Trans. Nonferrous Met. Soc. China 23(2013) 1556-1562
Fabrication of aluminum matrix composites reinforced with Al2ZrO5 nano particulates synthesized by sol-gel auto-combustion method
S. A. KHORRAMIE1, M. A. BAGHCHESARA2, D. P. GOHARI3
1. Department of Chemistry, North Tehran Branch, Islamic Azad University, Tehran 14358-84381, Iran;
2. Department of Metallurgy & Materials Engineering, Masjed Soleiman Branch, Islamic Azad University, Masjed Soleiman 64915-111, Iran;
3. Young Researchers and Elite Club, South Tehran Branch, Islamic Azad University, Tehran 14358-84381, Iran
Received 20 July 2012; accepted 29 November 2012
Abstract: Nanocrystalline Al2ZrO5 with particle size of about 38 nm was directly synthesized by combination of sol-gel auto-combustion and ultrasonic irradiation techniques from metal nitrates and glycine as precursors. The overall process involves formation of homogeneous sol, formation of dried gel and combustion process of the dried gel. Aluminum alloy matrix composites reinforced with 0.75%, 1.5% and 2.5% Al2ZrO5 nanoparticles were fabricated via stir casting method and the fabrication was performed at various casting temperatures. The resulting composites were tested for their nanostructure and present phases by SEM and XRD analysis. Optimum amount of reinforcement and casting temperature were determined by evaluating the density, hardness and compression strength of the composites. Al matrix alloy reinforced by Al2ZrO5 nanoparticles improves the hardness and compressive strength of the alloy to maximum values of BHN 61 and 900 MPa, respectively. The most improved mechanical properties are obtained with the specimen including 1.5% Al2ZrO5 produced at 850 °C.
Key words: metal matrix composites; Al2ZrO5; nanostructure; auto-combustion; ultrasonic; stir casting; mechanical properties
1 Introduction
Al2ZrO5 has attracted considerable attention because of its diverse practical applications in fuel-cell technology [1], as a catalyst or catalyst support [2], oxygen sensor [3], protective coating for optical mirrors and filters [4], nanoelectronic devices, thermal-barrier coating [5], ceramic biomaterial [6] and thermo luminescence UV dosimeter [7]. Al2ZrO5 nanostructure is one of the significant current interests in preparing piezoelectric, electrooptic, dielectric and nanocomposite materials [8]. Al2ZrO5 is classified as a wide band gap semiconductor and tends to become more conductive with increasing temperature.
Al2ZrO5 powders with nanostructured particles have been synthesized by various techniques [9,10]. Hydrothermal route is one of the most extensively employed techniques in the synthesis of metal oxide nanostructures [11]. Among these methods, induction plasma spheroidization of powders is a good way to study the melting, evaporation, solidification and oxidation behavior of the refractory metals such as tungsten and molybdenum. Also, it could be employed as an essential and individual technique in the similar type of works on metal/metal oxide materials [12-14].
Aluminum matrix composites possess many advantages such as low density, high strength and good wear resistance with the development of some non-continuous reinforcement materials, whisker, fibers or particles. In particular, the particulate reinforced aluminum matrix composites not only have good mechanical and wear properties, but also are economically viable [15].
There are many methods for fabrication of particulate reinforced metal matrix composites (MMCs) such as powder metallurgy [16], squeeze casting [17] and compocasting [18]. For the MMCs, molten metal mixing is a effective method while powder metallurgy is costly, and squeeze casting provides good infiltration quality of chopped performs [19].
In this work, Al2ZrO5 nanostructure was synthetized by combination of sol-gel auto-combustion and ultrasonic irradiation techniques using zirconium (IV) nitrate and aluminum nitrate as precursors. Moreover, Al2ZrO5 nanocomposite powders were incorporated into the molten A356 aluminum alloy via stir casting method under various conditions.
