Numerical simulation of intermediate phase growth in Ti/Al alternate foils
来源期刊:中国有色金属学报(英文版)2011年第3期
论文作者:刘江平 骆良顺 苏彦庆 徐严谨 李新中 陈瑞润 郭景杰 傅恒志
文章页码:598 - 603
关键词:热压;扩散偶;TiAl3;有限差分;数值模拟
Key words:hot pressing; diffusion couple; TiAl3; finite difference method; numerical simulation
摘 要:为研究固态Ti/Al扩散偶的扩散反应,将Ti/Al箔构成的扩散偶分别在525, 550, 575 和 600 °C退火1~40 h。实验结果表明TiAl3是Ti/Al界面处生成的唯一相。TiAl3的优先长大是界面热力学作用的结果。TiAl3相主要向Al箔一侧长大,其长大过程符合抛物线规律。在晶界扩散的基础上,用有限差分方法模拟TiAl3相的长大过程,模拟结果和实验结果吻合较好。
Abstract: To investigate the diffusion reaction between Ti/Al solid diffusion couple, Ti/Al alternate foils formed by hot pressing were annealed at 525, 550, 575 and 600 °C for time ranging from 1 to 40 h. The experimental results show that TiAl3 was the only observed phase at Ti/Al interface. The interface thermodynamics favored the preferential formation of TiAl3 in Ti/Al couple. The growth of TiAl3 layer occurred mainly towards Al foil side and exhibited a parabolic law. Using the interdiffusion coefficients calculated based on the contribution of grain boundary diffusion, the growth of TiAl3 was simulated numerically with the finite difference method, and the simulated results were in good agreement with the experimental ones.

LIU Jiang-ping, LUO Liang-shun, SU Yan-qing, XU Yan-jing, LI Xin-zhong,
CHEN Rui-run, GUO Jing-jie, FU Heng-zhi
School of Materials Science and Engineering, Harbin Institute of Technology, Harbin 15001, China
Received 17 May 2010; accepted 6 July 2010
Abstract: To investigate the diffusion reaction between Ti/Al solid diffusion couple, Ti/Al alternate foils formed by hot pressing were annealed at 525, 550, 575 and 600 °C for time ranging from 1 to 40 h. The experimental results show that TiAl3 was the only observed phase at Ti/Al interface. The interface thermodynamics favored the preferential formation of TiAl3 in Ti/Al couple. The growth of TiAl3 layer occurred mainly towards Al foil side and exhibited a parabolic law. Using the interdiffusion coefficients calculated based on the contribution of grain boundary diffusion, the growth of TiAl3 was simulated numerically with the finite difference method, and the simulated results were in good agreement with the experimental ones.
Key words: hot pressing; diffusion couple; TiAl3; finite difference method; numerical simulation
1 Introduction
Elemental powders or foils have been used to fabricate γ-TiAl sheets which have great potential in aerospace and automotive applications due to their high specific strength and excellent high temperature properties[1-2]. One route of producing γ-TiAl sheet with foil metallurgy is that the stacked foils are annealed below the Al melting point under proper pressure, followed by annealing at high temperature (above 1 200 °C) and cooled to room temperature then. The purpose of the first step is to consume Al foils by reactive diffusion. There were some studies on the diffusion reactions between elemental titanium and elemental aluminum, but the results varied with the thickness of Ti and Al[3-5]. If the Ti/Al multilayer with low period produced by direct current (DC) magnetron sputtering was subjected to proper heat treatment, the formation of TiAl3 can be avoided [3]. While for the diffusion couples with a thickness of 2 μm for both Ti and Al foils, only TiAl3 was found at the Ti/Al interface when a sample was annealed below 600 °C and only TiAl2 was found at 650 °C[4]. In the Ti-Al binary system, several intermetallic compounds appear as equilibrium phases in the temperature range of 525-600 °C: TiAl3, TiAl2, TiAl and Ti3Al. The absence of other compounds in Ti/Al diffusion couple were also published by other investigators[6-9]. In fact, the formation of only one compound also occurs in other binary thin films system, such as Zr-Al, Au-Al, Cu-Sn and Au-In systems[10]. Although there are several rules to predict the first phase formation in binary systems, none is suitable for the