
Removal of Co(��) from aqueous solutions by NKC-9 strong acid resin
XIONG Chun-hua(�ܴ���), FENG yu-jie(�����), YAO Cai-ping(Ҧ��Ƽ), SHEN Chen(�� ��)
Department of Applied Chemistry, Zhejiang Gongshang University, Hangzhou 310012, China
Received 8 June 2009; accepted 25 August 2009
Abstract: A strong acidic ion exchange resin (NKC-9) was used as a new adsorbent material for the removal of Co(��) from aqueous solutions. The adsorption isotherm follows the Langmuir model. The maximum adsorption capacity of the resin for Co(��) is evaluated to be 361.0 mg/g by the Langmuir model. It is found that 0.5 mol/L HCl solution provides effectiveness of the desorption of Co(��) from the resin. The adsorption rate constants determined at 288, 298 and 308 K are 7.12��10-5, 8.51��10-5 and 9.85��10-5 s-1, respectively. The apparent activation energy (Ea) is 12.0 kJ/mol and the adsorption parameters of thermodynamic are ��H�� =16.1 kJ/mol, ��S��=163.4 J/(mol��K), ��G��298 K=-32.6 kJ/mol, respectively. The adsorption of Co(��) on the resin is found to be endothermic in nature. Column experiments show that it is possible to remove Co(��) ions from aqueous medium dynamically by NKC-9 resin.
Key words: strong acid resin; ion exchange; Co(��); adsorption isotherm
1 Introduction
During the past decades, the presence of heavy metal ions in the water environment has received extensive attention due to the toxic effect on the human beings[1]. Cobalt is a valuable metal which has a great number of applications in different industrial and medicinal fields. The rising use of cobalt increases the possibility of its pollution to the environment, resulting in a potential risk to the environment. Removal of cobalt ions from wastewater is extremely important not only for their economic value, but also for reducing their contamination to the water environment. Various treatment techniques such as chemical precipitation, membrane separation, electrochemical reduction, adsorption and ion exchange have been developed to remove heavy metals from contaminated water[2-6]. Chemical precipitation is perhaps the most widely used method. However, it has the drawbacks of difficult sludge disposal and the diminished effectiveness when treating water with low heavy metal levels[7]. Membrane separation usually involves expensive materials and high operation costs[8]. Other methods such as electrodialysis and electrochemical precipitation have also been developed. However, their applications have been limited due to the high energy consumption[9]. On the other hand, as a cost-effective method, ion exchange process normally involves low-cost materials and convenient operations, and it has been proved to be very effective for removing heavy metals from aqueous solutions, particular for treating water with low concentration of heavy metals. Many studies have shown the superiority of ion exchange resins for metal ions removal or separation from aqueous solutions[10-14]. As one of these materials, macroporous strong acid resin (NKC-9) is an excellent adsorbent because of its strong mechanical stability and high chemical resistibility. NKC-9 resin contains the functional group of (��SO3H), which possesses not only protons that can exchange with cations, but also oxygen and sulfur atoms with a high affinity for heavy metal ions that can coordinate directly with metal ions. Therefore, the adsorption ability of NKC-9 resin used for enriching metal ion may be very strong. Besides, NKC-9 resin is also inexpensive and easy to regenerate.
Hitherto, little work has been published on the application of the NKC-9 resin for the removal of cobalt. In this work, NKC-9 strong acid resin was employed as a new adsorbent material for removal of cobalt ion from aqueous solutions. The effects of experimental conditions such as pH value, metal ion concentration and temperature were discussed to determine the optimum parameters for removal of Co(��) from aqueous solution by NKC-9 resin. The experimental results may be applied to wastewater treatment and cobalt refining in hydrometallurgy.
2 Experimental
2.1 Materials and instrument
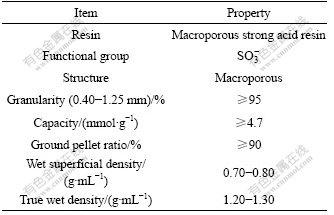
NKC-9 strong acid resin was provided by the Chemical Plant of Nankai University of China and its properties are shown in Table 1. Standard solution of Co(��) was prepared by dissolving CoSO4��7H2O (AR) in purified water. The buffer solution with pH 1-6 was prepared from HCl-KCl and HAc-NaAc, respectively. Other chemicals used were of analytical reagent grade. Co(��) ion concentration was determined with a flame atomic absorption spectrophotometer (M6 Thermo). DELTA 320 pH meter was used for measuring pH. The sample was shaken in the DSHZ-300A constant- temperature shaking machine.
