
Development of copper coatings on molybdenum powders by electroless plating technique
WANG Guang-jun(王光君), WANG De-zhi(王德志)
Schools of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Mo powders with average particle size of 3 μm were attempted to coat with copper by electroless plating technique. The effect of the solution composition and plating conditions on the electroless copper plating was studied. The uncoated and coated powders were subjected to the microstructural studies by SEM and the phases were analyzed by XRD. The results indicate that the Mo powders are coated with copper, at the same time, Mo-Cu composite powders with Cu content ranging from 15% to 85% (mass fraction) can be obtained. The optimal values of pH, HCHO concentration and temperature are in the ranges of 12-13, 22-26 ml/L and 60-65 ℃, respectively. The diffusion-shrinkage autocatalytic model is suggested for the growth mechanism of electroless coating over the surface.
Key words: Mo-Cu; electroless plating copper; powders; growth mechanism
1 Introduction
Mo-Cu composites have been investigated for many years due to its excellent properties such as electrical and thermal conductivity. Higher requirements are needed to enable the development of electronic devices and semiconductor with great power and better properties, such as electrical and thermal conductivity, strength, hardness, especially linear expansion coefficient and gas proof property[1-4].
The synthesis of advanced Mo-Cu composites requires new process. Melting is a classic method. For Mo-Cu composites, however, melting must be performed in a high-temperature arc furnace. This method is not suitable since it requires complex equipment and process, the components are difficult to control and the cost is high. Ordinary powder metallurgy (mixing, molding and sintering) can also be used to producing basic Mo-Cu composites, but the full densification can not be reached. The properties of the composite can not meet the demands of CMSH(Composite Materials for Substrate and Heat sink). Adding elements such as Ni can raise the densities of the composites, but it may have some bad influence on the electronic conductivity[5]. Infiltration is widely used to produce Mo-Cu and W-Cu composites. Although much higher densities can be obtained by this method, there are some disadvantages, e.g., the process is complex, the porosity is difficult to control, and the microstructures are coarser [6]. Many reports have been made on the use of chemical activation to improve the densities and properties of the alloys[7-8], but the disadvantages of the method include the influence of gas produced during sintering and the difficult control of the pore size due to the big shrinkage.
The surface coating[9-11] has been found to be beneficial to strengthen the interface and avoid direct contact of hardening phases. By depositing a uniform and continuous copper on the molybdenum particles at first, compacting the coated molybdenum powders and sintering the green compact or infiltrating them afterwards is a very ideal way to improve the homogeneity and uniformity, minimize compositional segregation and engineer a stronger molybdenum-matrix interface, thus ultimately improve the overall performance of molybdenum alloys.
In this work, copper was coated on the molybdenum powder surface with electroless plating technique and the effect of plating parameters on the electroless plating copper has also been studied.
2 Experimental
2.1 Pretreatment of molybdenum powder sample
Because of the poor bonding force between the molybdenum and copper, it is necessary to pretreat molybdenum powders before electroless plating. Pretreatment can create catalytic crystal centre on the surface of molybdenum powders, which makes the reaction occur on the surface of molybdenum powders.
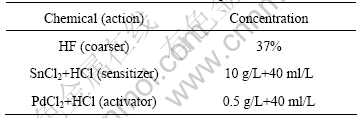
The powders were subjected to coursing for 20 min, sensitizing and activating for 10 min, respectively. The sample was washed three times with the deionized water before each process. The process details for the pretreatment are listed in Table 1.
