
Reaction diffusion in continuous SiC fiber reinforced Ti matrix composite
L? Xiang-hong(吕祥鸿), YANG Yan-qing(杨延清),
HUANG Bin(黄 斌), LUO Xian(罗 贤), LIU Yu-cheng(刘玉成)
State Key Laboratory of Solidification Processing, School of Materials Science and Engineering, Northwestern Polytechnical University, Xi’an 710072, China
Received 13 June 2006; accepted 15 November 2006
Abstract: SiC continuous fiber-reinforced pure Ti(TA1) matrix composites were fabricated by a vacuum hot pressing(VHP) method and then heat-treated in vacuum under different conditions. The interfacial reaction and the formation of interfacial phases were studied by using SEM, EDS and XRD. The results show that there exists reaction diffusion at the interface of SiC fibers and Ti matrix, and the concentration fluctuation of reaction elements such as C, Ti and Si appears in interfacial reaction layer. The interfacial reaction products are identified as Ti3SiC2, TiCx and Ti5Si3Cx. At the beginning of interfacial reaction, the interfacial reaction products are TiCx and Ti5Si3Cx. Along with the interfacial reaction diffusion, Ti3SiC2 and Ti5Si3Cx single-phase zones come forth in turn adjacent to SiC fibers, and the TiC+Ti5Si3Cx double-phase zone appears adjacent to Ti matrix, which forms discontinuous concentric rings by turns around the fibers. The formed interfacial phases are to be Ti3SiC2, Ti5Si3Cx and TiCx+Ti5Si3Cx from SiC fiber to Ti matrix. The interfacial reaction layer growth is controlled by diffusion and follows a role of parabolic rate, and the activation energy (Qk) and (k0) of SiC/TA1 are 252.163 kJ/mol and 7.34×10-3 m/s1/2, respectively.
Key words: SiC/TA1composite; reaction diffusion; interfacial phases; activation energy
1 Introduction
Due to their high specific strength and specific modulus, SiC fiber reinforced Ti alloy-matrix composites (TMCs) can be used for aero-engines[1]. The interfacial reaction taking place during the composite consolidation and high temperature service lead to the formation of some brittle compounds distributed in several layers at the interface. The brittle interfacial reaction products become the crack origination that makes the interface lose its stability and the ability of transfering the applied load[2].
Numerous studies have tried to analyze the interfacial reaction diffusion in Ti matrix composites reinforced with SiC continuous fiber[3-7]. The emphases are usually focused on the reaction thermodynamics between the matrix and the reinforcement, the growth dynamics of interfacial reaction product layer, the diffusion barrier coatings as well as the reaction product layers that prevent the diffusion of Ti, Si and C elements. Due to the different types of SiC fiber and Ti alloy matrix, there are many controversies in the formation types and formation sequence of the interfacial reaction products. For example, the interfacial reaction products from fiber to matrix are usually Ti3SiC2, Ti5Si3Cx, TiCx and Ti3Si for the SiC fiber without C coating[8-9]. For the SiC continuous fiber containing C-coating, before the C-coating is consumed completely, the interfacial reaction product is only TiCx[10]. For the SiC fiber coated by a C-rich layer containing SiC particles, such as SCS-6 SiC fiber, the interfacial reaction products are identified as TiCx, Ti5Si3Cx, TiCx and Ti3Si from the fiber to matrix[3,11]. For the selection of Ti alloy matrix, theoretically, all α, α+β and β Ti alloys can be considered the matrix materials. But when considering the fabricating technology and machining capability, α+β and β Ti alloys are usually chosen, i.e., Ti15V3Cr and Ti6Al4V. In addition, some Ti-Al intermetallic compounds such as Ti3Al and Ti2AlNb, having not only the oxidation resistance at high temperature, but also the ability of being rolled into foil, can be selected as matrix materials. Due to the different reaction intensity degrees between SiC fiber and the matrix, the interfacial reaction and the formation of interfacial products are different from each other.
In order to study the interfacial reaction as well as the formation of the interfacial reaction products between SiC fiber and Ti matrix, industrial pure Ti was used as the matrix, which was reinforced by SiC continuous fiber with no C-coating. The composite was fabricated and heat treated under different conditions, and the morphology and elemental distributions as well as components of interfacial reaction zone were analyzed with the SEM, EDS and XRD methods. The aim of the investigation is to forecast the formation of interfacial reaction products in SiC/Ti composite and the diffusion path of reactive elements, as well as to provide the basis for studying the interfacial reaction in Ti alloys and Ti-Al intermetallic compounds matrix composites reinforced with SiC continuous fiber.
2 Experimental
Continuous β-SiC fibers with no C-coating produced by a chemical vapour deposition(CVD) process were used as reinforcements. The fiber was about 100 ?m in diameter. The matrix material used for this experiment was industrial pure Ti (named TA1) foils with a thickness of about 50 ?m. Unidirectionally aligned composites of TA1 reinforced with SiC fibers were fabricated by solid-state foil-fiber-foil consolidation technique using a vacuum hot pressing(VHP) process to full density. The VHP process was done at 850 ℃ under a pressure of 50 MPa for 40 min. After its fabrication, the composites were heat-treated in vacuum at 800, 900 and 1 000 ℃, respectively.
Samples were cut from composites perpendicular to the fiber direction with a low-speed diamond saw. The cross section was prepared by grinding step by step with 400, 600, 1 000 and 1 200 grit silicon carbide metallographic papers, polishing with 1 ?m diamond paste, then etching for about 2 s with a solution made of 1 mL HF, 3 mL HNO3 and 7 mL H2O, and cleaning with water immediately. After sipping up water with filter paper, the samples were immerged in acetone or absolute alcohol for 3-5 min to get rid of water. Finally the dehydrated samples were dried by hot air, encased with qualitative filter paper and put into an airer for use later.
The morphologies of interfacial reaction products were examined with JSM 6460 scanning electron microscopy(SEM). The phases in the composites were identified by X-ray micro-area diffraction(XRD) equipped with Panalytical X’Pert PRO and the diameter of the micro-area was 100 ?m. The elements’ contents were analysed with Oxford INCA7574 energy dispersive spectrometer(EDS).
3 Results and discussion
3.1 Interfacial reaction of SiC/TA1
Figs.1-3 show the interfacial reaction zone morphologies of SiC/TA1. During the course of heat-treatment of the composites, the interfacial reaction zone becomes thicker and thicker along with prolonged time and enhanced temperature. At the same time, the interfacial reaction products tend to form discontinuously concentric rings around the fiber, which can be observed clearly in Figs.2 and 3. Fig.4 shows the EDS surface analysis result of interfacial reaction zone, and it can be found the interfacial reaction layers are formed by turns with carbide and silicide of titanium.

