
Electrochemical performance of organic film on copper surface by polymer plating of 6-mercapto-1, 3, 5-triazine-2, 4-dithiol monosodium
YE Qi(叶 奇), KANG Zhi-xin(康志新), LI Yuan-yuan(李元元)
School of Mechanical Engineering, South China University of Technology, Guangzhou 510640, China
Received 15 July 2007; accepted 10 September 2007
Abstract: The 6-mercapto-1, 3, 5-triazine-2, 4-dithiol monosodium (TTN) compound was used to fabricate an organic film on pure copper. The polymer plating process of TTN on pure copper in Na2CO3 aqueous solution and the growth mechanism of poly(6-mercapto-1, 3, 5-triazine-2, 4-dithiol) (PTT) film were studied by means of cyclic voltammetry. The polymer plating under galvanostatic mode at 0.05 mA/cm2 was conducted to generate PTT film on pure copper in the same electrolyte with different polymer-plating time. The film mass was determined by electronic balance and the insoluble fraction in tetrahydrofuran (THF) Is tested. The performance of organic film formed on copper surface was investigated preliminarily by potentiodynamic polarization and electrochemical impedance spectroscopy (EIS). It is found that a slight peak measured at 0 V vs SCE attributes to the oxidation of copper and generated Cu+ or/and Cu2+ to produce Cu-TTN complex, then a strong oxide peak is observed at 0.311 V vs SCE due to the polymerization of TTN for the increase of the film thickness. Electrochemical measurement results reveal that 10 min is an optimum polymer-plated time to obtain high quality film. The results of potentiodynamic polarization show that current density decreases from 1.85 μA/cm2 for bare copper to 0.168 μA/cm2 for polymer-plated copper while polymer-plated time is 10 min. The charge transfer resistances of bare copper and polymer-plated copper are 937 Ω?cm2 and 11.12 kΩ?cm2, respectively. The film capacitor for polymer-plated copper is as low as 1.82 μF?cm2. The EIS results confirm the results of potentiodynamic polarization and reveal that a homogenous and compact film is obtained by polymer plating technique.
Key words: copper; polymer plating; organic film; potentiodynamic polarization; electrochemical impedance spectroscopy
1 Introduction
In the microelectronics industry, there is a need for high dielectric materials in semiconductor, integrated circuit, telecommunication and many other important applications. However, traditional ceramic-based materials, such as piezoceramics, have suffered from both poor stability and flexibility despite their high dielectric constant[1]. Recently, polymeric materials have attracted much attention due to many desirable properties, such as flexibility, low density, low cost, good stability under stress, ease of processability in large area films, and ability to be molded into a desirable configuration[2]. As a simple, relatively inexpensive and convenient route for synthesizing polymers film, electrochemical polymerization has been applied in various research and application fields[3-6]. It is known that 6-substituted 1, 3, 5-triazine-2, 4(1H, 3H)-dithiones tautomerize to be 6-substituted 1, 3, 5-triazine-2, 4-dithiol, which has a thermally stable-triazine ring and two highly reactive thiol groups[7]. Author and co-workers has utilized electrochemical polymerization of this type triazine thiols compounds, referred to as polymer plating to prepare homogeneous, oriented and packed polymer films on metals such as magnesium alloys, iron, copper for directly adhering of metals to rubbers, inhibiting metallic corrosion and fabricating hydrophilic/ hydrophobic surfaces[8-12], especially, a perfluorinated-group-containing triazinethiols compound were used to get a very high dielectric constant (>200) film on aluminum, which can be a potential organic dielectric material in the future electronics industry[13].
Copper is a metal playing an important role in the microelectronics industries due to its high thermal and electrical conductivities, low cost, low resistivity and good electromigration performance[14]. In this work, a triazine thiols compound was used to prepare a organic film on the copper surface by polymer plating and its performance was evaluated preliminarily by electrochemical test.
2 Experimental
2.1 Materials
TTN monomer with thiol radicals (-SH) that have a strong affinity to bivalent metals was prepared by the method described in Ref.[15]. Na2CO3, NaCl, acetone and ethanol were used without any further treatment. Distilled water was employed as a solvent. Analytical reagents (AR) grade chemicals were utilized throughout the present study. The aqueous Na2CO3 solution was used as the supporting electrolyte. The concentrations of TTN and Na2CO3 were kept constant at 5 mmol/L and 0.15 mol/L, respectively.
