
Kinetics of acid-oxygen leaching of low-sulfur Ni-Cu matte at atmospheric pressure
FAN Chuan-lin(范川林), LI Bin-chuan(李斌川), FU Yan(符 岩), ZHAI Xiu-jing(翟秀静)
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 6 July 2009; accepted 5 January 2010
Abstract: The leaching of low-sulfur Ni-Cu matte in acid-oxygen (CuSO4-H2SO4-O2) solution at atmospheric pressure was researched. This matte was obtained from high grade Ni-Cu matte by magnetic separation, which mainly contained Ni-Cu alloy and a small quantity of sulfides. The effects of temperature, agitation speed, oxygen flow rate, particle size, acid concentration and concentration of copper ion were studied. It is found that the matte particles are leached by shrinking core mechanism and the leaching process is electrochemically controlled. In a temperature range of 30-60 ℃, the surface reaction is rate-limiting step, with an apparent activation energy of 41.9 kJ/mol. But at higher temperature (70-85℃), the rate process is controlled by diffusion through the product layer, with an apparent activation energy of 7.3 kJ/mol.
Key words: acid-oxygen leaching; Ni-Cu matte; Ni-Cu alloy; apparent activation energy; leaching kinetics
1 Introduction
Although nickel-copper mattes are conventionally treated by pyrometallurgical method, hydrometallurgical processes have gained an advantage for reducing the environmental impact of industrial activities and recovering valuable metals from such feeds in recent years[1]. However, the complex chemistry of Ni-Cu matte offers a challenge for the process control and optimization of its leaching process[2]. For many years, numerous studies on the leaching of Ni-Cu matte have been reported. The leaching behaviors of nickel-copper matte at atmospheric pressure were studied[3-6]. The studies on high pressure leaching of nickel-copper matte were also conducted[7-8]. Especially, a semi-empirical mathematical model for the acid-oxygen pressure leaching of Ni-Cu matte has been developed for the improvement of the pressure leaching process[2]. And the studies on the operations and improvements of the existing Ni-Cu matte refining processes have been made[9-12]. In these works, the leaching of Ni alloy in Ni-Cu matte was mentioned and it played an important role in the atmospheric pressure leaching of nickel- copper matte.
Low-sulfur Ni-Cu matte mainly contains Ni-Cu alloy and a small quantity of sulfides. And the dissolution of Ni alloy is the main leaching behavior during the leaching of Ni-Cu matte at atmospheric pressure. A detailed description of this leaching behavior is helpful for the process control and optimization of the leaching of Ni-Cu matte. For this reason, the leaching of low-sulfur Ni-Cu matte in acid-oxygen (CuSO4-H2SO4- O2) solution at atmospheric pressure was investigated in the present work. The effects of temperature, agitation, oxygen flow rate, particle size, acid concentration and concentration of copper ion were studied.
2 Experimental
The raw material used in this study was the low-sulfur Ni-Cu matte, which was obtained from high grade Ni-Cu matte by magnetic separation. From the X-ray diffraction analysis, the main phases present in the matte were nickel alloy, heazlewoodite (Ni3S2) and chalcocite (Cu2S) (Fig.1). Chemical analysis of the matte indicated the following elemental contents: Ni 48.1%, Cu 21.1%, Co 1.08%, Fe 5.56%, S 12.5% and precious metals with Ag 131 g/t, Au 32 g/t, Pt 112 g/t and Pd 74 g/t. A portion of the matte was screened in three different size fractions (74-150, 58-74, 45-58 μm) in leaching tests.

