
Thermal decomposition of ammonium hexafluoroaluminate and preparation of aluminum fluoride
HU Xian-wei, LI Lin, GAO Bing-liang, SHI Zhong-ning, LI Huan, LIU Jing-jing, WANG Zhao-wen
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 30 October 2010; accepted 27 May 2011
Abstract: The thermal decomposition process of (NH4)3AlF6 was studied by DTA-TGA method and the related thermodynamic data were obtained. The results show that AlF3 is obtained after three-step decomposition reaction of (NH4)3AlF6, and the solid products of the first two decomposition reactions are NH4AlF4 and AlF3(NH4F)0.69, respectively. The three reactions occur at 194.9, 222.5 and 258.4 °C, respectively. Gibbs free energy changes of pertinent materials at the reaction temperatures were calculated. Enthalpy and entropy changes of the three reactions were analyzed by DSC method. Anhydrous aluminum fluoride was prepared. The XRD analysis and mass loss calculation show that AlF3 with high purity can be obtained by heating (NH4)3AlF6 at 400 °C for 3 h.
Key words: ammonium hexafluoroaluminate; thermal decomposition; aluminum fluoride; thermodynamic data
1 Introduction
Anhydrous aluminum fluoride is commonly used in the scientific experiments related to aluminum electrolyte. However, aluminum fluoride from most reagent companies contains crystal water because of its hydrometallurgical preparing process. Therefore, it is important to develop the process for preparing anhydrous aluminum fluoride in the laboratory.
If heating aluminum fluoride containing crystal water, alumina will be formed because of the hydrolysis reaction of aluminum fluoride. Sublimation process under vacuum is commonly used for preparing anhydrous aluminum fluoride [1-4]. But in this process, the experimental equipments are strictly required. TIE et al [5] have put forward a new process in which aluminum fluoride with crystal water is mixed with ammonium acid fluoride and the mixture is then heated. Hydrogen fluoride generated by the decomposition of ammonium acid fluoride will inhibit the hydrolysis of aluminum fluoride, so the purity of aluminum fluoride will increase. This process has also been used by other researchers [6-9]. SCHOLZ and KEMNITZ [10] have proposed a mechanochemical synthesis method to prepare aluminum fluoride with ammonium fluoride used as fluorinating agent.
Anhydrous aluminum fluoride can be obtained by heating ammonium hexafluoroaluminate [11]. SHINN et al [12] have researched the thermal decomposition process of ammonium hexafluoroaluminate. According to their research results, ammonium hexafluoroaluminate decomposes by three steps. γ-AlF3 will be generated at 300 °C and then it will transform to α-AlF3 at 720 °C. BECK et al [13] have studied the thermal decomposition process of ammonium hexafluoroallate and ammonium hexafluoroindate which has a similar crystal form to ammonium hexafluoroaluminate. The processes are proved to be carried out step by step, and the crystal transformation of the final fluoride product is also observed.
However, the researches mentioned above are focused on the crystal transformation of permanent materials, and the research on the thermal decomposition of ammonium hexafluoroaluminate is not systematic. Thermodynamic data of the decomposition and the preparation technology of anhydrous aluminum fluoride have not been involved.
In the present study, the thermal decomposition process of ammonium hexafluoroaluminate was analyzed in detail by comprehensive thermal analysis technique. The reaction equations were determined, and the pertinent thermodynamic data were calculated. Subsequently, the process of preparing anhydrous aluminum fluoride in the laboratory was studied, and the reasonable preparing conditions were determined.
2 Experimental
The comprehensive thermal analysis experiments were carried out by a TA Instruments SDT 2960 thermal analysis instrument. The platinum crucible was used for holding samples. α-Al2O3 after calcination was used as reference material. About 10 mg of the sample was used for each measurement. The experiments were carried out under argon shield and the flow rate was controlled to 20 mL/min.
In the study on preparation of anhydrous aluminum fluoride, ammonium hexafluoroaluminate was held in a graphite crucible which was placed in a resistance furnace, and the experiments were carried out under argon shield.
3 Results and discussion
3.1 DTA-TGA analysis on (NH4)3AlF6
DTA-TGA experiments of (NH4)3AlF6 were carried out at four various heating rates of 1, 2, 5 and 10 °C/min, respectively. The scanned curves are shown in Fig. 1.
Each endothermic peak on the DTA curves in Fig. 1 corresponds to one step of decomposition reactions. There are three endothermic peaks on the DTA curves in Figs. 1(a), (b) and (c); but on the DTA curve in Fig. 1(d), only two endothermic peaks can be identified, which is attracted to that the heating rate is so high that the resolution of the curve is decreased. However, one can derive DTG curve from the TGA curve to improve the resolution. The turning points on the DTG curve are more easily to identify. DTG curve of (NH4)3AlF6 at heating rate of 10 °C/min is shown in Fig. 2. It is easy to identify that there are three obvious peaks on the DTG curve in Fig. 2, which correspond to the three steps of decomposition of (NH4)3AlF6, respectively. Meanwhile, on all TGA curves in Fig.1, there are four platforms, which also express the feature of three-step decomposition reactions.
The temperature corresponding to the extrapolated onset of peak on DTA or DTG curve is thought to be the starting temperature of the corresponding reaction. Therefore, the starting temperatures of the three-step decomposition reactions can be obtained by analyzing the DTA curves in Figs. 1(a)-(c) and DTG curve in Fig. 2.
The relationship curves between starting temperatures and heating rates are shown in Fig. 3. It can be seen in Fig. 3 that except for the second and third reactions corresponding to 5 °C/min of heating rate, there is a tendency that the starting temperature of the reaction is decreased with increasing the heating rate. For a lower heating rate, the heat transfer is more sufficient, so the whole system is closer to equilibrium. In contrary, for higher heating rate, the system is farther from equilibrium state, so the resolution of the instrument is decreased. The effect of heating rate on chemical reactions with mass change is more serious [14]. Therefore, it is thought that when the heating rate is equal to 0, the heat transfer is the most sufficient, and the whole system is the closest to heat equilibrium. Quadratic fit is made to the data points in Fig. 3 (the points corresponding to the second reaction and the third reaction for 5 °C/min of heating rate are not involved), as shown by the actual lines.