2 Experimental
Zirconium(IV) nitrate (Zr(NO3)4·3H2O), aluminum nitrate (Al(NO3)3·9H2O), glycine, NH4OH, n-butanol and Al powder were used as raw materials. All the reagents were obtained from Merck and used without further purification. Deionized water was used for all the experiments. Aluminum (A356) was obtained from Kian Alloy Compony (Kashan-Iran) as the metal matrix of composites. The chemical analysis of Al alloy is listed in Table 1.
Table 1 Chemical composition of A356 Al alloy (mass fraction, %)

Al2ZrO5 nanoparticles were prepared by sol-gel auto-combustion method. Appropriate amounts of analytical grade Zr(NO3 )4·3H2O and Al(NO3)3·9H2O were taken and mixed with glycine. The molar ratio of the nitrates to glycine was 1:3. The mixture was then dissolved in deionized water. The addition of glycine helped the homogenous distribution and segregation of the metal ions. A small amount of ammonium hydroxide was added carefully to the solution to change the pH value to 7. The solution was continuously stirred using a magnetic stirrer. The resultant gel was poured in a platinum crucible and heated at 400 °C. Finally, the as-burnt powder was calcined in air at 1100 °C for 4 h at a heating rate of 10 °C/min to obtain Al2ZrO5 nanoparticles. Then, the product was placed in ultrasonic irradiation bath with n-butanol as solvent for 15 min at room temperature.
In this work, nano Al2ZrO5 was used to improve the strength of A356 aluminum alloy matrix as ceramic reinforcing particle. The Al2ZrO5 powders were synthesized with particle size of about 38 nm. The Al2ZrO5 nano particle had monoclinic structure. Therefore, cast alloy (A356 Al) was selected. The Al2ZrO5 powders were mixed homogeneously with 0.75%, 1.5% and 2.5% (volume fraction) and Al powder at room temperature. The mixture was then placed in a stainless steel mold and 20 MPa pressure was used to form the Al2ZrO5 performs.
For casting Al-Al2ZrO5 composite, a resistance furnace equipped with a stirring system was used. After smelting the aluminum ingots, a given amount of keryolite was added to the molten Al alloy and stirred for a few minutes. Al2ZrO5 nanopowders with volume fraction of 0.75%, 1.5% and 2.5% were wrapped into the aluminum foils and added to the melt Al alloy to produce Al-Al2ZrO5 composite. The processing temperatures were chosen as 800 and 850 °C. Stirring was continued for another 10 min for homogeneous dispersion and to prevent the agglomeration of particulates. A metallic mold was used for casting. Finally, specimens fabricated under various conditions were prepared for subsequent microstructural and mechanical analyses.
X-ray diffractometer (Model: XPERT-MPD, Phylips) was used with Cu Kα radiation (λ=1.05406  ) and operated at 40 kV and 40 mA. The morphologies of the nanopowders and composites were analyzed by scanning electron microscope (SEM, Oxford CAMSCAN-MV2300), and specimens were polished and etched using Keller solution [20]. An ultrasonic bath at high frequency of 35 kHz and 240 W (made in Germany) was used at room temperature.
) and operated at 40 kV and 40 mA. The morphologies of the nanopowders and composites were analyzed by scanning electron microscope (SEM, Oxford CAMSCAN-MV2300), and specimens were polished and etched using Keller solution [20]. An ultrasonic bath at high frequency of 35 kHz and 240 W (made in Germany) was used at room temperature.
The bulk density of the samples was determined by the Archimedes method. The theoretical density was estimated by using mixture law. The compressive strength test was conducted in air at room temperature (Instron Universal Testing Machine-1195 Machine) according to ASTM-B557 [21]. At least five specimens were tested for each casting condition. The hardness of specimens was determined by the Brinell method. Hardness tests were taken under a load of 306.56 N and a punch diameter of 2.5 mm. Data of hardness were determined using at least ten indentations on two polished specimens.
