
Fabrication, lattice strain, corrosion resistance and mechanical strength of nanocrystalline nickel films
PAN Yong(�� ��)1, 2, ZHOU Yi-chun(���洺)1, 2, ZHOU Zhao-feng(����)1, 2,
HUANG Yong-li(������)1, 2, LIAO Yan-guo(����)1, 2, SUN Chang-qing(�ﳤ��)3
1. Faculty of Materials and Optoelectronics Physics, Xiangtan University,
Xiangtan 411105, China;
2. Key Laboratory of Low Dimensional Materials and Application Technology,
Ministry of Education, Xiangtan University, Xiangtan 411105, China;
3. School of Electrical and Electronic Engineering, Nanyang Technological University, Singapore 639798
Received 15 July 2007; accepted 10 September 2007
Abstract: Nanocrystalline nickel films of 17-40 nm grain sizes were prepared using pulsejet electrodeposition. Structure, corrosion and lattice strain were analysed by transmission electron microscope, electrochemical workstation and X-ray diffraction, revealing that with decreasing of grain size, the lattice strain, corrosion rate of the films are enhanced. The observations can be consistently understood in terms of the bond-order-length-strength correlation mechanism indicating that the shortened and strengthened bonds between the under-coordinated atoms modify the energy density and the atomic cohesive energy in the surface skins of the grains. The surface energy density gain is responsible for the residual atomic cohesive energy for the activation energy of corrosion. Additionally, a novel algorithm was proposed to extract the elastic-plastic properties of nickel films and results that the nickel film has much higher yield strength than bulk nickel.
Key words: pulse jet electrodeposition; nickel film; corrosion resistance; mechanical property
1 Introduction
Nanocrystalline materials with grain size smaller than 100 nm possess novel properties compared with their coarse polycrystalline counterpart, which have attracted tremendous interest of research[1-3]. Among various preparation techniques, the electrodeposition method has been developed rapidly. Electrodeposition has many advantages over other nano-processing techniques[4-6] including: 1) the capability for preparing pure metals, alloys and composites with grain sizes less than 100 nm; 2) the low cost and high efficiency in synthesizing these materials; 3) the relatively minor ��technological barriers�� to be overcome in transferring this technology from the research laboratory to existing electroplating and electroforming industries. It has been well demonstrated that under certain preparation conditions, electrodeposition can yield nickel films with nice nanocrystalline micro- structures[7-9].
In this work, the depositing nanocrystalline nickel films using the newly developed pulsejet electro- deposition was reported. The structure, lattice strain and corrosion resistance of the as-prepared nickel films were studied and the mechanism of the relation between microstructure and properties was analyzed.
Additionally, a number of methods based on experimental and numerical studies were proposed to extract mechanical properties from indentation load- displacement curves on the uncoated substrates[10-12]. However, little work has been conducted on the evaluation of the mechanical properties of the elastic- plastic thin films on tough substrates. In this work, a new theoretical/numerical algorithm for conical indentation to measure the elastic-plastic properties of nickel films on tough substrates was reported.
2 Experimental
The set-up of pulsejet electrodeposition, which was developed by us is shown in Fig.1. We applied a conventional Watts-type solution, as listed in Table 1. The bath temperature was maintained at (60��1)��. The pH was adjusted to 3.0 by adding diluted sulfuric acid. Before electroplating, the substrates were activated in diluted HCl solution. The pulse electrodeposition was performed using rectangular cathode current pulses as shown in Fig.2. The nanocrystalline nickel films were prepared using pulsejet plating on the wild steel substrates. D/max�CrA X-ray diffraction(XRD) and Tecnai-20 transmission electron microscope(TEM) were employed to examine the microstructure and lattice strain of the nickel films.

Fig.1 Schematic diagram of experimental setup: 1 Vertical- placed jetting tube; 2 Cathode substrate; 3 Electrodeposited bath; 4 Temperature control unit; 5 Thermometer; 6 Electrolytic bath; 7 Pulse power supply; 8 Magnetic pump; 9 Valve; 10 Flowmeter; 11 Plastic tube

Fig.2 Sketch of rectangular curve for cathodic current pulses
The electrochemical tests were conducted in the electrolyte of 0.1 mol/L H2SO4 and 1% NaCl using a platinum counter electrode and a saturated calomel electrode reference electrode. Weak polarization and Tafel tests were performed on electrochemical workstation (Solatron 1287) with the scan rates of 0.1 and 2 mV/s, respectively. The data were analyzed using the Corrview software (Solartron, UK). For evaluating the elastic-plastic properties of nickel films by our new theoretical/numerical algorithm, loading-displacement curves were obtained by means of nanoindentation tests on Triboindenter system (Hysitron, USA).
Table 1 Contents of electrolyte solution

3 Results and discussion
3.1 Microstructure
The TEM bright field image (Fig.3(a)) shows that the nickel films are composed of a large number of nanocrystals with an average grain size of approximately 30 nm for the nickel films obtained at the peak current density of 0.7 A/cm2. A selected-area electron diffraction pattern (Fig.3(b)) further confirms that the nickel films are face centred cubic (FCC) structure.

