五价锑在脱除铜电解液中砷、锑、铋杂质中的作用
来源期刊:中国有色金属学报(英文版)2014年第1期
论文作者:肖发新 曹 岛 毛建伟 申晓妮 任凤章
文章页码:271 - 278
关键词:五价锑;As;Bi;锑酸盐;铜电解液;脱除;净化
Key words:Sb(V); As; Bi; antimonate; copper electrolyte; removal; purification
摘 要:通过向含有45 g/L Cu2+、185 g/L H2SO4、10 g/L As和0.5 g/L Bi的铜电解液中加入Sb(V),研究五价锑对铜电解液中砷、锑、铋杂质脱除作用机理。过滤电解液,采用化学分析、SEM、TEM、EDS、XRD、FTIR等方法对沉淀渣的结构形貌和成分进行表征。结果表明,沉淀渣呈尺寸为50~200 μm的不规则块状,其化学成分主要为砷、锑、铋和氧。红外光谱检测表明,沉淀渣主要特征官能团为As―O―As、As―O―Sb、Sb―O―Bi、Sb―O―Sb和Bi―O―Bi。X射线衍射和电子衍射检测结果表明,沉淀渣由AsSbO4、BiSbO4和Bi3SbO7组成。锑酸盐的生成是五价锑脱除铜电解液中砷、锑、铋杂质的主要原因。
Abstract: The function mechanism of Sb(V) in As, Sb and Bi impurities removal from copper electrolyte was investigated by adding Sb(V) ion in a synthetic copper electrolyte containing 45 g/L Cu2+, 185 g/L H2SO4, 10 g/L As and 0.5 g/L Bi. The electrolyte was filtered, and the precipitate structure, morphology and composition were characterized by chemical analysis, SEM, TEM, EDS, XRD and FTIR. The results show that the precipitate is in the shape of many irregular lumps with size of 50-200 μm, and it mainly consists of As, Sb, Bi and O elements. The main characteristic bands in the FTIR spectra of the precipitate are As―O―As, As―O―Sb, Sb―O―Bi, Sb―O―Sb and Bi―O―Bi. The precipitate is the mixture of microcrystalline of AsSbO4, BiSbO4 and Bi3SbO7 by XRD and electronic diffraction. The removal of As, Sb and Bi impurities by Sb(V) ion can be mainly ascribed to the formation of antimonate in copper electrolytes.
Trans. Nonferrous Met. Soc. China 23(2013) 271-278
Fa-xin XIAO1,2, Dao CAO1, Jian-wei MAO1, Xiao-ni SHEN1, Feng-zhang REN1
1. School of Materials Science and Engineering, Henan University of Science and Technology, Luoyang 471003, China;
2. Henan Key Laboratory of Advanced Nonferrous Metal Materials, Henan University of Science and Technology, Luoyang 471003, China
Received 9 January 2012; accepted 20 May 2012
Abstract: The function mechanism of Sb(V) in As, Sb and Bi impurities removal from copper electrolyte was investigated by adding Sb(V) ion in a synthetic copper electrolyte containing 45 g/L Cu2+, 185 g/L H2SO4, 10 g/L As and 0.5 g/L Bi. The electrolyte was filtered, and the precipitate structure, morphology and composition were characterized by chemical analysis, SEM, TEM, EDS, XRD and FTIR. The results show that the precipitate is in the shape of many irregular lumps with size of 50-200 μm, and it mainly consists of As, Sb, Bi and O elements. The main characteristic bands in the FTIR spectra of the precipitate are As―O―As, As―O―Sb, Sb―O―Bi, Sb―O―Sb and Bi―O―Bi. The precipitate is the mixture of microcrystalline of AsSbO4, BiSbO4 and Bi3SbO7 by XRD and electronic diffraction. The removal of As, Sb and Bi impurities by Sb(V) ion can be mainly ascribed to the formation of antimonate in copper electrolytes.