3 Results and discussion
3.1 XRD and microstructural analyses
The XRD pattern of the Al2ZrO5 nanoparticles is shown in Fig. 1. The particle size of the samples is calculated by employing the Scherrer equation:
d=Kλ/βcosθ (1)
where θ is the angle between the incident and diffracted beams (°); β is the full width half maximum (rad); d is the particle size of the sample (nm); λ is the wavelength of the X-ray. The results of XRD confirm the formation of single phase of monoclinic Al2ZrO5 nanostructure at lower temperature. The grain size of the prepared Al2ZrO5 is found to be about 38 nm.
Figure 2 shows the SEM image of Al2ZrO5 nanoparticles. The results of phase analyses and the XRD pattern of A356 Al/Al2ZrO5 at 850 °C are shown in Fig. 3. The phases identified by XRD analysis are similar for all the composites. Although the intensities of the peaks are different, only Al2ZrO5, silicon (Si) and aluminum (Al) are detected. Also, no undesirable phases in the compositions are found.

Fig. 1 XRD pattern of Al2ZrO5 nanoparticles

Fig. 2 SEM image of Al2ZrO5 nanoparticles

Fig. 3 XRD pattern of A356 Al/Al2ZrO5 composite at 850 °C
The morphologies of A356 Al/Al2ZrO5 are shown in Figs. 4 and 5. The black matrix is aluminum and the white spots represent Al2ZrO5 nanoparticles. The grey area is silicon-rich interdendritic Al-Si eutectic. The phases are indicated by arrows on the images. It should be noted that nano-sized Al2ZrO5 particles are well dispersed in the matrix of aluminum and just a partial agglomeration in composites with 0.75%, 1.5% and 2.5% Al2ZrO5 can be detected in Figs. 4 and 5.

Fig. 4 SEM images of Al356/Al2ZrO5 composite specimens with 0.75% Al2ZrO5 prepared at 800 °C under different magnifications

Fig. 5 SEM images of Al356/Al2ZrO5 composite specimens with 1.5% (a, c) and 2.5% (b, d) Al2ZrO5 prepared at 800 °C
Similar microstructures are observed for composites fabricated at various casting temperatures and volume fraction of Al2ZrO5. The above-mentioned results, obtained from XRD and microstructural analyses, confirm the feasibility of stir casting method to produce such a kind of composite with homogenous distribution of reinforcement.
3.2 Density measurements
The effects of casting temperature and volume fraction of nano Al2ZrO5 on the density of Al-nano Al2ZrO5 composites produced by stir casting method are shown in Fig. 6. According to the mixture law, by increasing the volume fraction of Al2ZrO5 particles in aluminum, the density of the samples should be increased because the density of Al2ZrO5 is higher than that of aluminum.

Fig. 6 Density of composite specimens containing 0.75%, 1.5% and 2.5% Al2ZrO5 prepared at 800 and 850 °C
Considering that the density of the Al2ZrO5 is higher than that of the matrix alloy, the improvement in density of composites would occur by increasing the volume fraction of reinforcing particles. Therefore, the mixture law in composite materials confirms this phenomenon that the total density increases with the volume fraction of the second phase. Thus, the density of a composite is identified by the volume fraction of each material in the composite [22].
By increasing the content of reinforcement at constant temperature, the density of specimens is not increased remarkably. This may demonstrate the effect of low wettability and agglomeration at a high content of reinforcement, which leads to the formation of pores. Additionally, tensile stresses originated from the thermal expansion coefficient mismatch between metal matrix and rigid inclusions would normally form defects such as pores and dislocations around the particles.
The density of composites containing 1.5% and 2.5% Al2ZrO5 reduces by increasing temperature. This may be due to the air entrapment or increasing agglomeration by casting composites at temperatures above the melting point. However, this decreasing trend in the density of composite containing 1.5% and 2.5% Al2ZrO5 from 800 to 850 °C is not significant and it can be connived. It certainly should be noted that, temperature plays an important role in increasing the wettability of particles. Also, the mechanical properties of composite such as hardness and compressive strength could be enhanced due to the improvement of particle wettability.
