���±�ţ�1004-0609(2012)09-2656-06
CaCl2-Ca(OH)2-H2O��ϵ���������Ƶ�ƽ��Ũ��
����ƽ���� �գ�ʯ��������ϲ�ƣ��� �ǣ��̿��壬�� ��
(���ϴ�ѧ ұ���ѧ�빤��ѧԺ����ɳ 410083)
ժ Ҫ��������25��ʱCaCl2-Ca(OH)2-H2O��ϵ��c(CaCl2)��c(Ca(OH)2)ͼ��c(CaCl2)��pHֵͼ�������������ϵ��c(Ca(OH)2)��pHֵ����c(CaCl2)�����Ӷ����ϼ�С, c(Ca(OH)2)�仯������Debye-Huckel����ֵ�������c(CaCl2)��3.78 mol/Lʱ����ϵpHֵ��10.4����c(CaCl2)��2.57mol/Lʱ���г�����CaClOH���ɡ������㣬������CaCl2ĸҺ����ѭ������ʯ��ˮ132�Σ��о����Ϊʯ��ˮ���Ʊ�������þ������CaCl2ĸҺѭ������ʯ��ˮ�ṩ��һ�����������ݡ�
�ؼ��ʣ�CaCl2-Ca(OH)2-H2O��ʯ��ˮ��CaCl2ĸҺ��pHֵ��ѭ��
��ͼ����ţ�TF111.3���� ���ױ�־�룺A
Equilibrium concentration of calcium hydroxide in
CaCl2-Ca(OH)2-H2O system
LIU Wei-ping, XU Hui, SHI Xi-chang, YANG Xi-yun, CHEN Ya, CHENG Jun-feng, LI Gui
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: The c(CaCl2)��c(Ca(OH)2) chart and c(CaCl2)��pH value chart in CaCl2-Ca(OH)2-H2O system under 25 �� were plotted. The results represent that c(Ca(OH)2) and pH value decrease with the increase of c(CaCl2), the trend of c(Ca(OH)2) is supported by the calculation results of Debye-Huckel. When c(CaCl2)��3.78 mol/L, the pH value of the system is bigger than 10.4, and CaClOH is preciptated while c(CaCl2)��2.57 mol/L. Through calculation, the CaCl2 mother liquor can reuse 132 times for collocating lime water in theory. The research results provide a theoretical basis for the treatment and reuse of calcium chloride mother liquor for collocating lime water which is the precipitant in the process of synthesizing magnesium hydroxide.
Key words: CaCl2-Ca(OH)2-H2O; lime water; CaCl2 mother liquor; pH value; recycling
������Ŀ�����ҿƼ�֧�żƻ���ʮһ�塱�ش���Ŀ(2008BAB35B04)������ר�����ϴ�ѧǰ���о��ƻ��ص���Ŀ(2010QZZD003)
�ո����ڣ�2011-09-07�������ڣ�2012-01-16
ͨ�����ߣ��� �գ����ڣ���ʿ���绰��0731-88877352��E-mail: xuhui_0318@hotmail.com
������þ��Ϊһ����Ҫ����������ɹ㷺Ӧ������ȼ��[1-3]����ˮ����[4]����������ҽҩ���� ��[5-7]������������þ��;�㷺���ʶ����Ʊ��������о����ܹ�ע��
