J. Cent. South Univ. Technol. (2011) 18: 337-342
DOI: 10.1007/s11771-011-0701-y
Synthesis and characterization of polyfunctional aziridine/polyester microcapsules by multiple emulsion-solvent evaporation method
HU Jian-qing(胡剑青), ZHU Hai-jun(朱海军), TU Wei-ping(涂伟萍), WANG Feng(王锋)
School of Chemistry and Chemical Engineering, South China University of Technology,
Guangzhou 510640, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: Polyfunctional aziridine/polyester microcapsules as control-release waterborne cross-linker were synthesized by multiple emulsion-solvent evaporation method. The results show that, a lower surface free energy with shell polyester is more favourable for the formation of microcapsules. Full encapsulating microcapsules are synthesized with the polyester with a surface free energy of 34.5 mJ/m2. Shell-to-core feeding mass ratio has a significant influence on the morphology and core content of the resulting microcapsules. Well defined spherical microcapsules with uniform shell thickness and core content at around 22% are produced at a shell-to-core mass ratio of 1:1. When 2.5% of colloid stabilizer is used, hollow spherical microcapsules are obtained. A high solvent evaporation rate results in wrinkling and porosity of the microcapsules, and an evaporation rate equivalent to solvent elimination in about 2 h provides a uniform rate of surface hardening. The characterization of the microcapsules by SEM and FTIR demonstrates that polyfunctional aziridine is encapsulated at the centre of the microcapsule. The microcapsules synthesized can be broken at a high shear rate.
Key words: microcapsule; aziridine; polyester; multiple emulsion-solvent evaporation
1 Introduction
Encapsulation technology was originated from phenomena in nature, where many capsules with various dimensions exist. A cell that envelops its active ingredients with a cell wall can be a nano-scale capsule and bird’s egg and seed are examples of macroscopic capsules [1-2]. Microencapsulation is a micro-packaging technology which encloses micro-sized materials of solids, liquids or gasses in a shell of natural or synthetic polymer matrix, for the purpose of protecting unstable or susceptible functional materials [3-6]. Micro- encapsulation technology first caught researchers’ attention through the patent application of microcapsule for carbonless copying paper by NCR Company of America in 1953, and then was further explored through encapsulation of active ingredients
Raw materials for shell polyester of dimethyl phthalate, glycol, 1,3-propanediol and zinc acetate were all in chemical grade provided by Sinopharm Chemical Reagent Group. Emulsifiers of SPAN 80 was supplied by Aldrich-Sigma. Colloidal stabilizers of gelatin, polyvinyl pyrrolidone (K30) and Tween 80 were supplied by Aldrich-Sigma. Solvent of dichloromethane, in analytical reagent grade, was obtained from Sinopharm Chemical Reagent Group. Deionized water was self-produced. All materials were used as received.
2.2 Procedure of synthesis of microcapsules
Synthesis of microcapsules was preformed according to a process of multiple emulsion-solvent evaporation method in Fig.1, which included five steps described as follows.
1) Preparation of shell polyester by trans- esterification process
Dimethyl phthalate, glycol, 1,3-propanediol and zinc acetate were charged into a four-necked flask equipped with mechanical stirrer, thermometer, condenser and nitrogen inlet. The sample was maintained at 150 °C for 1 h, then it was heated to 220 °C at a heating rate of 20 °C/h. It was maintained at 220 °C for an additional 2-4 h. Finally, it was cooled to room temperature and the shell polyester was obtained.
2) Preparation of water in oil (W/O) emulsion
Shell polyester was dissolved into dichloromethane to produce a shell solution. XAMA-7, deionized water and SPAN 80 were added into the shell solution, then, a W/O emulsion was formed by stirring.
3) Preparation of W/O/W multiple emulsion
The colloidal stabilizers containing gelatin, polyvinyl pyrrolidone and Tween 80 were mixed into deionized water, then the colloidal stabilizers in water was charged into W/O emulsion. The mixture was dispersed by ultrasonication with a JY92-11 ultrasonic generator at a power setting of 200 W for 1 min and water in oil in water (W/O/W) multiple emulsion was obtained.