Table 1 Details of chemicals used for pretreatment
2.2 Copper coating process
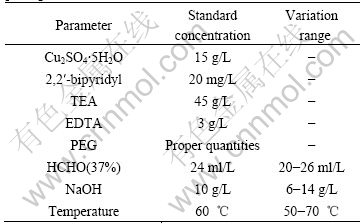
KONDO et al[12-13] have discovered that high quality coating can be obtained under the high speed with triethanolamine(TEA) as complex agent and accelerator. This experiment was taken cupric sulfate as metallic ions for coating, formaldehyde as reduction agent, TEA as chief complex agent, EDTA as assistant complex agent and 2,2′-bipyridyl and polyethylene glycol (PEG) as double stabilizers. The copper coating was obtained in an aqueous solution with the standard parameters and their variation ranges are listed in Table 2. In order to study the effect of one parameter, all the other parameters are kept constant at the value of the standard solution in Table 2, otherwise specified. The significant experimental results were obtained under the condition of keeping the other basic composition fixed, but changing the pH value, temperature, formaldehyde concentration and loadage, respectively. The obtained composite powders were washed three times with the deionzed water and oven dried at 100 ℃ under the nitrogen atmosphere.
Scanning electron microscopes (KYKY2800 and Sarion200) were used to examine the particle surface morphology. D/max2500 X-ray diffractometer was used for diffraction studies. The composition of deposits was determined by chemical analysis. The coating rate is expressed by the mass increment in a unit time, namely v=?m/t, in which ?m shows the mass change from mass of copper coated powders after dried (m) to that of copper uncoated powders(m*).
Table2 Standard parameters and their variation ranges in plating solution
3 Results and discussion
3.1 Parameters affecting copper coating
3.1.1 Effect of pH value on copper coating
As the capability of formaldehyde as a reduction agent is strongly dependent on the pH value of the solution, i.e. the higher the pH value, the stronger the reduction capability of formaldehyde. There is a threshold pH value above which the reduction initiates. The effect of pH value on coating is shown in Fig.1. It can be seen from Fig. 1 that this threshold value is about 12. The reduction reaction is very low or even terminates at a pH value less than 12. However, formaldehyde has a strong tendency to decompose at high pH value through following reaction[14]:
2HCHO+OH-→HCOO-+CH3OH
Therefore, the pH value in this study is kept between 12-12.5, at that time NaOH concentration is about 10 g/L.
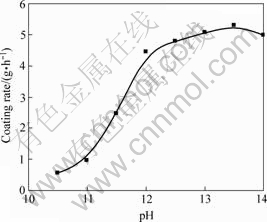
Fig.1 Effect of pH value on coating rate
3.1.2 Effect of temperature on copper coating
The catalytic chemical reactions are usually dependent on the temperature. The result indicates that as the temperature increases, the dissociation and the release rate of metallic ions from complex increase, therefore, the plating rate increases. But if the temperature is too high, the surface quality and the stability of coating solution become poor due to the fast reaction. On the other hand, the surface quality is also poor at the temperatures below 50 ℃. In this case enough energy needed to make the plating go on smoothly can not be provided. Therefore, the suitable temperature is 60-65 ℃.
3.1.3 Effect of HCHO concentration on copper coating
The coating rate exponent increases with the increment of the concentration of HCHO, however the stability of coating solution decreases. It is suggest that the concentration of HCHO should be kept at 22- 26 mL/L. The coating rate is very slow at the concentration of HCHO less than 20 mL/L, however when the concentration of HCHO is higher than 28 mL/L, the color of coating is unusual and there is copper precipitating in the coating solution.
3.1.4 Effect of loadage on copper coating
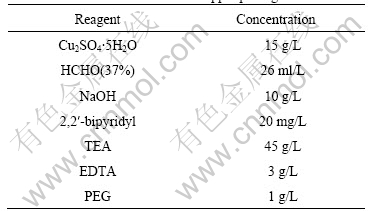
The copper coated powder with different contents of copper can be obtained through changing the loadage. The parameters of the electroless copper coating used in this study are given in Table 3.
Table 3 Parameters of electroless copper plating
The relationship between the copper content and loadage is shown in Fig.2. The results show that Mo-Cu composite powders can be obtained with a rang content of copper from 15%-85% (mass fraction). When the loadage increases continuously to acquire composite powders with content of copper below 15%, there are large numbers of gray powders appearing after coating, which illuminates that vast molybdenum powders have been uncoated. Nevertheless, while the loadage is too low, the stability of coating solution is too poor. Fig.3 shows the XRD results of the Mo-Cu composite powders with different copper contents. It can be seen that with the increment of the content of copper, the intensity of copper peak increases. One of the noticeable points of XRD pattern is that there is no peak of Cu2O phase appearing, which shows that Cu2O contained in copper coating is very small.