Fig.1 X-ray diffraction pattern of low-sulfur Ni-Cu matte
Leaching tests were performed in a 1.5 L glass vessel placed in a water bath with temperature control. The cover of the vessel was fitted with baffles, a gas inlet, a thermometer and a variable speed stirrer. Gas was supplied by an oxygen pump and a nitrogen pump with adjustable flow meters. And oxygen partial pressure was controlled by adjusting the flow rate ratio of oxygen to nitrogen. In an experimental procedure, 0.8 L solution with predetermined concentrations of copper sulfate and sulfuric acid was added to the leaching vessel and heated to the desired temperature. Then, gas was injected into the pulp with required flow rate and oxygen partial pressure. Stirrer was set to the required speed after 3.20 g matte sample was added. The typical test conditions excepted for a special mention for all tests were particle size of 74-150 μm, sulfuric acid concentration of 0.5 mol/L, copper ion concentration of 10 g/L, temperature of 80 ℃, stirring speed of 500 r/min and gas flow rate per litre slurry of 2.0 L/min with oxygen partial pressure of 100 kPa.
At predetermined time intervals, samples were taken using a pipette and immediately filtered. The solution samples were analyzed for Ni content using inductively coupled plasma (ICP). The fraction of nickel extraction was calculated for examining the leaching rate of the matte.
3 Results and discussion
3.1 Chemical reactions
Based on the literature data for Ni-Cu matte leached in acid-oxygen solution (CuSO4-H2SO4-O2) and the characterization of the leaching liquor and residues obtained[5-7], the following chemical reactions were chosen to study the leaching process of the low-sulfur Ni-Cu matte at atmospheric pressure.
During the leaching process, the main reactions identified for nickel leaching from the matte are
Ni(Fe)+H2SO4+1/2O2→NiSO4(FeSO4)+H2O (1)
2Ni3S2+3/2O2+3H2SO4→3NiSO4+Ni3S4+3H2O (2)
Ni3S2+Ni+4CuSO4→4NiSO4+2Cu2S (3)
Ni(Fe)+CuSO4→NiSO4(FeSO4)+Cu (4)
3.2 Effect of temperature
To determine the effect of temperature, the leaching tests of the matte were conducted in the temperature range of 30-85 ℃ with typical test conditions described above. The effect of temperature is significant on the leaching rate of nickel in the temperature range of 30-60 ℃, while slight in the temperature range of 70-85 ℃ (Fig.2). Significant effect of temperature indicates a leaching process controlled by surface reaction. And slight effect of temperature suggests that the leaching process is generally controlled by a transport process.
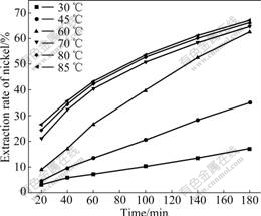
Fig.2 Effect of temperature on nickel extraction from matte
3.3 Effect of agitation rate
A leaching system controlled by transport through a product layer indicates that agitation has a slight effect on the leaching rate. Tests with various agitation speeds were performed with typical test conditions described above. The results are concluded as shown in Fig.3. It is found that the extraction rate of nickel has little dependence on agitation. This agrees with the characteristic of product layer controlled leaching process.

Fig.3 Effect of agitation speed on nickel extraction from matte
3.4 Effect of oxygen partial pressure and gas flow rate
For the acid-oxygen leaching of the matte (Eqs.(1) and (2)), effective oxygen partial pressure is important and mainly determined by gas flow rate and oxygen partial pressure for a special leaching system. Tests were carried out with various gas flow rates and oxygen partial pressures under the typical conditions described above. With the gas flow rate of 1.0 L/min, the extraction rate of nickel increases when oxygen partial pressure increases from 60 kPa to 80 kPa, while it has little effect on the extraction rate under 80-100 kPa (Fig.4). As shown in Fig.5, gas flow rate is significant for the leaching rate of the matte. At atmospheric pressure, gas flow rate is a key factor for effective oxygen partial pressure and the oxidation-reduction potential of leaching system. These also indicate that the leaching of nickel is electro- chemically controlled through half-reaction.

