J. Cent. South Univ. Technol. (2008) 15: 318-323
DOI: 10.1007/s11771-008-0060-5
Surface modification of magnesium hydroxide by γ-aminopropyltriethoxysilane
LUO Wei(罗 伟), FENG Qi-ming(冯其明), OU Le-ming(欧乐明), LIU Kun(刘 琨)
(School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China)
Abstract: Magnesium hydroxide(MH), which is commonly used as a halogen-free flame retardant filler in composite materials, was modified by silanization reaction with γ-aminopropyltriethoxysilane (γ-APS) in aqueous solution at different pH values (pH range from 8.0 to 12.0). The surface properties of grafted γ-APS on MH surface as a function of solution pH value were studied using elemental analysis, Fourier transform infrared spectroscopy and zeta potential measurement. The results show that hydrolysis and condensation of γ-APS are activated in alkaline solution and lead to multilayer adsorption of γ-APS molecules on the surface of MH. The type of adsorption orientation of γ-APS on MH surface is a function of coverage density that is altered by changing solution pH value. At low coverage density (e.g. 55 nm-2), γ-APS molecules are preferentially adsorbed to the surface with the silicon moiety towards the surface, and increasing coverage density (e.g. 90 nm-2) leads to parallel orientation. At an even higher coverage density (e.g. 115 nm-2), γ-APS molecules bond to the surface with the amino moiety towards the surface.
Key words: magnesium hydroxide; γ-aminopropyltriethoxysilane; surface modification; orientation
1 Introduction
Magnesium hydroxide(MH) has been applied in numerous areas including antiacids, absorbents, fertilizer additives[1], filler materials[2], precursors for magnesium oxide[3], flame retardants[4] and smoke suppressants[5]. In the field of halogen-free flame retardant composites, MH is widely used by virtue of its high decomposition temperature (300-320 ℃)[6].
The disadvantage of this filler, however, is the high level (more than mass fraction of 60%) required to achieve the desired flame retardant effect[7]. Such high level causes filler-matrix interfacial properties to be the major determinant of composite mechanical properties. Addition of fillers in such high contents affects nega- tively the rheological and mechanical properties of the composite, such as increasing melt viscosity, reducing elongation at break and impact strength[6]. In order to minimize this effect, surface modification of MH is an important method to improve their compatibility. It has been reported that MH can be successfully modified with fatty acids, unsaturated polymeric acids[8], titanate coupling agents[9] and silanes[10]. Although there have been some reports about the preparation of silane-modified MH particles, the study of the surface properties of this kind of material still remains to be a challenge. In addition, the effect of functional groups on the surface properties of the particles is not clearly understood. In this work, MH particles were modified by γ-aminopropyltriethoxysilane (γ-APS) and various experimental techniques were used to investigate the surface properties of the modified particles.
2 Experimental
2.1 Materials and procedures
2.1.1 Materials
MH powder (>98%, mass fraction) from Shandong Haihua Company Limited was further dried in a vacuum oven at 100 ℃ for 24 h prior to each silanization experiment in order to remove any physisorbed water from the surface. The average particle size with an average surface area of 32.23 m2/g was determined to be 9.83 μm by CILAS laser particle size analyzer based on the BET method using nitrogen as carrying gas. γ-APS 95% purchased from Wuhan Huachang Applied Technology Institute was used as received. All inorganic chemicals were of analytical grade. Deionized water was used in all of the experiments.
2.1.2 Synthesis of modified magnesium hydroxide
In a 500 mL three-neck flask, the initial pH value of 100 mL water was adjusted by NaOH solution. Then an excess γ-APS (10 mL, about 40 mmol) was added into the solution under vigorous stirring for 30 min. After that, 10 g MH was mixed with the solution. The reaction mixture was stirred at 25 ℃ for 4 h. Excess γ-APS was removed by filtration and washed with ethanol, and then with deionized water. The filter cake was dried under vacuum at 100 ℃ for 4 h. The modified materials were aminopropyl-magnesium hydroxide(APMH).
2.2 Characterization
2.2.1 Elemental analysis
The mass fraction of N in APMH was analyzed by using a Nitrogen/oxygen tester (TC 436, LECO, USA). From the obtained N content in APMH, the amount of γ-APS per gram of APMH can be calculated directly.