Fig. 1 DTA-TGA curves of (NH4)3AlF6 at different heating rates: (a) 1 °C/min; (b) 2 °C/min; (c) 5 °C/min; (d) 10 °C/min

Fig. 2 DTG curve of (NH4)3AlF6 at heating rate of 10 °C/min

Fig. 3 Relationship between starting temperature and heating rate
The extrapolated starting temperatures can be obtained by substituting the heating rate in the fitted equations with 0. The obtained starting temperature values are listed in Table 1.
(NH4)3AlF6 will decompose during the heating process and NH4F will be generated. NH4F will further decompose to HF and NH3. The other products of the decomposition reactions of (NH4)3AlF6 will be the combination of NH4F and AlF3 with different mole ratios or pure AlF3. The three decomposition reaction equations can be obtained according to the mass loss of each reaction. It can be seen from the four TGA curves in Fig.1 that there is little effect of heating rate on mass loss. Therefore, in the present work, only the TGA curve of 1 °C/min is analyzed typically. The mass loss of each reaction is obtained from corresponding starting temperature of the next reaction. The temperature at which mass loss in the TGA curves does not change any more is used as that for the third reaction.
Table 1 Starting temperature and solid product of decomposition reactions of (NH4)3AlF6

The amount of the mass loss equals that of HF and NH3, which could be regarded as the amount of NH4F according to stoichiometry. For 1 mol (195 g) of (NH4)3AlF6, the mass of NH4F can be obtained according to the mass loss of each decomposition, and then the amount of substance can be calculated. Based on the above results, the molecular formulas of the solid products of the three decomposition reactions can be obtained, which are also listed in Table 1.
In the solid product of the first decomposition reaction of (NH4)3AlF6, the molar ratio between NH4+ and AlF4- is 0.97. Considering the experimental error, the ratio is thought to be 1. Therefore, it is thought that the solid product of the first decomposition reaction is NH4AlF4.
The molecular formulas of the solid product of the second decomposition reaction is (NH4)0.69AlF3.69. The molar ratio between AlF3 and NH4F in the material is 1:0.69 according to stoichiometry. This material only exists at high temperature and its structure is very unstable [12].
Based on the above discussion, the three-step decomposition reaction equations are obtained as follows:
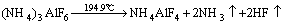 (1)
(1)