Key words: Sb(V); As; Bi; antimonate; copper electrolyte; removal; purification
1 Introduction
In the smelting and refining of copper, it is difficult to remove the impurities completely by the pyrometallurgical process [1,2]. Thus, copper anode needs to be electro-refined to meet the requirement for purity in most application fields. During the electrolytic refining of copper, some of the impurities contained in the anode dissolve into the electrolyte and accumulate in the form of metal ions. These impurities typically include antimony, arsenic and bismuth. Since their standard reduction potentials are very close to that of copper, these impurities are liable to be electrodeposited at cathode. The practice of purifying the electrolyte so as to keep their concentrations constantly below the prescribed levels proves to be very important to achieve the desired quality of cathode copper.
Up to now, most major copper refineries still employ electrowinning to treat electrolyte in their daily operation, while this conventional method has a number of disadvantages, such as difficult materials handling, energy consumption, formation of toxic arsine gas, and loss of high value copper in a low recycle product [3,4]. It was reported that a part of the impurities As, Sb and Bi dissolved from the anode can spontaneously precipitate from the electrolyte to the anode slimes or residue during the electrorefining of copper [1,5,6]. The novel purification technology of copper electrolytes based on these co-precipitation reactions among As, Sb and Bi impurities in bath has been put forward [7] and well applied in copper smelting industries [8]. However, the purification mechanism has not been fully understood up to now.
It was generally regarded that the arsenate formed from As(V), Sb(III) and Bi(III) ions was the main reason for the removal of these impurities [1,8-10]. Although the formation of arsenate well explains the impurities removal from electrolyte, the contents of As, Sb, and Bi in the anode slime do not always agree with the stoichiometry of the arsenates [11]. Furthermore, it was reported that the Sb content of larox residue was remarkably higher than those of As and Bi [12], which indicates that Sb element plays an important role in impurities removal from electrolyte, and it cannot be simply explained by formation of arsenate. Besides, it was found that As(III) can react with Sb(III) and Sb(V) to form antimony arsantimonate by adding copper arsenite in the electrolyte, resulting in the decline of Sb concentration in the electrolyte [6,13]. Nevertheless, in an accidental experiment, it was found that the precipitation reactions among As, Sb and Bi occurred even in the absence of Sb(III) ion in copper electrolyte. Obviously, this phenomenon cannot be adequately explained by arsenates or arsenato antimonates.
In this work, the purification experiments of synthetic electrolyte containing Cu2+, H2SO4, As(III), As(V), Sb(V) and Bi(III) were carried out to study the effects of Sb(V) ions on the removal of As, Sb and Bi. Also the removal mechanism of As, Sb and Bi by Sb(V) ion in the co-precipitation reactions was investigated by characterizing the sediment structure.
2 Experimental
First, As(III), As(V), Sb(V) and Bi(III) stock solutions were prepared. The sample of As2O3 was dissolved in water under heating and stirring to obtain the As(III) stock solutions [12]. The sample of Bi2O3 was added in concentrated sulfuric acid, stirred and heated to prepare the Bi(III) stock solution. The As(V) stock solution was prepared by adding hydrogen peroxide to the As(III) stock solution under stirring. Besides, a sample of Sb2O3 was firstly dissolved in concentrated sulfuric acid and diluted with water, and then oxidized with hydrogen peroxide to obtain the Sb(V) stock solution. A small amount of hydrochloric acid was added to promote Sb2O3 dissolving. The excess H2O2 was decomposed by boiling the solution for about 30 min. The total content of the elements was determined by atomic absorption spectroscopy (AA700, American PerkinE Corp).
The electrolyte used in the experiments contained the following basic components: 44-46 g/L Cu2+, 180- 185 g/L H2SO4, and As, Sb and Bi impurities. The contents of As and Bi in the synthetic electrolytes were adjusted according to the commercial electrolyte [6] and the initial concentrations of the synthetic electrolytes are listed in Table 1. The purification of the electrolyte was carried out by heating the electrolyte to 65 °C under stirring at 300 r/min for 4 h. The purified electrolyte was then filtered to collect the precipitate for structural characterization. The removal amount of impurity was calculated according to the impurities concentrations before and after purification experiments.