Fig.3 TEM images of nickel films deposited at peak current density Jp=0.7 A/cm2: (a) Bright-field; (b) Electron diffraction pattern
Using the Bragg diffraction and the Sherry formula, we can derive the lattice strain and the grain size from the XRD profiles (Fig.4(a)). It is derived that the average grain size decreases from 44 nm to 17.6 nm when the peak current density increases from 0.32 to 0.96 A/cm2. However the average grain size increases to 32.8 nm when the peak current density continues increasing to 1.28 A/cm2 because of the rapid deposition rate and substrate heating effect[13]. From Fig.4(b), it can be seen that the average lattice strain increases from 0.282% to 0.555% and then drops to 0.449% when the peak current density increases from 0.32 to 1.28 A/cm2, showing clearly the size dependence of lattice strain because of the increased portion of the under-coordinated atoms between which the bonds become shorter and stronger[1].

Fig.4 XRD patterns of nickel films deposited at different peak current densities (a) and grain size and lattice strain under different peak current densities (b) (GS: grain size LS: lattice strain)
It is known that the core of the grain might form when the electrode potential deviates from the balance potential to an exact value called over potential. Because of the pulse nature between the electrolyte solution and the cathode substrate, pulsejet electrodeposition can not only thin the diffusion layer so as to increase the limit current density but also facilitate the formation of new grains and slow down the growth of existing grains. Consequently, when the peak current density increases from 0.32 to 0.96 A/cm2, the average grain size of the nickel films decreases from 44 nm to 17.6 nm because the increase of the current density can lead to the increase of over potential and the forming rate of the grains. However, if the current density continue increasing, the pulse interval is too short to make the Ni2+ near the cathode supplying in time in addition to the effect of substrate heating. So the density of Ni2+ continues decreasing, which results in lowering the rates of ion discharging and grains forming. This decrease is more apparent than the over potential. In addition, the high current will raise the substrate temperature that determines the critical size of the deposited grains[13]. So grains of nickel films grow bigger and bigger.
3.2 Corrosion resistance
From the Tafel curves (Fig.5(a)) of nickel films deposited under different conditions, it can be seen that the open circuit potential is positively highest when the peak current density reaches 0.32 A/cm2, which means that nickel films deposited at the peak current density of 0.32 A/cm2, or nickel films with larger grain sizes, are more corrosion resistant. We also observed a small passivation behavior on anode for nickel films deposited at the peak current density value of 0.96 A/cm2. From the weak polarization curve (Fig.5(b)), it can be seen that the slopes of weak polarization curves under different peak current densities have no much difference. Only the curve of the film deposited at the peak current density of 0.96 A/cm2 has lower slope and shows less polarization impedance than others. The relation between the average corrosion rate and the peak current density (Fig.5(c)) is obtained by means of Tafel curve extrapolation and the Sturn method. Both methods produce the same trend. When the peak current density increases from 0.32 to 0.96 A/cm2, the corrosion current density increases, which means that the corrosion resistance of the film decreases with increasing grain size. However, when the current density continues increasing to 1.28 A/cm2, the corrosion current density drops to 62.8 A/cm2 and so the corrosion resistance is enhanced, which is in good accordance with the observation. So the nickel films with the smallest grain size exhibit the highest corrosion current density and the worst corrosion resistance. The size-depressed corrosion rate can be attributed to the