The copper (purity 99.9%) substrates (30 mm×50 mm×0.5 mm) were cut from a piece of copper plate. The substrates were polished with a series of emery papers, followed by thorough ultrasonic degrease in acetone and distilled water, then dried in air. Prior to any experiment, the substrates were treated as described and freshly used with no further storage.
2.2 Polymer plating
The polymer plating of TTN was performed using an IM6ex electrochemical workstation. The electrolytic cell was equipped with a working electrode (copper plate), two counter electrodes (stainless steel plates) and a reference electrode (saturated calomel electrode, SCE). All potentials given were referenced to SCE. Cyclic voltammetry was conducted from rest potential to 1.1 V at 0.02 V/s in Na2CO3 aqueous solution with or without TTN monomer, and the number of cycles was 5. Polymer plating was conducted with 0.05 mA/cm2 of current density at 25℃ in 0.15 mol/L Na2CO3 aqueous solution of TTN (5 mmol/L) without agitation. After polymer plating, the working electrode was removed from the electrolyte and rinsed with distilled water and ethanol, and dried in air.
2.3 Measurements
The organic film mass was determined by the difference in plate mass before and after polymer plating, using a Sartorius CP225D electronic balance (measuring accuracy was 0.01 mg), and was normalized in μg/cm2. Film performances measured by corrosion tests were carried out in a three-electrode type cell using the reference electrode (SCE) in a luggin capillary and the counter electrode (large area graphite plate). Electrochemical measurements were carried out using potentiodynamic polarization technique and EIS in 3.5% NaCl aqueous solution at 25℃ and in open air with an IM6ex electrochemical workstation. Potentiodynamic polarization tests were always conducted between -0.4 V and 0 V at the scan rate of 0.5 mV/s. Electrochemical impedance measurement was recorded at the corrosion potential following 1 h of immersion time under open-circuit condition. An excitation voltage of 5 mV was used in all tests. The frequency of the potential perturbation varied from 50 kHz to 10 mHz. All impedance data were fitted to appropriate equivalent circuits using Zsimpwin software.
3 Results and discussion
3.1 Polymer plating process on copper surface
The copper electrode was first polarized in 0.15 mol/L aqueous Na2CO3 solution (without TTN monomer) by continuously cycling the electrode potential between rest potential and 1.1 V versus SCE at a potential scan rate of 0.02 V/s, in order to understand the different processes occurred at the copper electrode surface. The cyclic voltammograms of the first to the fifth scan recorded during the polarization of the copper electrode in 0.15 mol/L Na2CO3 aqueous solution is shown in Fig.1(a). It is found that there is no obvious oxide peak before 0.8 V, and a sharp increase in current is observed due to oxygen evolution and/or oxidation of the metal substrate after 0.8 V. However, a very slight oxide peak measured at -0.006 V is seen while the curve is magnified in the 1st cycle as presented in Fig.1(b). This can be explained by the production of Cu2O or/and CuO attributed to the oxidation of copper surface. After polarized in blank Na2CO3 solution, the color of copper surface is almost the same to the surface prior to treatment.
Two anodic peaks were observed when the copper electrode was polarized in the aqueous Na2CO3 solution

Fig.1 Cyclic voltammograms of copper plate in 0.15 mol/L Na2CO3 solution: (a) CV curves of 1-5 cycle between open circuit potential φocp and 1.1 V; (b) Magnified curves of 1st cycle between φocp and 0.1 V
with TTN monomer (Fig.2). The peak A measured at 0 V is similar to the feature in the case of blank Na2CO3 solution, and the copper surface is covered with a slight yellow film after potential scan from φocp to 0 V. It is supposed that the film is produced by the reaction of Cu+ or/and Cu2+ with TTN and peak A marked as results from the oxidation of copper to generate Cu-TTN complexes. Besides this, the peak B measured at 0.311 V, which does not exist in blank solution, reveals that the oxidation and the polymerization of TTN takes place[10]. The anodic current increases after 0.8 V, which is attributed to the dissolution of copper and the generation of oxygen. The visual inspection of the copper surface after the 5th scan reveals the formation of a deep yellow colored PTT film.
