
Soft-chemical synthesis and high-temperature electrochemical
characteristics of VO2
ZHOU Ke-chao(周科朝), CAO Du-meng(曹笃盟), LI Zhi-you(李志友)
State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
Received 30 August 2005; accepted 24 November 2005
Abstract: VO2 powder was synthesized by a new soft-chemical method using formaldehyde as a reductant. The influences of pyrolysis temperature and time on the phase and morphology of grains were investigated by using thermal gravimeter/differential thermal analysis(TG/DTA), X-ray diffraction(XRD), Fourier transform infrared(FTIR) spectroscopy and scanning electron microscope(SEM). The positive electrode discharge performances of Li-B/LiCl-KCl/VO2 thermal battery at 500 ℃ were studied. The results show that the product is mainly non-crystal when the precursor of VO2 is heated below 300 ℃. VO2(B) appears and transits into VO2(R) irreversibly as the heating temperature rises. The open-circle voltage of VO2 is 2.6 V (vs Li-B), and the initial discharge voltage of 100 mA/cm2 at 500 ℃ is 2.52 V. The specific capacities of 100 mA/cm2 and 200 mA/cm2 at cut-off voltage of 1.4 V are 449 A・s/g and 539 A・s/g, respectively.
Key words: VO2 powder; synthesis; electrochemical characteristics; thermal battery
1 Introduction
In 1986, LVO (lithiated vanadium oxide) was first presented by FAUL et al[1-3] as positive electrode materials for thermal battery, which is a mixture of γ-LiV2O5 and VO2, but the morphology and crystallography of VO2 in the mixture were not clear. Up to date it is known that VO2 has six phases: VO2(A), VO2(B), VO2(C), VO2(M), VO2(T) and VO2(R)[4].The metastable VO2(B) phase has a layered structure and shows good performance as a positive host for rechargeable lithium cells with aqueous electrolytes [5]. To our knowledge using VO2(B)/VO2(R) (rutile) mixed phases as positive electrode materials for thermal battery has not been reported.
The general methods for VO2 powder preparation are to heat a mixture of V2O3 (or V) and V2O5 corresponding to VO2 composition in an evacuated silica tube at 700 ℃ for 2 d [6-8], or to reduce V2O5 powder in a platinum crucible in a CO2 atmosphere[6] at 1 227 ℃ for 3 d, or to melt V2O5 in a platinum crucible at 1 350 ℃ in a nitrogen atmosphere[9]. Some milder methods to prepare VO2 powder have been developed recently, such as pyrogenation[10-12] and soft chemistry[5, 13-15], or hydro-thermal methods[4,16,17]. Sol-gel routes[5, 13, 18, 19] and laser-insured vapor-phase aggradation[20] have also been used. But these methods have several disadvantages: long reactive times and high temperature; complexity of experimental equipments and contamina- tion by impurity phases.
In this study, the authors report a novel and very simple method to prepare a fine vanadium dioxide powder by heat-treatment of the black precursor powder, which is prepared by a solution hydrolysis processing by using formaldehyde as a reductant. The influences of the reactive temperature and time on XRD, appearance of the phase and morphlogy were studied. The positive electrode discharge performances of resultant VO2(B)/ VO2(R) mixed phase at 500 ℃ have also been investi- gated.
2 Experimental
2.1 Synthesis of VO2 powder
V2O5 (analytic reagent) was slowly added into dilute hydrogen chloride solution. In the process of reaction, the solution was agitated continuously by a magnetic stirrer. A transparent yellow VOCl3 solution was formed after the V2O5 had completely dissolved.Appropriate concentrated formaldehyde was added into the above VOCl3 solution. After reactions at 80 ℃ for 24 h, a blue VOCl2 solution was formed. A bulky black gel of high viscosity was obtained after slowly vaporizing the solution at 60 ℃ by hydrolysis of VOCl2. During the hydrolysis, the pH of the solution showed acidity. The black precursor, which can easily absorb moisture in the atmosphere and change to a green color, was obtained after the gel had been desiccated in vacuum at 200 ℃ for 2 h. The precursor was then ground and processed by heat treatment in an argon atmosphere (the purity>99.999%) at different temperatures for different times.
2.2 Measurements
The product was characterized by X-ray powder diffraction carried out on a Rigaku D/max2550VB+ diffractometer with Cu Kα radiation (40 kV, 300 mA, λ=0.154 06 nm, 10?≤2θ≤80?). Thermogravimetric analysis(TGA) and differential thermal analysis(DTA) were performed by TAS100 analyzer (12.8 mg, argon, and 10 ℃/min). Morphology of the powder was studied by using a JSM-5600LV scanning electron microscopy. Fourier transform infrared(FTIR) spectroscopic measure- ments were made with a NEXUS470 instrument.