4) Solvent evaporation
The W/O/W multiple emulsion was then transferred into an air-tight reactor equipped with nitrogen protection, reflux condensation and agitation, and the temperature was raised to 40 °C to evaporate the solvent. After 2-3 h of reaction, a microcapsule slurry was obtained.
5) Post-treatment of microcapsule slurry
The microcapsule slurry obtained was centrifuged at 5 000 r/min for 10 min in order to remove other residual components and foams. The sedimentation layer was collected and washed three times with deionised water and separated by suction filtration. The solid mass obtained was then dried for 24 h at 40 °C under vacuum to obtain microcapsules.
2.3 Characterization of samples
2.3.1 Analysis of surface free energy
The static contact angles of deionized water on shell polyester film were measured with a PGX dynamic/static measuring machine. The free surface energy of polyester films was calculated with the static contact angles of water on relevant polyesters according to ASTM D5946 method.
2.3.2 SEM analysis
The sample obtained was pre-coated with a 20 nm- thick layer of gold, using an ion coater, on the surface as a reflecting layer. Then, a JEOL JSM-820 scanning electronic microscope was used to observe the surface morphology.
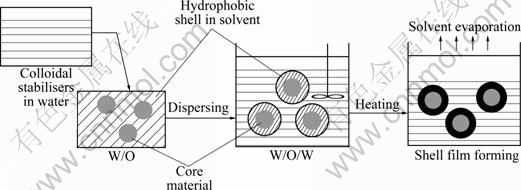
Fig.1 Schematic diagram of synthesis of microcapsules by multiple emulsion-solvent evaporation
2.3.3 Thermo gravimetric analysis (TGA)
A TA instrument TGA 2050 thermo gravimetric analyzer was used to characterize the thermal stability of the sample obtained under nitrogen atmosphere. Analyzes were conducted at a heating rate of 10 °C/min in the temperature range of 0-500 °C.
2.3.4 Fourier transform infrared analysis (FTIR)
A Perkin-Elmer Spectrum-2000 Fourier transform infrared spectrometer was adopted to characterize the chemical structure of microcapsules obtained. The range of wavenumber of the FTIR spectrum was set at 500- 4 000 cm-1.
3 Results and discussion
3.1 Effects of surface free energy of polyester
Polyesters, PE-1 and PE-2, are used as shell materials for the preparation of microcapsules to study the effect of surface free energy, respectively. The static contact angles of water on polyesters PE-1 and PE-2 films and the surface free energy of relevant polyesters calculated according to ASTM D5946 are shown in Fig.2. The morphologies of microcapsules using PE-1 and PE-2 as shell materials are presented in Fig.3.
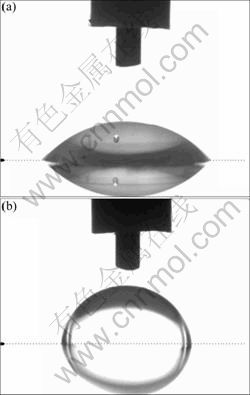
Fig.2 Pictures showing static contact angles of deionized water on PE-1 (a) and PE-2 (b)
It can be seen from Fig.2 that the static contact angles of water on polyesters PE-1 and PE-2 are 49.2° and 83.4°, respectively. According to ASTM D5946 method, the surface free energies of PE-1 and PE-2 calculated from the value of static contact angles are 47.6 and 34.5 mJ/m2, respectively. From Fig.3, it is found that the microcapsules using PE-1 as the shell show irregular morphologies and most of the microcapsules are seriously ruptured, while those using PE-2 as the shell are well-defined spherical. A lower surface free energy of shell polyester is more favourable for the formation of microcapsules [20]. Full encapsulating microcapsules can be synthesized by the polyester with a surface free energy of 34.5 mJ/m2.