Ti-Al system. There has been much interest in formulating rules to predict the first phase formation and the subsequent phase sequence in binary metal-silicon and metal-metal thin film systems. WALSER and BENE[11] stated that the first compound nucleated in planar binary reaction couples is the most stable congruently melting compound adjacent to the lowest-temperature eutectic on the bulk equilibrium phase diagram. This rule is relatively successful in predicting the first phase formation in metal-silicon systems. RONAY[12] proposed a rule for the first phase nucleation taking into account of the central eutectic, diffusing species and interfacial free energy. These two models failed to work in Ti-Al system due to the absence of eutectic. Later, PRETORIUS et al[10] suggested that the effective heat of formation (EHF) could be used directly to predict the first phase formation and the subsequent phase sequence. The EHF model is successfully applied to some metal-Si and metal-Al systems, but it fails to predict the first phase in some systems where congruent and non-congruent compounds are simultaneously present. LAIK et al[13] proposed a modification to the EHF model (MEHF) by incorporating a congruency factor. However, none of the above-mentioned rules can predict the first phase formation in Ti-Al system. XU et al[6] successfully predicted the first phase formation among TiAl3, TiAl and Ti3Al in Ti/Al diffusion couple, without considering TiAl2, which is also an equilibrium phase in Ti-Al system. More recently, based on the thermodynamic and kinetic factors (including nucleation and growth), LIU et al[14] proposed a new model to predict the phase formation sequence at the interface between two metals with different structures. This model was successfully applied on the interfacial reactions in Ni-Sn, Cu-In, Cu-Sn and Co-Sn diffusion couples.
In this study, Ti/Al diffusion couples are prepared by hot processing alternating layers of titanium and aluminum foils, and annealing experiments are performed to identify the controlling mechanisms of the reactive diffusion. The first phase selection in Ti-Al system is explained from interface thermodynamic and kinetic point of view, and the growth of TiAl3 is simulated numerically.
2 Experimental
Elemental titanium foils (purity 99.6%, 0.030 mm thick) and elemental aluminum foils (purity 99.6%, 0.023 mm thick) were employed. The Ti and Al foils were etched in 10% HF and 15% NaOH solutions, respectively, water flushed, alcohol cleaned, dried and cut into samples with size of 60 mm×60 mm. Alternating layers of titanium and aluminum foils were stacked with outer layers of titanium foils. Hot pressing was conducted at 610 °C for 3 h under 30 MPa in vacuum of about 10-2 Pa. The vacuum hot-pressed laminates were cut into smaller samples with size of 6 mm×6 mm for subsequent heat treatments. Samples were assembled in a stainless steel sample holder, which can provide pressure during sequential heat treatments. Al2O3 powders were used to prevent the interaction between the sample holder and the samples. The sample holder with sample was placed in a quartz tube, evacuated to a vacuum of 10-2 Pa, sealed, heat-treated in a furnace with a temperature limit of 1 600 °C and then air-cooled.
The thickness of the reaction layers at Ti/Al interfaces was measured from the scanning electron microscope (SEM) micrographs of the cross-sections. Under each condition, an average thickness of the reaction layer was obtained from 20 individual measurements. Annealing experiments were carried out at 525, 550, 575 and 600 °C, with diffusion time varying from 1 to 40 h, respectively. An SEM (Hitachi-S4700) equipped with energy dispersive X-ray spectrometer (EDS) and X’Pert Pro diffractometer with Cu Kα (λ= 0.154 06 nm) were used to identify the intermetallic compounds formed during both vacuum hot-pressing and subsequent annealing treatments.