Table 1 General description and properties of adsorbent
2.2 Experimental method
2.2.1 Batch studies
Batch adsorption experiments were conducted by placing a desired amount of treated resin in a 100 mL conical flask containing solutions with various concentrations of metal ions. The sample pH was adjusted to the desired value with buffer solution. The flasks were agitated at 100 r/min in a DSHZ-300A shaking machine at the ambient temperature to reach the equilibrium. Aliquot samples were taken from the flask at appropriate time intervals until adsorption equilibrium. The residual concentration of the metal ions in the aqueous phases was determined by a flame atomic absorption spectrophotometer (lamp current 7.5 mA, slit width 0.5 nm and wavelength 240.7 nm). The adsorption capacity (qe) and distribution coefficient (D) were calculated with the following formulas, respectively:
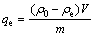 (1)
(1)
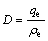 (2)
(2)
where ��0 and ��e are the initial and equilibrium concentrations of Co(��) in solution, V is the total volume of solution and m is the resin mass.
2.2.2 Desorption studies
Desorption of metal ions was performed by mixing Co(��) loaded resin and HCl eluent solution of different concentrations in a conical flask, stirring under 100 r/min at 298 K for 24 h. The final metal ion concentrations in the aqueous phase were similarly analyzed as described above. The desorption ratio (E) was calculated as follows:
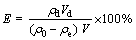 (3)
(3)
where ��d is the concentration of the solutes in the desorption solutions; and Vd is the volume of the desorption solution.
2.2.3 Column studies
The fixed-bed experiments were carried out in a water-jacketed glass column with an inner diameter of 3.0 mm and a full length of 200 mm. An aliquot of the fresh NKC-9 resin (300 mg) was packed into the column. The aqueous solution with 0.165 mg/mL Co(��) ions was then fed to the top of the bed at a flow rate of 0.27 mL/min until the breakthrough curve was completed. The samples in the outlet were taken at the preset time intervals and the concentrations of Co(��) ions were similarly determined as above. In addition, dynamic desorption procedures were also carried out. With respect to the desorption of Co(��) from the resin, the best concentration of the eluent solution selected in the batch desorption experiments was used.
3 Results and discussion
3.1 Effect of pH on distribution coefficient (D)
There are several factors that influence the uptake of metal ions by the adsorbent. Among these factors, the pH of the sample solution is the most important parameter for the effective uptake of metal ions. In this work, the effect of the pH of the sample solution was evaluated in a pH range of 1-6. As shown in Fig.1, the maximum distribution coefficient was obtained when pH is 4 with HAc-NaAc and it decreased by either raising or lowering the pH. The low distribution coefficient of the Co(��) ion at low pH may result from the low retention efficiency of the adsorbent due to the occupation of the active sites of the strong acid ion-exchanger by proton, whereas the hydrolysis of Co(��) at high pH probably accounts for the decrease of the absorbance of the Co(��) ion due to the diminution of free ions[15]. Thus, all the following experiments were carried out at pH 4.
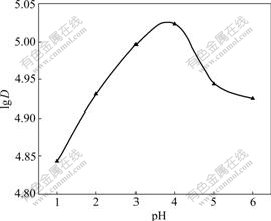
Fig.1 Effect of pH on distribution coefficient of Co(��) (Resin 15.0 mg, T=298 K, ��0=10/30 mg/mL)
3.2 Adsorption isotherm curve
The parameters of equilibrium isotherms often provide some insight into the sorption mechanism of the adsorbent. There are many equations for analyzing experimental adsorption equilibrium data. In this work, the experimental results obtained for the adsorption of Co(��) on NKC-9 resin under the optimum pH 4 and oscillation frequency 100 r/min were simulated by the Langmuir and Freundlich isotherm models[16-17], respectively:
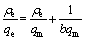 (4)
(4)
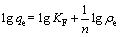 (5)
(5)
where qe is the equilibrium sorption amount; ��e is the equilibrium concentration; qm is the maximum adsorption capacity of Langmuir; b is the Langmuir constants; n is a constant indicating the Freundlich isotherm curvature; and KF is the Freundlich sorption coefficient.
The results are shown in Fig.2 and Table 2. It was found that the Langmuir model fitted the results better than the Freundlich model with the R2 values greater than 0.98 (Table 2). This suggests that the adsorption of Co(��) ions by NKC-9 resin is of monolayer-type and agrees with the observation that the metal ion adsorption from an aqueous solution usually forms a layer on the adsorbent surface.
