
Dynamic corrosion of copper-nickel sulfide by Acidithiobacillus ferrooxidans
TONG Lin-lin(佟琳琳), JIANG Mao-fa(姜茂发), YANG Hong-ying(杨洪英),
YU Juan(俞 娟), FAN You-jing(范有静), ZHANG Yao(张 耀)
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 3 April 2008; accepted 16 October 2008
Abstract: The dynamic corrosion process of bio-oxidation of copper-nickel sulfide from Karatungk in northern Xinjiang Province of China was studied. The polished wafer of the copper-nickel sulphide was used to carry on a series of oxidation corrosion experiment by Acidithiobacillus ferrooxidans. The changes of superficial corrosion appearance and the mineral dynamic corrosion process were discovered by microscope observation. Then, the galvanic cell model was established, and the bio-oxidation activation order of typical copper-nickel sulphide minerals was ascertained as pyrrhotite>pentlandite>chalocopyrite.
Key words: copper-nickel sulphide; bio-oxidation; dynamic corrosion; galvanic effect
1 Introduction
Bio-oxidation is an advanced technique of mineral processing. This method has become the research interest of hydrometallurgy in recent years[1-5]. Previous researches of bio-oxidation were mainly focused on breeding of dominant bacteria[6-8], influence of bio-oxidation, optimization and control of leaching process[9-10] and bio-metallurgical processes of different mineral by bacteria. In fact, the bio-oxidation system involves interface problems such as bacterial/ mineral and mineral/mineral[11], so researching the surface morphology changes can help us to reveal oxidation mechanism of bio-oxidation. Chalcopyrite is the most common copper sulfide in the copper-nickel ores. The oxidation of the secondary copper sulfide was easy but that of the primary copper sulfide was difficult, so secondary copper sulfide (chalcocite, covelline, etc.) was applied in bio-oxidation at present, while the primary copper sulfide (chalcopyrite) was still treated with pyrometallurgical method[12]. Resource of chalcopyrite is widely distributed in nature, but it is difficult to treat with bio-oxidation. The purpose of this work is to show the characteristics of dynamic corrosion of the typical copper-nickel sulfide mineral pairs from the microscopic point of view.
The mechanisms in bio-oxidation of chalcopyrite and the main associated minerals, pentlandite and pyrrhotite, were obtained. Direct and indirect bio- oxidation reactions in bacterial oxidation process of chalcopyrite, pentlandite and pyrrhotite take place as follows[13-14]:
Chalcopyrite:
4CuFeS2+17O2+2H2SO4
4CuSO4+2Fe2(SO4)3+2H2O (1)
CuFeS2+2Fe2(SO4)3→CuSO4+5FeSO4+2S0 (2)
Pentlandite:
8(Fe, Ni)9S8+141O2+26H2SO4
18Fe2(SO4)3+36NiSO4+26H2O (3)
2(Fe, Ni)9S8+18Fe2(SO4)3→45FeSO4+9NiSO4+16S0 (4)
Pyrrhotite:
4Fe1-xS+(9-3x)O2+(2-6x)H2SO4
(2-2x)Fe2(SO4)3+(2-6x)H2O (5)
Fe1-xS+(1-x)Fe2(SO4)3→3(1-x)FeSO4+S0 (6)
Oxidation of Fe2+:
4FeSO4+2H2SO4+2O2 2Fe2(SO4)3+2H2O (7)
2Fe2(SO4)3+2H2O (7)
Oxidation of S0:
S0+H2O+1.5O2 H2SO4 (8)
H2SO4 (8)
2 Experimental
2.1 Mineral samples
The mineral samples for this research were collected from Karatungk copper-nickel sulfide mineral belt in northern Xinjiang of China. Chalcopyrite (molecular formula CuFeS2) and pentlandite (molecular formula (Fe, Ni)9S8) were the most important minerals in the copper-nickel sulfide. At the same time, they often co-exist with pyrrhotite (molecular formula Fe1-xS, generally x=0±0.223, range of corresponding composition FeS-Fe11S12)[15].