Fig.4 Effect of oxygen partial pressure on nickel extraction from matte

Fig.5 Effect of gas flow rate on nickel extraction from matte
3.5 Effect of particle size
The matte with three particle size distributions was leached with the typical conditions described above. The influence of particle size distribution of the matte on the kinetics of nickel extraction is presented in Fig.6. The extraction rate of nickel decreases with increasing the particle size. The faster rate of nickel extraction observed with the finer particle size may be attributed to the larger surface area and thinner product layer presented by the finer particles. In addition, the morphology and structure of the product layer on particle surface may also have an effect on the extraction rate of nickel.

Fig.6 Effect of particle size distribution on nickel extraction from matte
3.6 Effect of acid concentration
To determine the effect of acid concentration, kinetic tests were performed with different acid concentrations under the typical conditions described. It is found that the nickel extraction with acid concentrations of 0.3 and 0.7 mol/L is faster than that with 0.5 mol/L (Fig.7). This is probably due to more oxygen dissolved in lower acid solution[13], while higher acid may lead to direct reaction between the nickel alloy and sulfuric acid with releasing hydrogen gas. For an industry practice, acid-oxygen leaching of nickel- copper matte is conducted in a lower acid solution. Therefore, nickel alloy dissolution is generally described by Eq.(1).

Fig.7 Effect of acid concentration on nickel extraction from matte
3.7 Effect of copper ion concentration
A small amount of copper ion acts as catalyst for the leaching of Ni-Cu matte, and it has been determined in previous works[14]. In this work, however, it seems that copper ion acts as a reactant for cementation by Eqs.(3) and (4). Kinetic tests were performed with different copper ion concentrations under the typical conditions described above. The results are concluded as shown in Fig.8. It can be seen that, the increase of initial copper ion concentration leads to increasing of the extraction rate of nickel. This indicates that cementation has a little contribution to the nickel extraction in such an acid- oxygen solution.

Fig.8 Effect of initial copper ion concentration on nickel extraction from matte
3.8 Kinetics of nickel leaching from matte
In this work, the well-known shrinking-particle model was chosen to represent the change during the leaching process[15]. The kinetic equations are as follows.
For reactions controlled by surface reaction:
 (5)
(5)
For reactions controlled by transport through a product layer:
 (6)
(6)
where Xb is the extraction rate of nickel; and k is a rate constant. Detailed description of these kinetic models can be found in other works[15-16].
The activation energy of the reactions was determined from the Arrhenius equation (Eq.(7)):
 (7)
(7)
where k is the reaction rate constant; A is the frequency factor; R is the gas constant; and E is the activation energy of the reaction.
A plot of model fitting for nickel extraction versus time based on Fig.2 is shown in Fig.9. It is found that the leaching system is controlled by surface reactions in the temperature range of 30-60 ℃ and transport through a product layer in the temperature range of 70-85 ℃. From the slopes of these plots in Fig.9, rate constants (k/min) were determined and plotted against 103/T (Arrhenius plot) as presented in Figs.10 and 11. Two distinct apparent activation energies, namely 7.3 kJ/mol at high temperatures (70-85 ℃) and 41.9 kJ/mol for lower temperatures (30-60 ℃) were obtained. These values of the activation energy indicated that the rate of the reaction was probably controlled by transport through a product layer at higher temperatures. At low temperatures, the activation energy was quite high, indicating that the rate was controlled by a surface reaction.

Fig.9 Plot of data fitting of nickel extraction (Xb) vs time at various temperatures based on Fig.2