3.3 Mechanical properties
3.3.1 Hardness test
The effects of the casting temperature and volume fraction of Al2ZrO5 nano particles on the hardness of prepared composites are shown in Fig. 7. It is observed that the hardness of Al2ZrO5-reinforced composite is higher than that of the unreinforced alloy. It can be attributed to the higher hardness of the ceramic particles compared to the aluminum alloy matrix. The hardness of the composite depends on the hardness of the reinforcement and the matrix. Also, increasing the volume fraction of Al2ZrO5 nano particles at 800 °C results in the enhancement of hardness, because the number and total surface area of Al2ZrO5 particles increase with increasing Al2ZrO5 particle volume fraction [23,24].
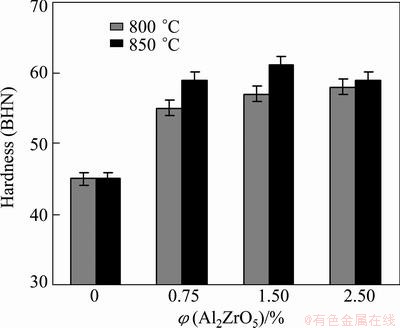
Fig. 7 Hardness of Al alloy and composite specimens containing 0.75%, 1.5% and 2.5% Al2ZrO5 prepared at 800 and 850 °C
In addition, for the composites cast at 850 °C, the maximum hardness is achieved when they contain 1.5% Al2ZrO5. However, the composites containing over 1.5% Al2ZrO5 show a decreasing trend of hardness, which may be caused by the presence of more pores and the air entrapment in composites with higher content of Al2ZrO5 and increasing temperature.
From the other point of view, increasing temperature improves the wettability of particles (with molten metal) and strengthens metal–ceramic bond at interfaces, so with better wettability, the properties of composite change, especially, mechanical ones. Figure 7 shows these effects clearly. It is seen that the hardness of the composites is improved with increasing casting temperature. Therefore, at a constant amount of Al2ZrO5, composites cast at lower temperatures have lower hardness, which causes poor mechanical properties The maximum hardness observed is BHN 61 of specimen with 1.5% Al2ZrO5 cast at 850 °C.
3.3.2 Compression test
The effects of casting temperature and volume fraction of nano Al2ZrO5 on the compressive strength of Al-nano Al2ZrO5 composites are shown in Fig. 8. As demonstrated, with increasing volume fraction of reinforcing particles, the value of the compressive strength initially increases up to 1.5% and then decreases at both casting temperatures compared with the unreinforced matrix alloy. The initial enhancement seems to be due to work-hardening behavior. This could be related to the effects of elastic properties of ceramic particles and the inhibition response of plastic deformation of matrix by them; ceramic particles can only deform elastically while aluminum matrix can deform plastically. If the boundary is assumed to be strong, ceramic particles could prevent plastic deformation of the matrix, leading to higher work- hardening rate [24].
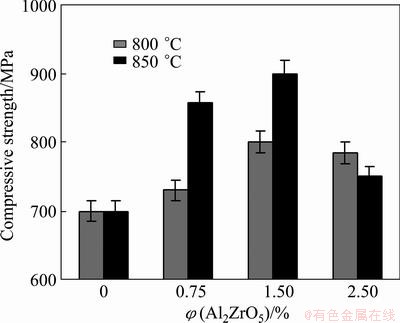
Fig. 8 Compressive strength of Al alloy and composite specimens containing 0.75%, 1.5% and 2.5% Al2ZrO5 prepared at 800 and 850 °C
On the other hand, the difference between the coefficients of thermal expansion of ceramic and metal matrix may result in stress concentrations and thus high density of dislocations, as a result, the strength of the aluminum matrix increases accordingly. This confirms the obvious effect of Al2ZrO5 particles on the strengthening of composites [25].
Also, a decreasing trend of compressive strength is resulted from the formation of pores caused by the incorporation of higher content of Al2ZrO5 and air absorption/entrapment during casting process. The maximum compressive strength observed is 900 MPa of the specimen with 1.5% Al2ZrO5 cast at 850 °C.