2.1.1 Mineral testing methods
The mineral species was identified using Leica DMRXP polarized optical microscope by process mineralogy method. EPMA-1600 electron probe micro-analysis (EPMA, SHIMADZU, Institute of Metal Research, Chinese Academy of Sciences) was applied to studying the mineral compositions. The conditions of full spectrum scanning qualitative analysis were as follows: accelerating voltage 15 kV, beam current 10 nA, count time 10 s, beam diameter 20 μm, and analyzer crystals RAP, PBST, PET and LiF. The conditions of spectrum quantitative analysis were as follows: accelerating voltage 15 kV, beam current 10 nA, count time 10 s and beam diameter 20 μm, analyzer crystal of PET for S with FeS2 as the standard sample, and analyzer crystal of LiF for Co, Fe, Ni with pure Cu as the standard sample, respectively.
2.1.2 Mineral species and compositions
Identification with process mineralogy method shows that the mineral mainly contains metal sulfides of pentlandite (Pe) , pyrrhotite (Pyr), chalcopyrite (Cp) and gangue minerals of chrysolite, pyroxene, biotite and plagioclase.
Regions of chalcopyrite, pentlandite and pyrrhotite identified by process mineralogy method were analyzed by electron probe micro-analysis. Fig.1 shows the result of qualitative analysis. There were elements Fe, S, Ni and Co in the region of pentlandite, elements Fe, S, and Cu in the region of chalcopyrite and elements Fe, S, and Ni in the region of pyrrhotite. Table 1 lists the result of quantitative analysis.
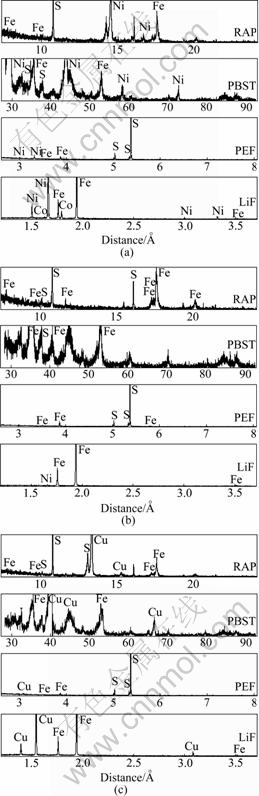
Fig.1 Results of qualitative analysis by EPMA: (a) Pentlandite; (b) Pyrrhotite; (c) Chalcopyrite
Table 1 Results of quantitative analysis by EPMA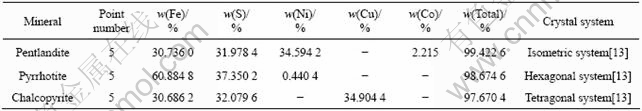
2.2 Bacterial sample
Acidithiobacillus ferrooxidans(A.f) were used in experiment. Culture medium was 9K liquid medium ((NH4)2SO4 3 g/L, KCl 0.1 g/L, K2HPO4 0.5 g/L, MgSO4・7H2O 0.5 g/L, Ca(NO3)2 0.01 g/L, FeSO4・7H2O 44.2 g/L), adjusted to pH 1.80 with 2 mol/L sulfuric acid. Before experiment, activated culture was conducted to bacteria for three times, and the concentration was above 108 cell/mL.
This experiment was carried out in a 3 L tank with a mechanic agitator at 950 r/min. When bacteria grow into stationary phase, minerals were added into tank with initial potential of 590 mV and initial pH of 1.8. The certain field of minerals was observed by Germany Leica DMRX microscope at a certain time interval.
3 Results
3.1 Dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite mineral pair
In the bio-oxidation corrosion experiment, pyrrhotite and chalcopyrite showed a sharp contrast. Before bio-oxidation, pyrrhotite crystal surface was smooth in pinkish brown and strong metallic luster. Chalcopyrite crystal surface was smooth in brass yellow and strong metallic luster. After the bio-oxidation, their microscopic characteristics were changed obviously, as shown in Fig.2.

Fig.2 Microscopic characteristics of Pyr/Cp mineral pair after bio-oxidation: (a) 0 h; (b) 24 h; (c) 48 h; (d) 72 h; (e) 96 h; (f) 120 h; (g) 144 h; (h) 168 h; (i) 244 h; (j) 288 h
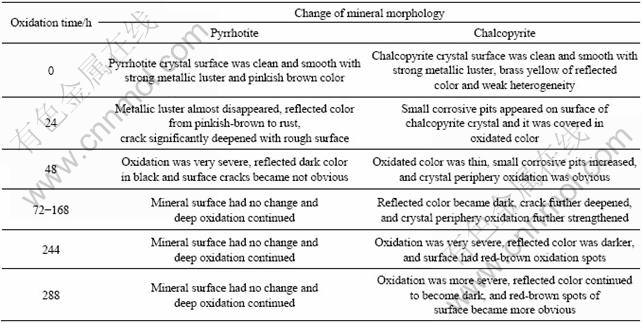
Pyrrhotite and chalcopyrite showed different oxidation behaviors in the process of bio-oxidation, as listed in Table 2.