3 Results and discussion
As shown in Fig.1(a), the interfaces between the bonded Ti and Al foils are fairly straight, indicating a good bonding between Ti and Al foils. At large magnification, thin reaction layers with an average thickness of 0.5 μm can be observed in Fig.1(b). Micrographs of cross-sections in samples are all composed of three regions, i.e. grey dark grey, and dark regions. EDS analysis on samples with wider dark grey regions elucidates that the compositions of the grey, dark grey and dark regions on each micrograph correspond to Ti, TiAl3 and Al, respectively. The formation of TiAl3 is also confirmed by XRD analysis (Fig.2) on cross-sections of the samples annealed at 600 °C for 3 h. Further experimental results suggest that in the temperature range of 525-600 °C, TiAl3 is the only compound observed after reactive diffusion before Al foils are consumed. EDS analysis shows that with the growth of TiAl3 layer, Al foils are consumed, and TiAl2, TiAl and Ti3Al exist between Ti and TiAl3. Comparing the sample annealed at 550 °C for 1.5 h (Fig.3(a)) with the one annealed at 550 °C for 16 h (Fig.3(b)), it can be found that the thickness of Al foils shrinks more

Fig.1 BSE images of cross-section of laminates hot-pressed at 610 °C for 3 h (a) and TiAl3 with average thickness of 0.5 μm appeared at Ti/Al interface (b)

Fig.2 XRD pattern of cross-section in samples annealed at 600 °C for 3 h

Fig.3 SEM micrographs of interface zone of samples annealed at 550 °C for 1.5 h (a) and for 16 h (b)
obviously than Ti foils. Obviously, TiAl3 grows mainly towards Al foils. This observation is consistent with the work of XU et al[6] and LUO et al[7]. The Ti-TiAl3 interface is rough, which may be caused by the difference in size of TiAl3 grains along the interface. EDS analysis shows that under all annealing conditions, there are few Al atoms in Ti foils, except for a small region where Ti foil meets the TiAl3 layer. Figure 4 shows the concentration profiles of the cross-section of
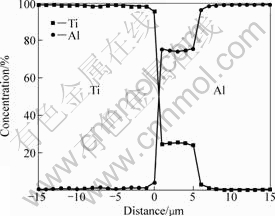
Fig.4 Concentration profiles of cross section of sample annealed at 575 °C for 6 h
the sample annealed at 575 °C for 6 h. The same results occur in the work of Van LOO and RIECK[8] on bulk Ti/TiAlx (x=1, 2) diffusion couples. Considering the solubility of Al in Ti is about 12% (molar fraction) in the temperature range studied in this work, it can be concluded that no Al atoms diffused into Ti foils through TiAl3, i.e., all Al atoms diffusing from Al foils are used to synthesize TiAl3.
Nucleation driving force is mainly decided by the combination of the interfacial energy and the volume Gibbs energy. The interfacial energy associated with new phase is nucleation resistance and exert main effect on nucleation at the very early stage. The compound with the lowest associated interface energy will nucleate more easily than the others. CHAKRABORTY et al[15] also proposed that interface thermodynamics can play a decisive role in the appearance/disappearance/ suppression of reaction phases diffusion couple. It is shown below that the interfacial energy also induces the initial formation of TiAl3.
The interfacial energy of the initial A/B interface (γA-B, A or B can be either Ti or Al), surface energy of phase AB (γAB, AB can be Ti3Al, TiAl, TiAl2 or TiAl3) and interfacial energy of AB/A(γA-AB) can be determined by equations described in Ref.[16]. Some parameters, such as the enthalpy change upon solution of 1 mole A in an infinitely large reservoir of B, the molar volume of atoms, the surface energy and surface fraction of atoms can be found in Refs.[15, 17-18]. The results calculated by methods mentioned above show that at all temperatures in the present work, TiAl3 has the lowest interfacial energy. For example, the new formed interfacial energies at 600 °C due to the formation of TiAl3, TiAl2, TiAl and Ti3Al are 450, 480, 534 and 598 mJ/m2, respectively, indicating that TiAl3 nucleates firstly in Ti/Al couple. This result is consistent with the experimental results.