Ŀǰ����ҵ������������þ�ķ����а������ռ��ʯ���鷨�ȣ�������Ϊ�������������İ�����ˮ�Ի�����Ⱦ��ͬʱ�����ڻӷ����д̼�����ζ����ɹ����Ͷ�������˷������ܵ����ơ��ռ�����ɱ��ϸߡ�ʯ���鷨�ɱ��ϵͣ����ܹ��˲�Ʒ���Ƚϵ͵�ƿ�����÷����нϺõ��г�Ӧ��ǰ������±ˮ����������þ�ȿ�ʯ����������þ������������[8]����ˣ��о���ʯ��ˮ��±ˮΪԭ���Ʊ�������þ�¹��վ�����Ҫ���塣ʯ��ˮ���Ʊ�������þʱ���������Ȼ���ĸҺ�������Ȼ���ĸҺѭ������ʯ��ˮ�ǽ���ʯ��ˮ���ɱ�����Ч;�����������Ȼ���ĸҺ����ʯ��ˮ�IJ���ѭ�����Ȼ��ƻ���ʯ��ˮ�в��ϸ�����ѭ����һ���������Ȼ���ĸҺ�����˼�������ʯ��ˮ��Ŀǰ���Ȼ���ĸҺѭ������ʯ��ˮ�Ĺ����о����٣������������о���δ��������
�����������õ°��ݿ˶�������̽��CaCl2- Ca(OH)2-H2O��ϵ���Ȼ���Ũ�ȶ����������ܽ�ȵ�Ӱ�죬ͬʱ�ⶨ����ϵpHֵ�ı仯���ɣ��������Ȼ���ĸҺ����ʯ��ˮ����ѭ���Ĵ�����Ϊ�Ȼ���ĸҺѭ������ʯ��ˮ�ṩ����ָ����
1 ʵ��
1.1 �Լ�������
��Ҫ�Լ���Ca(OH)2(���������㶫��ͷ����¤����������)��CaCl2(���������㶫��ͷ����¤����������)���Ҷ������������(����������ҩ���Ż�ѧ�Լ�����˾����)��NaOH(�ż���������л������ֻ�ѧ�Լ�������)����-����(������������л�ѧ�Լ��о�������)������ˮ�ȡ�
��Ҫ������PHS-3C����pH�ƣ�AUY220�͵��ӷ�����ƽ��JBV-���Ƶ���ٽ�������HH-601��������ˮԡ��SHZ-CD��ѭ��ˮʽ������ձõȡ�
1.2 ʵ�鷽��
����һ��Ũ�ȵ��Ȼ�����Һ�������˳��Ӻ�װ��������ƿ�У�����25 ��ĺ�������ˮԡ���У��̶�������̨��������轰������ת��Ϊ300 r/min���ҡ��õ��ӷ�����ƽ��ȡһ�����ķ������������ƣ�����������ƿ�У���Ӧ�ﵽƽ���ȡ�����˺������������Ũ�Ȳ���EDTA���Ƶζ����ⶨ�����������ӵIJⶨ���õζ����ⶨ�����Ȼ�����Һ��ΪAi��Һ����Ai��Һ�м�������������ƣ��ﵽ�ܽ�ƽ�����˵���Һ��ΪBi��Һ��
2 ʵ��ԭ��
2.1 ��Һ�ܽ�ƽ����ܶȻ��ͻ�Ȼ�
��һ���¶��£���������ˮ���ܽ�ﵽ����ʱ����ﵽ���ܽ�ƽ��[9]������һ�ָ�������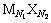 �����ܽ�ƽ����Ա�ʾΪ
�����ܽ�ƽ����Ա�ʾΪ
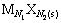

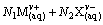 (1)
(1)
���ﵽƽ��ʱ��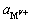 ��
��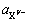 �ֱ�Ϊ�����Ӻ����������ܽ�ƽ��ʱ�Ļ�ȣ�
�ֱ�Ϊ�����Ӻ����������ܽ�ƽ��ʱ�Ļ�ȣ�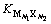 Ϊ����ˮ�е��ܽ�ƽ�ⳣ���������¶Ⱥ�����ǿ���йأ��ʳ�Ϊ����ʻ�Ȼ��������Թ����ܽ�ȼ����������ӵ�ˮ�⣬��ֻ������Һ������ǿ�ȵ�Ӱ�죬���ɽ��л�Ȼ����㡣����˽���������CaCl2-Ca(OH)2-H2O��Ԫ��ϵ��
Ϊ����ˮ�е��ܽ�ƽ�ⳣ���������¶Ⱥ�����ǿ���йأ��ʳ�Ϊ����ʻ�Ȼ��������Թ����ܽ�ȼ����������ӵ�ˮ�⣬��ֻ������Һ������ǿ�ȵ�Ӱ�죬���ɽ��л�Ȼ����㡣����˽���������CaCl2-Ca(OH)2-H2O��Ԫ��ϵ��
3.2 25 ��ʱCaCl2-Ca(OH)2-H2OϵCa(OH)2ƽ��Ũ�ȵļ���CaCl2-Ca(OH)2-H2Oϵ��һ���ɺ��й�ͬ������Ca2+���Ρ����ˮ��ɵ���Ԫ��ϵ����Ca(OH)2�DZ��͵ģ���Ũ��Ϊc(Ca(OH)2)����CaCl2û�б��ͣ���Ũ��Ϊc(CaCl2)����Ca(OH)2 ���ܽ�ƽ�ⳣ��Ϊ����ʽ(12)���ɵõ���Һ���Ȼ���Ũ������������Ũ�ȵĹ�ϵ��
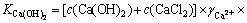
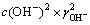 (2)
(2)
��c(CaCl2)����֪�ģ���c(Ca(OH)2)δ֪����CaCl2��Ũ��Ϊc(CaCl2)ʱ������ʽ(2)���c(Ca(OH)2)��

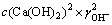 (3)
(3)
25 ��ʱ��KSP=[Ca2+][OH-]2=5.5��10-6[10]����Debye-Huckel��ʽ[11]����Ca2+��OH-�Ļ��ϵ����
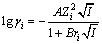 (4)
(4)
ʽ�У�riΪ���Ӱ뾶��A��BΪϵ�������ܼ����¶Ⱥͽ�糣���йأ����[11]֪25 ��ʱ��A=0.511 5��B= 0.329 1��r(Ca2+)=0.6 nm��r(OH-)=0.35 nm��
��Һ����ǿ��I:
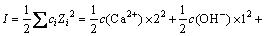
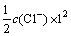 (5)
(5)
ʽ�У�ciΪi���ӵ����ʵ���Ũ�ȣ�ZiΪi���ӵĵ���������н�ʽ(9)����ʽ(4)�пɵ�ʽ(10)��(11)��
c(Ca2+)=c(Ca(OH)2)+c(CaCl2) (6)
c(OH-)=2c(Ca(OH)2) (7)
c(Cl-)=2c(CaCl2) (8)
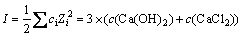 (9)
(9)
�����ӻ�����ӣ�
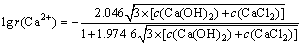 (10)
(10)
���������ӵĻ�����ӣ�
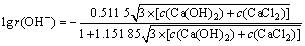 (11)
(11)
��ʽ(10)��(11)����ʽ(3)��ʽ(12)��
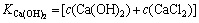 ��
��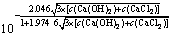 ��4��c(Ca(OH)2)2��
��4��c(Ca(OH)2)2��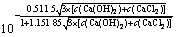 (12)
(12)
3 ���������
3.1 ƽ��ʱ���ȷ��
��0.48 mol/L���Ȼ�����Һ�м�������������ƣ���Һ���Ϊ800 mL���¶�Ϊ25 �棬�����ٶ�Ϊ300 r/min��Ϊ����������Һ��CO2��Ӧ��ˮ�ֵ�����Ӱ��ʵ�������ձ��ý����ͱ���Ĥ�ܷ⡣��һ��ʱ���ȡ������������OH-Ũ�Ⱥ�pHֵ�ı仯�����ʵ������ͼl��ʾ����ͼ1��֪����Ӧ2 h��OH-Ũ�Ⱥ�pHֵ�������ֲ��䣬�����ܽ�ﵽƽ�⡣
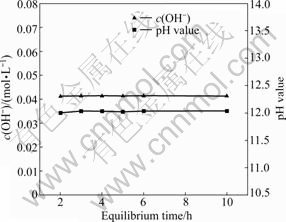
ͼl c(OH-)��pHֵ��ƽ��ʱ��Ĺ�ϵ
Fig. 1 Relationships between c(OH-), pH value and equilibrium time
3.2 25 ��ʱCaCl2-Ca(OH)2-H O��ϵ��Ca(OH)2ƽ��Ũ��
OH-ƽ��Ũ�ȵı仯��������ͼ2��ʾ����ͼ2��֪�������Ȼ���Ũ�ȵ����ӣ��������Ƶ�Ũ�Ȳ����½���
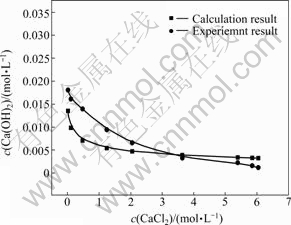