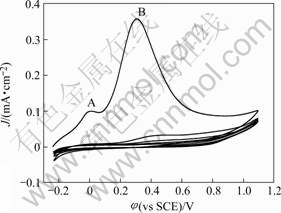
Fig.2 Cyclic voltammograms of copper plate in 0.15 mol/L Na2CO3 solution containing 5 mmol/L TTN
Fig.3 shows the relationship between film mass and polymer-plated time after galvanostatic polymer plating is conducted at 0.05 mA/cm2. The film mass reaches peak at 10 min and a slight decrease is observed. It is supposed that the organic film is destroyed after 10 min due to a long time exposure to high potential of film. In Fig.4, it can be observed that the THF-insoluble fraction
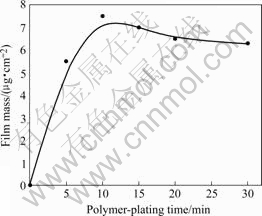
Fig.3 Effect of polymer-plating time on film mass in galvanostatic polymer plating
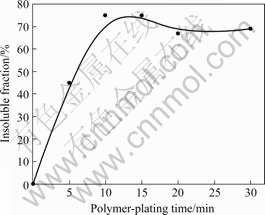
Fig.4 Effect of polymer-plating time on THF-insoluble fraction in polymer plating of TTN on copper plates
is much higher after 10 min than before, suggesting that the outer polymerized surface layer of the organic film has a higher THF-insoluble fraction than the inner layer near the interface between the formed film and the copper substrates. However, it can not be close to 100 % due to the absorption of TTN monomer on top film surface, which is not easily removed by rinsing but soluble in THF.
3.2 Electrochemical performance of organic films on
copper surface
Corrosion of metals involves the transfer of electrical charges in aqueous solution at the metal electrolyte interface. Corrosion protection is often afforded isolating the metal from the corrosive environment using polymer films[16]. Potentiodynamic polarization technique and Tafel extrapolation, and EIS were used to assess the protective properties of film in 3.5% NaCl solution.
The potentiodynamic polarization curves for bare copper and PTT plated copper (10 min) is shown in Fig.5.
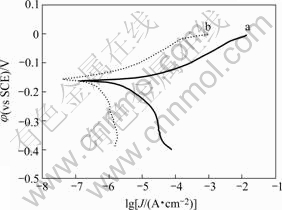
Fig.5 Polarization curves for copper plates in 3.5% NaCl solution: (a) Bare copper; (b) Polymer-plated copper (10 min)
It is clearly observed that the corrosion current density (Jcorr) decreases from 1.85 μA/cm2 for bare copper to 0.168 μA/cm2 for polymer-plated copper. The protection efficiency (Ep) was calculated by using the expression:
Ep=100×(1-J/J0) (1)
where J and J0 denote the corrosion current density of PTT film-plated copper and bare copper, respectively. The protection efficiency calculated from potentiodynamic polarization data is 90.9%. Table 1 presents the potentiodynamic polarization measurement results with different polymer-plated time. It can be seen that the organic film reduces the corrosion rate of copper and 10 min is an optimum time for polymer plating to obtain high quality film. These results reveal that the PTT film acts as a protective layer on copper and improves the overall corrosion performance. The corrosion protection offered by PTT film to copper may be due to the fact that the plated polymer is strongly adherent and uniformly covers the entire copper surface.
Table 1 Potentiodynamic polarization measurement results

To better evaluate film quality, electrochemical impedance measurements were performed at the open circuit potential. In Fig.6 electrochemical impedance data in 3.5% NaCl solution were presented for the bare and PTT plated copper plates (10 min). The equivalent circuits presented in Fig.7 were used to simulate the impedance R, constant phase Q and Warburg impedance W, which may result from diffusion. Rs is solution resistance, while Rct is resistance to charge transfer process. In a chloridze-containing medium, the anodic dissolution of copper proceeds via a two-step metal oxidation at potentials close to the open circuit potential as follows:
Cu+Cl-→CuCl+e- (2)
CuCl+Cl-→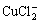 (3)
(3)
The dissolution process is controlled by the diffusion of 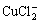 through the solution, so Warburg impedance is observed in Nyquist plots of bare copper[17-19].
through the solution, so Warburg impedance is observed in Nyquist plots of bare copper[17-19].