Table 2 Dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite mineral pair
3.2 Dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite/pentlandite mineral pairs
Before bio-oxidation, pyrrhotite crystal surface was smooth in pinkish brown and strong metallic luster; pentlandite crystal surface was smooth in light milk yellow and strong metallic luster; and chalcopyrite crystal surface was smooth in brass yellow and strong metallic luster. After the bio-oxidation, their microscopic characteristics were changed obviously, as shown in Fig.3.
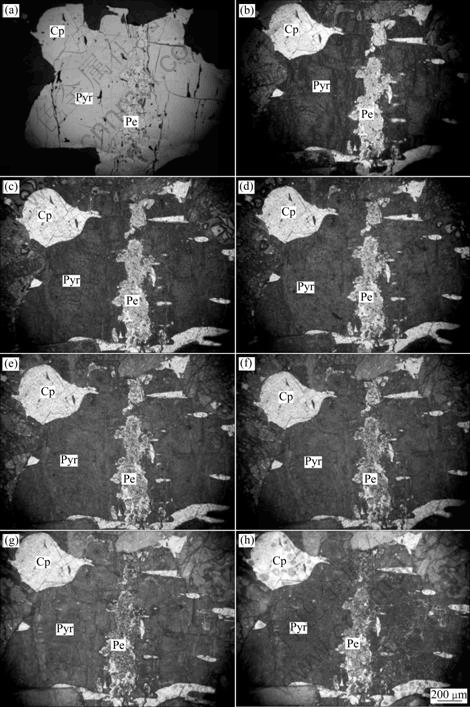
Fig.3 Microscopic characteristics of Pyr/Cp/Pe mineral pairs after bio-oxidation: (a) 0 h; (b) 24 h; (c) 48 h; (d) 72 h; (e) 96 h; (f) 120 h; (g) 192 h; (h) 288h
Pyrrhotite, chalcopyrite and pentlandite showed different oxidation behaviors in the process of bio-oxidation, as listed in Table 3.
Table 3 Dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite/pentlandite mineral pairs

4 Discussion
4.1 Analysis of bio-oxidation pyrrhotite/chalcopyrite mineral pair
The experiment showed that oxidation rate of pyrrhotite was much faster than chalcopyrite. This was consistent with the view of GOTTSCHALK et al[16] that one sulfide will be significantly oxidated because of different static potentials, while the other will be protected for galvanic effect. In acidic solution, when two sulfides of different static potentials contacted, mineral of high static potential as the cathode will be protected[17-22]. Static potentials obtained by Tafel polarization curves of chalcopyrite, pentlandite and pyrrhotite in bacterial system of three kinds of minerals were 0.318, 0.326 and 0.352 mV, respectively, that is
φCp>φPe>φPyr (Equipment: Shanghai Chenhua Equipment Company: CHI660C Electrochemical workstation). According to the operating principle of galvanic cell, redox reaction occurred on the two poles of cell, and some cations in electrode and the electrolyte solution carried through chemical reaction. Galvanic cell model of pyrrhotite/chalcopyrite mineral pair in bio-oxidation is drawn in Fig.4.
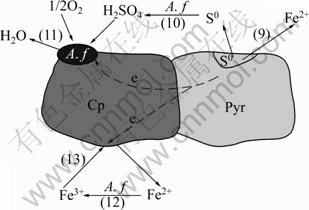
Fig.4 Galvanic cell model of pyrrhotite/chalcopyrite mineral pairs in bio-oxidation
In the bio-oxidation system, pyrrhotite and chalcopyrite formed an galvanic cell. Pyrrhotite had strong electrochemical activity as anode on which oxidative and corrosiveness occurred. Comparatively, chalcopyrite had strong electrochemical inertia as cathode which was protected. The reaction equations are as follows:
Fe1-xS→(1-x)Fe2++S0+2(1-x)e (anode-oxidation) (9)
S0+2O2+2e (10)
(10)
2H++1/2O2+2e H2O(cathode-revivification) (11)
H2O(cathode-revivification) (11)
Fe2+ Fe3+ (12)
Fe3+ (12)
Fe3++e→Fe2+ (cathode-revivification) (13)
A. f were absorbed to the surface of chalcopyrite, strengthened electron behavior of O2, and Fe2+ produced from pyrrhotite was oxidated to Fe3+ by bacteria. Fe3+ got electron on the surface of chalcopyrite. Fe3+, as oxidant, strengthened the dissolution of pyrrhotite. Furthermore, elemental sulfur produced from pyrrhotite was oxidated to SO42- by bacteria, which reduced the clag of the production layer on the surface for the dissolution of pyrrhotite.