ͼ2 ����ֵ��ʵ��ֵ�Ĺ�ϵ
Fig. 2 Relationship between calculation result and experiment result
���õ°��ݿ˶������ɼ��㲻ͬ�Ȼ���Ũ������������Ũ�ȣ�����������ͼ2�л������ͼ2��֪�������õ���������Ũ�ȱ仯���ƺ�ʵ�ʽ��һ�£����������ۼ���ʱ�����˹����ܽ�ȼ����������ӵ�ˮ�⣬������ֵ����һ����ƫ�ͬʱ������c(CaCl2)��2.57 mol/Lʱ��CaCl2��Ca(OH)2��Ӧ������CaClOH����ˣ���ͼ2��ʾ����c(CaCl2)��2.57mol/L�������Ȼ���Ũ�ȵ����ӣ���������Ũ����Ϊ���뷴Ӧ�����Ͻ��ͣ���c(CaCl2)��3.3 mol/L���������Ƶ�ʵ��ֵ������ֵ�͡�
3.3 CaCl2-Ca(OH)2-H2O��ϵ��pHֵ�ı仯����
ʯ��ˮ���Ʊ�������þ�����У�ͨ����������������Һ����pHֵ�����Ʒ�Ӧ���̵Ľ��У�pHֵ��������þ�ijɺ˺ͽᾧ�������̵�Ӱ��ܴ�[12-14]����ˣ��о�B��Һ���Ȼ���Ũ������ҺpHֵ֮��Ĺ�ϵ�dz���Ҫ��
��ͼ3��ʾ�������Ȼ���Ũ�ȵ����ӣ�B��Һ��pHֵ�dz��½����ơ��������������Ȼ���Ũ�����ӣ�������Һ�и��������ӣ�����ͬ����ЧӦ����������Ũ�ȱ�Ȼ�½����Ʊ�������þ�����з�Ӧ�յ�pHֵ����10.4[14]���ʵ�B��ҺpHֵ����10.4ʱ�����������Ʊ�������þ�ķ�Ӧ��ͼ3��a����д����ԣ�a���Ȼ���Ũ��Ϊ3.78 mol/L��pHֵΪ10.45�����Ȼ�
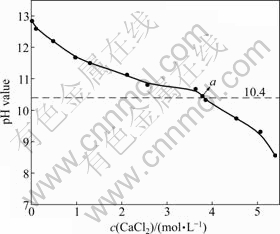
ͼ3 B��Һc(CaCl2)��pHֵ�Ĺ�ϵ
Fig. 3 Relationship between c(CaCl2) and pH value of B solution
��ĸҺѭ������ʯ��ˮ������Ȼ���Ũ��Ϊ3.78 mol/L��ʵ�ʷ�Ӧ�����У�����ͨ���ⶨ��ӦĸҺ�Ȼ���Ũ�Ⱥ�pHֵ���ж��Ȼ���ĸҺ�Ƿ���Խ�����һ��ѭ������ʯ��ˮ��
3.4 CaCl2-Ca(OH)2-H2O��ϵ�ᾧ�������
��ͼ4��ʾ, B1~B7��Һ������Ũ��ʼ�մ��ڶ�Ӧ��A1~A7��Һ������Ũ�ȣ�����A8��Һ��B8��Һ������Ũ�ȶ�Ϊ2.57 mol/L���˺�B9~B11��Һ������Ũ��ʼ��С�����Ӧ��A9~A11��Һ������Ũ�ȡ�������ͼ5��ʾ��B1~B11��Һ��pHֵʼ���Ǹ���A1~A11��Һ��pHֵ������B1~B11��Һ�ж������������ܽ⡣

ͼ4 c(Ca2+)Ai��c(Ca2+)Bi�Ĺ�ϵ
Fig. 4 Relationship between c(Ca2+)Ai and c(Ca2+)Bi
��ˣ���c(Ca2+)A��2.57 mol/Lʱ��c(Ca2+)B��c(Ca2+)A������ΪCaCl2��Ca(OH)2��Ӧ������CaClOH����B9��Һ������ո���ýᾧ�XRD������ͼ6��ʾ��
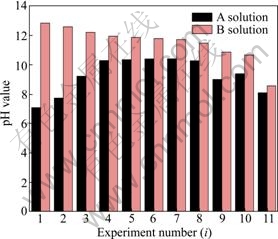
ͼ5 pH(Ai)��pH(Bi)�Ĺ�ϵ
Fig. 5 Relationship between pH(Ai) and pH(Bi)
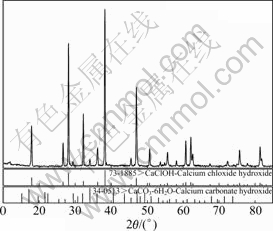
ͼ6 �ᾧ���XRD��
Fig. 6 XRD pattern of crystal
��ͼ6��ʾ���ᾧ������Ҫ��CaClOH��CaCO3? 6H2O���ֹ��ࡣCaCO3?6H2O������ʵ����ʯ��ˮ������Ӵ���ԭ����ʽ(13)��ʾ�������ױ���[15-17]��CaClOH������ԭ����ʽ(14)��ʾ��
Ca(OH)2+5H2O+CO2��CaCO3?6H2O�� (13)
CaCl2+Ca(OH)2��2CaClOH�� (14)