From Fig.6, it can be seen that there is very good agreement between the experimental and simulated data generated using the circuits. The adopting values in the two cases of Rct are 937 Ω?cm2 and 11.12 kΩ?cm2,

Fig.6 Nyquist plots for copper plates in 3.5% NaCl solution: (a) Bare copper; (b) Polymer-plated copper (10 min)
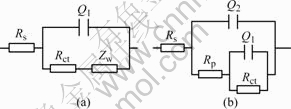
Fig.7 Equivalent circuits used for fitting impedance data: (a) Bare copper; (b) Polymer-plated copper
respectively. The impedance of the constant-phase Q, is defined as ZCPE=A(jω)-α, where ω is frequency of impedance, A is magnitude and α is exponential term(0≤α≤1), while Q1 describes a capacitance at double layer and Q2 represents the film capacitance[20]. In the case of PTT-plated copper, the capacitance of Q2 is as low as 1.82 μF?cm2 while α is 0.985 7, very close to 1, indicating that a homogeneous, compact film is formed on copper surface and no significant corrosion reaction on the copper substrate. The relationship between Rct and polymer-plated time is presented in Fig.8, and the trend coincides and confirms the results of potentiodynamic polarization.
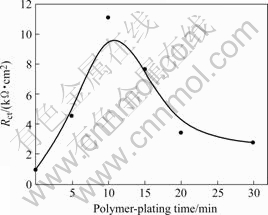
Fig.8 Relationship between polymer-plating time and charge transfer resistance (Rct)
4 Conclusions
1) The polymer plating process of an organic compound of 6-mercapto-1, 3, 5-triazine-2, 4-dithiol monosodium (TTN) analyzed by using cyclic voltammetry reveals that there exists two oxide peaks corresponding to the oxidation of copper and TTN. The generation of poly(6-mercapto-1, 3, 5-triazine-2, 4- dithiol) film (PTT) is divided into two steps. The first step is the reaction between Cu+ or/and Cu2+ and TTN monomer to produce Cu-TTN complexes, and the polymerization between TTN monomers is the second step for the increase of film thickness.
2) Potentiodynamic polarization and EIS are employed to evaluate the performance of organic film. Potentiodynamic polarization results show that the organic film can reduce the corrosion rate from 1.85 μA/cm2 for bare copper to 0.168 μA/cm2 for polymer-plated copper while polymer-plated time is 10 min. The simulation results of EIS show that charge transfer resistances of bare copper and polymer-plated copper are 937 Ω?cm2 and 11.12 kΩ?cm2, respectively. EIS results reveal that the film on copper surface is homogeneous and compacted, therefore polymer plating can be an effective way to fabricate high quality organic film on copper surface. This organic film may have good dielectric property.
References
[1] YAN X Z, GOODSON T. High dielectric hyperbranched polyaniline materials[J]. J Phys Chem B, 2006, 110(30): 14667-14672.
[2] CHUNG T C, PETCHSUK A. Synthesis and properties of ferroelectric fluoroterpolymers with Curie transition at ambient temperature[J]. Macromolecules, 2002, 35(20): 7678-7684.
[3] FENELON A M, BRESLIN C B. The electrochemical synthesis of polypyrrole at a copper electrode: Corrosion protection properties[J]. Electrochim Acta, 2002, 47(28): 4467-4476.
[4] LI C, SHI G Q, LIANG Y Q, YE W, SHA Z L. High-quality poly(p-phenylene) film prepared by electrochemical polymerization of benzene at a stainless steel electrode[J]. Polymer, 1997, 38(20): 5023-5026.
[5] GOSPODINOVA N, TERLEMEZYAN L. Conducting polymers prepared by oxidative polymerization: polyaniline[J]. Prog Polym Sci, 1998, 23(8): 1443-1484.
