusivity (Dl(m)��0) were adopted for the simulations. The simulated solid composition profiles of two solutes with the cooling rates of 0.23, 10 and 100 K/s are compared with the Scheil predictions in Fig.2. Note that the solid compositions of both solutes Cu and Mg predicted by the Scheil model are relative lower at the early solidification stage but higher at the late stage. This is due to the fact that the Scheil prediction was obtained with the complete mixing liquid, while the numerical simulations involved the limited liquid diffusivity using the realistic liquid diffusion coefficients. With the increase of solidification rate, the extent of the inhomogeneous solute distribution in liquid will increase. Consequently, it is understandable that the difference between the simulated data and the Scheil profile will increase with solidification rate. In case of very low cooling rate of 0.23 K/s, the MCA calculated data are nearly superposed on the Scheil profiles.
In order to investigate the effect of solid diffusion
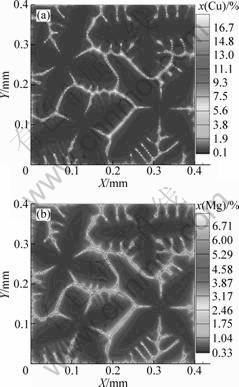
Fig.1 Simulated equiaxed dendrite morphology and solute fields of Cu (a) and Mg (b) for Al-3.9%Cu-0.9%Mg alloy solidified with 10 K/s
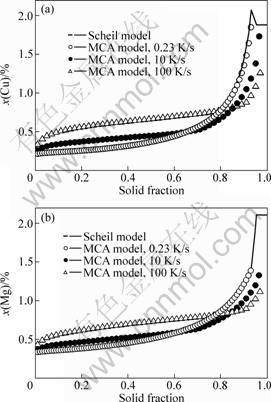
Fig.2 Comparison between Scheil model and present model for predicting solute concentration profiles of Cu (a) and Mg (b) as function of solid fraction for an Al-3.9%Cu-0.9%Mg alloy
on the solute microsegregation, the simulations were performed with zero and non-zero solid diffusivity for the cooling rate of 0.23 K/s. A finite liquid diffusivity (Dl(m)��0) was adopted for all simulations. The results are presented in Fig.3. The experimental data measured from a directionally solidified specimen[10] are also included in the figures for comparison. It can be seen that for the case of Cu composition, the effect of back diffusion is not so obvious. On the other hand, for the solute Mg, the composition profile with non-zero solid diffusivity is higher at the early stage but lower later than that obtained with zero solid diffusivity, indicating the evident effect of back diffusion. Regarding the comparison with the experiment, the calculated data are mostly found below the measured data. Nevertheless, they exhibit a same tendency of the solute compositions increasing with solid fraction. Particularly, the calculated Mg solute profile with non-zero solid diffusivity is nearly in parallel with the measured profile. The discrepancies between the calculated and measured data might be caused by some reasons. For example, the inaccuracy of the diffusion coefficients used in the calculations will directly influence the simulated solute profiles. The present model assumes the local equilibrium and no diffusion across the SL interface. However, non-equilibrium solute redistributions are likely to occur in the practical situations, leading to the
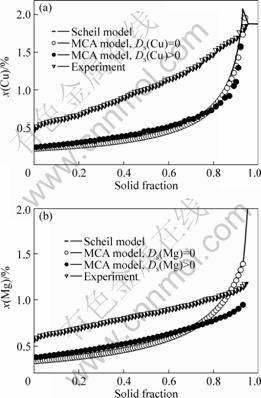
Fig.3 Predicted and measured concentration profiles of Cu (a) and Mg (b) for Al-3.9%Cu-0.9%Mg alloy solidified with 0.23 K/s as a function of the solid fraction
effect of more back diffusion. Moreover, in the present model the solute fields are considered to be controlled by diffusion in both liquid and solid phases, and convective mass transport is not taken into account. It is known that in the real cases interdendrite flow of liquid caused by natural buoyancy and solidification shrinkage is an unavoidable phenomenon, which may alter the local solutal transfer. The discrepancies might also be attributed to experimental measurement difficulties and uncertainties. Finally, the experiment data were measured from a three-dimensional specimen, whereas the simulations were carried out in a two-dimensional domain.
The model was applied to simulate the formation of multi equiaxed dendrites in an Al-3.9%Cu-0.9%Mg alloy. The simulation was carried out in a calculation domain divided into 300��300 cells with a cell size of 3 ��m. Nineteen nuclei having the randomly assigned preferred growth orientations ��0 ranging from 0? to 90? with respect to the horizontal direction were randomly distributed on the domain. The temperature of the domain was assumed to be uniform and cooled down from the liquidus temperature of 919 K to the binary eutectic temperature of 792 K with a constant cooling rate of 0.78 K/s. Fig.4 presents the simulated evolution of multi-equiaxed dendritic growth. The different colors represent grains with different crystallographic orientations. It can be seen that at the beginning of solidification process, most dendrites develop more or less homogeneously aligning their crystallographic orientations, while the growth of a few main arms is suppressed by the nearby dendrite. As the solidification process proceeds, the interaction among the dendrites becomes more evident, which modifies the morphology and growth rate of individual dendrites. Finally, the dendrites impinge on each other to form grain boundaries. The Cu solute map of the last picture in Fig.4 is compared with the experimental result in Fig.5. The experimental one shows the morphology in a transverse section of a directionally solidified Al-3.9%Cu-0.9%Mg alloy with 0.78 K/s[10]. Notice that the outside shell of dendrites exhibits the higher composition. The white color along the grain boundaries indicates the remaining liquid at the end of primary a dendrite formation, which will transform to binary and ternary eutectics at the end of solidification.