Table 1 Initial compositions of synthetic copper electrolytes

The composition of the precipitate was determined by chemical methods and energy dispersive spectrometry (EDS) (Genesis 60S, EDAX Company). Microscopic observations were carried out by transmission electron microscopy (JEM-2100, Japanese Electron Company) and scanning electron microscopy (JSM-5610LV, JEOL Corp). The FTIR spectrum was obtained by the KBr disk method with a VERTEX70 (JANPAN) spectrophotometer operating in the wavenumber range of 4000-400 cm-1. The X-ray diffraction (XRD) patterns were recorded on a diffractometer (D8 ADVANCE, German Electron Company) with Cu Kα X-ray radiation at 35 kV and 20 mA.
3 Results and discussion
3.1 Influence of initial Sb(V) ion concentration on removal of As, Sb and Bi
The purification experiments of electrolytes were carried out according to the above experimental procedure and the removal amount of As, Sb and Bi was calculated. The influence of initial Sb(V) ion concentration on the removal amount and removal rate of Sb is shown in Fig. 1. The influence of initial Sb(V) concentration on the removal amounts of As and Bi is shown in Fig. 2.

Fig. 1 Influence of initial Sb(V) concentration on removal amount and removal rate of Sb

Fig. 2 Influence of initial Sb(V) concentration on removal amounts of As and Bi
As seen in Figs. 1 and 2, the precipitation reactions occur only when the initial Sb(V) ion concentration reaches 0.8 g/L. The removal amounts of As, Sb and Bi increase with increase of initial Sb(V) concentration, and remain nearly constant with the further increase in Sb(V) ion concentration after it exceeds 1.2 g/L. The removal rates of Sb and Bi reach 76.83% and 79.2%, respectively, at Sb(V) ion concentration of 1.2 g/L. The purification time was prolonged to obtain the higher impurities removal rate. It was found that the removal rates of As, Sb and Bi reached 15.26%, 88.61% and 91.01%, respectively after stirring for 16 h under the same conditions. This result indicates that the increase of purification time can improve the purification efficiency. Obviously, Sb(V) ion has a distinct effect on the removal of As, Sb and Bi from electrolytes. Similar result was found by WANG et al [5]. They proposed that the increase of the oxidation rate of antimony from Sb(III) to Sb(V) in copper electrolyte can improve the percentages of the impurities As, Sb and Bi dissolved from the copper anode and then the impurities deposited into the anode slimes increased.
Precipitate structural characterization was performed to identify the removal mechanism of As, Sb and Bi impurities by Sb(V) ion. Meanwhile, in order to compare and clarify the structure, the structure of industrial filter residue obtained from the Larox filter in the copper electrolysis plant of Hubei Daye Copper Smelter of China was also characterized in this work.
3.2 Precipitate structure characterization
The SEM images and EDS patterns of precipitates formed in the synthetic electrolytes containing 10 g/L As, 1.2 g/L Sb(V) and 0.5 g/L Bi(III) are shown in Fig. 3.

Fig. 3 SEM images (a), (b) and EDS analysis (c) of synthetic precipitate
From Fig. 3, it can be clearly seen that the synthetic precipitate is in the shape of many irregular lumps with size of 50-200 μm, and it mainly consists of As, Sb, Bi and O elements. The precipitate can deposit quickly at the bottom of the container because of the large size, which accelerates the precipitate removal from copper commercial electrolytes. The exact compositions of the synthetic precipitate and industrial filter residue obtained from the commercial electrolyte of Daye Copper Smelter were determined by chemical analysis, and the results are listed in Table 2.