Fig.3 SEM micrographs of microcapsules using PE-1 (a) and PE-2 (b) as shell materials
3.2 Effects of shell-to-core feeding mass ratio
To investigate the effect of shell-to-core feeding mass ratio on the morphology and encapsulation efficiency of microcapsule obtained, the shell-to-core mass ratio of 3:1, 1:1 and 1:3 are controlled to prepare microcapsules. The relevant microcapsules are characterized by SEM and TGA and the analysis results are presented in Fig.4 and Fig.5, respectively.
It can be seen from Fig.4 that the ruptured microcapsules prepared at a shell-to-core ratio of 3:1 appear as hollow structure with large shell thickness and a shell-to-core ratio of 1:1 can produce very well-defined spherical microcapsules with more uniform shell thickness. While at the shell-to-core ratio of 1:3, it is found that no stable hollow spherical microcapsules but some hemispherical and irregular colloids are obtained. From Fig.5, it can be seen that the microcapsules prepared at the shell-to-core mass ratios of 3:1 and 1:1 both start to decompose at around 160 °C and reach a plateau of decomposition between 200 °C and 220 °C, respectively. Hereafter, both microcapsules show rapid mass loss with the increase of temperature. The samples prepared at the shell-to-core mass ratio of 1:3 start the decomposition from about 185 °C and then loss mass rapidly without platform because they are composed of polyester and fail to form fully encapsulated microcapsules. It is considered that the mass loss from starting decomposition temperature to the temperature of the plateau is attributed to the encapsulated core material, so the mass loss might correspond to the core content, which is around 12.5% for microcapsules prepared at a shell-to-core ratio of 3:1 and around 22% for microcapsules prepared at a shell-to-core ratio of 1:1.

Fig.4 SEM micrographs of microcapsules prepared at shell-to- core mass ratios of 3:1 (a), 1:1 (b) and 1:3 (c)
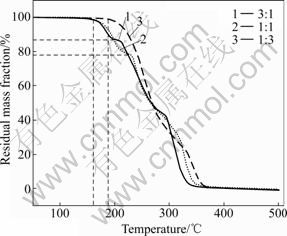
Fig.5 TGA curves of microcapsules prepared at different shell-to-core feeding mass ratios
3.3 Effects of colloidal stabilizer amount
In this work, the colloidal stabilizer consists of gelatin, polyvinyl pyrrolidone K30 and Tween 80 with the mass fractions of 70%, 20% and 10%, respectively. To study the effect of colloidal stabilizer amount, samples are prepared with the colloidal stabilizer of about 0.5%, 2.5% and 5.0% in the total colloid protective solution, respectively. The SEM morphologies of samples obtained are shown in Fig.6.

Fig.6 SEM micrographs of samples prepared with colloidal stabilizer of 0.5% (a), 2.5% (b) and 5.0% (c)
As shown in Fig.6, when 0.5% of colloid stabilizer is used, no scattered particles but agglomerated colloids are produced. When 2.5% of colloid stabilizer is used, hollow spherical microcapsules can be obtained. Moreover, when the amount of colloidal stabilizer is raised to 5.0%, separated particles can be manufactured, but the surface of samples become wrinkle and the hollow structure of samples is collapsed. It can be concluded that colloidal stabilizer will improve the dispersion of W/O emulsion in water phase. When the colloidal stabilizer amount is too low, W/O/W multiple emulsion can hardly be stably formed, which results in the agglomerated colloid. But if too much colloidal stabilizer is used, spare colloidal stabilizer will be located onto the interface between oil and water phase, which improves the stability of W/O/W multiple emulsion but at the same time softens the polyester shell and results in the collapse of hollow structure and the wrinkling of shell polyester.
3.4 Effects of solvent evaporation rate on surface morphology
The rate of solvent evaporation will affect the surface morphology of microcapsules. In this work, the microcapsules are prepared at a fast and slow evaporation rate which is equivalent to solvent elimination in 0.5 h and 2 h, respectively. The surface morphologies of both microcapsules are analyzed by SEM and the results are presented in Fig.7.