2.3 Electrochemical behaviors of VO2 at 500 ℃
The electrochemical measurements of Li-B/LiCl- KCl/VO2 (obtained at 500 ℃ and 2 h, and the mass ratio between VO2(B) and VO2(R) is about 3∶7) pellet single cell were performed at 500 ℃ and two voltage vs time curves were monitored by an x-y function recorder. The mass of positive pellet was 0.4 g (active materials 70%). The diameter of the electrodes was 17.5 mm and two pieces of molybdenum plates (thickness 1.0 mm) were used as the collector. The process has been described elsewhere in detail[21].
3 Results and discussion
Fig.1 shows the evolution of the X-ray diffraction patterns of the sample heated at various temperatures for 2 h. The precursor dried at 200 ℃ in vacuum shows a broad hump around 2θ=28?-30? without any discernible reflections, indicting the amorphous nature of the precursor. The sample heated at 300 ℃ shows the development of reflections corresponding to VO2(B), which also corresponds to a weak exothermic peak of DTA curve at 302 ℃. The reflections of VO2(B) and VO2(R) become more clearly defined on raising the temperature to 400 ℃, which indicates that the layered-structure metastable VO2(B) has begun to transform irreversibly to the thermodynamically more stable tetragonal rutile VO2(R) as the temperature increases. It is well known that at room temperature and below, the stable form of VO2 is a monoclinic distortion of the rutile structure, which undergoes a metal-insulator transition at 67 ℃ that is a result of a structure change to the normal tetragonal rutile type. The reflections corresponding to the rutile phase become more prominent as the heating temperature increases to 500 ℃, while the intensity of VO2(B) decreases. TSANG et al[13] have reported the transformation of nano- crystallite VO2(B) to rutile-type VO2(R) at 320 ℃, with a complete conversion around 500 ℃. So the VO2(B) obtained by this method shows higher thermal stability and still appears at least up to 500 ℃, which is consistent with the results obtained by GUI et al[4] from the evolution of phases of needle-like nano-crystallites VO2・H2O. The peaks become sharper with the temperature rising, indicating the degree of crystallinity of the product increases further.

Fig.1 XRD patterns of vanadium dioxide powder obtained at different heating temperatures for 2 h
Fig.2 shows the influences of heating time at 400 ℃ on the crystal structure. The resultant product is mainly amorphous because the heating time of 1h is too short to become crystallization. The crystal reaction takes place as the heating time extends to 2 h, in which both VO2(B) phase and VO2(R) phase are obtained. Although the phases are the same as those of 2 h as further prolong time, the reflections and intensity of VO2(B) and VO2(R) become sharper and swell, respectively, indicating the crystal degree of product further increases as the heating time postpones.
To understand the evolution of phases upon thermal treatment, the precursor powder was subjected to thermogavimetric analysis(TGA) and differential thermal analysis(DTA) in a flowing argon atmosphere (Fig.3). The DTA curve shows a strong, broad endothermic peak around 94 ℃, indicating the temperature of the loss and evaporation of free water absorbed by the precursor powder exposed to air before the examination, which corresponds to a mass loss of about 10% of the TGA. A mass of about 20% corresponds to the loss of absorbing and bound water between 100-350 ℃. The two weak exothermic peaks around 244 ℃ and 302 ℃ may correspond to the transformation to VO2(B) and VO2(R), respectively. But, because no sharp exothermic peaks are observed, these transformations may occur over a wide temperature range. These phase transformation temperature is consistent with the previous literatures[5, 15]. The mass shows no apparent change after 350 ℃.

Fig.2 XRD patterns of vanadium dioxide powder obtained by different heating time at 400 ℃

Fig.3 TG and DTA curves of precursor of VO2
The FT-IR spectra of V2O5 and products obtained at different temperatures are compared in Fig.4. The characteristic lines of V2O5 are at 1 020 cm-1, 826 cm-1,597 cm-1, 477 cm-1, of which the line at 1 020 cm-1 originates from the shortest ν(V== O)(0.158 nm) bond of V2O5 and disappears quickly in the course of phase transition[5]. The other lines result from the ν(V―O―V) vibration. As the ionic radius of V (Ⅳ) (0.060nm) is larger than that of V (Ⅴ) (0.059nm), the V―O bond length increases with the consequent softening of the vibration. The comparison of the IR spectra of VO2 with V2O5 shows a gradual shift to lower wave numbers and a varying intensity as the vanadium ionic radius becomes larger. This behavior may be related to the increased weakness of the surface V==O bonds and to the decreased oxidation state at the surface[22]. The intense line at 826 cm-1 of V2O5 is not observed in the spectra of VO2. The line at 1 620 cm-1 of precursor and resultant product at 400 ℃ for 2 h stems from the δ(O―H) bond, which is in accordance with a slight loss of mass of the TGA curve above 400 ℃ and disappears with increasing temperature and prolonging time, indicating the wet of precursor has been completely decomposed.
