
Microstructure and properties of nanocrystalline nickel coatings prepared by pulse jet electrodeposition
TIAN Zong-jun(田宗军)1, WANG Dong-sheng(王东生)2, WANG Gui-feng(王桂峰)1,
SHEN Li-da(沈理达)1, LIU Zhi-dong(刘志东)1, HUANG Yin-hui(黄因慧)1
1. College of Mechanical and Electrical Engineering, Nanjing University of Aeronautics and Astronautics,
Nanjing 210016, China;
2. Department of Mechanical Engineering, Tongling College, Tongling 244000, China
Received 10 December 2009; accepted 24 March 2010
Abstract: The fabrication of nanocrystalline nickel coatings was conducted by pulse jet electrodeposition on the substrate of 45# carbon steel. The effects of average current density on the surface morphology, microstructure, average grain size and microhardness of nickel coatings were investigated by scanning electron microscopy (SEM), X-ray diffractometry (XRD) and microhardness measurement. In addition, the corrosion resistances of coating and substrate were compared. It is revealed that the nickel coatings prepared by pulse jet electrodeposition exhibit a fine-grained structure with a smooth surface and a high density, although some pores and defects are still present in coatings. With the increase of average current density, the average grain size of nickel coatings is reduced at first and then increased. The coating with the optimum compactness, the smallest average grain size (13.7 nm) and the highest microhardness are obtained at current density of 39.8 A/dm2. The corrosion resistance is obviously increased for the coatings prepared by pulse jet electrodeposition; however, the corrosion rate is increased after a certain period due to the penetration of the corrosive media.
Key words: pulse jet electrodeposition; nanocrystalline nickel coating; microstructure; microhardness; corrosion resistance
1 Introduction
Along with the soaring development of economy and technology over last decades, modern industry poses higher demands on the materials capable of resisting extreme environment such as high temperature, wear and corrosion. As a common process used to protect a target material, surface coating technique organically combines advantages of both the substrate and coating material for purpose of high mechanical properties (such as strength and toughness) and environmental service demands (such as wear and corrosion resistance). With ultrafine grain size and extremely large specific surface area, nanocrystalline materials exhibit a number of unique physical, chemical as well as mechanical properties, taking on a feasible way to enhance performance of coating[1-2].
In recent years, jet electrodeposition technique, used for nanocrystalline coatings or bulk metals, has gained a wide range of applications with great prospects in the coming decades[3-4]. Based on the electrochemical principle, jet electrodeposition enables promoting the liquid mass transfer velocity, decreasing the thickness of diffused layer and increasing the limiting current density, leading to an increase of deposition rate by tens or even hundreds of times over conventional plating ways. Jet electrodeposition provides a totally new approach to rapidly produce nanocrystalline pure metal, alloy, as well as composite coatings. Another assisting measure for electrochemical process is pulse plating. Compared with direct current deposition, pulse plating takes some advantages, such as better uniformity of composition and thickness, higher density, wear and corrosion resistance, and lower porosity, inner-stress and impurity content [5-6]. Nowadays, pulse jet electrodeposition (PJED), combining jet electrodeposition and pulse plating, has gradually become a new trend and an important supplement in electrodeposition technique.
In the present study, the nanocrystalline nickel coatings were prepared by PJED on a substrate of 45# carbon steel and the influence of hydrogen evolution on the deposits was studied as well. The effects of average current density on the surface morphology, microstructure, average grain size and microhardness of the coating were discussed. The anti-corrosion behaviors of the finished coatings together with substrates were studied too.
2 Experimental
2.1 Theory of pulse jet electrodeposition
Theoretically, pulse jet electrodeposition is a plating method that incorporates pulse modulated power with jet electrodeposition system[7]. Through a nozzle, plating solution is continuously and directly sprayed onto a cathode surface, exciting a continuous liquid strike between aqueous solution and substrate. This kind of strike not only lowers diffused layer thickness and prompts limiting current density, but also retains depositing quality as usual even under the limiting current density. In general, there are two types of pulse modulated powers available, that is constant-current and constant-voltage pulses. The metal ions of the plating solution on the cathode surface are deposited during the pulse on-time (ton). In off-time (toff), metal ions concentration will be fully recovered. If ton is set short enough, a very high peak current density will be achieved and play a highly stimulating role in increasing overpotential, enhancing coating quality and reducing negative impact of hydrogen evolution[8].