According to Figs. 7 and 8, among the composites with different amounts of Al2ZrO5 cast at two different temperatures, the one which has maximum hardness shows the highest compressive strength (1.5% Al2ZrO5, 850 °C). Therefore, it can be concluded that composite with 1.5% Al2ZrO5 content cast at 850 °C represents the maximum mechanical properties and it can be considered as the optimum fabrication condition.
4 Conclusions
1) A356 aluminum alloy reinforced with Al2ZrO5 nanocomposite powders is fabricated via stir casting method. Reinforcing particles are well distributed in the matrix of composites. However, partial agglomeration is observed in composites with increasing Al2ZrO5 content.
2) The mechanical properties such as hardness and compressive strength are improved. Also, composite containing 1.5% Al2ZrO5 fabricated at 850 °C shows improved hardness and compressive strength, in comparison with other specimens.
References
[1] PARK S, VOHS J M, GORTE R J. Direct oxidation of hydrocarbon in a solidoxide fuel cell [J]. Nature, 2000, 404(6775): 265-269.
[2] LI Y W, HE D H, CHANG Z X, SU C L, LI J R, ZHU M J. Effect of calcium salts on isosynthesis over ZrO2 catalysts [J]. Molecular Catalysis A, 2001, 175(1-2): 267-271.
[3] SUBBARAO E C, MAITI H S. Science and technology of zirconia [J]. Advanced Ceramics, 1988, 24(1): 731-737.
[4] ZHANG Q, SHEN J, WANG J, WU G, CHEN L. Sol-gel derived ZrO2-SiO2 highly reflective coatings [J]. Inorganic Materials, 2000, 2(4): 319-322.
[5] WRIGHT P K, EVANS A G. Mechanisms govering the performance of thermal barrier coating [J]. Current Opinion in Solid State & Materials Science, 1999, 4(1): 25-30.
[6] PICONI C, MACCAURO G. Zirconia as a ceramic biomaterial [J]. Biomaterials, 1991, 20(1): 1-6.
[7] SALAS P, ROZA-CRUZ E D, DIAZ-TORRES L A, CASTANO V M, MELENDREZ R, BARBOZA-FLORES M. Monoclinic, ZrO2 as a broad spectral response thermoluminescence UV desmeter [J]. Radiation Measurements, 2003, 37(2): 187-193.
[8] TUAN W H, CHEN J R, HO C J. Critical zirconia amount to enhance the strength of alumina [J]. Ceramics International, 2008, 34(8): 2129-2133.
[9] KUMARI L, DU G H, LI W Z, SELVA VENNILA R, SAXENA S K, WANG D Z. Synthesis, microstructure and optical characterization of zirconium oxide nano structures [J]. Ceramics International, 2009, 35(6): 2401-2408.
[10] PRADEEP A, PRIYADHARSINI P, CHANDRASEKARAN G. Production of single phase nano size NiFe2O4 particles using sol-gel auto combustion route by optimizing the preparation conditions [J]. Materials Chemistry and Physics, 2008, 112(2): 572-576.
[11] SOMIYA S, AKIBA T. Hydrothermal zirconia powders: A bibliography [J]. European Ceramic Society, 1999, 19(1): 81-85.
[12] JIANG Xian-liang, BOULOS M. Induction plasma spheroidization of tungsten and molybdenum powders [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(1): 13-17.
[13] HANNA J, ATTIA Y A. Advances in fine particle processing [M]. New York: Elsevier Publishers, 1990: 472.
[14] BOIRE-LAVIGNE S, MOREAU C, SAINT-JACQUES R G. Taguchi analysis of the influence of plasma spray parameters on the microstructure of tungsten coatings [C]//International Symposium on Developments and Applications of Ceramics and New Metal Alloys. Quebec City, 1993: 473-485.
[15] SAHIN Y. The effect of sliding speed and microstructure on the dry wear properties of metal-matrix composites [J]. Wear, 1998, 214(1): 98-106.