Table 2 Compositions of synthetic precipitate and industrial filter residue
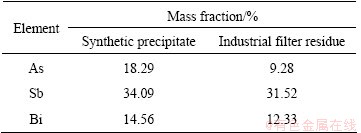
From Table 2, it can be found that the total content of As, Sb and Bi in synthetic precipitate is 66.94%, which indicates that As, Sb and Bi are the key elements in the synthetic precipitate. The mole ratio n(As):n(Sb):n(Bi) is approximately 3.5:4.0:1 in the synthetic precipitate. The contents of Sb and Bi in the industrial filter residue are approximately close to those of synthetic precipitate, while the content of As is much lower. The difference in the contents between the precipitate and industrial filter residue may be caused by the higher concentration of initial As (10 g/L) contained in the synthetic electrolytes. Besides, the existence of Sb(III), Pb, Sn, Zn and Fe elements, which are not present in the synthetic electrolyte, may also result in the difference.
The XRD measurement of the synthetic precipitate and the industrial filter residue was performed, and the results are shown in Fig. 4.

Fig. 4 XRD patterns of synthetic precipitate and industrial filter residue
XRD patterns shown in Fig. 4 reveal that although the crystal performance of the precipitate is not so good, some main crystalline diffraction reflections are detected at the main diffraction angles of 20.9°, 25.49°, 26.67°, 27.83°, 28.88°, 29.54°, 32.40°, 33.32°, 33.95°, 39.82°, 42.81°, 43.85°, 46.06°, 53.48° and 57.14°. XRD patterns of AsSbO4, BiSbO4 and Bi3SbO7 were identified according to the PDF pattern numbers 73-0875, 86-0126 and 45-0925, respectively. The same phases were also found in the XRD pattern of the industrial filter residue.
The TEM observation was carried out to get the further information about the structure of precipitate, and the results are shown in Fig. 5.
It can be seen from Figs. 5(a), (c) and (e) that the precipitate is composed of dendritic and lump particles. The size distributions of the lump particles differ substantially and range from dozens of microns to parts of a micron. The diameter of the dendritic particles is 10-20 nm and its length reaches 100 nm. These particles can agglomerate together to form large irregular lump precipitate as shown in SEM images (Fig. 3). Electron diffraction patterns shown in Figs. 5(b), (d) and (f) were taken from regions in Figs. 5(a), (c) and (e), respectively. Electron diffraction patterns indicate that the precipitate contains crystalline phases. The inter-planar distances of Figs. 5(b) and (d) were calculated according to the basic formula of electron diffraction and the values of R which is the distance between the central and diffraction spots that measured in electron diffraction images, and the results are listed in Tables 3 and 4, respectively.
In Table 3, the calculated inter-planar distances d1, d2, d3 and d4 are 3.9364, 2.0379, 1.9178 and 1.2366 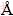 , respectively, which are close to the theoretical values of the crystalline AsSbO4 based on PDF 73-0875. Therefore, this phase of Fig. 5(b) can be inferred to be crystalline AsSbO4. Similarly, the precipitate of Fig. 5(c) can be inferred to be crystalline BiSbO4.
, respectively, which are close to the theoretical values of the crystalline AsSbO4 based on PDF 73-0875. Therefore, this phase of Fig. 5(b) can be inferred to be crystalline AsSbO4. Similarly, the precipitate of Fig. 5(c) can be inferred to be crystalline BiSbO4.
TEM results further confirm that the synthetic precipitate contains AsSbO4 and BiSbO4, which is consistent with the XRD results. The diffraction pattern in Fig. 5(f) displays that the diffraction points have no regular arrangement, which is too complex to be calibrated. This diffraction pattern indicates that with the proceeding of co-precipitation reactions, the grains of the precipitate form and assemble, and some of them begin to accumulate directionally.
Table 3 Inter-planar distances of crystal shown in Fig. 5(b) compared with AsSbO4

Table 4 Inter-planar distances of crystal shown in Fig. 5(d) compared with BiSbO4

Fig. 5 TEM images showing morphologies (a), (c), (e) and corresponding electron diffraction patterns (b), (d), (f) of precipitates obtained from synthetic electrolyte
The FTIR spectra of the synthetic precipitate and industrial filter residue recorded in the wavenumber of 4000-400 cm-1 are given in Fig. 6. And the FTIR spectrum of Bi2O3 recorded in the wavenumber range of 4000-400 cm-1 is shown in Fig. 7.