4 Numerical simulation of TiAl3 phase
4.1 Model and assumptions
A numerical model employing finite difference methods (FDM) was developed to solve the diffusion equations with the purpose of evaluating the kinetics of TiAl3 layer. Similar approaches were proved successfully in previous studies of diffusion controlled layer growth[19-20]. The model yielded interphase- interface positions and concentration profiles as functions of time. The growth of TiAl3 layer may be characterized as a finite, three-phase, binary and diffusion-controlled process. Figure 5 shows the concentration profile and the definition of the space grid terms used in the numerical formulation of the partial differential equations. Partial differential equations describe the change in concentration within each phase and the velocity of the interfaces.

Fig.5 Concentration profile and definition of space grid terms used in numerical simulation (ξ0, ξ3 are centers of Al foil and Ti foil, respectively)
The concentration with time within each phase follows Fick’s law:
![]() (4)
(4)
where C is the aluminum concentration; t is the diffusion time; x is the displacement and ![]() is the interdiffusion coefficient of the αi phase (i=0, 1, 2; αi=Al, TiAl3, Al)
is the interdiffusion coefficient of the αi phase (i=0, 1, 2; αi=Al, TiAl3, Al)
The flux balance equation for the interphase interface is
![]()
 (5)
(5)
where ![]() and
and ![]() (i=1, 2) are the aluminum concentrations in (αi-1) phase in equilibrium with αi phase, aluminum concentration in αi phase in equilibrium with (αi-1) phase, respectively; ξi- and ξi+ are the negative side and positive side of ξi, respectively.
(i=1, 2) are the aluminum concentrations in (αi-1) phase in equilibrium with αi phase, aluminum concentration in αi phase in equilibrium with (αi-1) phase, respectively; ξi- and ξi+ are the negative side and positive side of ξi, respectively.
It should be noted that ξ0 and ξ3 are the centers of Al foil and Ti foils, respectively. The moving velocities of ξ0 and ξ3 are zero and the fluxes across these positions are zero due to the symmetry, i.e. the boundary condition is
![]() (6)
(6)
Assumptions made in determining the solutions to the partial differential equations include: 1) interdiffusion coefficients are independent on composition within each phase; 2) interface concentrations are obtained from equilibrium phase diagram.
4.2 Calculation of interdiffusion coefficients
As known, there are no available interdiffusion coefficients of Ti, Al and TiAl3 in the temperature range in the present work. The interdiffusion coefficient ![]() in a binary alloys consisting of components A and B is related to the tracer-diffusion coefficients,
in a binary alloys consisting of components A and B is related to the tracer-diffusion coefficients, ![]() and
and ![]() by Darken-Manning equation[21]:
by Darken-Manning equation[21]:
![]() (7)
(7)
where Ci and Di are the molar fractions and intrinsic diffusion coefficient of component i, respectively. The thermodynamic factor Φ is defined as:
![]() (8)
(8)
where aB is the thermodynamic activity of component B. In present work, Φ, which is determined by means of CALPHAD calculations, varies between 1 and 1.06 in both Al(Ti) and Ti(Al) solid solutions. The vacancy wind factor is usually taken as 1. Ti tracer diffusion coefficient in pure Al or solid solution Al(Ti) (referred to as ![]() ) has never been studied. The atomic radius difference and valence difference between the atoms of solute and solvent affect the diffusion of impurities in pure metals[22]. For elements in the same group of the periodic table, the difference of factors mentioned above can be neglected, so the diffusion coefficients of atoms in the same group in the same metal differ slightly. The validity of this deduction can be verified from the diffusion coefficients of some impurities in Al[23]. Fortunately, the diffusion of Zr, another element in IVA group, in Al was studied. As a result, the tracer diffusion coefficient of Ti in Al is assumed to be equal to that of Zr in Al. The tracer-diffusion coefficients of Ti and Al atoms in Ti can be obtained in Ref.[24], and the intrinsic coefficients of these two atoms in TiAl3 can be obtained in Ref.[4]. It should be noted here that these diffusion coefficients are lattice diffusion coefficients. This is very important in this numerical simulation, which will be discussed below.