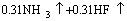 (2)
(2)
 (3)
(3)
3.2 Gibbs free energy change of pertinent materials
It is thought that Eqs. (1)-(3) are at equilibrium state, that is, the Gibbs free energy change is 0.
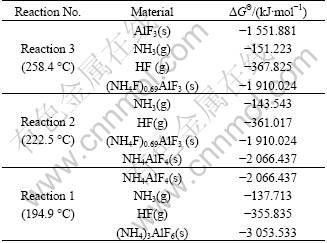
Though there is no data on the Gibbs free energy changes of (NH4)3AlF6, (NH4F)0.69AlF3 and NH4AlF4 in the literatures, the Gibbs free energy change of the three materials at the reaction temperatures can be calculated based on Eqs. (1)-(3). The Gibbs free energy change of AlF3, NH3 and HF at the reaction temperatures can be obtained by using HSC Chemistry 5.1, as listed in Table 2. The calculated data of pertinent materials are also listed. During the calculation process, the Gibbs free energy changes of (NH4F)0.69AlF3 at 258.4 °C and 222.5 °C are thought to be the same. Likewise, the Gibbs free energy changes of NH4AlF4 at 222.5 °C and 194.9 °C are thought to be the same.
Table 2 Gibbs free energy changes of pertinent materials in decomposition reactions of (NH4)3AlF6
3.3 Enthalpy and entropy change of decomposition reactions of (NH4)3AlF6
It is well known that the area of the peak enclosed by the DSC curve and the baseline is proportional to the enthalpy change as follows:
ΔH=KA (4)
where ΔH is the enthalpy change in J/g; A is the area of the peak enclosed by the DSC curve and the baseline in J/g; K is the proportional constant, which needs to be calibrated by the reference material.
BREUER and EYSEL [14] have researched the checking standard materials for DSC experiments. In the present study, metallic Sn and AgNO3 are chosen to calibrate the value of K.
The heating rate of the DSC experiments was set to be 5 °C/min, the mass of both the two samples were as same as possible. The DSC curves of Sn and AgNO3 are shown in Figs. 4 and 5, respectively.
The endothermic peak on the DSC curve in Fig. 4 corresponds to the melting of Sn. On the DSC curve in Fig. 5, there are two endothermic peaks,and they correspond to the polymorphic transition and the melting of AgNO3, respectively [15].

Fig. 4 DSC curve of Sn at heating rate of 5 °C/min
Fig. 5 DSC curve of AgNO3 at heating rate of 5 °C/min
The calibration of K was carried out by the melting peaks of metallic Sn and AgNO3. The values of K were calculated as 1.70 and 1.62, respectively. The difference between the two calculated values is small and the average value of 1.66 is used to calculate the enthalpy of decomposition reactions of (NH4)3AlF6.
In the DSC measurement of (NH4)3AlF6, the heating rate was also set as 5 °C/min and the mass used was as close to that of the former scannings as possible. The DSC curve of (NH4)3AlF6 is shown in Fig. 6.
By using Eq. (4), the enthalpy changes of the three decomposition reactions can be calculated and the results are listed in Table 3.
The relationships among entropy change, enthalpy change and Gibbs free energy change are described by
ΔH =ΔG + T・ΔS (5)
where ΔH, ΔG, ΔS, and T are the enthalpy change, Gibbs free energy change, entropy change, and temperature, and their units are J/g, J/g, J/(g・°C), and °C, respectively.

Fig. 6 DSC curve of (NH4)3AlF6 at heating rate of 5 °C/min
Therefore, the entropy changes of the three decomposition reactions can be calculated and the results are also listed in Table 3.
Table 3 Enthalpy and entropy change of decomposition reactions of (NH4)3AlF6