Fig.10 Arrhenius plot of data presented in Fig.9 in temperature range of 30-60 ℃

Fig.11 Arrhenius plot of data presented in Fig.9 in temperature range of 70-85 ℃
4 Conclusions
1) Under the applied test conditions, the kinetics of the leaching of nickel from the matte is influenced by process parameters such as temperature, oxygen flow rate, particle size distribution and acid concentration. Variations in the stirring speed, oxygen partial pressure and copper ion concentration appear to have little or no effect on the leaching kinetics.
2) The matte particles are leached by shrinking core mechanism and the leaching process is electrochemically controlled. The surface reaction is the rate-limiting step in the temperature range of 30-60 ℃, with an apparent activation energy of 41.9 kJ/mol. At higher temperatures (70-85 ℃), however, the rate process is controlled by diffusion through the product layer, with an apparent activation energy of 7.3 kJ/mol.
References
[1] SHEN Yong-feng, XUE Wen-ying, LI Wei. Selective recovery of nickel and cobalt from cobalt-enriched Ni-Cu matte by two-stage counter-current leaching [J]. Separation and Purification Technology, 2008, 60(2): 113-119.
[2] PROVIS J L, van DEVENTER J S J, RADEMAN J A M, LORENZEN L. A kinetic model for the acid-oxygen pressure leaching of Ni-Cu matte [J]. Hydrometallurgy, 2003, 70(1/3): 83-99.
[3] SYMENS R D, QUENEAU P B, CHOU E C, CLARK F F. Leaching of iron-containing copper-nickel matte at atmospheric pressure [J]. Canadian Metallurgical Quarterly, 1979, 18(2): 145-153.
[4] FILMER A O. The non-oxidative dissolution of nickel mattes in aqueous acidic solution [J]. Journal of the Southern African Institute of Mining and Metallurgy, 1981, 81(3): 74-84.
[5] LAMYA R M, LORENZEN L. A study of factors influencing the kinetics of copper cementation during atmospheric leaching of converter matte [J]. Journal of the Southern African Institute of Mining and Metallurgy, 2005, 105(1): 21-27.
[6] LAMYA R M, LORENZEN L. Atmospheric acid leaching of nickel-copper matte from Impala Platinum Refineries [J]. Journal of the Southern African Institute of Mining and Metallurgy, 2006, 106(6): 385-395.
[7] DUTRIZAC J E, CHEN T T. A mineralogical study of the phases formed during the CuSO4-H2SO4-O2 leaching of nickel-copper matte [J]. Canadian Metallurgical Quarterly, 1987, 26(4): 265-276.
[8] RADEMAN J A M, LORENZEN L, van DEVENTER J S J. The leaching characteristics of Ni-Cu matte in the acid-oxygen pressure leach process at Impala Platinum [J]. Hydrometallurgy, 1999, 52(3): 231-252.
[9] PLASKET R P, ROMANCHUK S. Recovery of nickel and copper from high grade matte at Impala Platinum by the Sherritt process [J]. Hydrometallurgy 1978, 3(2): 135-151.
[10] HOFIREK Z, KERFOOT D G E. The chemistry of the nickel-copper matte leach and its application to process control and optimization [J]. Hydrometallurgy, 1992, 29(1/3): 357-381.
[11] HOFIREK Z, NOFAL P J. Pressure leach capacity expansion using oxygen enriched air at RBMR (Pty) Ltd [J]. Hydrometallurgy, 1995, 39(1/3): 91-116.
[12] FUGLEBERG S, HULTHOLM S E, ROSENBACK L, HOLOHAN T. Development of the Hartley Platinum leaching process [J]. Hydrometallurgy, 1995, 39(1/3): 1-10.
[13] NARITA E, LAWSON F, HAN K N. Solubility of oxygen in aqueous electrolyte solutions [J]. Hydrometallurgy, 1983, 10(1): 21-37.
[14] MULAK W. Kinetics of dissolution of synthetic heazlewoodite (Ni3S2) in nitric acid solution [J]. Hydrometallurgy, 1985, 14(1): 67-81.
[15] LEVENSPIEL O. Chemical reaction engineering [M]. New York: Wiley, 1972: 1-31.
[16] SOHN H Y, WADSWORTH M E. Rate processes of extractive metallurgy [M]. New York: Plenum, 1979: 133-241
Foundation item: Project(50774020) supported by the National Natural Science Foundation of China
Corresponding author: FAN Chuan-lin; Tel: +86-24-83687729; E-mail: smmnf@qq.com
DOI: 10.1016/S1003-6326(09)60273-9
(Edited by YANG Bing)