) has never been studied. The atomic radius difference and valence difference between the atoms of solute and solvent affect the diffusion of impurities in pure metals[22]. For elements in the same group of the periodic table, the difference of factors mentioned above can be neglected, so the diffusion coefficients of atoms in the same group in the same metal differ slightly. The validity of this deduction can be verified from the diffusion coefficients of some impurities in Al[23]. Fortunately, the diffusion of Zr, another element in IVA group, in Al was studied. As a result, the tracer diffusion coefficient of Ti in Al is assumed to be equal to that of Zr in Al. The tracer-diffusion coefficients of Ti and Al atoms in Ti can be obtained in Ref.[24], and the intrinsic coefficients of these two atoms in TiAl3 can be obtained in Ref.[4]. It should be noted here that these diffusion coefficients are lattice diffusion coefficients. This is very important in this numerical simulation, which will be discussed below.
It has been known that grain boundary (GB) provides high diffusivity (‘short circuit’) paths in metals and GB diffusion plays a key role in the diffusion of polycrystal[24]. Like lattice diffusion, GB diffusion usually follows an Arrhenius law with boundary diffusion activation energy Qb and pre-exponential factor D0b. Qb is two orders of magnitude less than the lattice diffusion activation energy Qv[25], more exactly, Qb/Qv≈0.4-0.6, and the pre-exponential factors are not much different. D0b is usually viewed as equal to D0v (pre-exponential factor for lattice diffusion). To evaluate true diffusion coefficient in polycrystal, the effective diffusion coefficient related to the volume diffusion coefficient Dv and grain diffusion coefficient Db is based on the equation[25]
Deff=fDb+(1-f)Dv (9)
where f is the volume fraction of GBs in the polycrystal, i.e. f=qδ/d; q is a numerical factor depending on the grain shape (q=1 here); δ is the GB size (assuming δ=0.5 nm usually) and d is the grain size[25]. The average Ti grain size in the normal direction of the foil is about 7 μm and TiAl3 grain size varies between 3 and 4 μm. For samples annealed for a long time, in the consideration of small grain size at the early stage of annealing, the average grain size in numerical simulation is taken as 3 μm. All effective interdiffusion coefficients needed in numerical simulation can be calculated based on the analysis above and shown in Table 1, and the interface concentrations are determined from Ref.[26].
Theoretically speaking, TiAl3 layer in Ti/Al diffusion couple should grow with the increase of annealing time, which was proved by previous research work[2, 4, 6]. However, in the present work, the diffusion may be caused by the difference in pressure on diffusion couples only at 550 °C. The predicted TiAl3 phase growth in diffusion couple was compared with the experimental data of present work at 550 °C and the work by XU et al[6] at temperatures of 520, 575 and 600 °C, respectively. Figures 6(a) and (b) show the calculated TiAl3 layer thickness and experimental values with standard deviations as a function of annealing time at temperatures of 520, 550, 575 and 600 °C. To be distinguished clearly, the results of 520 and 600 °C are plotted in Fig.6(a) and the others are plotted in Fig.6(b). The simulation results agree well with the experimental values and the growth of TiAl3 exhibits a parabolic law, indicating that the growth of TiAl3 is diffusion controlled.