4��Conclusions
A MCA model has been coupled with the thermodynamic and phase equilibrium calculation engine PanEngine for simulating the microstructures and microsegregation in ternary alloy system. The model calculates the evolution of the SL interface directly from
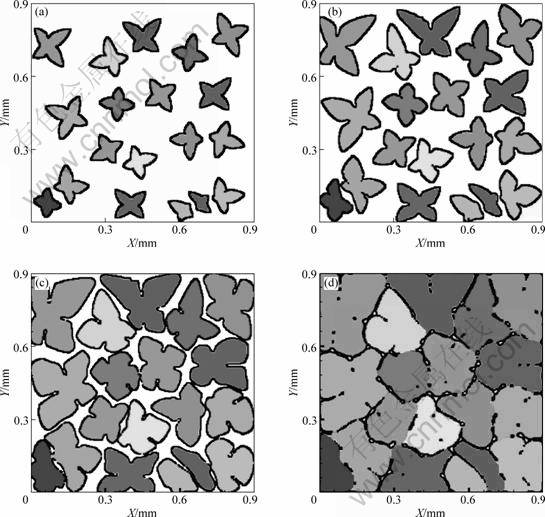
Fig.4 Simulated equiaxed dendrite evolution of Al-3.9%Cu-0.9%Mg alloy solidified with 0.78 ��/s after 4.33 s (a); 9.38 s (b); 29 s (c) and 164 s (d)(Calculation domain 300��300 with mesh size of 3 ��m)

Fig.5 Dendrite morphologies of Al-3.9%Cu-0.9%Mg alloy solidified with 0.78 ��/s: (a) Experiment[10]; (b) Simulation
the difference between the local equilibrium liquidus temperature and local actual temperature. Based on the interface liquid compositions of two solutes, which are determined by numerically solving the mass transport equation in the whole domain, the local equilibrium liquidus temperature and interface equilibrium solid compositions of two solutes are calculated with the aid of PanEngine. The model simulated composition profiles of two solutes as a function of solid fraction for an Al-3.9%Cu-0.9%Mg alloy were compared with the predictions of the Scheil model. Upon decreasing cooling rates, the simulated results with the zero solid diffusivity and the finite liquid diffusivity were increasingly close to the Scheil profiles, illustrating that the model can predict reasonably the microsegregation patterns depending on the involved solidification conditions. At the cooling rate of 0.23 K/s, the calculated solid composition profile of Mg solute with non-zero solid diffusivity was initially higher and later lower than that obtained with zero solid diffusivity, indicating the effect of back diffusion on the microsegregation. However, the simulated back diffusion of Cu solute is not evident in this alloy. The model is also able to reproduce very realistic dendritic morphology which is compared well with the experimental observation.
References
[1] Nastac L. Numerical modeling of solidification morphologies and segregation patterns in cast dendritic alloys[J]. Acta Mater, 1999, 47(17): 4253-4262.
[2] Shin Y H, Hong C P. Modeling of dendritic growth with convection using a modified cellular automaton model with a diffuse interface[J]. ISIJ Int, 2002, 42(4): 359-367.
[3] Zhu M F, Hong C P. A modified cellular automaton model for the simulation of dendritic growth in solidification of alloys[J]. ISIJ Int, 2001, 41(5): 436-445.
[4] Xu Q Y, Liu B C. Modeling of as-cast microstructure of Al alloy with a modified cellular automaton method[J]. Mater Trans, 2001; 42(11): 2316-2321.
[5] Li Q, Guo Q Y, Li D Z, Qian B N, Li D M, Li R, Zhang P W. Continuous method for describing dendrite evolution during solidification[J]. Chin Phys Lett, 2004, 21(1): 143-145.
[6] Belteran-Sanchez L, Stefanescu D M. A quantitative dendrite growth model and analysis of stability concepts[J]. Metall Mater Trans A, 2004, 35A(8): 2471-2485.
[7] Zhu M F, Lee S Y, Hong C P. Modified cellular automaton model for the prediction of dendritic growth with melt convection[J]. Phys Rev E, 2004, 69(61): 061610-1-061610-12.
[8] Wheeler A A, Boettinger W J, McFadden G B. Phase-field model for isothermal phase transitions in binary alloys[J]. Phys Rev A, 1992, 45(10): 7424-7439.
[9] Granasy L, Pusztai T, Warren J A, Douglas J F, Borzsonyi T, Ferreiro V. Growth of ��dizzy dendrites�� in a random field of foreign particles[J]. Nature Materials, 2003, 2(2): 92-96.
[10] Xie F Y, Kraft T, Zuo Y, Moon C H, Chang Y A. Microstructure and microsegregation in Al-rich Al-Cu-Mg alloys[J]. Acta Mater, 1999, 47(2): 489-500.
(Edited by ZHAO Jun)
Foundation item: Projects (50371015 and 50471030) supported by the National Natural Science Foundation of China; Project (DMR-0309468) supported by NSF through the FRG
Corresponding author: ZHU Ming-fang; Tel: +86-25-83793355; E-mail: zhumf@seu.edu.cn