[16] ZAMZAM M, ROS D, GROSCH J. Fabrication of P/M in situ fibre composite materials. Part I: Formation of fibrous structure [J]. Key Engineering Materials, 1993, 79-80(1): 235-246.
[17] COOKE P S, WERNER P S. Pressure infiltration casting of metal matrix composites [J]. Materials Science & Engineering A, 1991, 44(1-2): 189-193.
[18] LYOD D J. Particle-reinforced aluminium and magnesium matrix composites [J]. International Materials Reviews, 1994, 39(1): 1-23.
[19] MANSUR L J, MORTENSEN A, CORNIC J A, FLEMINGS M C. Infiltration of fibrous performs by a pure metal. Part II: Experiment [J]. Metallurgical Transactions A, 1998, 20(4): 2547-2553.
[20] EDITOR MILLS K. ASM Handbook: Metallographic techniques and Microstructures Al alloys [M].Formerly Ninth Edition. New York: ASTM International, 1992: 354.
[21] The american society for testing materials; standard test method of tension testing wrought and cast aluminum and magnesium alloy products—B557. Handbook of ASTM Standards [M]. Philadelphia, USA: ASTM International, 1994: 91.
[22] SPOWART J E, MARUYAMA B, MIRACLE D B. Multi-scale characterization of spatially heterogeneous systems: Implications for discontinuously reinforced metal–matrix composite microstructures [J]. Materials Science and Engineering A, 2001, 307(1-2): 51-66.
[23] DAS S, DAS S, DAS K. Abrasive wear of zircon sand and alumina reinforced Al-4.5wt% Cu alloy matrix composites – A comparative study [J]. Composite Science and Technology, 2008, 67(3-4): 746-751.
[24] ZHANG Q, ZHANG H, GU M, JIN Y. Studies on the fracture and flexural strength of Al/Sip composite, [J]. Materials Letters, 2004, 58(27-28): 35-45.
[25] GANESH V V, CHAWLA N. Effect of particle orientation anisotropy on the tensile behavior of metal matrix composites: experiments and microstructure-based simulation [J]. Materials Science and Engineering A, 2005, 391(1-2): 342-353.
溶胶-凝胶自蔓燃法制备Al2ZrO5纳米颗粒增强铝基复合材料
S. A. KHORRAMIE1, M. A. BAGHCHESARA2, D. P. GOHARI3
1. Department of Chemistry, North Tehran Branch, Islamic Azad University, Tehran 14358-84381, Iran;
2. Department of Metallurgy & Materials Engineering, Masjed Soleiman Branch, Islamic Azad University, Masjed Soleiman 64915-111, Iran;
3. Young Researchers and Elite Club, South Tehran Branch, Islamic Azad University, Tehran 14358-84381, Iran
摘 要:采用金属硝酸盐和甘氨酸做前驱体,通过溶胶–凝胶自蔓燃法和超声波辐射合成颗粒尺寸为38 nm的Al2ZrO5纳米颗粒。制备过程包括均匀溶胶的形成、凝胶干燥和干燥凝胶的燃烧。在不同温度下,通过搅拌铸造制备Al2ZrO5含量分别为0.75%、1.5% 和 2.5%(体积分数)的纳米颗粒增强铝基复合材料。通过SEM和XRD对所制得的复合材料进行结构和相分析。通过评估材料的密度、硬度和抗压强度确定最佳的强化相含量和铸造温度。实验结果表明:Al2ZrO5纳米颗粒增强铝基复合材料的硬度和抗压强度得到提升,其最大值分别达到BHN 61和 900 MPa。在850 °C下制备的含有1.5% Al2ZrO5的复合材料具有最佳的力学性能。
关键词:金属基复合材料;Al2ZrO5;纳米结构;自蔓燃;超声波;搅拌铸造;力学性能
(Edited by Jing-hua FANG)
Corresponding author: M. A. BAGHCHESARA; E-mail: amsara2000@gmail.com
DOI: 10.1016/S1003-6326(13)62630-8