2.2.2 FTIR
Fourier transform infrared(FTIR) spectrum analysis of samples was carried out by using a Nicolet NEXUS 670 spectrophotometer with special KBr discs prepared after mixing 1% sample with high-purity KBr. Analysis was performed in the transmittance mode in the wavenumber range of 4 000 - 400 cm-1, with a resolu- tion of 2 cm-1 and accumulation of 32 scans.
2.2.3 Zeta potential
A zeta potential analyzer (Delsa 440sx, Beckman Coulter, USA) was used to measure ζ-potentials of MH and APMH particles at (25±1) ℃. The particles were suspended in aqueous solution (0.4 g/L) for 2 h. In all experiments an ionic strength of 0.01 mol/L was kept with NaCl. For each sample, approximately 50 mL of supernatant was used to fill the cell. The ζ-potential data were based on the analysis of three measurements on each sample.
3 Results and discussion
3.1 Elemental analysis
In the studied pH range in water, amino silanes hydrolyze into silanol (SiOH group) rapidly, whereas the rate of the condensation reaction of the silanol into siloxane (Si―O―Si) obviously depends on the pH value. In acidic pH range, the hydrolysis process of silane is essentially activated whereas in basic pH range both hydrolysis and condensation are activated[11]. The coverage density of γ-APS molecules on the surface of MH particles can be calculated by following formula:
 (1)
(1)
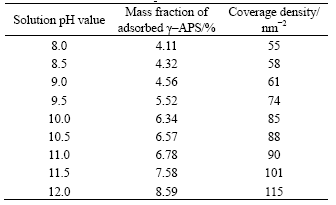
where ρ, w, SBET, Ar(N) and NA are the coverage density of γ-APS molecules on the surface of MH, the mass fraction of adsorbed N atoms in γ-APS, the BET surface area of MH particles, the relative atomic mass of N and Avogadro constant, respectively. From Table 1, it can be seen that the coverage density increases with increasing pH value of solution. This may be due to the structure of the coating layer, involving multilayer formation or a change in the molecular orientation on the surface[12]. Taking into account the fact that one γ-APS molecule covers approximately 0.5 nm2[13] and the results in Table 1, it suggests that both hydrolysis and condensation of γ-APS are activated in alkaline solution and lead to multilayer adsorption of γ-APS molecules on the surface of MH.
Table 1 Coverage density of γ-APS molecules on APMH particles at different solution pH values
3.2 FTIR characterization
The molecular structure of silane is a function of the solution pH value and differences in the molecular structure of the silane may greatly affect the reactivity of functional groups. The structure of usual silane in aqueous solution has been studied in various environments by FTIR spectroscopy[14].
The FTIR spectrum of γ-APS is given in Fig.1(a). The bands at 3 370 and 3 297 cm-1 are assigned to ―NH2 asymmetric and symmetric stretching, respectively, whereas the weak band at 3 179 cm-1 may be due to hydrogen bonded amine groups. The bands at 2 974 and 2 885 cm-1 are characteristics of ―CH3 asymmetric and symmetric stretching vibrations in the ethoxy group of γ-APS[15] and the band at 2 927 cm-1 is assigned to asymmetric stretching mode of ―CH2 in alkyl and ethoxy group. The broad band at 1 577 cm-1 is assigned to the bonding mode of free amine groups that are the characteristics of γ-APS. The bands at 1 167, 1 103, 1 079 and 959 cm-1 are the characteristics of Si―O―C vibrations.
The FTIR spectrum of γ-APS after hydrolysis is shown in Fig.1(b). The bands at 2 974 and 2 885 cm-1 that are characteristics of ―CH3 asymmetric and symmetric stretching vibrations in the ethoxy group of γ-APS disappear after silanization in water. The characteristic bands of Si―O―C in γ-APS at 1 167, 1 103, 1 079 and 959 cm-1 also disappear. Two new broad bands at 1 118 and 1 039 cm-1, characteristics of siloxane bonds, appear instead, suggesting that most ethoxy groups are hydrolyzed.