Fig.1 SEM micrographs of interfacial reaction zone of SiC/TA1 composite: (a) 800 ℃ for 94 h; (b) 800 ℃ for 206 h; (c) 800 ℃ for 500 h

Fig.2 SEM micrographs of interfacial reaction zone of SiC/TA1 composite: (a) 900 ℃ for 73 h; (b) 900 ℃ for 150 h; (c) 900 ℃ for 240 h

Fig.3 SEM micrographs of interfacial reaction zone of SiC/TA1 composite: (a) 1 000 ℃ for 40 h; (b) 1 000 ℃ for 91 h; (c) 1 000 ℃ for 260 h

Fig.4 EDS surface analysis results of interfacial reaction zone (1 000 ℃ for 91 h): (a) C; (b) Si; (c) Ti; (d) Grey image
Figs.5-7 are EDS line and point analysis results of interfacial reaction zone. Seen from the figures, the concentration fluctuation of reactive elements such as C, Ti and Si appears in interfacial reaction layers, which indicates that the Si and C atoms diffuse from SiC fiber to the matrix, and the Ti atoms diffuse in the opposite direction. From EDS line analysis result as well as Tables 1 and 2, the interfacial reaction zone is mostly TiCx+ Ti5Si3Cx duplex phase zone, and the ternary phase Ti3SiC2 may come into being adjacent to SiC fiber. As soon as the annealing time is long enough, the interfacial reaction layer is thick enough, and the Ti5Si3Cx single phase zone appears between the ternary Ti3SiC2 and Ti5Si3Cx+TiCx duplex phase zone.
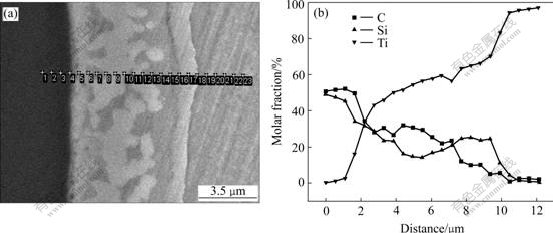
Fig.5 Position and EDS line analysis result of interfacial reaction zone (800 ℃ for 500 h): (a) SEM micrograph; (b) Linear analysis