Table 1 Interface concentrations and calculated interdiffusion coefficients
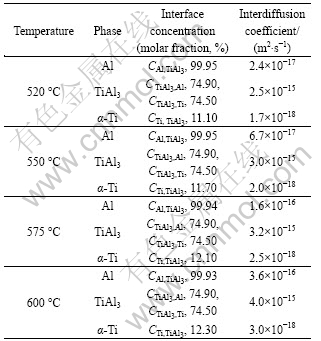
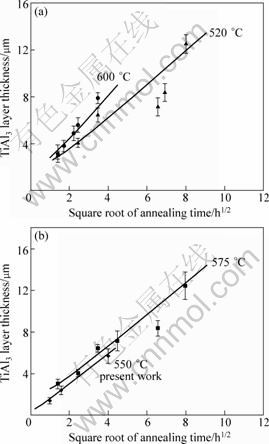
Fig.6 Numerical simulation and experimental results: (a) 520 and 600 °C[6]; (b) 575 °C[6] and 550°C(present work)
5 Conclusions
1) Only TiAl3 formed at the Ti/Al interface when Ti/Al foils were annealed at the temperature range of 525-600°C, which was explained from the interface thermodynamic point of view.
2) The growth of TiAl3 layer took place mainly on Al foils and followed the parabolic law.
3) The growth of TiAl3 phases was simulated successfully based on a numerical model employing finite difference method.
References
[1] LIU Zhi-jian, QU Xuan-hui, HUANG Bai-yun. TiAl alloys prepared by thermal extrusion of elemental powder mixtures [J]. Transactions of Nonferrous Metals Society of China, 1997, 7: 96-100.
[2] JIANG Yao, HE Yue-hui, TANG Yi-wu, LI Zhi, HUANG Bai-yun. Fabrication of Ti-Al alloy sheets by element powder cold roll forming and reactive synthesis [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(9): 1501-1507 (in Chinese).
[3] RAMOS A S, VIEIRA M T. Kinetics of the thin films transformation Ti/Al multilayer→γ-TiAl [J]. Surface and Coatings Technology, 2005, 200(1-4): 326-329.
[4] TARDY J, TU K N. Solute effect of Cu on interdiffusion in Al3Ti compound films [J]. Physical Review B, 1985, 32(4): 2070-2081.
[5] RAMOS A S, VIEIRA M T, GRZONKA J, SIM?ES S, VIEIRA M F. Production of intermetallic compounds from Ti/Al and Ni/Al multilayer thin films-A comparative study [J]. Journal of Alloys and Compands, 2009, 484(1-2): 335-340.
[6] XU L, CUI Y Y, HAO Y L, YANG R. Growth of intermetallic layer in multi-laminated Ti/Al diffusion couples [J]. Materials Science and Engineering A, 2006, 435-436(5): 638-647.
[7] LUO Jian-guo, ACOFF V L. Using cold roll bonding and annealing to process Ti/Al multi-layered composites from elemental foils [J]. Materials Science and Engineering A, 2004, 379(1-2): 164-172.
[8] Van LOO F J J, RIECK G D, Diffusion in the titanium-aluminium system-II. Interdiffusion in the composition range between 25 and 100 at.% Ti [J]. Acta Metallurgica, 1973, 21(1): 73-84.
[9] ROMANKOV S, SHA W, ERMAKOV E, MAMAEVA A. Characterization of aluminized layer formation during annealing of Ti coated with an Al film [J]. Journal of Alloys and Compounds, 2006, 420(1-2): 63-70.
[10] PRETORIUS R, VREDENBERG A M, SARIS F W, REUS R. Prediction of phase formation sequence and phase stability in binary metal-aluminum thin-film systems using the effective heat of formation rule [J]. Journal of Applied Physics, 1991, 70(7): 3636-3646.
[11] WALSER R M, BEN? R W. First phase nucleation in silicon-transition-metal planar interfaces [J]. Applied Physics Letters, 1976, 28(10): 624-625.
[12] RONAY M. Reinvestigation of first phase nucleation in planar metal-Si reaction couples [J]. Applied Physics Letters, 1983, 42(7): 577-579.
[13] LAIK A, BHANUMURTHY K, KALE G B. Intermetallics in the Zr-Al diffusion zone [J]. Intermetallics, 2004, 12(1): 69-74.