3.4 Preparation of anhydrous AlF3 by heating (NH4)3AlF6
According to the above thermal analysis of (NH4)3AlF6, one will get anhydrous AlF3 if (NH4)3AlF6 is heated to a high enough temperature and held for a long enough time.
40 g of (NH4)3AlF6 was heated to 400 °C, and then held for 3 h. The remained solid sample was 16.70 g, and the mass loss was calculated as 58.25%. Considering the error, it is thought that (NH4)3AlF6 is decomposed to AlF3 thoroughly.
The XRD pattern of the remaining sample is shown in Fig. 7. It indicates that the decomposition product is AlF3. Therefore, it is thought that anhydrous AlF3 with high purity can be prepared by heating (NH4)3AlF6.
4 Conclusions
1) During the heating process of (NH4)3AlF6, three-step decomposition reactions occur as follows:
(NH4)3AlF6 NH4AlF4+2NH3↑+2HF↑
NH4AlF4+2NH3↑+2HF↑
NH4AlF4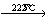 (NH4F)0.69AlF3+0.31NH3↑+0.31HF↑
(NH4F)0.69AlF3+0.31NH3↑+0.31HF↑
(NH4F)0.69AlF3 AlF3+0.69NH3↑+0.69HF↑
AlF3+0.69NH3↑+0.69HF↑
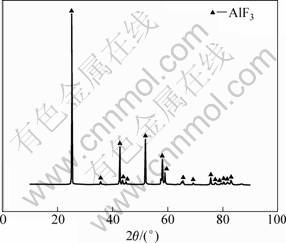
Fig. 7 XRD pattern of thermal deposition product of (NH4)3AlF6 at 400 °C
2) The Gibbs free energy changes of (NH4)3AlF6, NH4AlF4, AlF3(NH4F)0.69 at corresponding temperatures, and enthalpy and entropy changes of the three decomposition reactions are obtained.
3) High purity anhydrous AlF3 can be obtained by heating (NH4)3AlF6 to 400 °C and maintaining for a long enough time.
References
[1] ZHANG Jin-sheng, QIU Zhu-xian. Research on preparation of anhydrous aluminum fluoride by sublimation process under vacuum [J]. Light Metals, 1988(6): 10-13. (in Chinese)
[2] GILBERT B, MATERNE T. Reinvestigation of molten fluoroaluminiate Raman spectra: The question of the existence of AlF52- ions [J]. Applied Spectroscopy, 1990, 44(2): 299-305.
[3] TIXHON E, ROBERT E, GILBERT B. Molten KF-AlF3 system: A study by Raman spectroscopy [J]. Applied Spectroscopy, 1994, 48(12): 1477-1482.
[4] TIXHON E, ROBERT E, GILBERT B. The molten MF-AlF3-MCl system(M=K, Na): A study by Raman spectroscopy [J]. Vibrational Spectroscopy, 1996, 13(1): 91-98.
[5] TIE Jun, QIU Zhu-xian, LU Gui-min, SUN Ting. A new process for preparing anhydrous aluminium fluoride [J]. Nonferrous Metals, 1994, 46(2): 49-51. (in Chinese)
[6] GAO Bing-liang, LI Dan, SHI Zhong-ning, WANG Zhao-wen, REN Bi-jun. Determination of cryolite ratio of low-temperature aluminum electrolyte [J]. Rare Metals, 2009, 28(S1): 713-715.
[7] GAO B L, LI D, SHI Z N, WANG Z W, REN B J. Determination of cryolite ratio of aluminum electrolyte [C]//Light Metals 2010. Seattle: Minerals, Metals & Materials Soc, 2010: 569-573.
[8] CHEN Wei. Dissolution behavior of aluminum dross in aluminum electrolyte [J]. Non-ferrous Mining and Metallurgy, 2010, 26(3): 38-40. (in Chinese)
[9] KAN Hong-min. Study on low temperature aluminum electrolysis [D]. Shenyang: Northeastern University, 2008. (in Chinese)
[10] SCHOLZ G, KEMNITZ E. Mechanochemical synthesis of AlF3 with NH4F as fluorinating agent―Does it work? [J]. Solid State Sciences, 2009, 11(3): 676-682.
[11] LIU Jian-liang. Preparation of aluminium fluoride from ammonium ice dregs [J]. Chemical Production and Technology, 2005, 12(4): 6-7. (in Chinese)
[12] SHINN D B, CROCKET D S, HAENDLER H M. The thermal decomposition of ammonium hexafluoroferrate (III) and ammonium hexafluoroaluminate: A new crystalline form of aluminum fluoride [J]. Inorganic Chemistry, 1966, 5(11): 1927-1933.
[13] BECK L K, KUGLER B H, HAENDLER H M. The thermal decomposition of ammonium hexafluorogallate and ammonium hexafluoroindate: New crystalline forms of gallium fluoride and indium fluoride [J]. Journal of Solid State Chemistry, 1973, 8(4): 312-317.
[14] BREUER K H, EYSEL W. The calorimetric calibration of differential scanning calorimetry cell [J]. Thermochimica Acta, 1982, 57(3): 317-329.
[15] LI Yu-zeng. Thermal Analysis [M]. Beijing: Tsinghua University Press, 1987: 239-241. (in Chinese)
氟铝酸铵的热分解及氟化铝的制备
胡宪伟,李 琳,高炳亮,石忠宁,李 欢,刘敬敬,王兆文
东北大学 材料与冶金学院,沈阳 110004
摘 要:采用DTA-TGA法研究了(NH4)3AlF6的热分解过程并获得了相关的热力学数据。结果表明,(NH4)3AlF6经三步分解后,固态产物为AlF3,前两步分解的固态产物分别为NH4AlF4和AlF3(NH4F)0.69,三级反应的分解温度分别为194.9,222.5和258.4 °C;计算了3个反应温度下相关物质的吉布斯自由能变化,采用DSC法测量了3个反应的焓变和熵变。制备了无水氟化铝,XRD分析和失重分析表明,(NH4)3AlF6在400 °C下保温3 h可以得到高纯度的AlF3。
关键词:氟铝酸铵;热分解;氟化铝;热力学数据
(Edited by LI Xiang-qun)
Foundation item: Project (51004034) supported by the National Natural Science Foundation of China; Project (N090302009) supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: HU Xian-wei; Tel: +86-24-83686464; E-mail: huxw@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(11)60977-1