Fig.6 Position and EDS point analysis result of interfacial reaction zone (900 ℃for 240 h): ⅠTi5Si3Cx+TiCx;Ⅱ Ti5Si3Cx; Ⅲ Ti3SiC2

Fig.7 Position and EDS point analysis result of interfacial reaction zone (1 000 ℃ for 260 h)
Table 1 EDS point analysis results of interfacial reaction layers of SiC/TA1 (mole fraction, %)

Table 2 EDS point analysis results of interfacial reaction layers of SiC/TA1(molar fraction, %)

Figs.8-10 show the X-ray micro-area diffraction patterns of the interfacial reaction zone, and it indicates obviously that in all cases the interface phases existing in the composites contain TiCx and Ti5Si3Cx. Along with the enhanced annealing temperature and time, the diffraction intensity of Ti3SiC2 becomes more and more obvious. When it is heat treated at lower temperature, the diffraction peak is very tiny, or even does not appear. As shown in Fig.8, when SiC/TA1 composite is heat treated at 800 ℃ for 206 h, there is no Ti3SiC2 diffraction peak.

Fig.8 X-ray micro-area diffraction patterns of SiC/TA1 after heat treatment at 800 ℃

Fig.9 X-ray micro-area diffraction patterns of SiC/TA1 after heat treatment at 900 ℃

Fig.10 X-ray micro-area diffraction patterns of SiC/TA1 after heat treatment at 1 000 ℃
3.2 Formation of interface phase
The thermochemical compatibility of fiber/matrix interface is of primary concern in SiC continuous fiber-reinforced Ti matrix composites. Interface stability in multi-component system is controlled by a number of chemical, thermodynamic and kinetic factors[12]. The interfacial reaction products of SiC/TA1 were identified as Ti3SiC2, TiCx and Ti5Si3Cx, and C atoms may carry on long distance diffusion to react with the matrix or form interstitial solid solution[13]. Reaction of C and Si with Ti matrix is thermodynamically favored under the present consolidation and heat-treatment conditions:
C+Ti→TiC (1)
3Ti+Si+2C→Ti3SiC2 (2)
3SiC+8Ti→3TiC+Ti5Si3 (3)
5Ti+3Si→Ti5Si3 (4)
For SiC/ TA1 composite the free energy change of the four reactions above are about -170, -1 356, -895.4, -578.5 kJ/mol at 1 000 K[14], respectively. Hence, from the point of view of thermodynamics, the reactions are spontaneous between the SiC fibers and the matrix during the consolidation and the heat treatment of the composites.
Fig.11 shows the schematic representation of interface reaction products formation of SiC/TA1 composite. Generally, the interfacial reaction is a reaction-controlled process in its very early stage, and the interfacial products are Ti5Si3Cx and TiCx according to Eqn.(3), TiCx is adjacent to Ti side, while Ti5Si3Cx is adjacent to SiC side, respectively[15]. As long as the interfacial reaction layer becomes thicker, its growth is controlled by diffusion, and Ti, Si and C atoms inter-diffuse through interfacial reaction layer. Since the initial TiCx isn’t continuous, the inner Ti5Si3Cx and the outer TiCx contact with Ti matrix simultaneously. That is, the diffusion from SiC fiber to Ti matrix may through Ti5Si3Cx and TiCx, or only through Ti5Si3Cx. At the same time, the diffusion coefficient of Ti atoms is much less than that of C and Si atoms[16-17]. Consequently, the advanced speed of the reaction layer/matrix interface may be controlled by the diffusion of Ti atoms. Only a few of Ti atoms diffusing out can the zone in front of reaction layer/matrix be come to the concentration points of Ti5Si3Cx and TiCx in Ti-Si-C ternary phase diagram. Therefore, as soon as the concentration in front of the Ti5Si3Cx/Ti interface comes to that of TiCx, then TiCx forms subsequently. In the same way, when the concentration in front of the TiCx/Ti interface comes to that of Ti5Si3Cx, then Ti5Si3Cx forms. Thus the interfacial reaction products layer appears to be discontinuously concentric rings around the fibers. Along with the diffusion of C and Si atoms and the Ti5Si3Cx layer becoming even thicker, there are no more Ti atoms arriving at SiC/Ti5Si3Cx interface, but excessive C atoms pile up there, so as to form the ternary phase, Ti3SiC2. In addition, C atoms may carry on long distance diffusion to react with the matrix and form TiC in matrix. The formed interfacial phases are SiC, Ti3SiC2, Ti5Si3Cx, TiCx+Ti5Si3Cx and Ti from SiC fiber to Ti matrix.