��ˣ����Ȼ���ĸҺŨ�ȴ���2.57 mol/Lʱ����ĸҺ���ʺ�����ʯ��ˮ����Ϊ�����ɼ�ʽ�Ȼ���(CaClOH)������������Ƶ���ʧ�ͳɱ�����ߣ�ͬʱҲ�ή�Ͳ���������þ�Ĵ��ȡ���ʱ����ͼ3��֪����c(Ca2+)=2.57ʱ��B��ҺpHֵΪ10.81��
3.5 �Ȼ���ĸҺѭ�������ļ���
���Ʊ�������þ��Ӧ����pHֵ���ǣ��Ȼ���ĸҺŨ�ȵ���3.78 mol/L��������ʯ��ˮ���������Ȼ���ĸҺŨ�ȹ����������������ɼ�ʽ����ƣ�����������Ƶ���ʧ�����Դӳɱ��Ͽ��ǣ����Ȼ���ĸҺŨ�ȴ���2.57 mol/Lʱ������������ʯ��ˮ��
MgCl2+Ca(OH)2��Mg(OH)2��+CaCl2 (15)
ʯ�ҷ��Ʊ�������þԭ����ʽ(15)��ʾ�������һ�η�Ӧʱʯ��ˮŨ��Ϊ0.019 5 mol/L�������������Ȼ�þ����������1:1���С�ԭ��Ϊ5 mL ��95 g/L MgCl2��Һ��1 L��0.019 5 mol/Lʯ��ˮ����Ӧ��ɺ�ĸҺ�Ȼ���Ũ��Ϊ0.019 4 mol/L�����跴ӦĸҺ����ʧ��ÿ�εõ�1.005 L��ӦĸҺ������ʯ��ˮ��Ϊ1.005 L��ȡ1 L������һ��ѭ����ͨ�����㣬��֪��ӦĸҺ����ѭ��132�Ρ�
3 ����
1) 25 ��ʱ��CaCl2-Ca(OH)2-H2O��ϵ�辭2 h�ﵽƽ�⡣
2) 2 5��ʱ��CaCl2-Ca(OH)2-H2O��ϵ����������Ũ�������Ȼ���Ũ�ȵ����߶����ͣ����õ°��ݿ˶������ɼ����Ȼ���Ũ������������Ũ�ȵĹ�ϵ������ʵ��ֵ�ͼ���ֵ���ϡ�
3) 25 ��ʱ��CaCl2-Ca(OH)2-H2O��ϵ���Ȼ���Ũ��С��3.78 mol/Lʱ����ҺpHֵ��10.4��
4) 25 ��ʱ��CaCl2-Ca(OH)2-H2O��ϵ���Ȼ���ĸҺŨ��С��2.57 mol/Lʱ����CaClOH�����ɡ�
5) �ӳɱ��ۺϿ��ǣ�ȷ���Ȼ���ĸҺŨ��С��2.57 mol/L��pHֵ����10.81ʱ��������������ʯ��ˮ�����ݷ�Ӧ��ģ�ͣ��������Ȼ���ĸҺ����ѭ������ʯ��ˮ132�Ρ�
REFERENCES
[1] ������, �� ��. ��������ȼ�����о���չ[J]. ���ջ���, 2002, 30(4): 20-22.
XU Wang-sheng, ZHANG Yi. Research progress of novel inorganic five retarding agent [J]. Jiangsu Chemical Industry, 2002, 30(4): 20-22.
[2] KIM S. Flame retardancy and smoke suppression of magnesium hydroxide filled polyethylene [J]. Journal of Polymer Science Part B: Polymer Physics, 2003, 41(9): 936-944.
[3] HORNSBY P, WANG J, ROTHON R, JACKSON G, WILKINSON G, COSSICK K. Thermal decomposition behaviour of polyamide fire-retardant compositions containing magnesium hydroxide filler [J]. Polymer Degradation and Stability, 1996, 51(3): 235-249.
[4] ������. ������þ�ڹ�ҵ��ˮ�����е�Ӧ��[J]. ��ҵˮ����, 2000, 20(2): 1-4.
GUO Ru-xin. The applications of magnesium hydroxide to industrial wastewater treatment [J]. Industrial Water Treatment, 2000, 20(2): 1-4.