[14] LIU Hua-shan, WANG Hang, ZHU Wen-jun, TAO Xiao-ma, JIN Zhan-peng. Prediction of formation of intermetallic compounds in diffusion couples [J]. Journal of Materials Research, 2007, 22(6): 1502-1511.
[15] CHAKRABORTY J, WELZEL U, MITTEMEIJER E J. Interdiffusion, phase formation, and stress development in Cu-Pd thin-film diffusion couples: Interface thermodynamics and mechanisms [J]. Journal of Applied Physics, 2008, 103(11): 113512.
[16] BENEDICTUS R, B?TTGER A, MITTEMEIJER E J. Thermodynamic model for solid-state amorphization in binary systems at interfaces and grain boundaries [J]. Physical Review B, 1996, 54(13): 9109-9125.
[17] SCHUSTER J C, PALM M. Reassessment of the binary Aluminum-Titanium phase diagram [J]. Journal of Phase Equilibria and Diffusion, 2006, 27(3): 255-277.
[18] MIEDEMA A R, CH?TEL P F, BOER F R. Cohesion in alloys-fundamentals of a semi-empirical model [J]. Physica B+C, 1980, 100(1): 1-28.
[19] JAN C H, CHEN C P, CHANG Y A. Growth of intermediate phases in Co/Si diffusion couples: bulk versus thin-film studies [J]. Journal of Applied Physics, 1993, 73(3): 1168-1179.
[20] HICKEL A J, HECKEL R W. Kinetics of phase layer growth during aluminide coating of nickel [J]. Metallurgical and Materials Transactions A, 1975, 6(3): 431-440.
[21] IKEDA T, ALMAZOUZI A, NUMAKURA H, KOIWA M. Single-phase interdiffusion in Ni3Al [J]. Acta Materialia, 1998, 46(15): 5369-5376.
[22] XIA Li-fang, ZHANG Zhen-xing. Diffusion in metals [M]. Harbin: Press of Harbin Institute of Technology, 1988: 98-102. (in Chinese)
[23] BRANDES E A, BROOK G B. Smithells Metals Reference Book-7th Edition [M]. Bath: The Bath Press, 1992: 13-118.
[24] MISHIN Y, HERZIG C. Diffusion in the Ti-Al system [J]. Acta Materialia, 2000, 48(3): 589-623.
[25] MISHIN Y, HERZIG C. Grain boundary diffusion: recent progress and future research [J]. Materials Science and Engineering A, 1999, 260(1-2): 55-71.
[26] ZHANG F, CHEN S L, CHANG Y A, KATTNER U R. A thermodynamic description of the Ti-Al system [J]. Intermetallics, 1997, 5(6): 471-482.
刘江平,骆良顺,苏彦庆,徐严谨,李新中,陈瑞润,郭景杰,傅恒志
哈尔滨工业大学 材料科学与工程学院,哈尔滨 150001
摘 要:为研究固态Ti/Al扩散偶的扩散反应,将Ti/Al箔构成的扩散偶分别在525, 550, 575 和 600 °C退火1~40 h。实验结果表明TiAl3是Ti/Al界面处生成的唯一相。TiAl3的优先长大是界面热力学作用的结果。TiAl3相主要向Al箔一侧长大,其长大过程符合抛物线规律。在晶界扩散的基础上,用有限差分方法模拟TiAl3相的长大过程,模拟结果和实验结果吻合较好。
关键词:热压;扩散偶;TiAl3;有限差分;数值模拟
(Edited by FANG Jing-hua)
Foundation item: Project (50771041) supported by the National Natural Science Foundation of China; Project (05-0350) supported by the New Century Excellent Talents in University, China
Corresponding author: SU Yan-qing; Tel: +86-451-86417395; E-mail: suyq@hit.edu.cn
DOI: 10.1016/S1003-6326(11)60756-5