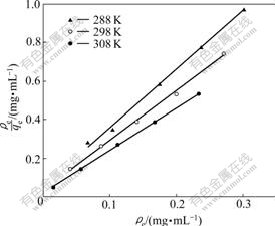
Fig.2 Langmuir isotherm curve (Resin 15.0 mg)
Table 2 Linearity relation of ��e/qe and ��e

3.3 Effect of contact time and determination of sorption rate constant
Fig.3 shows the time-dependent behaviors of the removal of Co(��) from aqueous solution by NKC-9 resin. It is clear that the uptake of Co(��) increased as the contact time increased. The removal amount of Co(��) ions increased rapidly during the first a few hours, and then increased slowly until the equilibrium state was reached. The equilibrium for the removal of Co(��) was reached after 11 h. A further increase in contact time had a negligible effect on the removal amount. The initial adsorption rate was very high, and this may be due to the greater number of resin sites available for the adsorption of metal ions. With the remaining vacant surface sites decreasing, the adsorption rate slowed down due to the formation of repulsive forces among the metals on the solid surface and in the liquid phase.
According to the Brykina method[18], the sorption rate constant k can be calculated:
-ln(1-F)=kt+B, F=qt/qe (6)
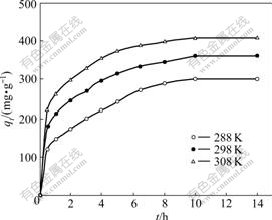
Fig.3 Effect of contact time on adsorption (��0=20/60 mg/mL, resin 30.0 mg)
where qt is the sorption capacity at time t and qe is the sorption capacity in equilibrium. The sorption rate constant can be found from the slope of the straight line (Fig.4). The calculated sorption rate constants (k) at 288, 298 and 308 K are 7.12��10-5, 8.51��10-5, 9.85��10-5 s-1, respectively.
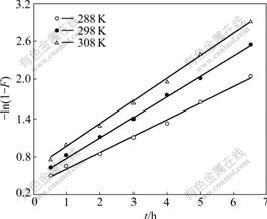
Fig.4 Determination of adsorption rate constant (��0=20/60 mg/mL, resin 30.0 mg)
According to the Arrhenius formula:
lg k=�CEa/(2.30RT)+lg A (7)
the slope of straight line, which was attained by plotting lg k versus 1/T (Fig.5), and calculated by linear fitting, yields the apparent activation energy of Ea=12.0 kJ/mol. It can be seen from the rate constant that the adsorption speed accelerates when the temperature rises within the scope of experimental temperature.
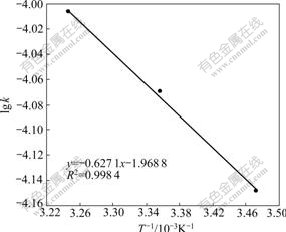
Fig.5 Relationship between lg k and 1/T (��0=20/60 mg/mL, resin 30.0 mg)
3.4 Effect of temperature on distribution ratio and determination of thermodynamic parameters
The effect of temperature on the adsorption of Co(��) by resin was studied over the range of temperature from 288 K to 308 K. The result shown in Fig.6 obviously indicates that it is favorable for the adsorption with the temperature going up, which implies that the adsorption process is an endothermic process[19]. The van Hoff equation[20] given below can be used to calculate the enthalpy changes associated with the adsorption process of the metal ions:
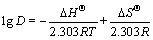 (8)
(8)
where R is the universal gas constant; D is the distribution coefficient; and T is the absolute temperature. The plot of lg D versus 1/T gives a straight line, from which ��H�� (the enthalpy variation) and ��S�� (the entropy variation) are deduced from the slope and intercept of the line, respectively. And the free energy variation, ��G��, was calculated from
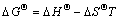 (9)
(9)
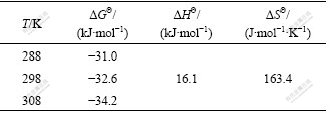
The thermodynamic parameters of the sorption of Co(��) were calculated and the results are given in Table 3. The positive values of ��H�� indicate the endothermic characteristic of the solid phase extraction and sorption process, while the negative value of ��G�� indicates the spontaneous nature of Co(��) sorption. The positive entropy change (��S��) value corresponds to an increase in the degree of freedom of the adsorbed species.