From Fig. 6, it can be seen that the FTIR band at 2434.19 cm-1 is due to CO2 in the air, the band at 1231.82 cm-1 is the bending vibration absorption spectrum of As―OH and Sb―OH [14,15], the band at 1061.62 cm-1 is the anti-stretching vibration absorption spectrum of As―OH [16], the band at 788.55 cm-1 is the anti-stretching vibration absorption spectrum of As―OX (X=As, Sb) [17], the band at 625.63 cm-1 is the anti-stretching vibration absorption spectrum of Sb―OH, and the band at 502.56 cm-1 is the anti-stretching vibration absorption spectrum of Sb―OY (Y=As, Sb, Bi) [14,15]. The band at 450.10 cm-1 is not only inferred as the bending vibration absorption spectrum of O―As―O [18], but also is probably in association with antimony atom [16]. Besides, it can also be inferred as the absorption spectrum of Bi―O―Bi since there is a similar band at 446.52 cm-l in the FTIR spectrum of Bi2O3, as shown in Fig. 7.
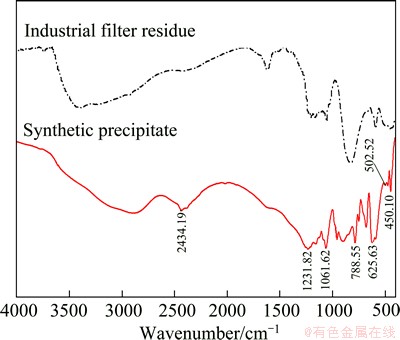
Fig. 6 FTIR spectra of synthetic precipitate and industrial filter residue
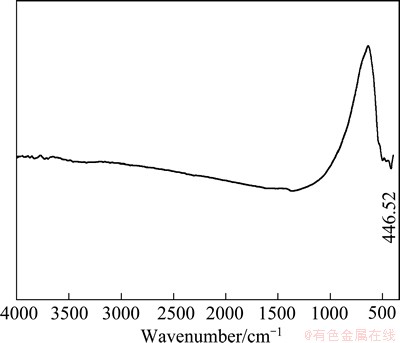
Fig. 7 FTIR spectrum of Bi2O3
Therefore, the main valence bands of As―O―As, As―O―Sb, Sb―O―Bi, Sb―O―Sb and Bi―O―Bi are associated with the structure of the synthetic precipitate. All these bands can be found in the structures of the antimonates of BiSbO4 and AsSbO4. Meanwhile, Sb―O―Bi and Bi―O―Bi were discovered in the crystal structure of Bi3SbO7 [19]. These results further confirm the results of the XRD and TEM. Similarly, the As―O―As, As―O―Sb, Sb―O―Bi, Sb―O―Sb and Bi―O―Bi bands are also found in the FTIR spectra of industrial filter residue.
3.3 Mechanism analysis
The structural analysis shows that the precipitate is the mixture of microcrystalline of AsSbO4, BiSbO4 and Bi3SbO7. It is found that As(III), Sb(V) and Bi(III) exist in the form of AsO+, HSb(OH)6 and Bi3+, respectively in the copper electrolyte according to the thermodynamic calculation [6]. Besides, during the electrorefining of anode copper, antimony dissolves as antimonious acid (HSbO2) and oxidizes to antimonic acid (HSb(OH)6) by the atmospheric oxygen dissolved in the copper refining electrolyte [20]. And Bi(III) is also discovered in the form of BiO+ in the electrolyte [8,21]. Therefore, the following reactions may occur in the process of electrolyte purification:
AsO++HSb(OH)6=AsSbO4↓+H++3H2O (1)
Bi3++HSb(OH)6=BiSbO4↓+3H++2H2O (2)
3BiO++HSb(OH)6=Bi3SbO7↓+3H++2H2O (3)
The mixture of compounds forms and deposits in the electrolyte according to reactions (1)-(3), which leads to the removal of As, Sb and Bi impurities from copper electrolyte.