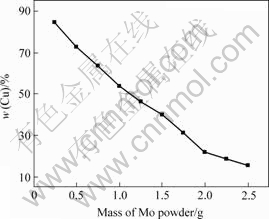
Fig.2 Relationship between copper content and loadage
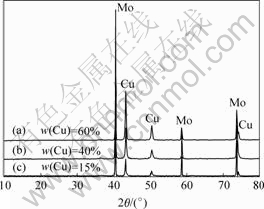
Fig.3 XRD results of Mo-Cu composite powder with different copper contents
3.2 SEM characterization
From the SEM micrographs of uncoated powders, it is seen that uncoated powder particles are composed of individual molybdenum particles and clusters made up of fine molybdenum particles, all of the particles are polyhedral in shape (Fig.4). The copper coated powders in different stages are shown in Fig.5. The powders coated for 1 min (Fig.5(a)) are found to have many spherical particles adhering on the surface. Fig.5(b) shows the morphology of powders coated for 3 min, it can be seen that the spherical particles on surface of molybdenum powders grow further. Fig.5(c) shows that the spherical particles contact each other and cover the whole molybdenum powder when coating is finished. The powders’ shape changes from polyhedron to nearly sphere and the surface is smooth without obvious fluctuation when the reaction is over. The cross-sectional micrograph of coated powders is presented in Fig.6. It is evident that the molybdenum powders are coated with copper successfully and the powder’s shape changes from polyhedron to approximate sphere. However, the agglomeration nature of molybdenum powders leads to the inhomogenity of copper coating, namely, the copper coating of outer cluster is thicker than that of inner cluster. But it doesn’t influence the Mo-Cu composite powders on producing Mo-Cu composite materials. Copper can prevent molybdenum particles from contacting each other during the preparation of Mo-Cu composite materials

Fig.4 Morphology of raw Mo powders

Fig.5 SEM micrographs of coated powder in different stages: (a) Powder coated for 1 min; (b) Powder coated for 3 min; (c) Finished coated powder

Fig.6 Cross-sectional micrograph of coated powders
3.3 Growth model of electroless coating on molybdenum powder surface
Combining the reaction model of electroless coating on fly-ash cenosphere particles introduced by SHULKA et al[15] with the morphology of the copper coated powders in the different stages in this experiment, the diffusion-shrinkage autocatalytic model is created here, and is presented schematically in Fig.7. Supposing that the volume and shape of molybdenum powders don’t change throughout the process of the electroless copper plating. After the sensitization of the molybdenum powders, SnCl2 gets deposited on the molybdenum powder surface. Subsequent activation of the sensitized powders results in the deposition of Pd particles due to the reduction of PdCl2 by SnCl2 (Fig.7(a)). Because the size of Pd particles is much smaller than that of molybdenum powder, the molybdenum powder surface can be considered a plane approximately. The Cu coating begins at this point, when Cu atoms formed by the reduction of Cu2+ ions by HCHO are deposited on the molybdenum powder surface, the reaction is activated by Pd sites. Due to the deposited Cu atoms has analogous nature to Pd particles, subsequent Cu deposition is regarded as surface autocatalytic reaction. Consequently, the equal radius hemispheres taking Pd particle as the centers are formed (Fig.7(b)). With the quantity of copper deposition increment, the sum of active surface areas increases continuously. When the hemispheres are under the condition of being tangent, Cu particles contact with each other and cover the whole molybdenum powder. With the reaction going on, the copper coating grows thicker. When the reaction finishes, a homogeneous equal thick copper coating is produced (Fig.7(c)). As the temperature rises, the reaction runs up and reaction time at each stage shortens, which still accords with growth model.