4.2 Bio-oxidation pyrrhotite/chalcopyrite/pentlandite mineral pairs analysis
In process of oxidation of pyrrhotite/chalcopyrite/ pentlandite mineral pairs, three minerals had different static potentials, and formed galvanic cell. Galvanic cell model of pyrrhotite/chalcopyrite/pentlandite mineral pairs in bio-oxidation is drawn in Fig.5.

Fig.5 Galvanic cell model of pyrrhotite/chalcopyrite/pentlandite mineral pairs in bio-oxidation
In the bio-oxidation system, there were two galvanic cells. Pyrrhotite and chalcopyrite formed an galvanic cell, and pyrrhotite and pentlandite formed the other one. Pyrrhotite worked as anode, and chalcopyrite and pentlandite worked as cathode. Reaction equations were as follows:
Fe1-xS→(1-x)Fe2++S0+2(1-x)e (anode-oxidation) (14)
S0+2O2+2e (15)
(15)
2H++1/2O2+2e H2O (cathode-revivification)
H2O (cathode-revivification)
(16)
Fe2+ Fe3+ (17)
Fe3+ (17)
Fe3++e→Fe2+ (cathode-revivification) (18)
A. f were absorbed to the surfaces of chalcopyrite and pentlandite, strengthened electron behavior of O2, and Fe2+ produced from pyrrhotite was oxidated to Fe3+ by bacteria. Fe3+ got electron on the surfaces of chalcopyrite and pentlandite. Fe3+, as oxidant, strengthened the dissolution of pyrrhotite. Furthermore, elemental sulfur produced from pyrrhotite was oxidated to SO42- by bacteria, which reduced the clag of the production layer on the surface for the dissolution of pyrrhotite.
4.3 Comparison of bio-oxidation rate of main sulfides
The bio-oxidation corrosion experiment results showed that pyrrhotite was the most easily oxidized mineral, pentlandite was moderate, and chalcopyrite was the hardest in copper-nickel sulfide from Karatungk. Bio-oxidation rate of main sulfide was in the order of pyrrhotite>pentlandite>chalcopyrite.
5 Conclusions
1) Bio-oxidation rate of pyrrhotite was the fastest among the three minerals, which came after by pentlandite, and the slowest was chalcopyrite. Bio-oxidation rate sequence of main sulfide from Karatungk was pyrrhotite>pentlandite>chalcopyrite.
2) In dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite mineral pair, pyrrhotite and chalcopyrite formed an galvanic cell, with pyrrhotite worked as anode, and chalcopyrite worked as cathode.
3) In dynamic corrosion process of bio-oxidation of pyrrhotite/chalcopyrite/pentlandite mineral pairs, there were two galvanic cells. Pyrrhotite and chalcopyrite formed an galvanic cell, while pyrrhotite and pentlandite formed the other one. Pyrrhotite was anode, chalcopyrite and pentlandite were cathodes.
References
[1] KUMARI A, NATARAJAN K A. Development of a clean bioelectrochemical process for leaching of ocean manganese nodules [J]. Minerals Engineering, 2002, 15(1/2): 103-106.
[2] NESTOR D, VALDIVIA U, CHAVES A P. Mechanisms of bioleaching of a refractory mineral of gold with Thiobacillus ferrooxidans [J]. International Journal of Mineral Processing, 2001, 62(1/4): 187-198.
[3] KUMARI A, NATARAJAN K A. Electrochemical aspects of leaching of ocean nodules in the presence and absence of microorganisms [J]. International Journal of Mineral Processing, 2002, 66(1/4): 29-47.
[4] MAZUELOS A, PALENCIA I, ROMERO R, RODR?GUEZ G, CARRANZA F. Ferric iron production in packed bed bioreactors: Influence of pH, temperature, particle size, bacterial support material and type of air distributor [J]. Minerals Engineering, 2001, 14(5): 507-514.
[5] LIU Z, BORNE F, RATOUCHNIAK J, BONNEFOY V. Genetic transfer of IncP, IncQ and IncW plasmids to four Thiobacillus ferrooxidans strains by conjugation [J]. Hydrometallurgy, 2001, 59(2/3): 339-345.