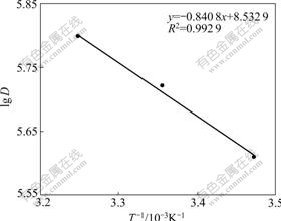
Fig.6 Relationship between lgD and 1/T (��0=10/30 mg/mL, resin 15.0 mg)
Table 3 Thermodynamic data calculated for adsorption of Co(��) on NKC-9 resin at different temperatures
3.5 Desorption studies
Adsorption of metal ions on any sorbent can be obtained by physical adsorption, chemical adsorption, ion-exchange or combination of all. If adsorption is controlled by physical bonding, then the loosely bound metal ions can be easily desorbed with distilled water. However, if the adsorption process is controlled by chemical bonding, ion exchange or combination of both, then desorption can be affected by stronger eluents like acid or alkali solution. Thus, desorption study can give a clear idea about the mechanism of adsorption and is useful in recycling of the adsorbent and recovery of metals. In this work, desorption of Co(��) ions with various concentrations of HCl solution (0.1, 0.5, 1.0, 2.0 and 3.0 mol/L) was carried out. It was found that the recovery of Co(��) was 97.7% when the HCl solution concentration was 0.1 mol/L, and the maximum recovery, of 100%, was achieved with 0.5, 1.0, 2.0 and 3.0 mol/L HCl eluent solutions, respectively, which indicates that the Co(��) adsorbed by the resin can easily be desorbed and thus can be used repeatedly in Co(��) adsorption.
3.6 Column studies
3.6.1 Dynamic adsorption curve
The fixed bed column operation allows more efficient utilization of the adsorptive capacity than the batch process. One of the main tools used in the investigation of the efficiency in adsorption columns is the breakthrough analysis. Total sorption capacity of metal ion (q0) in the column for a given feed concentration and flow rate is calculated by[21]
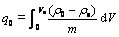 (10)
(10)
where ��0 and ��e are metal ion concentrations in the influent and effluent, respectively; m is the total mass of the sorbent loaded in the column; and Ve is the volume of metal solution passed through the column. The maximum sorption capacity value q0 was obtained to be 319.9 mg/g by graphical integration.
The Thomas model was selected for describing the breakthrough for the fixed bed sorption column since it could be written in a simple form allowing a quick and easy interpretation of the results. The Thomas model can be expressed by[21]:
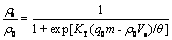 (11)
(11)
where KT is the Thomas rate constant and �� is the volumetric flow rate. The linearized form of the Thomas model is
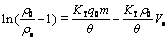 (12)
(12)
The kinetic coefficient KT and the sorption capacity of the bed, q0, can be determined from a plot of ln[(��0/��e)-1] against t at a given flow rate (Fig.7). The Thomas equation coefficients for Co(��) sorption are KT=1.67��10-2 mL/(min��mg) and q0=305.4 mg/g. The theoretical predictions based on the model parameters are compared with the observed data in Fig.8.
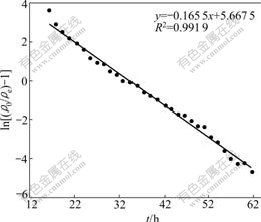
Fig.7 Linear plots of ln[(��0/��e)-1] vs t by application of Thomas model (Resin 300.0 mg, ��0=0.165 mg/mL, flow-rate 0.27 mL/min)
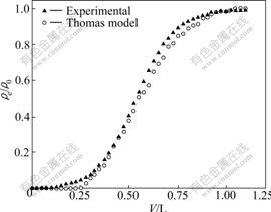
Fig.8 Experimental and predicted breakthrough curves using Thomas model for Co(��) adsorption by NKC-9 resin (Resin 300.0 mg, ��0=0.165 mg/mL, flow-rate 0.27 mL/min)
The Thomas model was found in a relatively good fitness with breakthrough curves for adsorption of Co(��) on NKC-9 resin with the high R2 value (0.991 9), and the theoretical q0 value was very close to the experimental one. Therefore, it can be concluded that the experimental data fitted well with the Thomas model.
3.6.2 Dynamic desorption curve
Once the column reached exhaustion, efficient elution of adsorbed solute from resin in column is essential to ensure the recovery of metal ions as well as the reuse of resin for repeated adsorption/desorption cycles. In this work, 0.5 mol/L HCl was used to elute Co(��). Desorption curve (Fig.9) was obtained by plotting the effluent concentration (��e) versus elution volume from the column at a flow rate of 0.2 mL/min. Less sorption flow rate indicates that the volume of elution is less which helps for easy handling and the concentration is high so that economical metal recovery is possible.