The FTIR spectra of MH and APMH samples modified at different pH values are shown in Fig.2. In

Fig.1 FTIR spectra of samples: (a) γ-APS; (b) γ-APS after hydrolysis
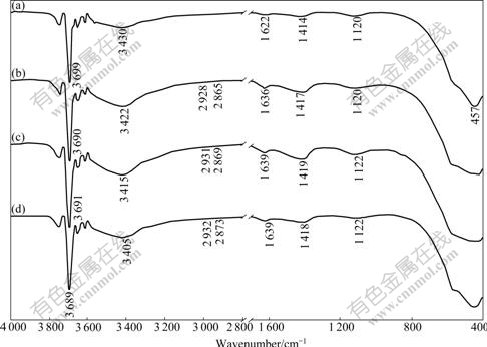
Fig.2 FTIR spectra of samples: (a) MH; (b) APMH modified at pH=8.0; (c) APMH modified at pH=10.9; (d) APMH modified at pH=12.0MH samples (Fig.2(a)), the strong peak at around 457 cm-1 is assigned to the Mg―O stretching vibration. The band at 3 699 cm-1 is assigned to the hydroxyl stretch of MH[16]. The band at 1 622 cm-1 is attributed to the ―OH stretching mode in water[17]. A broad band at 3 430 cm-1 is attributed to the surface adsorbed ―OH groups mentioned above[18].
As shown in Figs.2(b)-(d), the band at 3 430 cm-1, characterized by the effect of ―OH stretching vibration, is shifted after interaction with γ-APS molecules. This indicates the formation of hydrogen bonds through the N atom of the amino group with water[19]. From the comparison of APMH and γ-APS, it is easy to see the disappearance of peak at 2 974 cm-1 (Fig.1(a)) that is assigned to the ethoxysilane. For APMH with the dis- appearance of the peak at 2 974 cm-1, the bands at 2 926 and 2 861 cm-1 (Fig.1(b)) assigned to the asymmetric and symmetric stretching of ―CH2 in alkyl chain are shifted. Note that the band at 3 370 cm-1 (Fig.1(a)) assigned to free ―NH2 asymmetric stretching disappears on the spectra of MH silanized in water because condensation of γ-APS is activated in alkaline solution.
As can be understood from the indication of spectroscopic results, the organosilane molecules adsorbed on MH surface are coordinated to the Lewis acidic sites or surface hydroxyls by H-bonding interaction through nitrogen lone pairs.
3.3 Zeta potential
Since bifunctional silane γ-APS is able to be adsorbed to the surface via both functional groups creating different possible orientations for an adsorbed molecule within an organosilane film, three distinct pH adsorption regimes, i.e. pH ranges of 8-10, 10-11 and 11-12 (see Fig.3).
3.3.1 MH modified by γ-APS in pH range of 8-10
In pH range of 8-10, it is hypothesized that the molecules are preferentially adsorbed to the surface with the silicon moiety towards the surface by long range electrostatic attraction according to the known electro- chemical behavior of the adsorption system[15]. The pH value of the isoelectric point(IEP) of MH is found to be 10.9 (Fig.3(a)), which is in good agreement with electrophoretic measurements of POKROVSKY and SCHOTT[20] who reported pHIEP=11. From the literature about water suspension of brucite[20], the surface will be effectively positively charged in pH range of 8-10 with dominant species of MgOH2+, electrically neutral in pH range of 10-11 with dominant species of MgOH0 and negatively charged in pH range of 11-12 with dominant species of MgO-.
Supposing the pKa values of the silanol and amine moieties equal 3-4 and 10-11, respectively[21], the silanol group will be negatively charged throughout the pH range of interest, whereas the charge of the amine group will change from positive to neutral as pH changes

Fig.3 Influence of solution pH value on ζ-potential at 25 ℃ in 0.01 mol/L NaCl solution (Vertical dashed line denotes pH value of the IEP of MH particles, and errors are calculated from standard deviations of the measurements): (a) MH; (b) APMH modified at pH=8.0; (c) APMH modified at pH=10.9; (d) APMH modified at pH=12.0
from acid to basic. Therefore, as summarized in Table 2, for pH values between 8 and 10, the silanol group is negatively charged, whereas the surface and the amine group are positively charged and hence an orientation with the silanol group at the interface is formed. This orientation is also due to the low coverage density, as shown in Table 1, which permits that the adsorbed molecules have enough space to allow the propyl chains to occupy a locus of orientation at various angles.
Table 2 Electrochemical behavior of MH and γ-APS in aqueous solution

The pH-dependence on ζ-potential of APMH (modified at pH=8.0) determined in 0.01 mol/L NaCl solution is presented in Fig.3(b). The surface charge decreases with increasing pH value and remains a positive ζ-potential due to the protonation of the amino moiety of the molecules. The fact that the modification of MH with γ-APS causes a shift of IEP to a more basic value compared with MH itself shows that the surface has gained a more basic characteristic because the molecule attached to the surface has dissociable basic molecule groups[22]. But the pH value of IEP was not determined in this work.