Based on the analysis above, PJED increases crystal nucleating rate due to the higher current density, giving rise to the formation of nanocrystalline coatings.
2.2 Experimental equipment
The cathode substrate material was made of 45# carbon steel (50 mm×20 mm×10 mm in size) and the anode material was nickel rode (99.9%). Prior to electrodeposition, steel plates were cleaned through a careful polishing and emerged in alcohol solution to get rid of contaminants. Based on the theory of PJED, a device for purpose of rapid fabrication of nanocrystalline nickel coating was designed, as illustrated in Fig.1.
The system is composed of CNC unit, electrolyte circulation unit, pulse power supply unit and temperature-control system. CNC system was comprised of industrial PC, software and servo system, etc. Electrolyte circulation unit takes the control of electrolyte supplement.
2.3 Composition of electrolyte and deposition processing parameters
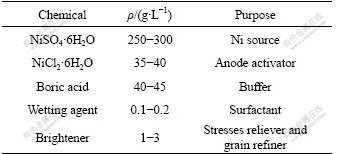
The experimental compositions of the electrolytes and operating parameters are listed in Table 1. Among adding ingredients, nickel sulfate worked as the main salt; nickel chloride worked as the anodic activating agent. Boric acid worked as the buffer reagent that serves to maintain the pH value in electrolytic solution, restrain the precipitation of hydroxides occurring on the cathode, raise the cathode polarization and improve the morphology of deposits. Additionally, as an important organic additive in producing nanoscaled-nickel. Brightener could also brighten the coating surface and reduce the grain size of the coatings.

Fig.1 Schematic diagram of pulse jet electrodeposition
Table 1 Composition of electrolyte and process parameters
A group of rectangular current pulses with various average current densities of 9.9, 29.8, 39.8, 49.7 and 59.7 A/dm2, respectively, were used in plating. Other process conditions are listed in Table 2.
Table 2 Process conditions of pulse jet electrodeposition

2.4 Microstructure characterization and performance test
The surface microstructure of the coatings was characterized by scanning electron microscopy (SEM, LEO-1530VP, Oxford). The average size of crystallites was calculated with the Scherrer formula according to XRD peak broadening. The XRD detection was carried out by a X-ray diffractometer (XD-5A, Japan) under conditions of radiation source of Cu Kα, wavelength of 0.154 178 nm, voltage of 40 kV, current of 60 mA, scanning speed of 4 (?)/min and step size of 0.02?.
Microhardness test was undergone by a HVS-1000A Vickers microhardness tester under a load of 100 N in a dwelling time of 15 s. The distance between the indentations was kept at least three times as large as the diagonal size of indentation in case of residual stress interfering with neighboring indentations measurement [9].
The corrosion resistance was measured by a mass-loss mensuration with each value derived from the average of three specimens. The specimens were cut into 20 mm×10 mm×10 mm blocks and a specified 20 mm× 10 mm area of each block was to be corroded. Before corrosion, they were mechanically polished with 800 grit emery paper, and degreased in organic solvent, ethanol and distilled water in turn. Then, the area for corrosion was dried and measured by the micrometer and the rest part was protected by a layer of epoxy resin. The corrosion process was carried out in a 10% HCl aqueous solution for a total of 68 h. The sample was taken from the corrosion solution at a certain period to weigh the mass-loss by a precision electronic balance with an accuracy of 1×10-4 g. Corrosion resistance is reflected by the corrosion rate, which can be expressed by the ratio of corrosion mass loss to the product of corrosive area and time.
3 Results and discussion
3.1 Morphology of nanocrystalline nickel coating
The cross-sectional SEM image of the nickel coating is shown in Fig.2. As illustrated, no microcracks are observed in the highly-dense nickel coating with a uniform deposit thickness of around 60 μm. However, there presents a clear interface between coating and substrate.

Fig.2 SEM morphology of cross section of coating prepared with current density of 39.8 A/dm2
In general, during the entire cathode reaction, metal ion deposition is companied by the appearance of hydrogen[8]. Hydrogen appearance is an inevitable side reaction, which is harmful because it not only reduces the cathodic current efficiency and costs more electricity power consumption, but also impacts the coating property. However, this disadvantage can be improved by a joint action of pulse modulation and jet electrolytic solution. The pulse jet electrodeposition dramatically decreases the thickness of cathode diffused layer, improves the mass transfer process, reduces the concentration polarization of the hydrogen evolution reaction, and brings down the overpotential by hydrogen evolution[5]. This can be proved by the study of hydrogen evolution in nickel electrodeposition[6].