[6] LI Hong-xu, DONG Qing-hai, CANG Da-qiang, WANG Dian-zuo. Thermophilic microorganism sulfolobus growth and leaching chalcopyrite properties [J]. Journal of University of Science and Technology Beijing, 2007, 29(1): 20-24. (in Chinese)
[7] WANG Jian-wei, WANG Mo-hui, YUAN Yuan. The research situation on leaching of ores using mixed bacteria [J]. Multipurpose Utilization of Mineral Resources, 2007(5): 24-27. (in Chinese)
[8] HALLBERG K B, DOPSON M, LINDSTR?M E B. Arsenic toxicity is not due to a direct effect on the oxidation of reduced inorganic sulfur compounds by Thiobacillus caldus [J]. FEMS Microbiology Letters ,1996 , 145(3): 409-414.
[9] WANG Ying-ru, ZHONG Kang-nian, WEI Yi-he. Optimization of biooxidation pretreatment process of hard-to-leach gold ore [J]. Metal Mine, 2003(10): 40-49. (in Chinese)
[10] CHEN Shi-guan. Bioleaching and its application in nonferrous metallurgy [J]. Shanghai Nonferrous Metals, 2000, 21(3): 137-146. (in Chinese)
[11] LIU Jian-she, WANG Zhao-hui, GEN Mei-mei, QIU Guan-zhou. Progress in the study of polyphase interfacial interactions between microorganism and mineral in bio-hydrometallurgy [J]. Mining and Metallurgical Engineering, 2006, 26(1): 40-44. (in Chinese)
[12] LI Ji-bi. Application status of process for copper hydrometallurgy in domestic and abroad [J]. Hydrometallurgy of China, 2007, 26(1): 13-16. (in Chinese)
[13] SHU Rong-bo, RUAN Ren-man, WEN Jian-kang. Review on passivation of chalcopyrite during bioleaching process [J]. Chinese Journal of Rare Metals, 2006, 30(3): 395-400. (in Chinese)
[14] DENG Jing-shi. Studies on moderate thermophile intensifying pentalandite leaching [D]. Kunming: Kunming University of Science and Technology, 2002: 11-12. (in Chinese)
[15] PAN Zhao-lu. Crystallography and mineralogy [M]. Beijing: Geological Press, 1994: 28-34. (in Chinese)
[16] GOTTSCHALK V, BUEHLER H. Oxidation of sulfides [J]. Econ Geol, 1910, 5: 28-35.
[17] BERRY V K, MURR L E, HISKEY J B. Galvanic interaction between chalcopyrite and pyrite during bacterial leaching of low-grade waste [J]. Hydrometallurgy, 1978, 3(4): 309-326.
[18] MEHTA A P, MURR L E. Fundamental studies of the contribution of galvanic interaction to acid-bacterial leaching of mixed metal sulfides [J]. Hydrometallurgy, 1983, 9(3): 235-256.
[19] WOODS R. Recent advances in electrochemistry of sulfide mineral flotation [J]. Trans Nonferrous Met Soc China, 2000, 10(s1): 26-29.
[20] MAHMOOD M N, TURNER A K. The selective leaching of zinc from chalcopyrites-phalerite concentrates using slurry electrodes [J]. Hydrometallurgy, 1985, 14(3): 317-329.
[21] SANTOS L R G, BARBOSA A F, SOUZA A D, LE?O V A. Bioleaching of a complex nickel-iron concentrate by mesophile bacteria [J]. Minerals Engineering, 2006, 19(12): 1251-1258.
[22] CRUZ R, LUNA-S?NCHEZ R M, LAPIDUS G T, GONZ?LEZ I, MONROY M. An experimental strategy to determine galvanic interactions affecting the reactivity of sulfide mineral concentrates [J]. Hydrometallurgy, 2005, 78(3/4): 198-208.
Foundation item: Project(2006AA06Z127) supported by the National Hi-tech Research and Development Program of China; Projects(50674029, 50274024) supported by the National Natural Science Foundation of China; Project(20060145015) supported by Doctoral Program Foundation of Ministry of Education of China; Project(20052030) supported by the Natural Science Foundation of Liaoning Province, China
Corresponding author: JIANG Mao-fa; Tel: +86-24-83680373; E-mail: jiangmf@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(08)60292-7
(Edited by YANG Bing)