3.3.2 MH modified by γ-APS in pH range of 10-11
In pH range of 10-11, the elemental analysis data indicate that there is an increasing tendency for molecules to be adsorbed. It is hypothesized that the propyl chains are parallel to the surface and once again it is in agreement with the prediction based on the electrochemical measurements. The result of APMH (modified at pH value of 10.9) surface charge determined in 0.01 mol/L NaCl is shown in Fig.3(c). The positive surface charge decreases slightly with increasing pH value and the ζ-potential is smaller than that of APMH modified at pH value of 8.0. Still, the pH value of IEP is not observed either. Since this pH range is close to the IEP of the MH surface, the surface net charge will be close to neutral and hence no preferential adsorption of either moiety will be expected. It will be reasonable to assume that the dipolar organosilane molecules will bond to the surface by minimizing the overall charge of the system, hence adsorbing with a silanol group close to amino group.
3.3.3 MH modified by γ-APS in pH range of 11-12
In pH range of 11-12, results in Fig.3(d) demonstrate that the surface charge becomes negative and increases with increasing pH value because of the ionization of the silanol. The presence of amino and hydroxyl groups, containing hetroatoms with neucleophilic characters such as N and O, in the γ-APS will contribute to additional chemiadsorption process involving the unshared lone pair of electrons of these atoms and the vacant d orbits of the metal hydroxide surface atoms to form a coordinate type of bond. The absorbability of these atoms depends primarily on their electronegativity[23]. The less steric hindrance effect of the amino group as well as its less electronegativity leads to the higher organosilane coverage density (Table 1). It seems reasonable to speculate that γ-APS molecules bond to the surface with the amino moiety towards the MH surface.
4 Conclusions
1) Coverage density of γ-APS molecules on APMH particles increases form 55 nm-2 to 115 nm-2 with increasing solution pH value from 8.0 to 12.0. Hydrolysis and condensation of γ-APS are activated in alkaline solution and lead to multilayer adsorption of γ-APS molecules on the surface of MH.
2) The appearance of the bands (2 926 and 2 861 cm-1) assigned to the asymmetric and symmetric stretching of ―CH2 in alkyl chain and the disappearance of the band (3 370 cm-1) assigned to free ―NH2 asymmetric stretching confirm the condensation of γ-APS in aqueous solution and therefore lead to multilayer adsorption on MH surfaces.
3) The type of adsorption orientation of γ-APS molecules on MH surface is a function of coverage density. γ-APS molecules preferentially are adsorb to the surface with the silicon moiety towards the surface at a relatively low coverage density (e.g. 55 nm-2), and increasing coverage density (e.g. 90 nm-2) by changing solution pH value leads to parallel orientation. Whereas at an even higher coverage density (e.g. 115 nm-2), γ-APS molecules bond to the surface with the amino moiety towards the MH surface.
4) From this work, it can be concluded that the present study established the relationship between the adsorption orientation of amine silane on metal hydroxide surface and the coverage density. Also, efforts have been made to explain the adsorption features observed through detailed experimentation.
References
[1] BOOSTER J L, van SANDWIJK A, REUTER M A. Conversion of magnesium fluoride to magnesium hydroxide [J]. Minerals Engineering, 2003, 16(3): 273-281.
[2] KARIDAKIS T, AGATZINI-LEONARDOU S, NEOU-SYNGOUNA P. Removal of magnesium from nickel laterite leach liquors by chemical precipitation using calcium hydroxide and the potential use of the precipitate as a filler material [J]. Hydrometallurgy, 2005, 76(1/2): 105-114.
[3] HENRIST C, MATHIEU J P, VOGELS C, RULMONT A, CLOOTS R. Morphological study of magnesium hydroxide nanoparticles precipitated in dilute aqueous solution [J]. Journal of Crystal Growth, 2003, 249(1/2): 321-330.
[4] ROTHON R N, HORNSBY P R. Flame retardant effects of magnesium hydroxide [J]. Polymer Degradation and Stability, 1996, 54(2/3): 383-385.
[5] YEH J T, YANG H M, HUANG S S. Combustion of polyethylene filled with metallic hydroxides and crosslinkable polyethylene [J]. Polymer Degradation and Stability, 1995, 50(2): 229-234.