As reported by WU et al [22], BiSbO4 was synthesized by a direct hydrothermal reaction of Bi(NO3)3・5H2O and Sb2O3 in NaOH solution. During the synthesis, Sb2O3 is oxidized to  in the presence of O2 and NaOH. As the reaction continues, the obtained precursors diffuse to each other, leading to the formation of nuclei and further crystal growth of the final product, which manifests that Sb(V) can combine with Bi(III) to form BiSbO4. Although the synthesis conditions are different, it verifies that BiSbO4 can be generated in the solution containing Sb(V) and Bi(III).
in the presence of O2 and NaOH. As the reaction continues, the obtained precursors diffuse to each other, leading to the formation of nuclei and further crystal growth of the final product, which manifests that Sb(V) can combine with Bi(III) to form BiSbO4. Although the synthesis conditions are different, it verifies that BiSbO4 can be generated in the solution containing Sb(V) and Bi(III).
Moreover, it was found that a large amount of AsSbO4 in the Southwire Anode Slimes is composed of small well formed individual crystals. Similar material was detected in the slimes from Huttenwerke Kaiser [23]. It was found that they precipitated in the slime layer or on the walls of the refining cells. This finding further demonstrates that antimonate can be generated in copper electrolyte.
According to the results above, Sb(V) can combine with As(III) or Bi(III) ions to form AsSbO4, BiSbO4 and Bi3SbO7 in the synthetic and commercial copper electrolytes. The formation of antimonate results in the decline of the concentrations of As, Sb and Bi impurities in electrolyte. Therefore, the main contribution of the Sb(V) ion to the removal of As, Sb and Bi impurities can be ascribed to the formation of antimonate in copper electrolytes.
When Sb(V) ion concentration is too low (0.4 g/L), the As, Sb and Bi elements exist in the form of free ion, the ion solubility product constant Ksp is less than those of AsSbO4, BiSbO4 and Bi3SbO7. Therefore, there is no precipitation in the bath and the impurities of As, Sb and Bi cannot be removed from the electrolyte. With the increase in the concentration of Sb(V) ion, the formation of antimonate based on Eqs. (4)-(6) is speeded up and the percentages of the impurities As, Sb and Bi deposited into the precipitate increase, resulting in the increase of impurities removal amount. When Sb(V) concentration exceeds 1.2 g/L, the formation of antimonate is saturated, and therefore the removal amounts of As, Sb and Bi remain nearly constant.
In order to demonstrate the role of Sb(V) ion in the impurities removal, the purification experiment was carried out by adding 1.2 g/L Sb(V) ion in commercial copper electrolyte. Before the purification experiment, a proper amount of reductant was added in the electrolyte to adjust the As(V) ion to As(III) ion. After stirring for 4 h, it is found that the concentrations of As, Sb and Bi decrease from 12.584, 0.934 and 0.608 g/L to 10.637, 0.344 and 0.252 g/L, respectively. The results further verify that the As, Sb and Bi impurities can be removed by Sb(V) ion. While in the electrolyte, arsenic and antimony are in the three and five valent states, respectively [20]. And the Sb element mainly exists in the Sb(III) ion in the commercial copper refining electrolyte [12]. Therefore, a method needs to be proposed to accelerate the oxidation rate of Sb(III) ion in order to improve the purification efficiency of copper electrolytes.
4 Conclusions
1) The As, Sb and Bi impurities can be removed by Sb(V) ion and the removal rates of Sb and Bi reach 76.83% and 79.2%,respectively in a synthetic electrolyte containing 10 g/L As, 1.2 g/L Sb(V) and 0.5 g/L Bi(III) under stirring at 65 °C for 4 h. The longer the purification time is, the higher the impurity removal rates are.
2) The precipitate, mainly consisted of As, Sb, Bi and O elements, forms in the synthetic copper electrolyte. It is in the shape of irregular lump with size of 50-200 μm which is agglomerated by fine dendritic and lumps particles. The main characteristic bands in the FTIR spectra of the precipitate are As―O―As, As―O―Sb, Sb―O―Bi, Sb―O―Sb and Bi―O―Bi.