Fig.11 Schematic representation of interface reaction product formation of SiC/TA1 composite (Arrows show diffusion way of C and Si)
3.3 Growth kinetics of interfacial reaction products of SiC/TA1 composite
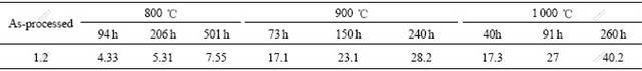
From Figs.1-3, it can be seen that the thickness of the reaction layer becomes thicker and thicker along with the enhanced heat-treated temperature and prolonged heat-treated time. The measured thickness of the interfacial reaction layer in SiC/TA1 composite is summarized in Table 3. The temperature is more effective than the time in the reaction layer growth, for example, the reaction layer thickness of 17.3 ?m is measured in SiC/TA1 heat-treated at 1 000 ℃ for 40 h, but it is only 7.55 ?m at 800 ℃ even for 500 h.
Table 3 Thickness of interfacial reaction layer (μm)
As mentioned above, the interfacial reaction is reaction-controlled in the early stage but soon becomes diffusion-controlled when a thin reaction layer forms. A diffusion-controlled growth may be satisfied with a parabolic law[8,13]:
l=kt1/2+b0 (5)
where l is the thickness of the interfacial reaction layer, k is a rate constant which is related to the diffusion coefficient of the components and to the thermodynamic properties of the interfacial reaction products, t is time and b0 is the original thickness of the reaction layer in the as-processed samples.
Fig.12 shows the relationship between the thickness of the reaction layer and the square root of the reaction time at different temperatures for SiC/TA1 composites.
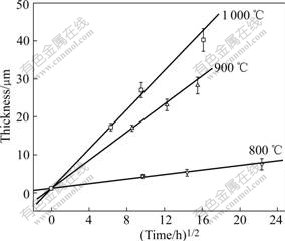
Fig.12 Interfacial reaction kinetic curves of SiC/TA1
The data points for each treatment temperature are fitted with a straight line to estimate a generalized reaction rate constant. The fitted equations are
l=0.281 6t1/2+1.331 39 (800 ℃) (6)
l=1.070 103t1/2+1.695 18 (900 ℃) (7)
l=2.474 08t1/2+2.389 47 (1 000 ℃) (8)
The thickness l is in ?m, and time t is in hour for the three equations.
These equations provide quantitative descriptions for the interfacial reactions of the composites, from which the reaction results at a specific time and temperature can be calculated. Furthermore, the fact that the k value increases with the increase of the reaction temperature reveals the importance of fabricating these composites at a lower temperature.
In addition the rate constant k in Eqn.(5) follows Arrhenius relation[8]:
k=k0exp(-Qk/2RT) (9)
where k0 is the pre-exponential factor, which is also related to the diffusion coefficient and to the thermodynamic properties, Qk is the growth activation energy, R is the gas constant (R=8.346 2 J/K), and T is the temperature.
Fig.13 shows the Arrhenius plot of the parabolic rate constant of SiC/TA1. The values k0 and Qk calculated according to Eqn.(6) are 252.163 kJ/mol, 7.34×10-3 m?s-1/2, respectively. The values are well fitted with those calculated by MARTINEAU[8].