[5] ������. þ�����������������е�Ӧ��[J]. �������뻯��, 2003, 32(3): 8-11.
GUO Ru-xin. Magnesium-agents and its application to flue gas desulfurization [J]. Sea-Lake Salt & Chemical Industry, 2003, 32(3): 8-11.
[6] κ����, �����. һ�ּ��߷�չǰ����þϵ��Ʒ�D������þ[J]. �������뻯��, 2003, 32(1): 26-29.
WEI Bing-ju, ZHANG Wan-feng. A king of development prospect magnesium product-magnesium hydroxide [J]. Sea-Lake Salt & Chemical Industry, 2003, 32(1): 26-29.
[7] ����÷, ����־. ������þ��������Ӧ�ü�����չ[J]. �������뻯��, 2006, 35(2): 15-20.
SONG Yan-mei, YI Shou-zhi. Progress on the production and application of Mg(OH)2 [J]. Sea-Lake Salt & Chemical Industry, 2006, 35(2): 15-20.
[8] ������. ʯ�ҷ�����������þ�����о�[J]. ��ҵ�뻯��, 2010, 39(6): 19-23.
WANG Lan-jun. Study on the craft of magnesium hydroxide by lime method [J]. Journal of Salt and Chemical Industry, 2010, 39(6): 19-23.
[9] ���Ǻ�, ������, ������, ������. Pitzer��ϲ�����HCl-NaCl-H2O��ϵ�ܽ��Ԥ���Ӱ��[J]. ������ѧѧ��, 2001, 17(1): 91-94.
LI Ya-hong, GAO Shi-yang, SONG Peng-sheng, XIA Shu-ping. Effects of pitzer mixing parameters on the solubility prediction of the phase system HCl-NaCl-H2O [J]. Acta Phys Chim Sin, 2001, 17(1): 91-94.
[10] ������, ������, �� ��. ��ѧ���������ֲ�(����)[M]. ����: ��ѧ��ҵ������, 2002.
LIU Guang-qi, MA Lian-xiang, LIU Jie. Physical property of chemistry & chemical engineering handbook (Inorganic volume) [M]. Beijing: Chemical Industry Press, 2002.
[11] Ҧ�ʱ�, �� ��, ��Ӣ��. ������ѧ�ֲ�[M]. �Ϻ�: �Ϻ���ѧ����������, 1985.
YAO Yun-bin, XIE Tao, GAO Ying-min. Handbook of physical chemistry [M]. Shanghai: Science & Technology Press of Shanghai, 1985.
[12] �����, ������, ¬�Ϸ�, ֣����, л ��, ������. pH ֵ��������þ����������Ӱ��[J]. ���̹���ѧ��, 2007, 25(4): 609-611.
LI Qiu-ju, LIU Hua-yan, LU Han-feng, ZHANG Min-zhu, XIE Jing, CHEN Yin-fei. Effects of pH on the growth of Mg(OH)2 crystals [J]. Journal of Materials Science & Engineering, 2007, 25(4): 609-611.
[13] PHILLIPS V A, KOLBE J L, OPPERHAUSER H. Effect of pH on the growth of Mg(OH)2 crystals in an aqueous environment at 60 �� [J]. Journal of Crystal Growth, 1977, 41(2): 228-234.
[14] �ߴ���, ����ɽ, ����ƽ, ���ٻ�. Ũ��ˮ-�Ʒ���ȡ������þ�����о�[J]. ��ҵ�뻯��, 2011, 40(1): 5-7.
GAO Chun-juan, ZHANG Yu-shan, HUANG Xi-ping, CAI Rong-hua. Study on producing magnesium hydroxide by concentrated seawater-calcium method [J]. Journal of Salt and Chemical Industry, 2011, 40(1): 5-7.
[15] ALLAL K M, DOLIGNIER J C, MARTIN G. Determination of thermodynamical data of calcium hydroxichloride [J]. Revue de l'Institute Francais du Petrole, 1997, 52(Compendex): 361-368.
[16] ALLAL K M, DOLIGNIER J C, MARTIN G. Determination of the residence time distribution of solid particles by a photometric method [J]. Chemical Engineering Research and Design, 1998, 76(5): 643-648.
[17] ALLAL K M, DOLIGNIER J C, MARTIN G. Reaction mechanism of calcium hydroxide with gaseous hydrogen chloride [J]. Revue del' Institute Francais du Petrole, 1998, 53(Compendex): 871-880.
(�༭ ��ѧ��)