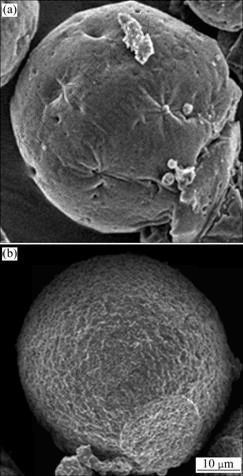
Fig.7 SEM micrographs of microcapsules prepared at solvent evaporation rate equivalent to solvent elimination in 0.5 h (a) and 2 h (b)
The microcapsules formed at a high solvent evaporation rate equivalent to solvent elimination in 0.5 h as seen from Fig.7(a) are smooth but with wrinkling and porosity in surface texture. The microcapsules formed by a slower solvent evaporation rate in Fig.7(b) are well defined spherical microcapsule with the surface of uniform textures of gradual solidification. A slower solvent evaporation rate equivalent to solvent elimination in 2 h leads to a uniform rate of surface hardening, which makes the surface of microcapsules formed less smooth and slightly bumpy.
3.5 Characterization of microcapsules
In order to characterize the chemical structure, microcapsules having a polyester with the surface free energy of 34.5 mJ/m2 as shell and XAMA-7 as core, are synthesized according to the process conditions of shell-to-core mass ratio of 1:1, 2.5% of colloid stabilizer and solvent evaporation rate equivalent to about 2 h. The morphology and composition of microcapsules are analyzed by SEM and FTIR.
Microcapsules can be broken at a high shear rate. The microcapsules prepared and one of the broken microcapsules analyzed by SEM are shown in Fig.8.

Fig.8 SEM micrographs of microcapsules (a) and one of broken microcapsules (b)
From Fig.8(a), it can be seen that microcapsules synthesized have a very well defined spherical structure and it can also be found from Fig.8(b) that the microcapsule is of uniform shell thickness and XAMA-7 is located at the centre of the microcapsule.
FTIR analysis of shell polyester, core XAMA-7 and microcapsules is carried out and relevant spectra are shown in Fig.9.
It can be seen from Fig.9 that the spectrum of microcapsules contains characteristic absorption peaks of shell polyester and core XAMA-7, which includes the characteristic peak of benzene ring in shell polyester at around 1 600 cm-1 and the ―C―N peak of aziridine in core XAMA-7 at around 1 658 cm-1. The FTIR spectra demonstrate that the shell polyester and the core XAMA-7 exist stably in the microcapsules.
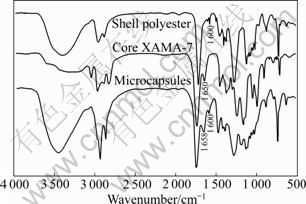
Fig.9 FTIR spectra of shell polyester, core XAMA-7 and microcapsules
4 Conclusions
1) A lower surface free energy of shell polyester is more favourable for the formation of microcapsules. Full encapsulating microcapsules can be synthesized by the polyester with a surface free energy of 34.5 mJ/m2.
2) Shell-to-core feeding mass ratio has a significant influence on the morphology and core content of the resulting microcapsules. It is found that at a shell-to-core mass ratio of 1:1, well defined spherical microcapsules with uniform shell thickness and core content at around 22% can be produced.
3) When 2.5% of colloid stabilizer is used, hollow spherical microcapsules can be obtained. It is also found that a high solvent evaporation rate results in wrinkling and porosity of the microcapsules.
4) An evaporation rate equivalent to solvent elimination in about 2 h provides a uniform rate of surface hardening.
5) The characterization of the physical and chemical structure of the microcapsules by SEM and FTIR demonstrates that the microcapsules synthesized has very well defined hollow structure and uniform shell thickness, and that polyfunctional aziridine is encapsulated at the centre of the microcapsule.
6) The polyfunctional aziridine/polyester microcapsules synthesized can be broken at a high shear rate, releasing the core materials of polyfunctional aziridine to provide self-crosslinking function.
References
[1] KONDO A. Microcapsule processing and technology [M]. New York: Marcel Dekker, 1979: 165.
[2] SPARKS R E. Microencapsulation [M]. New York: Marcel Dekker, 1989: 255.