Although the hydrogen evolution reaction will be promoted to a certain extent, the concentration polarization of the deposited nickel ion simultaneously decreases. As a result of the competition, a high cathode current efficiency can still be maintained. Furthermore, PJED will be able to considerably improve the stability of pH value by inhibiting pH level from rising around cathode, as a result, generating much less Ni(OH)2. Even if a small amount of basic colloidal salts still generate, they would be redissolved in the plating solution again when they grow and hardly attach to the cathode surface because of the continuous strike of the rushing electrolytes. Additionally, the strike between plating solution and cathode substrate also hinders hydrogen atoms from being adsorbed onto the electrode surface, lowering the concentration of hydrogen atoms at the cathode surface and largely decreasing hydrogen embrittlement in this area. So, it can be concluded that PJED is evidently in favor of creating high-quality nanocrystalline nickel coatings.
The SEM morphology of PJED nickel coatings (Fig.3) shows a comparison of coatings under various current densities. From a macro viewpoint, the deposit layer featuring variously-sized nanocrystalline grain is basically compact, but still contains some observable pores and defects. The coating prepared under a relatively low current density of 19.9 A/dm2 exhibits a rough surface and coarse grain size (Fig.3(a)). In contrast, as current density is increased, the deposit surface becomes smoother and smoother with gradually refined grains till the current density reaches 39.8 A/dm2. Then, with current increasing, the whole deposit layer undergoes a cellular-like growth, companied by a larger grain size, higher surface roughness, lower density and worsened surface flatness. At this point, concentration polarization increases and deposition process is controlled by diffusion. The reactive particles which diffuse from the deep solution are easy to arrive the small protrusion of the surface. The deposition surface will become rough by the fighting for ion.

Fig.3 SEM morphologies of PJED nickel coating prepared under different current densities: (a) 19.9 A/dm2; (b) 29.8 A/dm2; (c) 39.8 A/dm2; (d) 49.7 A/dm2; (e) 59.7 A/dm2
The cyclic and intermittent formation of diffusion layer in the pulse jet electrodeposition decreases its actual thickness and reduces the concentration polarization. In the meantime, its current density and overpotential are larger than those of direct electroplating by many times, which promotes the electrochemical polarization and the nucleation. It also makes the crystallization meticulous. Additionally, the existence of pulsing interval inhibits the crystal nucleus growing, and crystalline grains get refined, which makes plating have more perfect structure than direct electroplating[5].
3.2 Influence of current density on grain size and preferred orientation
The average size of crystallites was calculated with XRD peak broadening using the Scherrer formula[10].
According to the (111) and (200) peaks in Fig.4, the average grain sizes calculated by Scherrer formula are 25.1, 14.1, 13.7, 13.9 and 14.4 nm, respectively. It shows a declining-and-raising trend with the increase of average current density, similar to the result reported in Ref.[11], but quite different from that prepared under direct current electrodeposition. As pointed out in Ref.[12], the average grain size of the deposits goes up as the deposition current density increases.
Fig.4 also shows a shift on preferred orientation of grain from (111) to (200) and eventually to a strong (200) when the current density increases. PANGAROV[13] suggested that the preferred orientation of the electrodeposited grain was decided by cathode over- potential. According to the calculation, electrodeposited nickel is most likely to form (111)-like texture at a low over-potential; while with the increase of over-potential, the preferential orientation is (200), and then (220) texture. The appearance of (220) texture can be attributed to the high deposition over-potential resulted from the high current density peak. Also from Fig.4, the enhancement of (220) orientation with the increase of current density may be resulted from the hydrogen atoms absorption. Compared with the (200) crystal plane, (220) has a lower surface energy; therefore, hydrogen atoms are more easily adsorbed on the (220) crystal plane instead of (200) with the increase of current density. The adsorption of hydrogen inhibits crystal growth on the (220) crystal plane. In Ref.[14], it was found that the preferred orientation would change from (200) to (220) when the current density increased to a certain level.