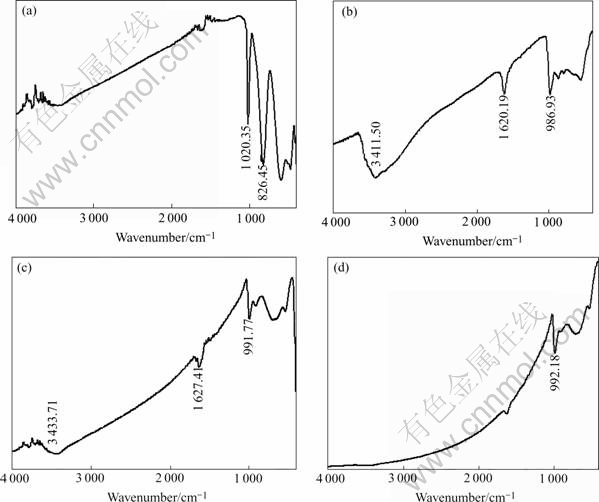
Fig.4 FTIR spectra of V2O5 and products obtained at different heating temperatures: (a) V2O5; (b) Precursor; (c) 400 ℃, 2 h; (d) 500 ℃, 2 h
Fig.5 shows the SEM photographs of the precursor and sample obtained at 500 ℃ for 2 h. The precursor is bulky. The particle size of precursor is far larger than 5 μm and consists of small and compact reuniting powder granules. This indicates that the sintering does not happen at 200 ℃ yet. The smaller grains in the sample obtained at 500 ℃ for 2 h loosely combine and the spacing between the grains is larger.
The discharge plots of VO2(B)/VO2(R) mixed phase material at different current densities at 500 ℃ are shown in Fig.6. The open-circle voltage of the single cell
of Li-B/LiCl-KCl/VO2 is 2.6 V. During the discharge process the transient voltage pinnacle does not exist in the beginning, which is very important to a thermal battery. About 9.1 min and 5.5 min can be achieved at the cut-off voltage of 1.4 V for the current density of 100 mA/cm2 and 200 mA/cm2, respectively. Then the specific capacities are 449 A・s/g, 539 A・s/g, respectively. When the current density is 100 mA/cm2 the initial work-voltage is 2.52 V, and then the voltage begins to decrease slowly because of the influences of the ohm resistance and polarization.

Fig.5 SEM photographs of precursor(a) and sample obtained at 500 ℃ for 2 h(b)

Fig.6 Electrochemical characteristics of VO2(B)/VO2(R) composite
The structure of VO2(B)/VO2(R) mixed phase material after discharge is shown in Fig.7. The following reactions take place as the cell makes up of a VO2(B)/ VO2(R) cathode and a Li-B alloy anode:
2Li+2VO2=V2O3+Li2O (1)
Li+VO2=LiVO2 (2)

Fig.7 XRD patterns of VO2(B)/VO2(R) composite after discharge
During the discharge process Li+ transfers from the negative to the positive via electrolyte and the redox reactions happen. V4+ of the positive materials has been deoxidized to V3+. The diffractions of KCl result from the molten salt of KCl-LiCl added into the positive materials.
4 Conclusions
Formaldehyde solution has been used as a reductant to obtain VO2 powder from vanadium pentoxide. VO2(B) obtained by the soft-chemical method shows higher thermal stability even at 500 ℃ for 2 h. The open-circle voltage of the single cell of Li-B/LiCl-KCl/VO2 is 2.6 V. During the discharge process the voltage is very stable and V4+ of the cathode materials is deoxidized to V3+. When the current density is 100 mA/cm2 the initial work-voltage is 2.52 V. The specific capacities of 100 mA/cm2 and 200 mA/cm2 at cut-off voltage of 1.4 V are 449 A・s/g and 539 A・s/g, respectively.
Acknowledgements
Professor LIU Zhi-jian is gratefully acknowledged by courtesy of supplying the negative material Li-B alloy for thermal battery.
References
[1] Faul I, Golder A J. Electrochemical cell structures and materials therefore [P]. USA 4596 752, 1986-06-24.
[2] Clark A J, Mckirdy I, Ritchie A G. Lithiated vanadium oxides (LVO) and vanadium dioxide (VO2) as thermal battery cathode material [A]. Attewell A, Keily T. Power Sources 15. Research and Development in Non-Mechanical Electrical Power Sources. The 19th International Power Sources Symposium [C]. Crowborough, UK: Int Power Sources Symposium Committee, 1995. 488.
[3] Gibbard H F. High temperature, high pulse power lithium batteries [J]. J Power Sources, 1989, 26(1-2): 81-91.
[4] Gui Z, Fan R, Chen X H, WU T C. A new metastable phase of needle-like nanocrystalline VO2・H2O and phase transformation [J]. J Solid State Chemistry, 2001, 157(2): 250-254.