Fig.13 Arrenius plot for parabola rate constant of SiC/TA1 composites
4 Conclusions
1) The interfacial reaction takes place between SiC reinforcement and Ti matrix, and the interfacial reaction products are identified as Ti3SiC2, TiCx and Ti5Si3Cx.
2) At the beginning of interfacial reaction, the interfacial reaction products are TiCx and Ti5Si3Cx, in which TiCx is discontinuous. Along with the enhanced heat-treatment temperature and prolonged heat-treatment time, Ti5Si3 forms in front of the TiCx/Ti interface, and TiCx forms in front of the Ti5Si3Cx/Ti interface, thus the interfacial reaction product layer presents the discontinuously concentric rings around the fibers. At the same time, the ternary phase, Ti3SiC2, may come into being between SiC fiber and the reaction products.
3) The interfacial reaction layer growth is controlled by diffusion and follows a role of parabolic rate, and the activation energy Qk and k0 of SiC/TA1 are 252.163 kJ/mol and 7.34×10-3 m?s-1/2, respectively.
References
[1] LU Pan-quan, ZHOU Sheng-nian. Research progress in continuous fiber reinforced titanium alloy matrix composites [J]. Aviation Production Engineering, 1994(6): 35-37. (in Chinese)
[2] YANG Y Q, DUDEK H J, KUMPFERT J. Interfacial reaction and stability of SCS-6SiC/Ti-25Al-10Nb-3V-1Mo composites [J]. Mater Sci Eng A, 1998, A246: 213-220.
[3] YANG Y Q, DUDEK H J, KUMPFERT J. Interface stability in SCS-6 SiC/Super α2 composites [J]. Scripta Materialia, 1997, 37(4): 503-510.
[4] MOGILEVSKY P, WERNER A, DUDEK H J. Application of diffusion barriers in composite materials [J]. Mater Sci Eng A, 1998, A242: 235-247.
[5] YANG Yan-qing, MA Zhi-jun, LI Jian-kang, L? Xiang-hong, AI Yun-long. Interfacial reaction and its effects on mechanical property of SiCf/super α2 composites [J]. Rare Metal Materials and Engineering, 2006, 35(1): 43-46. (in Chinese)
[6] ZHU Yan, YANG Yan-qing, SUN Jun. Calculation of activity coefficients for components in ternary Ti alloys and intermetallics as matrix of composites [J]. Trans Nonferrous Met Soc China, 2004, 14(5): 875-879.
[7] ZHU Yan, YANG Yan-qing. Activity coefficients in Ti alloys and intermetallics [J]. Rare Metal Materials and Engineering, 2003, 32(8): 600-603. (in Chinese)
[8] MARTINEAU P, PAILER R, LAHAVE M, NASLAIN R. SiC filament/titanium matrix composites regarded as model composites (Part 2): Fibre/matrix chemical interactions at high temperatures [J]. Journal of Materials Science, 1984, 19: 2749-2770.
[9] YANG Yan-qing, MA Zhi-jun, L? Xiang-hong. Microstructure and Micromechanics of the interface of TiC(f)/Ti-6Al-4V composites [J]. Rare Metal Materials and Engineering, 2006, 35: 301-304. (in Chinese)
[10] ZHANG G X, KANG Q, SHI N L, LI G P, LI D. Kinetics and mechanism of interfacial reaction in a SiCf/Ti composite [J]. J Mater Sci Technol, 2003, 19(5): 407-410.
[11] L? Xiang-hong, YANG Yan-qing, MA Zhi-jun, LI? Cui-xia, CHENG Yan, AI Yun-long. Kinetics and mechanism of interfacial reaction in SCS-6 SiC continuous fiber-reinforced Ti-Al intermetallic matrix composites [J]. Trans Nonferrous Met Soc China, 2006, 16(1): 77-83.
[12] XUN Y W, TAN M J, ZHOU J T. Processing and interface stability of SiC fiber reinforced Ti-15V-3Cr matrix composites [J]. Journal of Materials Processing Technology, 2000, 102: 215-220.
[13] YANG Y Q, DUDEK H J, KUMPFERT J. TEM investigations of the fiber/matrix interfaces in SCS-6 SiC/Ti-25Al-10Nb-3V-1Mo composites [J]. Composites Part A, 1998, 29A: 1235-1241.
[14] ZHU Yan. Study on the Interfacial Reactions of SiC Fiber Reinforced Ti-Matrix Composites [D]. Xi’an: Northwestern Polytechnical University, 2003. (in Chinese)
[15] NAKA M, FENG J C, SCHUSTER J C. Phase reaction and diffusion path of SiC/Ti system [J]. Metallurgical and Materials Transactions A, 1997, 28A: 1385-1390.
[16] SARIAN S. Diffusion of Ti in TiCx [J]. Journal of Applied Physics, 1969, 40(9): 3515-3520.
[17] VAN LOO F J J, BASTIN G F. On the diffusion of carbon in titanium carbide [J]. Metallurgical Transaction A, 1989, 20A: 401- 411.
Foundation item: Project(50371069) supported by the National Natural Science Foundation of China; Project(20030699013) supported by the State Education Ministry Doctoral Foundation of China; Project(04G53044) supported by the Foundation of Aviation Science of China
Corresponding author: L? Xiang-hong; Tel: +86-29-88486091; E-mail: lxhong71@sina.com.cn
(Edited by LI Xiang-qun)