Fig.5 Tafel curves of nickel films deposited at different current densities (a), weak polarization curves of nickel films deposited at different current densities (b) and relations between current density and corrosion resistance of nickel films (c)
lowered residual cohesive energy of the under- coordinated atoms in the grain boundaries and the increased portion of such under-coordinated atoms, according to the BOLS correlation mechanism[1]. Because of the lowered residual cohesive energy, the atoms in the grain boundaries possess higher energy and so these atoms exist in unstable states[13].
3.3 Mechanical strength
We proposed a novel algorithm to extract the elastic-plastic properties of nickel films on mild steel substrate. Firstly, the normalized loading and unloading work were computed from extensive finite element method(FEM) indentation simulations and fitted by smoothening the dimensionless functions. Then explicit relationships between the indentation parameters and the elastic-plastic properties of the thin films on tough substrates were established through extensive FEM approaches.
Equivalent elastic modulus Er was obtained by Eqns.(1) and (2)[14]. Stiffness S can be estimated from Eqn.(2)[15] according to the unloading curve. Contacting area is the function of contacting depth hc, which can be confirmed by an experiential rule:
 (1)
(1)
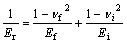 (2)
(2)
 (3)
(3)
According to the above three equations, the elastic modulus of nickel films and the wild steel substrate were calculated as 220.52 GPa and 170 GPa, respectively.
Based on the results of FEM, strain hardening exponent of nickel films was achieved by the Eqns.(4) and (5):
 (4)
(4)
 (5)
(5)
According to the above two equations, we calculated the yield strength and the strain rigidity exponent of nickel film as 2 353.56 MPa and 0.434, respectively. The stress��strain relation of nickel film is as follows:
�ҡ�2 353.56MPa
�ҡ�2 353.56MPa
 (6)
(6)
From Eqn.(6) we can conclude that nickel film has much higher yield strength than bulk nickel whose yield intensity is only 483-724 MPa[16].
4 Conclusions
1) The nanocrystalline nickel films with different grain sizes were fabricated by the pulsejet electro- deposition sep-up, which was developed by us.
2) The lower corrosion resistance, higher lattice strain of the smaller grains can be understood consistently in terms of the bond-order-length-strength correlation mechanism indicating that the under-coordinated atoms on the surface or grain boundaries take the responsibility of the observed phenomena.
3) A novel algorithm was proposed to extract the elastic-plastic properties of nickel films on mild steel substrate and the results show that the nickel films have much higher yield strength than bulk nickel.
References
[1] SUN Chang-qing. Size dependance of nanostructures: Impact of bond order deficiency [J]. Prog Solid State Chem, 2007, 35: 1-159.
[2] KIM S H, AUST K T, ERB U, GONZALEZ F, PALUMBO G. A comparison of the corrosion behaviour of polycrystalline and nanocrystalline cobalt [J]. Scripta Mater, 2003, 48(9): 1379-1384.
[3] WEI H, HIBBARD G D, PALUMBO G, ERB U. The effect of gauge volume on the tensile properties of nanocrystalline electrodeposits [J]. Scripta Mater, 2007, 57(11): 996-999.
[4] TAI H Y, SEUNG C Y, HYOUNG S K I. Tensile properties of electrodeposited nanocrystalline nickel [J]. Mater Sci Eng A, 2007 25(449/451): 836-840.
[5] HIBBARD G D, AUST K T, ERB U. Thermal stability of electrodeposited nanocrystalline Ni-Co alloys [J]. Mater Sci Eng: A, 2006, 433(1/2): 195-202.
[6] ERB U. Electrodeposited nanocrystals: Synthesis, properties and industrial applications [J]. Nanostructured Materials, 1995, 6: 553-538.
[7] EL-SHERIK A M, ERB U, PALUMBO G. Deviations from hall-petch behaviour in as-prepared nanocrystalline nickel [J]. Scripta Metall Mater, 1992, 27: 1185-1188.
[8] WANG Li-ping, ZHANG Jun-yan, GAO Yan, XUE Qun-ji, HU Li-tian, XU Tao. Grain size effect in corrosion behavior of electrodeposited nanocrystalline Ni coatings in alkaline solution [J]. Scripta Mater,2006, 55(7): 657-660.
[9] CZIRAKI A, FOGARASSY B, GEROCS I. Microstructure and growth of electrodeposited nanocrystalline nickel foils [J]. J Mater Sci, 1994, 29(18): 4771-4777.
[10] ZHAO Man-hong, NAGAHISA O, NORIMASA C, XI Chen. A new approach to measure the elastic-plastic properties of bulk materials using spherical indentation [J]. Acta Mater, 2006, 54(1): 23-32.
[11] ZHAO Man-hong, CHEN Xi, YAN Jin, ANETTE M K. Determination of uniaxial residual stress and mechanical properties by instrumented indentation [J]. Acta Mater, 2006, 54(10): 2823-2832.
[12] CASALS O, ALCALA J. The duality in mechanical property extractions from Vickers and Berkovich instrumented indentation experiments [J]. Acta Mater, 2005, 53(13): 3545-5361.
[13] SUN Chang-qing, SHI Y, LI C M. Size-induced undercooling and overheating in phase transitions in bare and embedded clusters [J]. Phys Rev B, 2006, 73: 075408.
[14] JOSLIN D L, OLIVER W C. A new method for analyzing data from continuous depth-sensing microindentation tests [J]. J Mater Res, 1990, 5(1): 123-126.
[15] OLIVER W C, PHARR G M. An improve technique for determining hardness and elastic modulus using load and displacement sensing indentation experiment [J]. J Mater Res, 1992, 7(6): 1564-1583.
[16] YANAKA M, TSUKAHARA Y, NAKASO N. Cracking phenomena of brittle films in nanostructure composites analysed by a modified shear lag model with residual strain [J]. J Mater Sci, 1998, 33: 2111-2119.
(Edited by LI Xiang-qun)
Foundation item: Project(2005AA01210) supported by the National High-Tech Research and Development Program of China; Projec(50531060) supported by the Key Program of National Natural Science Foundation of China; Project(10525211) supported by the National Natural Science Foundation of China for Distinguished Young Scholars; Project(076044) supported by the Ministry of Education of China; Project(10772157) supported by the National Natural Science Foundation of China
Corresponding author: ZHOU Yi-chun; Tel: +86-732-8293586; E-mail: zhouyc@xtu.edu.cn