[6] PATIL S, SAINKAR S R, PATIL P P. Poly(o-anisidine) coatings on copper: synthesis, characterization and evaluation of corrosion protection performance[J]. Appl Surf Sci, 2004, 225(1/4): 204-216.
[7] OISHI Y, KIM J J, NAKAMURA M, HIRAHARA H, MORI K. Synthesis of polysulfides containing s-triazine rings from 6-substituted amino-1, 3, 5-triazine-2, 4-dithiols and 1, 10- dibromodecane[J]. Macromol Rapid Comm, 1999, 20(5): 294-298.
[8] KANG Z X, MORI K, OISHI Y. Surface modification of magnesium alloys using triazine dithiols[J]. Surf Coat Tech, 2005, 195(2/3): 162-167.
[9] KANG Z X, LI Y Y, ZHONG C W, SHAO M, XIA W. Direct joining of acrylic rubber to cast iron with functional nanofilm by polymer plating[J]. Key Eng Mater, 2006, 315/316: 491-495.
[10] BABA H, KODAMA T, MORI K, HIRAHARA H. The corrosion inhibition of copper by potentiostatic anodization in triazinedithiol solutions[J]. Corros Sci, 1997, 39(3): 555-564.
[11] MORI K, KANG Z X, OISHI Y. Effect of 6-dioctylamino-1, 3, 5- triazine- 2, 4-dithiol concentration on the polymer plating on iron plates[J]. Polym J, 2005, 37(11): 862-865.
[12] KANG Z X, LI Y Y, MORI K. Application of polymer plating to inhibit corrosion of magnesium alloy[J]. Mater Sci Forum, 2005, 488/489: 661-664.
[13] MORI K, KANG Z X, WANG F, OISHI Y. Polymer plating of 6-(n-allyl-1, 1, 2, 2-tetrahydro-perfluorodecyl)amino-1, 3, 5-trazine-2, 4-dithiol monosodium on aluminum in magnetic field and its electrostatic capacity [J]. IEEE T Appl Supercon, 2004, 14(2): 1177-1180.
[14] ROSENBERG R, EDELSTEIN D C, HU C K, RODBELL K P. Copper metallization for high performance silicon technology[J]. Annu Rev Mater Sci, 2000, 30: 229-262.
[15] MORI K, HIRAHARA H, OISHI Y, KUMAGAI N. Effect of triazine dithiols on the polymer plating of magnesium alloys[J]. Mater Sci Forum, 2000, 350/351: 223-234.
[16] HERRASTI P, OCON P, IBANEZ A, FATAS E. Electroactive polymer films for stainless steel corrosion protection[J]. J Appl Electrochem, 2003, 33(6): 533-540.
[17] FENG Y, TEO W K, SIOW K S, TAN K L, HSIEH A K. The corrosion behaviour of copper in neutral tap water. Part I: Corrosion mechanisms[J]. Corros Sci, 1996, 38(3): 369-385.
[18] CICILEO G P, ROSALES B M, VARELA F E, VILCHE J R. Comparative study of organic inhibitors of copper corrosion[J]. Corros Sci, 1999, 41(7): 1359-1375.
[19] HU J M, ZHANG J Q, CAO C N. Determination of water uptake and diffusion of Cl- ion in epoxy primer on aluminum alloys in NaCl solution by electrochemical impedance spectroscopy[J]. Prog Org Coat, 2003, 46(4): 273-279.
[20] WU X J, MA H Y, CHEN S H, XU Z Y, SUI A F. General equivalent circuits for Faradaic electrode processes under electrochemical reaction control[J]. J Electrochem Soc, 1999, 146(5): 1847-1853.
(Edited by CHEN Can-hua)
Foundation item: Project(50673028) supported by the National Natural Science Foundation of China; project(04020090) supported by Guangdong Natural Science Foundation of Guangdong Province, China; Project(2005Z3-I0011) supported by Guangzhou International Science and Technology Cooperation Project
Corresponding author: KANG Zhi-xin; Tel: +86-20-87112948; E-mail: zxkang@scut.edu.cn