[3] BENITA S. Microencapsulation: Methods and Industrial applications [M]. New York: Marcel Dekker, 1996: 12.
[4] ARSHADY R. Microspheres, microcapsules and liposomes [M]. London: Citrus Books, 1999: 25.
[5] AO Z, YANG Z, WANG J F, ZHANG G Z, NGAI T. Emulsion- templated liquid core-polymer shell microcapsule formation [J]. Langmuir, 2009, 25: 2572-2574.
[6] BLAISZIK B J, CARUSO M M, McILROY D, MOORE J, WHITE S R, SOTTOS N R. Microcapsules filled with reactive solutions for self-healing materials [J]. Polymer, 2009, 50: 990-997.
[7] GHOSH S K. Functional coatings by polymer microencapsulation [M]. Wiley-VCH, 2006: 282.
[8] HOGAN J E, AULTON M. Pharmaceutical coating technology [M]. London: Taylor & Francis, 1995: 215.
[9] OLIVIER P H, NICOLAS J, LAHOUSSINE O. Real-time monitoring of fragrance release from cotton towels by low thermal mass gas chromatography using a longitudinally modulating cryogenic system for headspace sampling and injection [J]. Anal Chem, 2010, 82(2): 729-737.
[10] HU S H, TSAI C H, LIAO C F, LIU D M, CHEN S Y. Controlled rupture of magnetic polyelectrolyte microcapsules for drug delivery [J]. Langmuir, 2008, 24(20): 11811-11818.
[11] UTTAM M, SATISH P. Dual drug delivery microcapsules via layer-by-layer self-assembly [J]. Langmuir, 2009, 25(18): 10515-10522.
[12] STEVEN A C, STEPHEN L W. Modeling approach to assess clustering impact on release rates of pesticides from microencapsulated products [J]. J Agric Food Chem, 2009, 57(12): 5443-5451.
[13] BROWN E N, KESSLERS M R, SOTTOST N T. In situ poly(urea-formaldehyde) microencapsulation of dicyclopentadiene [J]. J Microencapsulation, 2003, 20: 719-730.
[14] XU Xiang, DONG Jie, LI Jie. Analysis of volatile components in propolis by solid-phase micro-extraction and GC-MS [J]. Food Industry Technology, 2008, 29(5): 57-60. (in Chinese)
[15] XIANG Z Y, LU Y C, ZOU Y, GONG X C, LUO G S. Preparation of microcapsules containing ionic liquids with a new solvent extraction system [J]. Reactive and Functional Polymers, 2008, 68: 1260-1265.
[16] YUAN Li, LIANG Guo-zheng. Synthesis and characterization of microencapsulated dicyclopentadiene with melamine-formaldehyde resins [J]. Coll Poly Sci, 2007, 285: 781-791.
[17] RULE J D, SOTTOS N R, WHITE S R. Effect of microcapsule size on the performance of self-healing polymers [J]. Polymer, 2007, 48: 3520-3529.
[18] YANG H, WU G, CHEN H Z, WANG M. Oil core/polymer shell microcapsules prepared by solvent evaporation technique [J]. Sciencepaper Online, 2009, 4(4): 288-291.
[19] LI Ming, ROUAUD O, PONCELET D. Microencapsulation by solvent evaporation state of the art for process engineering approaches [J]. International Journal of Pharmaceutics, 2008, 363(1/2/3): 26-39.
[20] TORZA S, MASON S G. Three-phase interactions in shear and electrical fields [J]. Journal of Colloid and Interface Science, 1970, 33(1): 67-83.
(Edited by YANG Bing)
Foundation item: Project(50903031) supported by the National Natural Science Foundation of China; Project(2009ZM0046) supported by the Fundamental Research Funds for the Central Universities in China; Project(N9100240) supported by the Foundation for Distinguished Young Talents in Higher Education of Guangdong Province, China
Received date: 2010-04-15; Accepted date: 2010-08-19
Corresponding author: HU Jian-qing, PhD; Tel: +86-20-87113486; E-mail: glennhu@scut.edu.cn