[6] TITELMAN G I, GONEN Y, KEIDAR Y, BRON S. Discolouration of polypropylene-based compounds containing magnesium hydroxide [J]. Polymer Degradation and Stability, 2002, 77(2): 345-352.
[7] HORNSBY P R, MTHUPHA A. Analysis of fire retardancy in magnesium hydroxide filled polypropylene composites [J]. Plastics Rubber and Composites Processing and Applications, 1996, 25(7): 347-355.
[8] LIAUW C M, ROTHON R N, LEES G C, IQBAL Z. Flow micro- calorimetry and FTIR studies on the adsorption of saturated and unsaturated carboxylic acids onto metal hydroxide flame-retardant fillers [J]. Journal of Adhesion Science and Technology, 2001, 15(8): 889-912.
[9] ZHU S, ZHANG Y, ZHANG Y X. Deformation and fracture of Mg(OH)-filled polyolefin composites under tensile stress [J]. Journal of Applied Polymer Science, 2003, 89(12): 3248-3255.
[10] HORNSBY P R, WATSON C L. Interfacial modification of polypropylene composites filled with magnesium hydroxide [J]. Journal of Materials Science, 1995, 30(21): 5347-5355.
[11] SANCHEZ C, RIBOT F, ROZES L, ALONSO B. Design of hybrid organic-inorganic nanocomposites synthesized via sol-gel chemistry [J]. Molecular Crystals and Liquid Crystals, 2000, 354: 731-746.
[12] VRANCKEN K C, van der VOORT P, POSSEMIERS K, VANSANT E F. Surface and structural properties of silica gel in the modification with [gamma]-aminopropyltriethoxysilane [J]. Journal of Colloid and Interface Science, 1995, 174(1): 86-91.
[13] VANSANT E F, VOORT P V D, VRANCKEN K C. Characterization and chemical modification of the silica surface [M]. Amsterdam: Elsevier, 1995.
[14] CULLER S R, ISHIDA H, KOENIG J L. Silane interphase of composites: Effects of process conditions on gamma- aminopropyltriethoxysilane[J]. Polymer Composites, 1986, 7(4): 231-238.
[15] XU Z, LIU Q, FINCH J A. Silanation and stability of 3-aminopropyl triethoxy silane on nanosized superparamagnetic particles (I): Direct silanation [J]. Applied Surface Science, 1997, 120(3/4): 269-278.
[16] FROST R L, KLOPROGGE J T. Infrared emission spectroscopic study of brucite [J]. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 1999, 55: 2195-2205.
[17] HE Z Q, LI X H, LIU E H, HOU Z H, DENG L F, HU C Y. Preparation of calcium stannate by modified wet chemical method [J]. Journal of Central South University of Technology, 2003, 10(3): 195-197.
[18] SEVIM A, TANIL A, DAVIES J E D. Adsorption of 2, 2′-bipyridyl onto sepiolite, attapulgite and smectite group clay minerals from anatolia: The FT-IR and FT-Raman spectra of surface and intercalated species [J]. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 1994, 18(2): 123-135.
[19] SABAH E, CELIK M S. Interaction of pyridine derivatives with sepiolite [J]. Journal of Colloid and Interface Science, 2002, 251: 33-38.
[20] POKROVSKY O S, SCHOTT J. Experimental study of brucite dissolution and precipitation in aqueous solutions: Surface speciation and chemical affinity control [J]. Geochimica et Cosmochimica Acta, 2004, 68(1): 31-45.
[21] THOMSEN L, WATTS B, DASTOOR P C. A NEXAFS orientation study of gamma-aminopropyltriethoxysilane on zinc oxide surfaces [J]. Surface and Interface Analysis, 2006, 38(7): 1139-1145.
[22] ALKAN M, TEKIN G, NAMLI H. FTIR and zeta potential measurements of sepiolite treated with some organosilanes [J]. Microporous and Mesoporous Materials, 2005, 84(1/3): 75-83.
[23] AWAD H S, ABDEL GAWAD S. Mechanism of inhibition of iron corrosion in hydrochloric acid by pyrimidine and series of its derivatives [J]. Anti-Corrosion Methods and Materials, 2005, 52(6): 328-336.
(Edited by CHEN Wei-ping)
Foundation item: Projects(50574104; 50574102) supported by the National Natural Science Foundation of China
Received date: 2007-10-20; Accepted date: 2007-12-10
Corresponding author: FENG Qi-ming, Professor, PhD; Tel: +86-731-8836817; E-mail: qmfeng@mail.csu.edu.cn