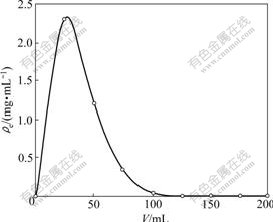
Fig.9 Dynamic desorption curve (flow-rate 0.20 mL/min)
It was observed that the total volume of eluent was 125 mL, and further desorption was negligible. Therefore, 125 mL 0.5 mol/L HCl could help for easy handling and recovering of Co(��) absorbed by 300 mg NKC-9 resin.
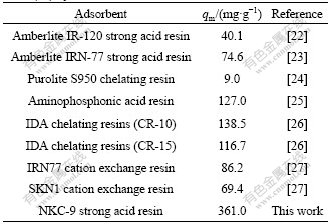
3.7 Comparison of maximum capacity of NKC-9 resin with some other adsorbents
A comparison of the maximum capacity of NKC-9 resin with that of some other adsorbents reported in literature is given in Table 4. Differences of metal uptake are due to the properties of adsorbents such as structure, functional groups and surface area.
Table 4 Comparison of maximum adsorption capacities (mg/g) of Co(��) by various adsorbents
4 Conclusions
1) Co(��) adsorption by NKC-9 resin is highly dependent on pH. In addition, initial Co(��) concentra- tion influences on the adsorption process.
2) The maximum adsorption capacity of the resin for Co(��) for the Langmuir model is evaluated to be 361.0 mg/g. And it is found that 0.5 mol/L HCl solution provides effectiveness of the desorption of Co(��) from NKC-9 resin.
3) Isotherm studies show that the adsorption process of NKC-9 resin for Co(��) follows the Langmuir model. This suggests that the adsorption of Co(��) by NKC-9 resin is of monolayer-type and agrees with the observation that the metal ion adsorption from an aqueous solution usually forms a layer on the adsorbent surface.
4) Thermodynamic parameters including standard enthalpy (?H��), standard entropy (?S��) and standard free energy (?G��) indicate that the adsorption of Co(��) on NKC-9 resin is a spontaneous reaction and is endothermic in nature.
5) Column experiments show that it is possible to remove Co(��) ions from aqueous medium dynamically.
References
[1] PEHLIVAN E, CETIN S. Sorption of Cr(��) ions on two Lewatit-anion exchange resins and their quantitative determination using UV�Cvisible spectrophotometer [J]. Journal of Hazardous Materials, 2009, 163 (1): 448-453.
[2] MAUCHAUFFEE S, MEUX E. Use of sodium decanoate for selective precipitation of metals contained in industrial wastewater [J]. Chemosphere, 2007, 69(5): 763-768.
[3] MELITA L, POPESCU M. Removal of Cr(��) from industrial water effluents and surface waters using activated composite membranes [J]. Journal of Membrane Science, 2008, 312(1/2): 157-162.
[4] AHMED BASHA C, BHADRINARAYANA N S, ANANTHARAMAN N, MEERA SHERIFFA BEGUM K M. Heavy metal removal from copper smelting effluent using electrochemical cylindrical flow reactor [J]. Journal of Hazardous Materials, 2008, 152(1): 71-78.
[5] SHU Zeng-nian, XlONG Chun-hua, WANG Xu. Adsorption behavior and mechanism of amino methylene phosphonic acid resin for Ag(��) [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(3): 700-704.
[6] XIONG Chun-hua, YAO Cai-ping. Adsorption behavior of gel-type weak acid resin (110-H) for Pb2+ [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(5): 1290-1294.
[7] ABROWSKI A D, HUBICKI Z, PODKO��SCIELNY P, ROBENS E. Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method [J]. Chemosphere, 2004, 56(2): 91-106.
[8] ERDEM E, KARAPINAR N, DONAT R. The removal of heavy metal cations by natural zeolites [J]. Journal of Colloid and Interface Science, 2004, 280(2): 309-314.
[9] KURNIAWAN T A, CHAN G Y S, LO W H, BABEL S. Physico-chemical treatment techniques for wastewater laden with heavy metals [J]. Chemical Engineering Journal, 2006, 118(1/2): 83-98.
[10] XIONG Chun-hua, YAO Cai-ping. Study on the adsorption of cadmium(��) from aqueous solution by NKC-9 resin [J]. Journal of Hazardous Materials, 2009, 166(2/3): 815-820.