3) The precipitate is composed of microcrystalline of AsSbO4, BiSbO4 and Bi3SbO7. The main role of Sb(V) ion in the removal of As, Sb and Bi impurities can be ascribed to the formation of antimonate in copper electrolytes.
References
[1] HOFFMANN J. The purification of copper refinery electrolyte [J]. Journal of the Minerals, Metals and Materials Society, 2004, 56(7): 30-33.
[2] WANG Shi-jie. Impurity control and removal in copper tankhouse operations [J]. Journal of the Minerals, Metals and Materials Society, 2004, 56(7): 34-37.
[3] NAVARRO P, ALGUACIL F J. Adsorption of antimony and arsenic from a copper electrorefining solution onto activated carbon [J]. Hydrometallurgy, 2002, 66(1-3): 101-105.
[4] DEORKAR N, TAVLARIDES L. A chemically bonded adsorbent for separation of antimony, copper and lead [J]. Hydrometallurgy, 1997, 46(1-2): 121-135.
[5] WANG Xue-wen, CHEN Qi-yuan, YIN Zhou-lan, WANG Ming-yu, XIAO Bing-rui, ZHANG Fan. Homogeneous precipitation of As, Sb and Bi impurities in copper electrolyte during electrorefining [J]. Hydrometallurgy, 2011, 105(3-4): 355-358.
[6] XIAO Fa-xin. Novel technology of purification of copper electrolyte and basic research [D]. Changsha: Central South University, 2008. (in Chinese)
[7] XIAO Fa-xin, ZHENG Ya-jie, WANG Yong, XU Wei, LI Chun-hua, JIAN Hong-sheng. Novel technology of purification of copper electrolyte [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(5): 1069-1074.
[8] ZHENG Ya-jie, XIAO Fa-xin, WANG Yong, LI Chun-hua, XU Wei, JIAN Hong-sheng, MA Yu-tian. Industrial experiment of copper electrolyte purification by copper arsenite [J]. Journal of Central South University of Technology, 2008, 15(2): 204-208.
[9] YANNOPOULOS J C, AGARWAL J C. Extractive metallurgy of copper: Pyrometallurgy and electrolytic refining [M]. New York: The Metallurgical Society of AIME, 1977: 512-524.
[10] KAMATH B P, MITRA A K, RADHAKRISHNAN S, SHETTY K. Electrolyte impurity control at chinchpada refinery of sterlite industries (India) limited [C]//Copper Electrorefining and Electrowinning. Montreal: Canadian Institute of Mining, Metallurgy and Petroleum, 2003: 137-150.
[11] WANG Xue-wen. Study on the mechanism of the formation and action of arsenate antimonic acid in copper electrorefining [D]. Changsha: Central South University, 2003. (in Chinese)
[12] WANG Xue-wen, CHEN Qi-yuan, YIN Zhou-lan, WANG Ming-yu, TANG Fang. The role of arsenic in the homogeneous precipitation of As, Sb and Bi impurities in copper electrolyte[J]. Hydrometallurgy, 2011, 108(3-4): 199-204.
[13] XIAO Fa-xin, ZHENG Ya-jie, WANG Yong, JIAN Hong-sheng, HUANG Xing-guan, MA Yu-tian. Purification mechanism of copper electrolyte by As(III) [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(5): 1275-1279.
[14] NAILI H, MHIRI T. X-ray structural, vibrational and calorimetric studies of a new rubidium pentahydrogen arsenate RbH5(AsO4)2 [J]. Journal of Alloys and Compounds, 2001, 315(1-2): 143-149.
[15] QURESHI M, KUMAR V, SYNTHESIS I R. X-ray and ion-exchange studies of some amorphous and semicrystalline phases of titanium antimonate: Separation of VO2+ from various metal ions [J]. Journal of Chromatography A, 1971, 62(3): 431-438.