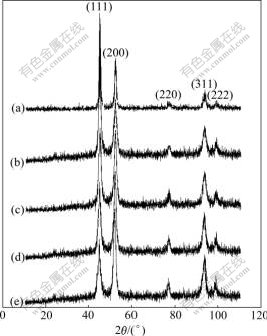
Fig.4 XRD patterns of coatings under different current densities: (a) 19.9 A/dm2; (b) 29.8 A/dm2; (c) 39.8 A/dm2; (d) 49.7 A/dm2; (e) 59.7 A/dm2
During metal electrodeposition, the crystal nucleation takes place only if the electrode potential deviates from the equilibrium potential to a certain negative value, i.e. the over-potential transiting to a certain positive value. Through the special interaction mechanism between the plating solution and substrate, PJED not only significantly decreases the diffusion layer thickness and promotes the limiting current density, but also generates a high cathode over-potential, which can reduce the critical nucleation radius and increase nucleation rate, and is quite helpful for forming a large amount of new crystal nucleus and suppressing the growth of existing crystallites simultaneously, resulting in a fine and compact structure[15]. As a result, the average grain size of nanocrystalline nickel coatings decreases from 25.1 to 13.7 nm with current density increasing from 19.9 to 39.8 A/dm2, which can be attributed to the presence of the raised over-potential and high crystal nucleation rate caused by increase of current density. However, if current density further increases, Ni2+ ion around cathode cannot be supplemented in time during a extremely short pulse interval, leading to a reduction of charged ions in this area. As a result, the reduction of nucleation rate due to the decrease of Ni2+ outweighs the increase caused by overpotential. In addition, the increase of overpotential results in the increase of hydrogen evolution, leading to the grain growth eventually. JIANG et al[12] also researched the influence of current density on the average grain size in PJED and drew a similar conclusion that the average grain size increases when the current density is higher than a certain level.
3.3 Microhardness
The microhardness of coatings under different current densities is presented in Table 3. Compared with the mean grain size, it can be found that microhardness increases with the reduction of grain size, in good agreement with the Hall-Petch empirical relationship.
Table 3 Microhardness of coating under different processing parameters

3.4 Corrosion resistance
In Fig.5, two corrosion rate―time curves correspond to the substrate and the sample coated by PJED nickel, respectively. As for the corrosion resistance, the PJED-nickel coated sample is much better than the bared substrate during the first 26 h of corrosion. After this period, however, the anti-corrosion behavior of coated material is worse than the substrate. This can be explained that, on one hand, the nickel coating with fined-grain and compact structure has a far lower corrosion rate than substrate material, on the other hand, a few pores and defects scattered on the nickel coating allow corrosive liquid to infiltrate into substrate and accelerate corrosion in the coming time.
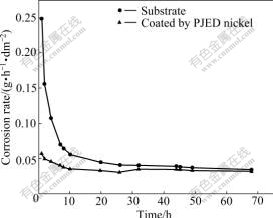
Fig.5 Curves of corrosion rate versus time
Currently, there are two different viewpoints on the coating corrosion mechanisms[16]. One argues that corrosion media come into substrate along the pores and defects of coatings to corrode and lead to its failure, while the other regards that the corrosion also exists inside coatings. Actually, the real corrosion failure mechanism is not completely clear now. Regardless, the microcracks, pores and defects on and inside coatings should be avoided as much as possible to improve corrosion resistance. Other proposed methods associated with anti-corrosion, including composite electro- deposition[1,17] and laser remelting treatment[18], are effective ways to improve the corrosion resistance.
4 Conclusions
1) Under given process parameters, the nickel coatings prepared by pulse jet electrodeposition exhibit some advantages, such as, fine grain, smooth surface and good bond with substrate, except for little pores and defects.
2) With the increase of average current intensity, the mean grain size undergoes a declining-and-rising trend. At an optimum current density of 39.8 A/dm2, the coating with finest grain size of 13.7 nm shows the better properties in the compactness and microhardness.
3) Processed by pulse jet electrodeposition, the corrosion resistance of target material was considerably improved, but the corrosion rate increases somewhat after 26 h in the presence of long-time corrosive media penetrating.
References
SRIRAMAN K R, GANESH SUNDARA RAMAN S, SESHADRI S K. Corrosion behaviour of electrodeposited nanocrystalline Ni-W and Ni-Fe-W alloys [J]. Materials Science and Engineering A, 2007, 460/461: 39-45.