[11] XIONG Chun-hua, YAO Cai-ping, WANG Li, KE Jia-jun. Adsorption behavior of Cd(��) from aqueous solutions onto gel-type weak acid resin [J]. Hydrometallurgy, 2009, 98(3/4): 318-324.
[12] DIZGE N, KESKINLER B, BARLAS H. Sorption of Ni(��) ions from aqueous solution by Lewatit cation-exchange resin [J]. Journal of Hazardous Materials, 2009, 167(1/3): 915-926.
[13] HAMDAOUI O. Removal of copper(��) from aqueous phase by Purolite C100-MB cation exchange resin in fixed bed columns: Modeling [J]. Journal of Hazardous Materials, 2009, 161(2/3): 737-746.
[14] ABO-FARHA S A, ABDEL-AAL A Y, ASHOUR I A, GARAMON S E. Removal of some heavy metal cations by synthetic resin purolite C100 [J]. Journal of Hazardous Materials, 2009, 169(1/3): 190-194.
[15] XIONG Chun-hua, YAO Cai-ping. Preparation and application of acrylic acid grafted polytetrafluoroethylene fiber as a weak acid cation exchanger for adsorption of Er(��) [J]. Journal of Hazardous Materials, 2009, 170(2/3): 1125-1132.
[16] LANGMUIR I. Adsorption of gases on plain surface of glass mica platinum [J]. Journal of the American Chemical Society, 1918, 40: 1361-1403.
[17] MUBASHIR H N, RAZIYA N, KALSOOM A, MUHAMMAD A H, AHMAD M K. Efficacy of modified distillation sludge of rose (Rosa centifolia) petals for lead(��) and zinc(��) removal from aqueous solutions [J]. Journal of Hazardous Materials, 2007, 147(3): 1006-1014.
[18] SHU Qin-zhang, WAN Guo-hou. Adsorption behavior of Pb(��) on montmorillonite [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2008, 320(1/3): 92-97.
[19] XIONG Chun-hua, YAO Cai-ping, WU Xiang-mei. Adsorption of rhenium(��) on 4-amino-1,2,4-triazole resin [J]. Hydrometallurgy, 2008, 90(2/4): 221-226.
[20] LYUBCHIK S I, LYUBCHIK A I, GALUSHKO O L, TIKHONOVA L P, VITAL J, FONSECA I M, LYUBCHIK S B. Kinetics and thermodynamics of the Cr(��) adsorption on the activated carbon from co-mingled wastes [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2004, 242(1/3): 151-158.
[21] TABAKCI M, YILMAZ M. Sorption characteristics of Cu(��) ions onto silica gel-immobilized calix[4]arene polymer in aqueous solutions: Batch and column studies [J]. Journal of Hazardous Materials, 2008, 151(2/3): 331-338.
[22] JUANG R S, LIN S H, WANG T Y. Removal of metal ions from the complexed solutions in fixed bed using a strong-acid ion exchange resin [J]. Chemosphere, 2003, 53(10): 1221-1228.
[23] KANG S Y, LEE J U, MOON S H, KIM K W. Competitive adsorption characteristics of Co2+, Ni2+, and Cr3+ by IRN-77 cation exchange resin in synthesized wastewater [J]. Chemosphere, 2004, 56(2): 141-147.
[24] DEEPATANA A, VALIX M. Recovery of nickel and cobalt from organic acid complexes: Adsorption mechanisms of metal-organic complexes onto aminophosphonate chelating resin [J]. Journal of Hazardous Materials, 2006, 137(2): 925-933.
[25] SHI Lin-mei, MO Jian-jun, XIONG Chun-hua. Study on sorption of Aminophosphonic acid resin for cobalt [J]. Nonferrous Metals, 2003, 155(1): 86-89. (in Chinese)
[26] DRAGAN E S, DINU M V, LISA G, TROCHIMCZUK A W. Study on metal complexes of chelating resins bearing iminodiacetate groups [J]. European Polymer Journal, 2009, 45(7): 2119-2130.
[27] RENGARAJ S, MOON S H. Kinetics of adsorption of Co(��) removal from water and wastewater by ion exchange resins [J]. Water Research, 2002, 36(7): 1783-1793.
Foundation item: Project(2008F70059) supported by Zhejiang Provincial Scientific and Technological Research Planning, China; Project(Z200907459) supported by the Key Grant of Education Department of Zhejiang Province, China
Corresponding author: XIONG Chun-hua; Tel: +86-571-88932083; E-mail: xiongch@163.com
DOI: 10.1016/S1003-6326(09)60269-7
(Edited by YANG Bing)