[5] Frédéric G, Jean-Christophe V, Jean-Raymond G. Nanocrystalline vanadium dioxide: synthesis and mid-infrared properties [J]. Optical Materials, 2000, 15(2): 111-114.
[6] Kimizuka N, Isahii M, Kawada M, KIMIZUKA N, ISHII M, KAWADA I, SAEKI M, NAKAHIRA M. Behaviour of vanadium dioxide single crystals synthesized under the various oxygen partial pressures at 1 500 K [J]. J Solid State Chemistry, 1974, 9(1): 69-77.
[7] Oppermann H, Reichelt W, Gerlach U, WOLF E, BRUCKNER W, MOLDENHAUER W, WICH I. The range of homogeneity of VO2 and the influence of the composition on the physical properties(1): The preparation of defined VO2 and the determination of its phase boundaries [J]. Physica Status Solidi (A) Applied Research, 1975, 28(2): 439-446.
[8] Brueckner W, Moldenhauer W, Wich H, WOLF E, OPPERMANN H, GERLACH U, REICHELT W. Range of homogeneity of VO2 and the influence of the composition on the physical properties [J]. Physica Status Solidi (A) Applied Research, 1975, 29(1): 63-70.
[9] Kucharczyk D, Niklewski T. Accurate X-ray determination of the lattice parameters and the thermal expansion coefficients of VO2 near the transition temperature [J]. Journal of Applied Crystallography, 1979, 12(4): 370-373.
[10] Stanley A L, Edward A T. Synthesis of vanadium oxide powders by evaporative decomposition of solutions [J]. J Am Ceram Soc, 1995, 78(1): 104-108.
[11] ZHENG Chen-mou, ZHANG Jie-li, LUO Guo-bin, YE Jian-qing, WU Ming-mei. Preparation of vanadium dioxide powders by thermolysis of a precursor at low temperature [J]. J Materials Science, 2000, 35(13): 3425-3429.
[12] ZHENG Chen-mou, ZHANG Xin-min, ZHANG Jian-hui, LIAO Kai-rong. Preparation and characterization of VO2 nanopowders [J]. J Solid State Chemistry, 2001, 156(2): 274-280.
[13] Tsang C, Manthiram A. Synthesis of nanocrystalline VO2 and its electrochemical behavior in lithium batteries [J]. J Electrochem Soc, 1997, 144(2): 520-524.
[14] Valmalette J C, Gavarri J R. High efficiency thermochromic VO2(R) resulting from the irreversible transformation of VO2(B) [J]. Materials Science and Engineering B, 1998, 54(3): 168-173.
[15] Kannan A M, Manthiram A. Synthesis and electrochemical evaluation of high capacity nanostructured VO2 cathodes [J]. Solid State Ionics, 2003, 159(3-4): 265-271.
[16] Gui Z, Fan R, Mo W, CHEN X, YANG L, ZHANG S, HU Y, WANG Z, FAN W. Precursor morphology controlled formation of rutile VO2 nanorods and their self-assembled structure [J]. Chem Mater, 2002, 14(12): 5053-5056.
[17] Douglas H, Jon Z, Christopher J W, LINDA M M, MICHAEL M J T, ROBERT C H. A new polymorph of VO2 prepared by soft chemical methods [J]. J Solid State Chemistry, 1998, 138(1): 178-182.
[18] LU Song-wei, HOU Li-song, GAN Fu-xi. Preparation and optical properties of phase-change VO2 thin films [J]. J Materials Science, 1993, 28: 2169-2177.
[19] Manivannan V, Goodenough J B. Low-temperature synthesis of rutile VO2 in aqueous solution using NH2OH・HCl as reducing agent [J]. Materials Research Bulletin, 1998, 33(9): 1353-1357.
[20] Toshiyuki O, Yasuhiro I, Kenkyu K R. Synthesis of SnO2, VO2 and V2O3 fine particles by laser-insured vapor-phase reaction [J]. J Photopolymer Science and Technology, 1997, 10(2): 211-220.
[21] LIU Zhi-jian, DUAN Wei, LI Zhi-you, HUANG Ying-hua, QU Xuan-hui, HUANG Bai-yun. Effects of cathode materials on discharge characteristics of Li-B alloy/FeS2 thermal battery [J]. Trans Nonferrous Met Soc China, 1999, 9(3): 530-534.
[22] Botto I L, Vassallo M B, Baran E J, MINELLI G. IR spectra of VO2 and V2O3 [J]. Materials Chemistry and Physics, 1997, 50(3): 267-270.
Foundation item: Project(50002015) supported by the National Natural Science Foundation of China
Corresponding author: ZOU Ke-chao; Tel: +86-731-8836264, E-mail: zhoukc2@mail.csu.edu.cn
(Edited by YUAN Sai-qian)