[1] WANG Li-ping, ZHANG Jun-yan, GAO Yan, XUE Qun-ji, HU Li-tian, XU Tao. Grain size effect in corrosion behavior of electrodeposited nanocrystalline Ni coatings in alkaline solution [J]. Scripta Mater, 2006, 55(7): 657-660.
[2] QIAO Gui-ying, JING Tian-fu, WANG Nan, GAO Yu-wei, ZHAN Xin, ZHOU Ji-feng WAND Wei. High-speed jet electrodeposition and microstructure of nanocrystalline Ni-Co alloy [J]. Electrochimica Acta, 2005, 51(1): 85-92.
[3] CHEN Jin-song, HUANG Yin-hui, LIU Zhi-dong, TIAN Zong-jun, ZHAO Yang-pei. Jet electrodeposition oriented by rapid prototyping [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(S3): 247-250.
[4] PELLICER E, GOMEZ E, VALLES E. Use of the reverse pulse plating method to improve the properties of cobalt-molybdenum electrodeposits [J]. Surface and Coatings Technology, 2006, 201(6): 2351-2357.
[5] BAHROLOLOOM M E, SANI R. The influence of pulse plating parameters on the hardness and wear resistance of nickel-alumina composite coatings [J]. Surface and Coatings Technology, 2005, 192(2/3): 154-163.
[6] TIAN Zong-jun, WANG Gui-feng, HUANG Yin-hui, LIU Zhi-dong, CHEN Jin-song. Fractal study on copper electrodeposition [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(1): 167-173. (in Chinese)
[7] JIANG Shan, PAN Yong, TANG Tian, ZHONG Yi-chun. Preparation of nanocrystalline nickel coating by pulse jet-electrodeposition [J]. Materials Protection, 2007, 40(3): 49-51. (in Chinese)
[8] LEIGH S H, LIN C K, BERNDT C C. Elastic response of the thermal spray deposits under indentation tests [J]. Journal of the American Ceramic Society, 1997, 80(8): 2093-2099.
[9] ZHANG Li-de, MOU Ji-mei. Nano-material and nano-structured [M]. Beijing: Science Press, 2001: 148. (in Chinese)
[10] TOTH-KADAR E, BAKONYI I, POGANY L, CZIRAKI A. Microstructure and electrical transport properties of pulse-plated nanocrystalline nickel electrodeposits [J]. Surface and Coatings Technology, 1996, 88(1): 57-65.
[11] JIANG Shan, PAN Yong, TANG Tian, ZHOU Yi-chun. Preparation and characterization of jet-electrodeposited nanocrystalline nickel coatings [J]. Natural Science Journal of Xiangtan University, 2004, 23(3): 61-65. (in Chinese)
[12] PANGAROV N A. Preferred orientations in electro-deposited metals [J]. Journal of Electroanalytical Chemistry, 1965, 9(1): 70-85.
[13] FANG Xiao-hong. Research progress for ultrasound-assisted electroplating of Ni and Ni-based composite coatings [J]. Materials Protection, 2008, 41(5): 58-60. (in Chinese)
[14] XIONG Yi, JING Tian-fu, ZHANG Chun-jiang, SHAO Guang-jie, YU Sheng-xue, ZHANG Fang, ZHANG Chun-lin. A study of jet electrodepositing nanometer crystalline nickel [J]. Plating and Finishing, 2000, 22(5): 1-4. (in Chinese)
[15] CELIK E, OZDEMIR I, AVCI E, TSUNEKAWA Y. Corrosion behaviour of plasma sprayed coatings [J]. Surface and Coatings Technology, 2005, 193(1/3): 297-302.
[16] YAO Ying-wu, YAO Su-wei, ZHANG Lu, WANG Hong-zhi. Electrodeposition and mechanical and corrosion resistance properties of Ni-W/SiC nanocomposite coating [J]. Materials Letters, 2007, 61(1): 67-70.
[17] GAO Ya-li, WANG Cun-shang, YAN Man, LIU Hong-bin. The resistance to wear and corrosion of laser-cladding Al2O3 ceramic coating on Mg alloy [J]. Applied Surface Science, 2007, 235(12): 5306-5311.
Foundation item: Project(50175053) supported by the National Natural Science Foundation of China
Corresponding author: TIAN Zong-jun; Tel: +86-25-84892520; E-mail: tianzj@nuaa.edu.cn
DOI: 10.1016/S1003-6326(09)60254-5
(Edited by YANG Bing)