[16] COLOMBAN P, DOREMIEUX-MORIN C, PIFFARD Y, LIMAGE M, NOVAK A. Equilibrium between protonic species and conductivity mechanism in antimonic acid, H2Sb4O11・nH2O [J]. Journal of Molecular Structure, 1989, 213: 83-96.
[17] YNENI S C B, TRAINA S J , WAYCHUNAS G A, LOGAN T J. Experimental and theoretical vibrational spectroscopic evaluation of arsenate coordination in aqueous solutions, solids, and at mineral-water interfaces [J]. Geochimica et Cosmochimica Acta, 1998, 62(19-20): 3285-3300.
[18] ZHAO Zong-yi. Mechanism of arsenic removal in oxidized Fe-As system [J]. Chinese Research Academy of Environmental Sciences, 1995, 15(1): 18-20. (in Chinese)
[19] DINNEBIER R E, IBBERSON R M, EHRENBERG H, JANSEN M. The crystal structures of the binary mixed valence compound Bi(III)3Bi(V)O7 and isotypic Bi3SbO7 as determined by high resolution X-ray and neutron powder diffraction [J]. Journal of Solid State Chemistry, 2002, 163(1): 332-339.
[20] PETKOVA E. Mechanisms of floating slime formation and its removal with the help of sulphur dioxide during the electrorefining of anode copper [J]. Hydrometallurgy, 1997, 46(3): 277-286.
[21] WANG Xue-wen, CHEN Qi-yuan, YIN Zhou-lan, XIAO Lian-sheng. Identification of arsenato antimonates in copper anode slimes [J]. Hydrometallurgy, 2006, 84(3-4): 211-217.
[22] WU Jian-jun, HUANG Fu-qiang,  Xu-jie, CHEN Ping. One-pot synthesis of BiSbO4 nanophotocatalyst with enhanced visible-light performance [J]. Cryst Eng Comm, 2011, 13: 3920-3924.
Xu-jie, CHEN Ping. One-pot synthesis of BiSbO4 nanophotocatalyst with enhanced visible-light performance [J]. Cryst Eng Comm, 2011, 13: 3920-3924.
[23] DUTRIZAC J E, CHEN T T. The role of hydrometallurgy in the recycling of zinc, copper and lead [J]. Acta Metallurgica Slovaca, 1998(1): 5-28.
肖发新1,2,曹 岛1,毛建伟1,申晓妮1,任凤章1
1. 河南科技大学 材料科学与工程学院,洛阳 471003;
2. 河南科技大学 河南省有色金属材料科学与加工技术重点实验室,洛阳 471003
摘 要:通过向含有45 g/L Cu2+、185 g/L H2SO4、10 g/L As和0.5 g/L Bi的铜电解液中加入Sb(V),研究五价锑对铜电解液中砷、锑、铋杂质脱除作用机理。过滤电解液,采用化学分析、SEM、TEM、EDS、XRD、FTIR等方法对沉淀渣的结构形貌和成分进行表征。结果表明,沉淀渣呈尺寸为50~200 μm的不规则块状,其化学成分主要为砷、锑、铋和氧。红外光谱检测表明,沉淀渣主要特征官能团为As―O―As、As―O―Sb、Sb―O―Bi、 Sb―O―Sb和Bi―O―Bi。X射线衍射和电子衍射检测结果表明,沉淀渣由AsSbO4、BiSbO4和Bi3SbO7组成。锑酸盐的生成是五价锑脱除铜电解液中砷、锑、铋杂质的主要原因。
关键词:五价锑;As;Bi;锑酸盐;铜电解液;脱除;净化
(Edited by Wei-ping CHEN)
Foundation item: Project (50904023) supported by the National Natural Science Foundation of China; Project (2010B450001) supported by the Natural Science Fund of Department of Education of Henan Province, China; Project (092300410064) supported by the Basic and Frontier Technologies Research Projects of Henan Province, China
Corresponding author: Fa-xin XIAO; Tel: +86-379-64231846; E-mail: xiaofaxin@yahoo.com
DOI: 10.1016/S1003-6326(14)63057-0