So the growth model of electroless coating on the molybdenum powder can be divided into three stages, namely incubation period for nucleation production, autocatalysis period and front edge meeting period.
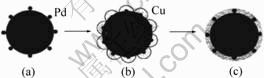
Fig.7 Model for Cu growth on Mo surface: (a) Initial stage; (b) Medium stage; (c) Final stage
4 Conclusion
1) Mo-Cu composite powders with a range content of copper from 15%-85% can be successfully obtained by electroless plating method.
2) During the electroless copper plating, the pH value, temperature and concentration of HCHO in the plating solution should be controlled within 12-12.5, 60-65 ℃ and 22-26 ml/L, respectively.
3) The diffusion-shrinkage autocatalytic model is suggested for the growth mechanism over the surface.
References
[1] MU Ke-qiang, KUANG Yong-geng. The property and application of Mo-Cu composite[J]. Metallic Functional Materials, 2002, 9(3): 26-29.
[2] LI Yun-ping, QU Xuan-hui, DUANG Bo-hua. Development of tungsten-copper (Molybdenum-copper) composite [J]. Materials Science and Engineering of Powder Metallargy, 2002, 7(1) 18-23.
[3] LU Da-ming. The exploration and application of Mo-Cu composites[J]. Powder Metallurgy Industry, 2000, 10(6): 30-33.
[4] ZHOU Jun, WANG Zhi-fa, ZHENG Qiu-bo, WU Hong. Study of the deformation behavior about 70MoCu composites[J]. China Molybdenum Industry, 2006, 30(6): 35-39.
[5] LI Han-feng, TANG Hui-yan, ZHANG Hai-yan, et al. Influence of activating element Ni on performance of Mo-Cu alloy[J]. Materials Science and Engineering of Powder Metallurgy, 2006, 11(3): 185-189.
[6] CAI Yi-xiang, LIU Bo-wu. Problem and solution in desification of W-Cu composite[J]. Powder Metallurgy Technology, 1999, 17(2): 138-144.
[7] LI Chao-lin, GERMAN R M. The properties of tungsten processed by chemically activated sintering[J]. Metall Trans A, 1983, 14: 2031-2041.
[8] JOHNSON J L, GERMAN R M. Chemically activated liquid phase sintering of tungsten-copper[J]. The International Journal of Powder Metallurgy, 1994, 30(1): 91-101.
[9] DIGIAMPAOLO A R, ORDONEZ J G, GUGLEMACCI J M, LIRA J. Electroless nickel-boron coatings on metal carbides[J]. Surf Coat Technol, 1997, 89: 127-131.
[10] WANG B, JI Z, ZIMONE F T, JANOWSKI G M, RIGSBEE J M. A technique for sputter coating of ceramic reinforcement particles[J]. Surf Coat Technol, 1997, 91: 64-68.
[11] YUAN Hai-long, FENG Yi. Electroless plating of carbon nanotube with copper[J]. The Chinese Journal of Nonferrous Metals, 2004, 14(4): 665-669.
[12] KONDO K, MURAKAWA K, NOMOTO K, ISHIKAWA F, ISIDA N, ISIKAWA J. Electroless copper plating solution and process for electrolessly plating copper. US 4834796[P]. 1989-05-30.
[13] KONDO K, AMAKUSA S, MURAKAWA K, KOJIMA K, ISHIDA N, ISHIKAWA J, ISHIKAWA F. Electroless copper plating solution and process for formation of copper film. US 5039338[P]. 1991-08-13.
[14] PENG X L. Preparation of nickel and copper coated fine tungsten powder[J]. Materials Science and Engineering A, 1999, 262: 1-8.
[15] SHUKLA S, SEAL S, AKESSON J. Study of mechanism of electroless copper coating of fly-ash cenosphere particles[J]. Appl Surf Sci, 2001, 181: 35-50.
(Edited by LONG Huai-zhong)
Foundation item: Project (50301017) supported by the National Natural Science Foundation of China
Corresponding author: WANG De-zhi; Tel: +86-731-8877221; E-mail: dzwang@mail.csu.edu.cn